Reference of Blubber Depth and Muscle Depth of Captive Common Bottlenose Dolphins (Tursiops truncatus)
- 1. Jiangxi Provincial Key Laboratory for Animal Health, College of Animal Science and Technology, China
- 2. Department of Veterinary Clinical Medicine, China Agricultural University, China
ABSTRACT
The reports on evaluation and formulation of blubber depth, and muscle depth of common bottlenose dolphin (Tursiops truncatus) with age and sex differences were scarce recently. Twenty-one healthy common bottlenose dolphins (male: 5; female: 16) in managed care were selected to study the association of blubber depth, muscle depth, body weight, plasma biochemistry, sex hormone within four years (2018-2021).The blubber depth and muscle depth of five anatomic sites (umbilical, dorsal, renal, uterovesical, and hepatic locations) were scanned and measured via ultrasonography. The results showed that umbilical blubber depth and umbilical muscle depth presented sex and age-related variations. For adults, there were significant differences in umbilical blubber depth between females and males. For females, the umbilical blubber depth and muscle depth revealed significant differences between the adults with pubescents; for males, the umbilical blubber depth and ratio of umbilical muscle depth to blubber depth demonstrated significant differences between adults and pubescents. Umbilical blubber depth of the adults (2.76 ± 0.34 cm) was the highest at five sites comparing umbilical with dorsal, renal, uterovesical, and hepatic blubber depth, respectively. The dorsal muscle depth was the thickest one. In summary, this study establishes baselines of the blubber depth, muscle depth with sex, age and anatomical sites variations in managed common bottlenose dolphins.
KEYWORDS
Common bottlenose dolphins, Tursiops truncates, Blubber depth, Muscle depth
CITATION
Yu J, Hu G, Xia Z (2022) Reference of Blubber Depth and Muscle Depth of Captive Common Bottlenose Dolphins (Tursiops truncatus). Ann Clin Pathol 9(1): 1160.
INTRODUCTION
Cetacean blubber layers are highly organized tissues consisting of adipocytes, collagen, microvessels, nerves, and elastin fibers, it contains as much as 80-90% lipids [1]. Blubber layers not only contributes to insulation, positive buoyancy, body streamlining, locomotion and heat stress prevention [2,3], but also is the main site of metabolic energy accumulation and mobilization [4,5]. There are three vertical stratifications in blubber layers: the superficial (outer), intermediate (middle), and deep (inner) layers [6].The inner layer is metabolically active while the outer and middle layers are relatively inert [7]. Blubber development and lipid metabolism are imperative for cetaceans survival. Struntz found blubber mass of common bottlenose dolphins increased with age, mean adipocytes size increased during postnatal growth, but the blubber were collected only at the level of mid-thorax, and there were no blubber data of other anatomic sites in dolphins [8]. Stephanie report the body composition of common bottlenose dolphin in sarasota bay from Atlantic [9], it presented a linear regression formula in two time periods, the blubber depth of dolphin were measured four anatomic sites, body composition of your and mature dolphins were compared, however, it did not provided the specific data of blubber depth, meanwhile, the your dolphins were less than and equal to 10 years, and did not divided the your dolphin with the juveniles and pubescents. Therefore, to fill the knowledge gap, we measured the blubber depths with five different anatomic sites of bottlenose dolphins.
In cetaceans, muscle masses play a critical role in locomotion and diving [10], which associated with upstroke and downstroke of cetaceans. Upstroke, which involves muscles that extend the tail, is powdered primarily by two epaxial muscles: muscle multifidus with its caudal extension (muscle extensor caudae medialis) and muscle longissimus with its caudal extension (muscle extensor caudae lateralis). Downstroke, which involve muscles that flex the tail and depression the fluke, is powdered by muscle hypaxalis lumborum with muscle flexor caudae lateralis) and muscle hypaxalis lumborum with muscle extensor caudae medialis [11]. Dorsal musculature of bottlenose dolphins is composed of longitudinal hypaxial and epaxial muscles associated with the vertebral column, including slow oxidative, fast glycolytic, and fast oxidative glycolytic muscle fibers [12,13]. Dearolf and his group indicated that muscle development of bottlenose dolphins was precocial. The percent of muscle mass to body mass in adult dolphins was 25.7% ± 4%, and the percent of blubber mass to body mass was 20.7 ± 2.9%, respectively; these finding suggested muscle and blubber masses accounted for about half of body composition [14]. Therefore, the estimation of body condition not only measure the blubber depth, but also muscle depth need to consider. In the previous study, muscle depth or muscle mass correlated with body condition were not studied, therefore, our research will elucidate these baselines and differences of muscle depth with age and sex in common bottlenose dolphins.
