In vitro 5?-Reductase Inhibitory Activity of Echinops echinatus: Possible Explanation for its Activity against Benign Prostatic Hyperplasia
- 1. Department of Pharmaceutical Sciences, Doctor Hari Singh Gour Central University, India
- 2. Research and Development Unit, Ying Zhi Agricultural and Industries Sdn Bhd, Malaysia
Abstract
Objective: Echinops echinatus Roxb. (Compositae) (EE) is a pubescent annual herb of 1-3 feet in height distributed throughout India. The aim of present study was to determine the 5α-reductase inhibitory potential of the extracts of EE and elaborate the previously reported studies on this plant regarding its effects on benign prostatic hyperplasia.
Methods: The in vitro studies measured the 5α-reductase inhibitory potential of the petroleum ether and ethanolic extracts, their fractions and finasteride by determining the concentration of testosterone in the reaction mixture using High Performance Liquid Chromatography. The column was eluted isocratically with a mobile phase of methanol: water (80:20) at a flow rate of 1.0 ml/min. The volume of sample injected in the column was 20 µl and the wavelength was 245 nm. The IC50 value i.e., the concentration of test compound required for 50% inhibition of the control conversion of 1 mM Testosterone, was calculated by regression analysis.
Result: Butanolic fraction of ethanolic extract was found to be the best 5α-reductase inhibitor.
Conclusion: On the basis of the present studies, it can be concluded that EE is a potential 5α-reductase inhibitor and its major mechanism of action against testosterone-induced benign prostatic hyperplasia is enzyme inhibition through the suggested mechanism.
Keywords
Antiandrogenic activity; Enzyme inhibition; 5α-reductase; Benign prostate hyperplasia
Citation
Nahata A, Agrawal M, Dixit VK (2017) In vitro 5α-Reductase Inhibitory Activity of Echinops echinatus: Possible Explanation for its Activity against Benign Prostatic Hyperplasia. J Urol Res 4(3): 1091.
ABBREVIATIONS
EE: Echinops echinatus; BPH: Benign Prostatic Hyperplasia; DHT: Dihydrotestosterone; EDTA: Ethylenediamine tetraacetic acid; HPLC: High Performance Liquid Chromatography; BSA: Bovine Serum Albumin
INTRODUCTION
Echinops echinatus is a xerophytic annual herb belonging to the family Asteraceae (Compositae). This herb is globally distributed in the pan tropics throughout India, Pakistan and Afghanistan. Within India, it is found practically throughout, ascending to 1500 meters on the hills. It is common on roadsides, grasslands and open forest areas [1,2]. Traditionally Echinops echinatus is used both internally and externally for the treatment of sexual disorders [3]. An aqueous paste of the root is applied in the lower abdominal region to hasten the process of delivery, also the patients are advised to take the paste internally for quick and safe delivery [4]. In case of patients having poor sexual vitality, aqueous paste of the root bark powder is applied externally on the male genital one hour before intercourse [5,6]. In an indigenous formula the powdered root is made into paste and is applied on the belly of a pregnant woman at the time of child birth, it is considered helpful for easy delivery of the baby [2,7- 9]. The plant is pungent, bitter, stomachic, antipyretic, analgesic; improves the taste, increases the appetite, stimulates the liver, useful in diseases of the brain, diseases of the heart, pains in the joints, inflammations. The root is abortifacient and aphrodisiac [1] and used in the hoarse coughs of children. Infusion is given in seminal debility, impotence, hysteria etc [10].
Nowadays, androgen–mediated diseases such as prostate cancer, hirsutism, acne, androgenetic alopecia and benign prostatic hyperplasia (BPH) have become serious problems [11,12]. Above all, BPH is one of the most common ailment seen in older men and the prevalence of histologically identifiable BPH for 60 year old males is greater than 50% and reaches approximately 90% by the age 85. It is characterized by increase in the size of the gland due to increase in the total number of stromal as well as epithelial cells [13]. In the prostate, dihydrotestosterone (DHT) is produced from testosterone by the enzyme 5α-reductase. DHT is a potent androgen that promotes growth of the prostate. Although the pathogenesis of BPH is not completely defined, but these androgens have been identified as playing an integral part in the disease process. Inhibition of the production or actions of DHT can result in the inhibition of the growth of the prostate gland. There is substantial clinical evidence that androgens and DHT play a key role in the development of BPH [14].
