Effect of Casearia Sylvestris Hydroalcoholic Crude Extract on Histological Parameters for Skin Reorganization in a Pre-Clinical Model of Postoperative Wound
- 1. Undergraduation in Medicine – Southern University of Santa Catarina (UNISUL), Brazil
- 2. Laboratory of Experimental Neuroscience (LANEX) – UNISUL, Brazil
- 3. Histopathology and Cytology Laboratory, Federal Institute of Santa Catarina (IFSC), Brazil
- 4. Post-Graduation Program in Health Science – UNISUL, Brazil
Abstract
The skin is the largest organ in the body, with a total area of about 2 m². It is estimated that one in Twenty-two people in the world suffers from an epithelial surgical insult, of which there is a large proportion of anomalous repairs, resulting from exacerbation of local inflammatory processes. Therapeutic strategies that minimize such conditions are of great interest, including that use of folkloric herbal medicines. Thus, the present experimental study intends to investigate the effect of CS on histological parameters of epithelial reorganization in a pre-clinical model of postoperative wound. To that end we maintained 40 male Swiss mice in controlled environmental conditions. On the first day the animals were submitted to a 5 mm plantar epithelial incision in the right hind paw, one stitch suture and 50 µg topic treatment solutions: dexamethasone 0,5 mg/mL, acetone 2,5% and hydroalcoholic crude extract of CS (HCE-CS) 30 mg/site every 24 hours for 5 days. Every day after the treatments the surgical wound were photographed and on the third and fifth day, the mice were euthanized, the hind paws were collected for histological analyzes. To compare all different treatment groups we used one-way ANOVA methodology, followed by post hoc Dunnett’s test for comparison. All animals shrank their scars on fifth day and in the group treated with HCE-CS we observed a larger dermal thickness and smaller scar proportions related to other groups on third day, showing an accelerated inflammation resolution.
Keywords
- Casearia
- Wound healing
- Surgical wound
- Skin
Citation
Costa DM, Pereira HL, Ferreira EC, Poeta Fermino PM, Piovezan AP (2022) Effect of Casearia Sylvestris Hydroalcoholic Crude Extract on Histological Parameters for Skin Reorganization in a Pre-Clinical Model of Postoperative Wound. J Dermatolog Clin Res 10(1): 1145.
ABBREVIATIONS
HCE-CS: Hydroalcoholic Crude Extract of Casearia Sylvestris; CS: Casearia Sylvestris
INTRODUCTION
The skin is the largest organ in the body, covering an area of about 2 m2; it provides a mechanical and biological barrier against chemical, physical and pathogenic insults [1,2]. After an injury to the skin, the body tries to restore its integrity to the original state before the injury, with the physiological result of wound healing being the formation of replacement tissue or a scar as result of any insult to the skin, including surgical procedures [3,4]. Operative interventions require a solution of continuity through the tissues and, therefore, an uncontaminated wound, in which healing depends on cellular and molecular events [5] hemostasis, inflammation, cell proliferation, granulation tissue formation, re-epithelialization, neovascularization, matrix deposition and remodeling [6,7]. It is estimated that 313 million surgeries were performed in 2012, an increase of 138% since 2004 [8], corresponding to one operation for every 22people.
These statistics are especially important if we consider that wounds when incorrectly repaired can cause physical, psychological and aesthetic discomfort, leading to psychological stress, depression and social withdrawal that lead to a reduced quality of life [9]. Some available treatment modalities that aim to reduce inflammation are the systemic or topical use of corticosteroids, radiotherapy, cryotherapy, compression therapy, and surgical methods that reduce skin tension [10,11]. However, currently available medical treatments are not fully effective. Defective skin regeneration has been understood as a result of continuous and localized inflammation of the reticular dermis layer, increased fibroblasts, blood vessels and collagen deposits, as well as raised pro-inflammatory flags [12]. CS plant is used by different communities, even indigenous tribes, to treat skin lesions [13]. Some properties of the crude aqueous extract of its leaves observed in pre-clinical studies are its anti-inflammatory activity [14], due to enzymes that inhibit the phospholipase A2, an enzyme category involved in the fatty acids supply important for skin homeostasis [15] and the ability to inhibit the infiltration of inflammatory cells into injured tissues [16]. Hereof, since the epidemiology of skin scars is frequent worldwide, with a great impact on the quality of life of affected patients, the present study aimed to investigate the possible influence (Figure 1) of topical treatment of the HCE-CS on different parameters observed in a post-operative wound model.