Ultrasonography is a simple and non-invasive technology used to scan dolphins and reveal normal and abnormal visceral organs. It can also verify gestation [15], predict an expecting parturition date [16], and measure blubber depth [17]. Still, there is no comprehensive report on dolphin blubber depth and muscle depth measurements. In this study, we scanned and measured the blubber depth and muscle depth at umbilical, dorsal, renal, uterovesical, and hepatic anatomical sites in common bottlenose dolphins.
Previous studies on cetaceans blubber were focused on pollution, cortisol, sex hormones, and trace metals, but references on gender, age, and anatomic sites differences are not presented and needed to facilitate assessment of nutritional conditions in healthy bottlenose dolphins. Therefore, the gender, age, and anatomic sites (umbilical, dorsal, renal, uterovesical, and hepatic locations) variation of blubber depth and muscle depth in healthy bottlenose dolphins remain elusive. This study will elucidate these baselines and differences Sukkriang reported there was correlation of body visceral fat rating with serum lipid profile and fasting blood sugar in obese adults [18], however, there was no report on correlation blubber depth and muscle depth with biochemical parameter and sex hormone, thus, to fill the knowledge gap, the present study were done.
MATERIALS AND METHODS
Experimental design
Twenty-one healthy common bottlenose dolphins (male: 5; female: 16) in managed care were selected to study the association of blubber depth, muscle depth, body weight, plasma biochemistry, sex hormone within four years (2018-2021). Age were classified by a previous study [19]. Based on the results of Kasuya’ report, ages were determined by birth dates or estimated from body length [20]. There were three age groups in this study: Juveniles: 0-5 y old; pubescents: 6-10 y old; adults: 10-30 y old. Because there was no geriatric dolphins (? 30 y old) lived in these aquariums, so the data were not included in this study.
Animal Care
All dolphins were reared in aquariums that were met the requirements of aquatic mammal rearing facility of the China National Industrial Standard (SC/T6073-2012). All measures were collected using voluntary behaviors without any constraints or drugs. Except of gestation and lactation periods, daily food intake was 2-6% of body weight, and diet consisted of herring (Clupea harengus, 30-40%), capelin (Mallotus villosus, 40-60%), and squid (Loligo spp., 5-10%) (fish was done quality inspection tests for every batch, mainly checked the energy content, heavy metal levels such lead, arsenic and mercury, total bacterial count and total volatile basic nitrogen). Water temperature was maintained between 19 to 23? for bottlenose dolphins. Natural photoperiod was main lighting in daily life, and the appetite, feeding, training, and swimming of dolphins were normal. Powerful life-support systems ensured that water quality at or above standards (China National Industrial Standard SC/T 9411- 2012). Environment enrichments offered according to facility and exhibit schedules.
Ultrasonography
Ultrasonography was performed at least monthly or bimonthly using a SonoSite MicroMAXX (SonoSite Inc., Bothell, WA, USA). Umbilical, dorsal, renal, uterovesical, and hepatic anatomical sites were selected to measure blubber and muscle depth using sagittal or transverse scans (Figures 1,2) with dolphins under voluntary participation during routine husbandry training via ultrasonography.
Figure 1 Ultrasonography locations at uterovesical, umbilical, and hepatic anatomical sites in a common bottlenose dolphin (Tursiops truncatus). Red line: uterovesical location; red cruciform: umbilical location; red pentagon: hepatic location.
Figure 2 Ultrasonography locations at dorsal and renal anatomical sites in a common bottlenose dolphin (Tursiops truncatus). Green ellipse: renal location; purple rectangle: dorsal location.
The umbilical, uterovesical and hepatic sites were examined by placing the transducer perpendicularly or longitudinally on the ventral side of dolphin body in a recumbent position. The hepatic site was located on the right side of abdomen, also paralleled to the pectoral flipper [21]. The dorsal sites were scanned the approximate junction of muscle multifidus and muscle longissmus, and the renal site were scanned in a lateral position. The renal site was located nearby the point of intersection of the transverse and horizontal planes, closely to the lateral midline. The blubber layer included dermis and subcutis, and the muscle layer was adjacent and internal to the blubber layer (Figure 3).
