Chronic Parvovirus B19 infection in a patient with Ewing’s sarcoma-a case report
- 1. Haematology Department, Indus Hospital and Health Network, Korangi campus, Karachi, Pakistan
- 2. Paediatric Oncology Department, Indus Hospital and Health Network, Korangi campus, Karachi, Pakistan
Abstract
Human parvo virus B19 (B19V) is a small single stranded DNA virus with known tropism and cytotoxicity for erythroid progenitors. Its infection is associated with various hematological disorders like aplastic crisis, erythroid hypoplasia, idiopathic thrombocytopenic purpura etc. Here we are presenting a case of Parvo virus B19 infection in a patient treated for Ewing’s sarcoma. A 13 year old boy diagnosed as Ewing’s sarcoma on gluteal mass biopsy, received 14 cycles of chemotherapy with few intermittent cycles of radiotherapy, presented with persistent anemia post treatment. Bone marrow biopsies before initiation and during the course of treatment showed no evidence of metastasis. Patient developed anemia during therapy which persisted even after completing the course of treatment. So again bone marrow examination was performed 4 months after the last cycle of chemotherapy to investigate anemia. It showed paucity of erythroid precursors and gaint basophilic pronormoblasts with intra-nuclear inclusions pathognomonic for Parvo virus B19. Serology for Parvo virus B19 antibodies was performed which reported positivity. This case highlights the morphology of human parvovirus B19 inclusions in a post chemotherapy immunocompromised child.
Keywords
Parvo virus B19; pure red cell aplasia; intranuclear inclusions
Citation
Meraj F, Javed O, Khan H, Devi P, Zia N (2022) Chronic Parvovirus B19 infection in a patient with Ewing’s sarcoma-a case report. J Hematol Transfus 9(1): 1103.
INTRODUCTION
Human Parvovirus B19 is a single stranded DNA (ssDNA) virus. Its infection leads to different clinical manifestations including transient aplastic anemia, pure red cell aplasia, erythema infectiosum (fifth disease), acute hepatitis, non-immune hydrops fetalis etc (1). It is found worldwide without ethnic or geographic background. It targets the erythroid precursors in the bone marrow by binding to P blood group antigen receptor, also called glycosphingolipid globoside (Gb4), triggering either cell lysis or apoptosis. Rare individuals who lack the P antigen are immune to parvovirus B19 infection (2-3). Transmission is usually through aerosol droplets inhalation containing the virus, but can also occur vertically from mother to fetus, through transfusion of blood products and organ-transplantation (4-5,9). Persistent infection tends to occur in immunocompromised patients which manifest as chronic anemia. Pediatric population have particularly a higher risk of getting the infection, since they may not yet have encountered the infection and thus lack immunity.5 This becomes significant in a patient receiving chemotherapy due to some malignancy as its infection and subsequent clinical presentation could mimic disease relapse or therapy-induced cytopenia, leading to unnecessary hospital admissions, blood transfusions and cessation of therapy.8 We describe a case of Parvovirus B19 infection in a child who was treated for Ewing’s sarcoma and developed persistent anemia during the course of treatment.
CASE PRESENTATION
A 13 years old male patient, diagnosed as Ewing’s Sarcoma on a gluteal mass biopsy. Initial bone marrow examination done for staging of disease was negative for metastasis. Imaging workup was unremarkable. Blood parameters were; hemoglobin 13.5 g/dl, white blood cell (WBC) count 12.97x10E9/L and Platelets 345 x10E9/L. The bone marrow aspirate was cellular and exhibited trilineage hematopoiesis with normal myeloid to erythroid ratio (M:E). The patient was started on AEWS1031 chemotherapy protocol. Reassessment bone marrow biopsy was done which was again negative for metastasis with normal myeloid to erythroid ratio. Blood counts done at follow ups remained stable till 2 months after initiation of chemotherapy and patient developed isolated anemia thereafter for which he received monthly transfusions. There was no history of any blood transfusion prior to the onset of disease. Reticulocyte count done on multiple occasions was persistently low. There was no history of rash or fever prior to the development of anemia. The patient received a total of 14 cycles of chemotherapy with intermittent radiotherapy sessions. However because of persistent anemia he remained on blood transfusions even after completion of therapy. A third bone marrow biopsy was performed 4 months after the completion of treatment, to investigate his anemia.
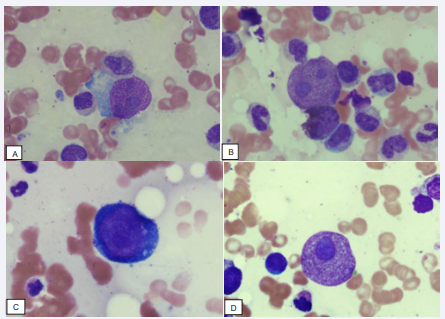
Bone marrow aspirate was spicular and cellular. It exhibited significant erythroid hypoplasia with absence of late erythroid precursors suggestive of maturation arrest. Myeloid to erythroid ratio was 12:1. Scattered giant pronormoblasts were seen having multiple intranuclear eosinophilic viral inclusions and varying degrees of nuclear membrane dissolution. Few also showed cytoplasmic vacuolations. Myelopoiesis was active and showed complete maturation spectrum. Adequate megakaryocytes were noted [Figure 1,2].
Figure 1 Giant pronomoblasts with intra-nuclear inclusions can be seen. Wright-Geimsa stained bone marrow asoirate slides, under 40x magnification lens.