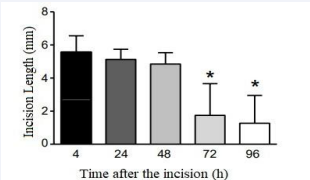
Figure 1 Time course of healing, observed by the reduction of the macroscopic incision extension
MATERIALS AND METHODS
Animals
The population consisted of forty healthy, male, 2 months old, Swiss mice from the Federal University of Santa Catarina bioterium. The animals were acclimatized to 22±2ºC, in a 12- hour light to 12-hour dark cycle, with access to food and water ad libitum at LANEX - UNISUL. Approximately 10 mice were accommodated per polypropylene box covered with sawdust. Mice that, after the surgical procedure, did not survive or presented signs of infections or bleeding in the postoperative wounds were excluded from the sample.
Ethics
The project was approved by the Ethics Committee on Animal Use (CEUA/UNISUL), under registration: 19.045.4.01.IV. All experiments were performed in accordance with the Care of Laboratory Animals Guide and Ethical Guide for Experimental Investigations in Conscious Animals.
HCE-CS
The plant was collected from the botanical Horto at the campus of UNISUL, in the city of Tubarão, Santa Catarina, Brazil and was identified with the assistance of Professor Jasper Zanco (Agronomy, UNISUL), by direct comparison with a voucher specimen deposited at the herbarium Laelia Purpurata at this University. Leaves were dried, minced and standardized through sieves, using particles ranging between 250 μm and 850 μm. The extract was produced at a ratio of 1:3 of plant and solvent with 70% ethanol used as extractor liquid. The plant was engorged in a minimal volume of ethanol and the remaining quantity of extractor liquid was subsequently added to this material. This process of maceration was dynamic, with the extractor liquid remaining in contact with the plant for 10 days under constant agitation in a closed container at room temperature. At the end of this process, the extract was filtered using a vacuum pump. HCE- CS was then concentrated in rotavapor under reduced pressure to evaporate the ethanol. This process continued with the extract
being deposited in a round bottom container which was placed within a water bath at a temperature of 35,1°C to 40,1°C until total ethanol evaporation had occurred. The concentrated HCE- CS was then frozen and lyophilized, and placed in posterior storage in a desiccator until required for use.
Postoperative Wound Induction
The model was adapted [17] for plantar incision with the animals under anesthesia induced via a nasal mask of 1%-2% isoflurane. After checking the animal state of consciousness, the right hind paw was cleaned by 10% povidone-iodine solution, on which a 5 mm longitudinal incision was made through the skin and fascia of the plantar part with a scalpel blade number 11. The incision started 2 mm from the proximal end of the calcaneus towards the toes. After the incision, the skin was sutured with 8.0 nylon threads and the suture covered with 10% povidone-iodine solution.
Treatment
All animals were treated once a day for 3 to 5 days, starting from the day of the plantar incision, with 20 µL/site of the following solutions: a) vehicle: 2.5% acetone solution; b) dexamethasone
0.05 mg/dL diluted in the vehicle; c) Casearia: HCE-CS at a dose of 30 µg/mL diluted in the vehicle. The allocation of animals in different groups was done randomly.
Macroscopic evaluation
From the first day of treatment to the last day, the animals were placed in an acrylic chamber and had their hind legs photographed, along with a ruler, by a Canon camera. Their paws were illuminated and fixed at a standard distance. After the images obtained, the measurements of the length of the scar with the Image J program were made (Figure 2).
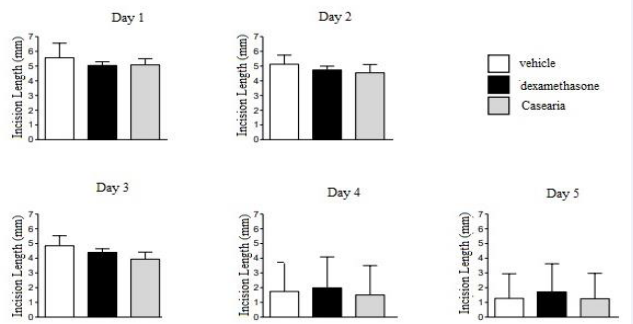
Figure 2 Macroscopic analysis of the influence of different treatments on the time course of healing.
Histology
On the third day and on fifth the animals were randomly chosen to be euthanized by intraperitoneal injection of 300 mg/ kg ketamine and 30 mg/kg xylazine, after loss of consciousness and death, the right hind legs of the mice were dissected and fixed in 10% formalin, decalcified with 3% formic acid, kept in ethanol 70%, dehydrated, clarified in xylene and embedded in paraffin at 58ºC. Subsequently, blocks were made; these were cut on a Leica RM 2025 microtome at a thickness of 5 μm. The sections were then collected in a thermostatic bath at 52ºC and placed on slides. For histomorphometry, the sections obtained were deparaffinized, hydrated, stained with hematoxylin and eosin, dehydrated and mounted. To obtain the images, the photos were taken using an Opticam camera connected to a Quimis optical microscope. The images were captured at 40x magnification and analyzed by the Image software assessing the microscopic length of the scar seen in cross-section and the thickness of the dermis.