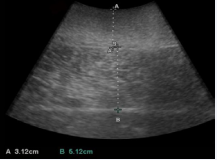
Figure 3 Illustration of blubber and muscle depth at the umbilical location in a common bottlenose dolphin (Tursiops truncatus). A: blubber depth; B: muscle depth.
Bottlenose dolphin epidermis was about 2 – 3 mm [22] and negligible in this research. The formula of the ratio of muscle to blubber depth is: RMB = muscle depth (cm) / blubber depth (cm).
Blood samples collection, centrifugation, storage, and tests
Blood samples were collected monthly at 8:30 - 10:00 in every fasted animal as schedules (i.e., no fish fed for 14h -16h, or before the first morning feed on sample collection days) from the caudal vascular bundle of the ventral or dorsal fluke using a disposable scalp vein set and a 10 mL syringe. Two to six blood samples were collected for each dolphin during research. Blood samples (2 mL) were placed in vacuum tubes (4-5 mL) for plasma biochemistry and sex hormone tests. Plasma samples were centrifuged at 1,000 g for 5 min by TRIAC Centrifuge (Becton Dickinson primary care diagnostics, Becton Dickinson and Company), and plasma was frozen and stored at -20º C until analysis. Laboratory tests were performed once or twice for each sample within 24 - 48h after blood collection. Plasma biochemistry and sex hormone parameters were determined using an Olympus biochemical analyzer (Olympus AU400, Tokyo, Japan).
Statistical analysis
All data were analyzed using SPSS software (Statistical Package for Social Science, 16.0, SPSS Inc., Chicago, Illinois, USA). The data were manifested as mean ± standard deviation (M ± SD). Blubber depth, and muscle depth data from different age, sex, and anatomical sites were analyzed via one-way ANOWA method. Pearson correlation coefficients for all variables were done by bivariate correlation analysis and stepwise linear regression. Differences statistically that were significant at P < 0.01 are presented for this study.
RESULTS
The umbilical blubber depth and muscle depth in bottlenose dolphins by age and sex
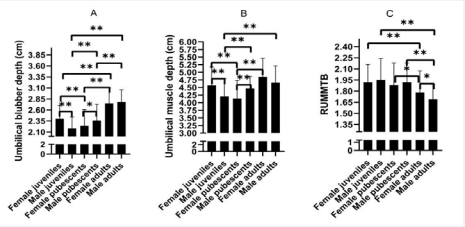
Umbilical blubber depth and muscle depth demonstrated sex and age variations (Figure 4).
Figure 4 Blubber and muscle depths at the umbilical location in healthy common bottlenose dolphins (Tursiops truncatus). A, umbilical blubber depth in dolphins; B, umbilical muscle depth in dolphins; C, ratio of umbilical muscle depth to blubber depth (RUMMTB) in dolphins. Female juveniles, n= 241; male juveniles, n = 32; female pubescents, n = 69; male pubescents, n = 64; Female adults, n = 31; male adults, n = 35. Comparison of blubber and muscle depths of one group with another group (? P<0.05; ??P<0.01); juvenile: 0-5 years old; pubescent: 6-10 years old; adult: 10-30 years old.
For females, the umbilical blubber depth of juvenile, pubescent, and adult was 2.40 ± 0.30, 2.24 ± 0.37, and 2.75 ± 0.35 (cm), respectively; the umbilical muscle depth of juvenile, pubescent, and adult was 4.57 ± 0.61, 4.14 ± 0.59, and 4.85 ± 0.61 (cm), respectively. For males, the umbilical blubber depth of juvenile, pubescent, and adult was 2.18 ± 0.26, 2.36 ± 0.37, and 2.78 ± 0.28 (cm), respectively; the umbilical muscle depth of juvenile, pubescent, and adult was 4.21 ± 0.59, 4.47 ± 0.39, and 4.66 ± 0.55 (cm), respectively.
We found that females umbilical blubber depth and muscle depth demonstrated a U type model with age, while males revealed a growing trend with age. For adults, there were significant differences in umbilical blubber depth between females and males (P < 0.01). For pubescents, the value of umbilical muscle depth of males was higher than that of females (P < 0.01). For juveniles, the values of umbilical blubber depth and muscle depth of females were higher than those of males (P < 0.01). For females, there were significant differences in the umbilical blubber depth and muscle depth values between adults and pubescents (P < 0.01); the umbilical blubber depth and muscle depth values of pubescents were much lower than those of juveniles(P < 0.01); the umbilical blubber depth and ratio of umbilical muscle depth to blubber depth (RUMMTB) values of adults revealed a significant difference from juveniles (P < 0.01); For males, adult umbilical blubber depth and RUMMTB values revealed a significant differences between adults and pubescents; the umbilical blubber depth and muscle depth values of pubescents were much higher than those of juveniles (P < 0.01); umbilical blubber depth, umbilical muscle depth, and RUMMTB of adults demonstrated a significant difference from juveniles (P < 0.01).