The present study is an elaboration of the studies conducted previously by our group on EE roots for determining their protective effects against BPH and correlating it with the in vitro 5α-reductase inhibitory activity of the test extracts [13].
MATERIALS AND METHODS
Plant material
Roots of EE were procured from local fields of Sagar (Madhya Pradesh, India). Plant was authenticated by the Department of Botany, Dr. Hari Singh Gour Vishwavidyalaya, Sagar (India). A herbarium has been deposited in the Department of Botany and the accession number of the specimen is Bot/Her./1370. Roots were cut in small pieces and dried in the sun to remove the moisture and coarsely powdered in a grinder.
Preparation of extracts
Coarsely powdered roots were fed in Soxhlet apparatus and extracted with petroleum ether (60-80°C) till complete extraction. The solvent from the extract was eliminated under reduced pressure which yielded 5.6 % w/w of petroleum ether extract .Then the defatted marc of the roots was extracted with ethanol (95% v/v) to obtain the ethanolic extract and the yield of ethanolic extract was found to be 6.25% w/w.
Fractionation of extract
Petroleum ether extract was shaken in a separating funnel with acetone to get the acetone soluble and acetone insoluble fractions of Petroleum ether extract. Ethanolic extract was suspended in water to get the water insoluble and water soluble fractions of the ethanolic extract. Water soluble fraction was further extracted with butanol in a separating funnel and aqueous and butanolic fractions were collected separately.
Drugs and chemicals
Finasteride was kindly supplied as a gift sample from Aurochem Laboratories Pvt. Ltd., Mumbai (India). Testosterone was obtained from Sun Pharma Advanced Research Center, Vadodara, Gujarat (India). Ethylenediamine tetraacetic acid (EDTA), sucrose and sodium phosphate were purchased from HiMedia Pvt. Ltd., Mumbai (India). Methanol, ethyl acetate and petroleum ether (60-80 °C) were purchased from Qualigens Fine Chemicals Pvt. Ltd. Mumbai (India). All other chemicals used in the study were of analytical grade.
In vitro studies
The in vitro studies measured the 5α-reductase inhibitory potential of the extracts, its fractions and finasteride by determining the concentration of testosterone in the reaction mixture using HPLC [12-16]. The detailed methodology is described in the following sections:
Preparation of enzyme solution
Human prostate (about 200 mg) supplied from the local hospital of Sagar was minced in small pieces and homogenized with 10 ml of medium A (containing 20 mM sodium phosphate (pH 6.5), 0.32 mM sucrose and 1mM EDTA). The homogenate was centrifuged at 4000 rpm for 15 minutes and the supernatant was used as a source of enzyme .The concentration of enzyme in the supernatant was determined through Bradford method of protein estimation [17].
Protein estimation
Preparation of Bradford’s Reagent: 10 mg of Comassie Brilliant Blue G-250 was dissolved in 5 ml of 95% ethanol. Add 10 mL of concentrated o-Phosphoric acid in it, and final volume was made up to 20 ml with distilled water. This stock solution was mixed with distilled water in a ratio of 1:4, and filtered through Whatman paper No. 1, before use.
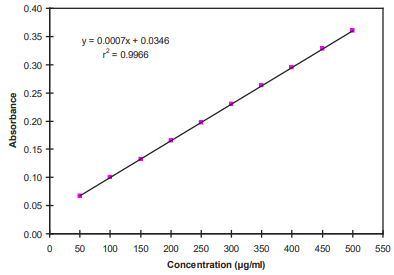
Standard curve of BSA: Standard solution (500 µg/ml) of Bovine serum albumin (BSA) was prepared in phosphate buffer. From this stock solution, aliquots from 50 µg/ml to 500 µg/ml were prepared. 4 ml of Bradford’s reagent was added to each aliquot and final volume was made up to 5 ml by phosphate buffer. This final mixture was incubated for 5 min. Absorbance were taken at 595 nm on UV spectrophotometer (GBC Cintra, Australia). Equation of straight line was found to be y=0.0007x+0.0346 and R2 = 0.9946.