Statistical Analysis
Data from independent samples were analyzed using the Graphpad Instat® 6.0 program and, first, evaluated for normality. To compare the different study groups, a one-way Ordinal Analysis of Variance (ANOVA) test was used followed by Dunnett’s post hoc, with data presented as mean ± standard error of the mean.
RESULTS AND DISCUSSION
cally significant reduction in the macroscopic length of the incisional lesion after the 4th day in all groups, when the mean values of this outcome changed from 5.5 ± 1.0 mm to 1, 7 ± 1.9 mm. On the other hand, this effect was not statistically significantly in any treatment group at the different time periods tested in this study. Regarding the histomorphometric analysis, the effect on the thickness of the dermis was greater in the group treated with HCE-CS with an average of 12.69 ± 1.92 µm, whereas in the group treated with dexamethasone was the lowest at 6.08 ± 1.54 µm and 6.47 ± 0.70 µm in the vehicle group. The transversal section scar microscopic length was longer in the vehicle group with 107.50 ± 14.80 µm, in the dexamethasone group the mean was 68.88 ± 12.46 µm and shorter in the HCE-CS with a mean of 58.28 ± 10.64 µm. Both measurements showed statistical significance, with a value of P = 0.0549 for the microscopic length of the cross section and a value of P = 0.0124 for the dermis thickness.
Through hydroalcoholic solutions of the macerated leaves of CS, medicinal plastids abbreviate the healing process through the improvement of capillary permeability, angiogenesis and cleaning of cellular debris [18]. Corroborating these findings, structurally in our research it was possible to visualize a faster return to dermal homeostasis of postoperative wounds from the third day of scar evolution, through a shorter scar length and greater dermal thickening.
It has already been pointed out in a previous study that HCE- CS promotes a reduction of edema in the ear of mice, in addition to reducing pro-inflammatory enzymatic activity, promoting
histological reorganization of the skin, with a decrease in the infiltration of mast cells [19]. A comparison of its efficacy and potency in both models is possible because the dose of the extract used in both studies was the same.
Another study, also edematogenic model, treated with a different species of Casearia, demonstrates medicinal properties common to the genus, related to share phytoconstituents, visualized by the plant ability to scavenge several free radicals, and a lower inflammatory infiltrate [20], histological data similar to the result found by our study. Another author [21] used a model of second-degree burns and assessed the beneficial effect for a biofilmform incorporating a hydroalcoholic CS extract. Despite the re-epithelialization and neovascularization improvements, no effect was found on scar width or new collagen formation, similarly to our macroscopic results. In the present study, the fact that HCE-CS increased the thickness of the treated skin, at first, may be intriguing, since the increase in this region indicates a greater activity of fibroblasts. However, it remains for further studies to investigate whether this is an initial effect of the plant to promote an improvement in the final outcome, since the microscopic wound length was reduced by the treatment.
CONCLUSION
In conclusion, the present study expanded the literature data on the effect of CS in a post-surgical skin lesion model, demonstrating the benefit on microscopic aspects related to skin reorganization (Figure 3,4).
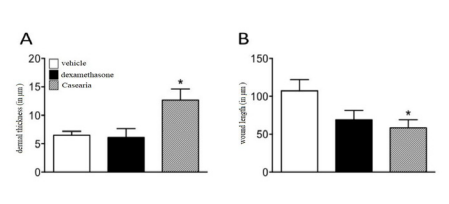
Figure 3 Influence of HCE-CS on microscopic parameters of skin reorganization. Results presented as a mean ± standard deviation of the mean (n=8). One-way ANOVA, followed by Dunnet’s, P ≤ 0.05.
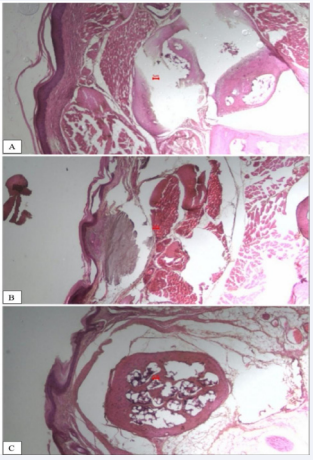
Figure 4 Histomorphometric analysis of the influence of HCE-CS on skin reorganization parameters (skin thickness and scar length). A: Casearia; B: vehicle; C: dexamethasone.