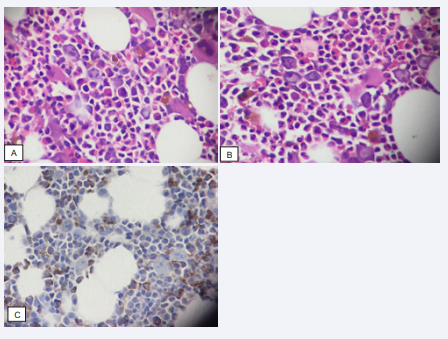
Figure 2 A, B: Giant erythroid precursors with intra-nuclear inclusions can be seen. H/E stained bone marrow trephine sections, under 20x magnification lens.
C: Immunohistochemical marker Glycophorin A on trephine section highlighted the few scattered erythroid precursors.
Bone marrow trephine exhibited overall cellularity of 40-45%. It showed normal distribution and morphology of myeloids and lymphoids with erythroid hypoplasia. Scattered giant erythroblasts were noted having prominent intranuclear inclusions. Glycophorin A immunohistochemistry was performed and was positive in few erythroblasts.
Serological workup for Parvovirus B19 was sent, on morphological suspicion, which came out to be positive (for both IgG and IgM). Thus the case was finalized as acquired pure red cell aplasia secondary to Parvovirus B19 infection.
DISCUSSION
Studies have shown that immunocompromised patients, particularly HIV positive cases, chemotherapy recipients and post-organ transplant patients, presenting with anemia have a high frequency of Parvovirus B19 infection (5-6,9). Hadir Ahmed El-Mahallawy et al. studied acute lymphoblastic leukemia patients during maintenance cycle of chemotherapy, presenting with anemia. 64% of the patients tested positive for serology (IgG or IgM parvovirusb19 antibodies), while 22% of those were also positive for virus’ DNA. In these cases hemoglobin ranged from 2.9 to 10 g/dl and bone marrow examination revealed giant normoblasts with absence of late erythroid precursors in most of them (5).
Natalia Calderia Loss Vincens and colleagues studied a renal transplant patient having complain of persistent unexplained anemia after transplantation (9). Hemoglobin in this case dropped from 11.8 g/dl at the time of hospital discharge to 8.9 g/dl and further to 6.0 g/dl after two months of transplantation. Reticulocyte count was 0.6%. Bone marrow biopsy was performed after excluding out the common causes of anemia and giving a trial of hematinics. Marrow showed erythroid hypoplasia, giant pronormoblasts and eosinophilic intranuclear inclusions with positive PCR for Parvovirus B19.
What made our case interesting initially is that cytopenia is a common finding in patients receiving chemotherapy. So the anemia was interpreted as drug induced. But persistent isolated anemia after the completion of therapy alerted regarding some underlying pathology. Diagnosis of the infection can be done in various ways which include immunostain against the virus’ capsid protein applied on trephine biopsy section, serological testing through ELISA for antiparvovirus B19 IgM and IgG antibodies and PCR for DNA detection (3). Although it has a characteristic morphology, it can be missed upon examination leading to diagnostic delay. Individuals with hematologic or solid-organ malignancies, transplant recipients and HIV infected hosts are particularly at risk of chronic bone marrow infection with Parvovirus B19. This results in persistent anemia requiring regular blood transfusions (7), which was exactly the scenario in our case. Some studies have shown beneficial effects of IVIGs in such condition (8).
This case highlights the need to investigate persistent anemia in immunocompromised individuals or those receiving chemotherapy. It is also important to emphasize the need for adequate training of hematopathologists in the recognition of characteristic morphological lesions.
REFERENCES
- Furukawa M, Kaji K, Masuda H, Ozaki K, Asada S, et al. “Severe Aplastic Anemia following Parvovirus B19-Associated Acute Hepatitis.” Case Reports Hepatol 2017: 1359486.
- Young NS, Brown KE. Parvovirus B19. N Engl J Med. 2004. 5; 350: 586-597
- Burns K, Parish CR. Parvoviridae. Knipe DM, Howley PM, eds. Fields Virology. 5th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2007. 2: 65.
- Rogo LD, Mokhtari-Azad T, Kabir MH, Rezaei F. Human parvovirus B19: a review. Acta Virol. 2014; 58: 199-213.
- El-Mahallawy, Hadir Ahmed MD*; Mansour, Tarek MD†; El-Din, Sahar Ezz BSc*; Hafez, Mohamed MD†; Abd-el-Latif, Soheir MD* Parvovirus B19 Infection as a Cause of Anemia in Pediatric Acute Lymphoblastic Leukemia Patients During Maintenance Chemotherapy, J Pediatr Hemato/Oncol. 2004; 2: 403-406.
- Kurtzman GJ, Ozawa K, Cohen B, Hanson G, Oseas R, Young NS. Chronic bone marrow failure due to persistent B19 parvovirus infection. N Engl J Med. 1987; 317: 287-294.
- Kurtzman G, Frickhofen N, Kimball J, Jenkins DW, Nienhuis AW, Young NS. Pure red cell aplasia of 10 years’ duration due to persistent parvovirus B19 infection and its cure with immunoglobulin therapy. N Engl J Med. 1989; 321: 519-523.
- Heegard ED, Schmiegelow K. Serologic study on parvovirus B19 infection in childhood acute lymphoblastic leukemia during chemotherapy: clinical and hematologic implications. J Pediatr Hematol Oncol. 2002; 24: 368–373.
- Vincens NC, Carminatti M, Franco MF, Hallack Neto AE, Pinheiro HS. Anemia crônica no pós-transplante renal: parvovirose B19 [Posttransplant chronic anemia: parvovirus B19]. J Bras Nefrol. 2012. Sep; 34: 303-308.