Estimation in enzyme solution: 1 ml of enzyme solution was incubated for 5 min with 4 ml of Bradford reagent .The absorbance was noted as 0.3072 and concentration was found to be 390.28 µg/ml from the standard curve of BSA.
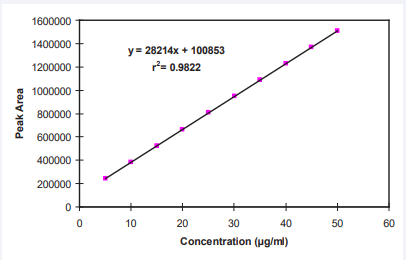
Standard curve of testosterone: A standard solution of Testosterone (500 µg/ml) was prepared in ethanol. Aliquots from 5 µg/ml to 50 µg/ml from this stock solution were taken and standard curve was plotted using HPLC (AT10, Shimadzu Corp. Kyoto, Japan). The peak area was determined at 245 nm by using mobile phase Methanol: Water (80:20) at the flow rate of 1 ml/min [14,16]. Retention time was found to be 6.35 min. Equation of straight line was found to be y=28214x+100853 and R2 = 0.9822. The standard curve is depicted in Figure 2.
Figure 2 Standard Curve for Testosterone.
Preparation of test materials: Solution of Extract (1 mg/ ml) was prepared in ethanol (95%) with gentle heating wherever necessary. The EDTA solution (10 mg/ml) was made in distilled water. Finasteride solution (10 µg/ml) was prepared in ethanol.
Determination of optimum concentration of enzyme: It was determined by keeping the concentration of substrate constant and varying the concentration of enzyme. Testosterone solution (1mM) was prepared in ethanol. The reaction mixture (1.5 ml) was prepared by adding testosterone solution (0.1 ml), and enzyme solution (0.1 to 1.0 ml) to sodium phosphate buffer (20 mM). The final compositions of mixtures were as follows:
Reaction Mixture = Testosterone + Enzyme Solution + Sodium Phosphate (1.5 ml) (0.1 ml) (0.1 to 1.0 ml) (up to 1.5 ml) (Figure 1).
Figure 1 Standard Curve of BSA.
The reaction mixture was incubated at 37°C for 1 hour. The reaction was terminated by the addition of 2 ml of ethyl acetate. The reaction mixture was then shaken vigorously for 1 minute and the ethyl acetate layer was separated. It was evaporated to dryness, and the residue was dissolved in 2 ml of methanol. Testosterone content in methanolic solution was estimated by HPLC (AT10, Shimadzu Corp. Kyoto, Japan). The column was eluted isocratically with a mobile phase of methanol: water (80:20) at a flow rate of 1.0 ml/min [18]. The volume of sample injected in the column was 20 µl and the wavelength was 245 nm. The concentration of enzyme that caused the maximum conversion of testosterone to dihydrotesterone was found to be 0.8 ml (Table 1).
Table 1: Determination of Optimum Concentration of Enzyme in prostate homogenate.
|
Table 1: Determination of Optimum Concentration of Enzyme in prostate homogenate. |
|||
|
S. No. |
Volume of enzyme solution (ml) |
Peak area |
Concentration of testosterone (µg/ml) |
|
1 |
0.1 |
680425 |
23.76 |
|
2 |
0.2 |
634539 |
22.13 |
|
3 |
0.3 |
582616 |
20.29 |
|
4 |
0.4 |
502616 |
17.46 |
|
5 |
0.5 |
426719 |
14.77 |
|
6 |
0.6 |
376530 |
12.99 |
|
7 |
0.7 |
322379 |
11.07 |
|
8 |
0.8 |
249974 |
8.50 |
|
9 |
0.9 |
295721 |
10.12 |
|
10 |
1.0 |
319171 |
10.96 |
This concentration has been referred to as optimum amount of enzyme solution required for the control conversion of testosterone to dihydrotesterone.
Determination of inhibitory concentration of extract: The reaction mixture (1.5 ml) was made by adding 0.1 ml of EDTA solution, 0.1 to 0.4 ml of extract solutions depending upon the group, optimum amount of enzyme solution (0.8 ml) and sodium phosphate (20 mM), to a final volume of 1.5 ml. The final composition of mixture was as follows:
Reaction Mixture (1.5 ml) =Testosterone (0.1 ml) +EDTA solution (0.1 ml) +Extract/fractions/finasteride standard (0.1 ml to 0.4 ml) +optimum amount of enzyme solution (0.8 ml) + Sodium phosphate (upto 1.5 ml).