Blubber depth and muscle depth at different anatomical sites for adult bottlenose dolphins
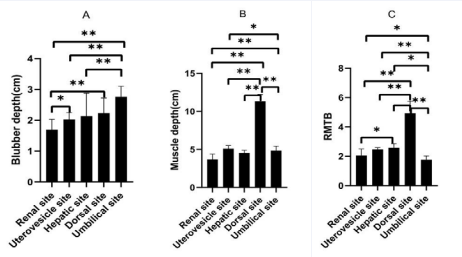
Blubber depth and muscle depth vary by anatomical sites in adult bottlenose dolphins (Figure 5).
Figure 5 Blubber depth and muscle depth of adult healthy common bottlenose dolphins (Tursiops truncatus) at five sites. A, blubber depth at five sites; B, muscle depth at five sites; C, ratio of muscle depth to blubber depth (RMTB) at five sites. Umbilical site, n = 271; dorsal site, n = 15; renal site, n = 10; uterovesical site, n = 6; hepatic site, n = 4; *P<0.05, **P<0.01.
Umbilical blubber depth (2.77 ± 0.32 cm) is the highest blubber depth at five sites (umbilical, dorsal, renal, uterovesical, and hepatic), and dorsal muscle depth (11.31 ± 0.91 cm) is the thickest muscle depth at these sites. Ratio of dorsal muscle depth to blubber depth was the highest (4.93 ± 0.82), while Ratio of umbilical muscle depth to blubber depth was the lowest (1.77±0.28). The ratio of dorsal muscle depth to umbilical muscle depth in adult bottlenose dolphins was more than double (2.23 ± 0.37).
DISCUSSION
In emaciated animal, lipid mobilization is evidenced by a reduction in adipocytes size of the intermediate and deep layers; In lactating animals, total blubber lipid content and the size of adipocytes in the deep layers decreased to meet the energy demands of calf [7]. Adipocytes count and adipocyte size in the middle layer was the highest or the largest of the three layers [23]. This may be the result of the microvascular distribution in blubber stratification One study shows that the microvascular characteristic of the outer layers in cetaceans resembled the adipose tissue of terrestrial mammals, but the microvascular percentage and microvascular branching of the middle and deep layers of T.truncatus were higher than that of other marine mammals [23]. This specific structure with abundant microvessels benefits lipid metabolism in T.truncatus. These findings are consistent with studies of harbor porpoises (Phocoena phocoena) and mysticete whales, where lipid deposition and metabolism of the inner layers are more active than that of outer ones [6]. Because the content of polyunsaturated fatty acid (PUFA) was the highest in the deep layer (inner layer) of harbor porpoises; Smith’s study illustrated the levels of monounsaturated fatty acids were greatest in the outer layer of common dolphins (Delphinus sp.), whereas levels of saturated and PUFA were greatest in the inner layer [24]. One report indicated the fat reserve was dramatically consumed during weaning in grey seal pups (Halichoerus grypus) [25]. Moreover, nerves may be involved in the lipid metabolism. One Japanese study indicated that there were nerve fiber bunches in the inner layer of blubber of common bottlenose dolphins and Pacific white-sided dolphins [26]. In conclusion, the blubber lipid of the deep layer in marine mammals may be mobilized first to meet the energy requirements of the physiological state or pathological condition, then the middle layer will be consumed and depleted.
The blubber development and metabolism play a crucial part in dolphin life. We found the umbilical blubber depth of common bottlenose dolphins increased with age. One study indicated that blubber depth increases after postnatal growth because of the size of adipocytes rather than the quantity [8]. Our results shown umbilical blubber depth and muscle depth presented age and sex variations. It was found the umbilical blubber depth and muscle depth of the female demonstrated a U type model with age, it manifested the umbilical blubber depth and muscle depth of the pubescents were the lowest in three age groups, while males revealed a growing linear association with age. The reason remains unclear. Some study indicated the blubber depth (BD) increased after postnatal growth by adipocytes size rather than adipocytes number [8], this matches our findings that the UMBD increase with age.