ACKNOWLEDGEMENTS
We thank LANEX, as well as the National Council for Scientific and Technological Development, since this analysis derives from another study previously approved by this institution, through the Program Scientific Initiation Scholarship.
REFERENCES
- Yousef H, Alhajj M, Sharma S. Anatomy, Skin (Integument). Epidermis 2018.
- Belkaid Y, Tamoutounour S. The influence of skin microorganisms on cutaneous immunity. Nat Rev Immunol. 2016; 16: 353-366.
- Wong R, Geyer S, Weninger W, Guimbertau JC, Wong JK. The dynamic anatomy and patterning of skin. Exp Dermatol. 2016; 25: 92-98.
- Khansa I, Harrison B, Janis JE. Evidence-Based Scar Management: How to Improve Results with Technique and Technology. Plast Reconstr Surg. 2016; 138: 165-178.
- Haensel D, Dai X. Epithelial-to-mesenchymal transition in cutaneous wound healing: Where we are and where we are heading. Dev Dyn. 2018; 247: 473-480.
- Gonzalez AC, Costa TF, Andrade ZA, Medrado AR. Wound healing - A literature review. An Bras Dermatol. 2016; 91: 614-620.
- Childs DR, Murthy AS. Overview of Wound Healing and Management. Surg Clin North Am. 2017; 97: 189-207.
- Weiser TG, Haynes AB, Molina G, Lipsitz SR, Esquivel MM, Uribe T, et al. Size and distribution of the global volume of surgery in 2012. Bull World Health Organ. 2016; 94: 201-209F.
- Kinani ME, Duteille F. Scar Epidemiology and Consequences. Textbook on Scar Management. 2020.
- Ward RE, Sklar LR, Eisen DB. Surgical and Noninvasive Modalities for Scar Revision. Dermatol Clin. 2019; 37: 375-386.
- Deflorin C, Hohenauer E, Stoop R, Van Daele U, Clijsen R, Taeymanns J. Physical Management of Scar Tissue: A Systematic Review and Meta- Analysis. J Altern Complement Med. 2020; 26: 854-865.
- Moretti L, Stalfort J, Barker TH, Abebayehu D. The interplay of fibroblasts, the extracelular matrix, inflammation in scar formation. J Biol Chem. 2022; 298: 101530.
- Bueno PCP, Abarca LFS, Anhesine NB, Giffoni MS, Pereira FMV, Torres RB, et al. Infraspecific Chemical Variability and Biological Activity of CS from Different Brazilian Biomes. Planta Med. 2021; 87: 148-159.
- Carvalho FA, Uchina HS, Borges FA, Oyafuso MH, Herculano RD, Gremião MPD, et al. Natural membranes of Hevea brasiliensis latex as delivery system for CS leaf components. Rev Bras Farmacogn. 2018; 28: 102-110.
- Yamamoto K, Miki Y, Sato M, Taketomi Y, Nishito Y, Taya C, et al. The role of group IIF-secreted phospholipase A2 in epidermal homeostasis and hyperplasia. J Exp Med. 2015; 212: 1901-1919.
- Albano MN, Silveira MR, Danielski LG, Florentino D, Petronilho F, Piovezan AP. Anti-inflammatory and antioxidant 429 properties of hydroalcoholic crude extract from CS. J Ethnopharmacol. 2013; 147: 612-617.
- Pogatzki EM, Raja SN. A mouse model of incisional pain materials and methods. Anesthesiology. 2003; 99:1023-7
- Luna MFM, Delgado JEC, Balderas VYM, Villagrana ARC. Medicinal Plant Extracts and Their Use As Wound Closure Inducing Agents. J Med Food. 2019; 22: 435-443.
- Heymanns AC, Albano MN, Silveira MR, Muller SD, Petronilho FC, Gainski LD, et al. Macroscopic, biochemical and hystological evaluation of topical anti-inflammatory activity of CS in mice. J Ethnopharmacol. 2020; 264: 113139.
- Campognara C, Brum EDS, Belke BV, Brum TF, Jesus RDS, Piana M, et al. Casearia decandra leaves present anti inflammatory efficacy in a skin inflammation model in mice. J Ethnopharmacol. 2020; 249: 450 112436.
- Campos EP, Trombini LN, Rodrigues R, Portella DL, Werner AC, Ferraz MC, et al. Healing activity of CS in second-degree scald burns in rodents. BMC Res Notes. 2015; 8: 269.







































































































