Reaction mixture was incubated at 37 °C for 60 min and reaction was terminated by addition of 2 ml of ethyl acetate. The mixture was vortexed for 1 min; the ethyl acetate layer was separated and evaporated to dryness. The residue was dissolved in methanol and made up to a volume of 2 ml with methanol. The testosterone content in methanol was determined by HPLC. The inhibitory concentrations for different test extracts were determined by regression analysis and relative testosterone concentrations were calculated in each case which indirectly depicted the ability of the extracts to inhibit the enzyme and hence the conversion of testosterone to dihydrotesterone. The comparison of all the tested extracts and finasteride standard has been shown in detail in table 2.
Table 2: 5α-Reductase Inhibitory Activity of Tested Extracts and Standard Finasteride
|
Table 2: 5α-Reductase Inhibitory Activity of Tested Extracts and Standard Finasteride. |
|||||||||
|
S.Noa. |
Extract solution (ml) |
Relative Testosterone Concentration (µg/ml) * |
|||||||
|
PEE |
WIF |
ASFPEE |
AIFPEE |
EE |
WSF |
BFEE |
FINA |
||
|
1 |
0.1 |
2.762 |
6.328 |
4.486 |
0.3144 |
4.189 |
5.8683 |
0.92 |
6.324 |
|
2 |
0.2 |
3.805 |
8.807 |
4.987 |
0.7018 |
5.206 |
6.3916 |
11.59 |
11.188 |
|
3 |
0.3 |
4.851 |
11.286 |
5.488 |
1.0892 |
6.223 |
6.9149 |
24.1 |
16.052 |
|
4 |
0.4 |
5.895 |
13.765 |
5.989 |
1.4766 |
7.24 |
7.4382 |
36.61 |
20.916 |
|
Abbreviations: *RV: Regressed Values; PEE: Petroleum Ether Extract; WIF: Water Insoluble Fraction; ASFPEE: Acetone Soluble Fraction of Petroleum Ether extract; AIFPEE: Acetone Insoluble Fraction of Petroleum Ether Extract; EE: Ethanolic Extract; WSF: Water Soluble Fraction; BFEE: Butanolic Fraction of Ethanolic Extract; FINA: Finasteride |
|||||||||
RESULTS
According to the requirement of the experiment, the standard curves for BSA was prepared spectrometrically (equation of straight line y=0.0007x+0.0346, R2 = 0.9946) and curve for testosterone was prepared via HPLC (equation of straight line y=v28214x+100853, R2 = 0.9822).
The optimum concentration of enzyme that caused the maximum conversion of testosterone to dihydrotesterone was found to be 0.8 ml. This amount referred to the volume of enzyme solution which is the most effective in converting testosterone to dihydrotesterone in the reaction mixture, hence depicting maximum conversion. As shown in table 1, enzyme solution (0.8 ml) has the ability to convert most of the testosterone to dihydrotesterone as compared to other volumes taken together from 0.1 to 1 ml. Hence it was selected for the experiment to achieve maximum conversion and to get the best results in terms of enzyme inhibitory activity by the test extracts/fractions.
able 2 shows the inhibitory activity of tested extracts, fractions and standard finasteride against 5α-reductase and clearly emphasize the mechanism of action by showing the relative testosterone concentration left in the reaction mixture after the completion of reaction process. Table 2 clearly states that the more the relative testosterone concentration in the reaction mixture, the more is the enzyme inhibition by the tested extract and hence the better inhibitory activity. Here butanolic fraction of the ethanolic extract (BFEE) has shown considerable 5α-reductase inhibition activity as compared to other tested extracts/fractions and the standard finasteride.