The blubber and muscle are pivotal tissues in cetacean life, the blubber depth and muscle depth varied in five locations, it indicated the blubber thickness of bottlenose dolphins is not homogenous. The value of UMBD of T.truncatus was strongly and positively correlated with that of UMMD. It suggests UMBD and UMMD have a synergistic effect on lipid and muscle metabolism for T.truncatus. When blubber accumulates or mobilizes, the muscle tissues also increase or decrease. We found that umbilical blubber depth was the thickest blubber depth in five locations, it indicated the umbilical blubber depth may be the primary location of blubber energy accumulation and mobilization, which presented species-related variation [27]. The dorsal muscle depth was the deepest muscle depth in five locations. Dorsal muscles of dolphins consist of the longitudinal epaxial muscles (longissimus and multifidus) which were the most powerful muscle to generate bending characteristic during dolphin swimming and jumping [28]. The muscle of umbilical and uterovesical site are rectus abdominis, our study found the umbilical muscle depth were higher than that of uterovesical muscle depth, it suggest the depth of rectus abdominis are variable in the belly. Moreover, the umbilical muscle depth is the second thickest muscle depth in five sites. The muscles of hepatic site and renal site are internal oblique, they also shown different muscle depth. Taken together, the muscle depth are not uniform, they varied with sites. We found ultrasonography is a practical and efficient tool to measure the blubber depth and muscle depth in dolphins to assess obesity or malnutrition of common bottlenose dolphins.
CONCLUSION
The blubber depth, and muscle depth in common bottlenose dolphins presented age and sex variations. Moreover, this study firstly found blubber depth and muscle depth varied at five anatomical sites. It suggested the body condition assessment is complex process, and need to get comprehensive data and information to evaluate animal welfare and fitness. However, the data collection is difficult and insufficient in practice environment and limit of logistical support. It is difficult to collect all parameters perfectly. Therefore, our results blubber depth and muscle depth could be useful in managed and wild environment to give some professional advices and decision-making.
REFERENCES
- Evans K, Hindell MA, Thiele D. Body fat and condition in sperm whales, Physeter macrocephalus, from southern Australian waters. Comp Biochem Physiol A Mol Integr Physiol. 2003; 134: 847-862.
- Bagge LE, Koopman HN, Rommel SA, McLellan WA, Pabst DA. Lipid class and depth-specific thermal properties in the blubber of the short-finned pilot whale and the pygmy sperm whale. J Exp Biol.2012; 215: 4330-4339.
- Heath ME, Ridgway SH. How dolphins use their blubber to avoid heat stress during encounters with warm water. Am J Physiol. 1999; 276: R1188-1194.
- Ackman RG, Hingley JH, Eaton CA, Sipos JC, Mitchell E D. Blubber fat deposition in mysticeti whales. Can J Zool. 1975; 53: 1332-1339.
- Jackson CH, Gales R, Virtue P, Nichols PD. Stratification, sex and ontogenetic effects on the lipid and fatty acid profiles in the blubber of sperm whales from Tasmanian waters. J Comp Physiol B. 2022; 192: 789-804.
- Koopman HN, Iverson SJ, Gaskin DE. Stratification and age-related differences in blubber fatty acids of the male harbour porpoise (Phocoena phocoena). J Comp Physiol B. 1996; 165: 628-639.
- Montie EW, Garvin SR, Fair PA, Bossart GD, Mitchum GB, McFee WE, et al. Blubber morphology in wild bottlenose dolphins (Tursiopstruncatus) from the Southeastern United States: influence of geographic location, age class, and reproductive state. J Morphol. 2008; 269: 496-511.
- Struntz DJ, McLellan WA, Dillaman RM, Blum JE, Kucklick JR, Pabst DA. Blubber development in bottlenose dolphins (Tursiops truncatus). J Morphol. 2004; 259: 7-20.
- Adamczak SK, Holser RR, Costa DP, McCabe EJB, Wells RS. Body composition of common bottlenose dolphin in sarasata bay, Florida. Frontier in Marine Science. 2021; 8: 1-12.
- Noren SR, Williams TM, Pabst DA, McLellan WA, Dearolf JL. The development of diving in marine endotherms: preparing the skeletal muscles of dolphins, penguins, and seals for activity during submergence. J Comp Physiol B. 2001; 171: 127-134.
- Berta A, Sumich JL, Kovacs KM. Marine Mammal: Evolutionary Biology.Third ed. United States of America: Academic Press; 2015.
- Kielhorn CE, Dillaman RM, Kinsey ST, McLellan WA, Gay DM, Dearolf JL, at al. Locomotor muscle profile of a deep (Kogia breviceps) versus shallow (Tursiops truncatus) diving cetacean. J Morphol. 2013; 274: 663-675.
- Kroeger JP, McLellan WA, Arthur LH, Velten BP, Singleton EM, Kinsey ST, at al. Locomotor muscle morphology of three species of pelagic delphinids. J Morphol. 2020; 281: 170-182.
- Dearolf JL, McLellan WA, Dillaman RM, Frierson D, Jr Pabst DA. Precocial development of axial locomotor muscle in bottlenose dolphins (Tursiops truncatus). J Morphol. 2000; 244: 203-215.
- Ivancic M, Gomez FM, Musser WB, Barratclough A, Meegan JM, Waitt SM, et al. Ultrasonographic findings associated with normal pregnancy and fetal well-being in the bottlenose dolphin (Tursiops truncatus). Vet Radiol Ultrasound. 2020; 61: 215-226.
- Lacave G, Eggermont M, Verslycke T, Brook F, Salbany A, Roque L, at al. Prediction from ultrasonographic measurements of the expected delivery date in two species of bottlenosed dolphin (Tursiops truncatus and Tursiops aduncus). Vet Rec. 2004; 154: 228-233.
- Lane EP, de Wet M, Thompson P, Siebert U, Wohlsein P, Plon S. A systematic health assessment of indian ocean bottlenose (Tursiops aduncus) and indo-pacific humpback (Sousa plumbea) dolphins incidentally caught in shark nets off the KwaZulu-Natal Coast, South Africa. PLoS One. 2014; 9: e107038.
- Sukkriang N, Chanprasertpinyo W, Wattanapisit A, Punsawad C, Thamrongrat N, Sangpoom S. Correlation of body visceral fat rating with serum lipid profile and fasting blood sugar in obese adults using a noninvasive machine. Heliyon. 2021; 7: e06264.
- Venn-Watson S, Jensen ED, Ridgway SH. Effects of age and sex on clinicopathologic reference ranges in a healthy managed Atlantic bottlenose dolphin population. J Am Vet Med Assoc. 2007; 231: 596- 601.
- Kasuya T, Izumisawa Y, Komyo Y, Ishino Y, Maejima Y. Life history parameters of common bottlenose dolphins off Japan. IBI Rep. 1997; 7: 71-107.
- Brook F, Bonn WV, Jensen E. Chapter 26 Ultrasonograhy. In: Dierauf LA, D Frances MG, editors. CRC Handbook of Marine Mammal Medicine CRC press; 2001. p. 601.
- Cozzi B, huggenberger S, Oelschlager H. Anatomy of Dolphins- Insights into body structure and functure. London: Academic Press is imprint of Elsevier; 2016.
- McClelland SJ, Gay M, Pabst DA, Dillaman R, Westgate AJ, Koopman HN. Microvascular patterns in the blubber of shallow and deep diving odontocetes. J Morphol. 2012; 273: 932-942.
- Smith HR, Worthy GA. Stratification and intra- and inter-specific differences in fatty acid composition of common dolphin (Delphinus sp.) blubber: implications for dietary analysis. Comp Biochem Physiol B Biochem Mol Biol. 2006; 143: 486-499.
- Bennett KA, Speakman JR, Moss SE, Pomeroy P, Fedak MA. Effects of mass and body composition on fasting fuel utilisation in grey seal pups (Halichoerus grypus Fabricius): an experimental study using supplementary feeding. J Exp Biol. 2007; 210: 3043-3053.
- Hashimoto O, Ohtsuki H, Kakizaki T, Amou K, Sato R, Doi S, et al. Brown adipose tissue in cetacean blubber. PLoS One. 2015; 10: e0116734.
- Zeng X, Ji J, Hao Y, Wang D. Topographical distribution of blubber in finless porpoises (Neophocaena asiaeorientalis sunameri): a result from adapting to living in coastal waters. Zool Stud. 2015; 54: e32.
- Long JH Jr, Pabst DA, Shepherd WR, McLellan WA. Locomotor design of dolphin vertebral columns: bending mechanics and morphology of Delphinus delphis. J Exp Biol. 1997; 200: 65-81.