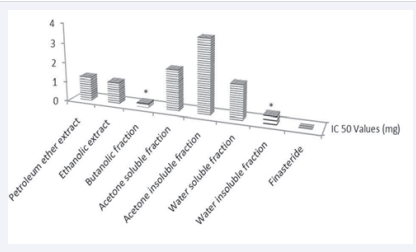
The IC50 value i.e., the concentration of test compound required for 50% inhibition of the control conversion of 1mM Testosterone, was calculated by regression analysis. Finasteride showed an IC50 vale of 0.267 ± 0.67 µg. Extracts exhibited IC50 values ranging from 0.223 ± 0.76 mg for butanolic fraction to 3.741 ± 0.79 mg for acetone insoluble fraction. Hence butanolic fraction showed a significant activity (P < 0.05) as compared to standard finasteride. Results obtained are shown in Figure 3 and suggest that butanolic fraction of ethanolic extract shows greater potential as compared to the pure ethanolic extract and other fractions as a 5α-reductase inhibitor.
Figure 3: 5α-Reductase inhibitory concentrations (IC50) of treated groups
*P < 0.05 as compared to finasteride treated group. One way ANOVA followed by Dunnett’s test.
Therefore EE can be considered a suitable candidate for 5α-reductase inhibitory activity against BPH.
DISCUSSION
The present study is an elaboration of our previous studies on Echinops echinatus (EE) and describes the methodology involved in the action of the drug and its extract towards BPH [13]. As discussed in our previous findings, EE has been established as a drug that can be used for the treatment and management of BPH, it was necessary to find the inhibitory capacity of individual extracts against 5α-reductase for inhibition of conversion of less harmful testosterone to more harmful dihydrotestorone. Testosterone is converted to more potent dihydrotestosterone by the enzyme 5α-reductase type 2 present in prostate homogenates [19]. Dihydrotestosterone is more harmful than testosterone and can cause double the damage caused by testosterone to the prostate. As this study was an elaboration of our previous studies on Echinops echinatus, it was felt necessary to know the effect of various extracts and fractions on 5α-reductase mediated conversion of testosterone to DHT. Hence in vitro analysis utilizing HPLC, as discussed in previous sections, was utilized and various samples were tested. Addition of petroleum ether extract, ethanolic extract and its fractions in reaction mixture found increased level of unchanged testosterone in the reaction mixture, suggesting inhibition of enzyme action by these test materials. Furthermore, the inhibition of conversion by these materials clearly reflects that the activity of 5α-reductase was blocked and therefore, more testosterone remains unchanged in the reaction mixture. This finding was confirmed by the IC50 values. Butanolic fraction and water insoluble fraction showed significant activity as compared to standard finasteride indicating their superiority over other tested extracts and hence better inhibitory activity against 5α-reductase. Butanolic fraction of EE has shown best activity. Finasteride which is a marketed 5α-reductase inhibitor was used a standard to compare the activities of the extracts and this proved to be very successful in finding the best extract out of the tested samples. Our previous claims about EE being useful in the treatment and management of BPH in experimental animals are further strengthened by the results of the present study which described the underlying mechanism of action. So the possible clinical implications of EE point towards androgenic disorders like BPH and prostate cancer.
It can be said that BPH should not be considered as a fatal disorder and sufficient herbal drugs are available in the market which can substantially reduce the progression of this disease without any side effects on the human body. Further it can be stated that a mixture of drugs with proven effects against BPH can be taken together for a better effect on the progression and treatment of this disease. One of such studies involving eleven different drugs with proven antiandrogenic activity including Echinops echinatus has been conducted previously and it was found that Ganoderma lucidum, Sphaeranthus indicus, Urtica dioica are some of the drugs which possessed significant inhibitory activity against 5α-reductase along with Echinops echinatus [20]. Hence their combinations can also be pretty useful in preparing polyherbal formulations for BPH.
CONCLUSION
On the basis of the present studies, it can be concluded that EE is a potential 5α-reductase inhibitor and its major mechanism of action against testosterone-induced benign prostatic hyperplasia is enzyme inhibition through the suggested mechanism. It can be considered as a drug useful for the treatment and management of BPH and other prostatic disorders.
REFERENCES
Kirtikar KR, Basu BD. Indian Medicinal Plants, vol. 2. Delhi: Periodical experts. 1975; 1415-1426.
10. Nadkarni AK. Indian Materia Medica, vol.1. Bombay: Popular Prakashan Pvt. Ltd.; 1976; 468.
18. Purdon MP, Lehman-McKeeman LD. Improved high-performance liquid chromatographic procedure for the separation and quantification of hydroxyl-testosterone metabolites. J Pharmacol Toxicol Methods. 1997; 37: 67-73.