Inflammatory Mediators of Hypertrophic Adenoids in Children with Otitis Media with Effusion
- 1. Department of Paediatric Laboratory Diagnostics, Medical University of Bialystok, Poland
- 2. Department of Paediatric Oncology and Hematology, Medical University of Bialystok, Poland
ABSTRACT
Adenoid is lymphoid tissues in the pharynx that play an important role in host defense against invading antigens. The structures of adenoid consist of well-defined microcompartments which all participate in the immune response: the follicular germinal center, the mantle zone, the interfollicular area and the crypt epithelium. Otitis media with effusion (OME) is a common middle ear disease in children, but etiology of this disease is not clearly understood. Immunologic reaction within adenoid, pro-inflammatory cytokines (Th1 and Th2), decrease of lymphocytes T and B, metaloproteinase may lead to chronic infection.
KEYWORDS
• Lymphocytes T and B
• Adenoid
• Cytokine
• VEGF
• MMP
• Apoptosis
CITATION
?elazowska-Rutkowska B, Latoch E (2022) Inflammatory Mediators of Hypertrophic Adenoids in Children with Otitis Media with Effusion. Ann Otolaryngol Rhinol 9(2): 1284.
INTRODUCTION
Adenoid
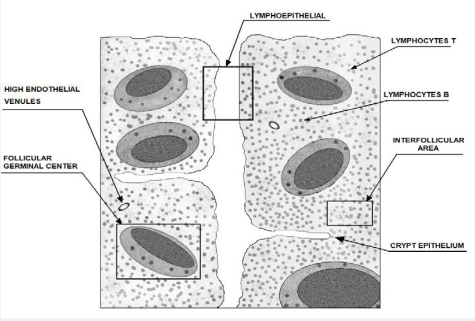
Adenoid play an important role in host defenses of the upper respiratory tract against invading antigens. Lymphoid tissue develops after birth. It reaches maximum size during early childhood and starts to regress at the age of 8-10 years [1]. These tissues have four histological compartments which are very important in the immune response: the follicular germinal center, the mantle zone, the interfollicular area and the crypt epithelium [2] (Figure 1).
Figure 1: The schema construction of adenoid with lymphoid interval.
Germinal center reaction is characterized by large amounts of rapid clonal expansion and is generally polarized into two distinct zones. The dark zone is proximal lymphocytes T and contains proliferating B cells, called centroblasts, while the light zone contains noncykling B cells called centrocytes. Lymphocytes T provide help to B cells via CD40-CD40L interaction and cytokine production. Lymphocytes B provide –MHC class II complexes in the context to stimulatory production T cells and cytokine. Dendritic cells (DC) are found in the region between T and B cell zone [3]. The role of dendritic cells is to participate in polarization of immunological response to Th1 or Th2 which is necessay of rolling inflammatory process in middle ear [4]. Analysis of plasmoid PDCs (BDCA-2+ , CD 123+ , CD 11c) and myeloid MDCs (BDCA-1, CD1c+ , CD 11c++) DC in adenoid and blood in patients with adenoid hypertrophy and otitis media with effusion prove the higher levels of PDCs in adenoid than in the peripheral blood [5].
The etiology of hypertophic adenoid in children with or without otitis media with effusion is not exactly known. The literature shows that there is an increased expression of several inflammatory response mediators of adenoid which may lead to hypertrophy and chronic infections and eustachian tube dysfunction [2].
The role of Th1 and Th2 cytokines in hypertrophic adenoid
Effusion fluid in the middle ear contained acid phosphatase, mucin, alkaline phosphatase, cytokines, creatine phosphokinase and immunoglobulin producing cells [6]. Bacteria can trigger cytokine secretion of macrophages and monocytes as well as TLR expression. Gram–negative bacteria tend to induce expression of IL-6, IL-10, IL-8 but gram-positive bacteria tend to induce expresion of TNF-α, IFN-γ and IL-12 [7]. Subgroup T helper cellsTh17 producting proinflammatory IL-17 and developmentally, different from Th1 and Th2 cells, which play an import ant role in antimicrobiol immunity [8].
In respond to infection adenoid starts production in crypt epithelium proinflammatory Th1 cytokines, including TNF-α, IFN-γ, IL-2 and later secrete Th2 type cytokine ( IL-4, IL-5, IL-6, IL10, IL-13) [8]. Todorovic et al. demonstrated higher production of Th-1 cytokines (TNF-α, IFN-γ) in recurrent tonsillitis compared in tonsillar hypertrophy. In both forms of tonsilitis the production of Th-1 type cytokine was higher compoared to the production of Th-2- type cytokines. Bernstein found a decrease in the synthesis of Th1 cytokines IFN-γ and IL-2 in lymphoid tissue of patients with hypertrophic adenoids and recurrent otitis media. This author suggested that persistence of Th1 cytokines over Th2 could contribute to chronic inflammatory of hypertophic adenoid [9]. High concentrations of TNF-α, IL-8, IL-1β and IL-6 were found in children with chronic otitis media with effusion but in the non-atopic group. In the non-atopic group with bacterial isolate, TNF-α and IL-1β levels positively correlated with the number of past acute otitis media. High concentrations of the proinflammatory cytokine are involved locally in the zone of inflammation [10]. Lymphocytes T CD4+ and T follicular helper (Tfh) are responsible for assisting B lymphocytes to generate antibodies. These cells interact with ligands on the surface of lymphocytes B and start secretion of IL-21 and IL-4, IL-6, IL-8 and IgE which may be involved in the onset of adenoidal hypertrophy combined with secretory otitis media in children [11].
In our study we demonstrated higher IL-5 and TNF-α production in cell cultures stimulated with PHA in hypertophic adenoid in children with media with effusion than in hypertophic adenoid. There were no differences in the levels of cytokines 6, 8 and 10 in supernatants of cells isolated from two groups. Increased IL-5 production in middle ear effusion may be beneficial for the host, because this cytokine is important in the stimulation of the production of antibodies that would be responsible for a humoral immune response directed against potential pathogens [12].
The role of VEGF in hypertrophic adenoid
Pro-inflammatory cytokines such as TNF-α, transforming growth factor-beta (TGF-β), IL-1 can induce production of VEGF (vascular endothelial growth factor) from epithelial cells, vascular endothelial cells or monocytes. Berse et al. suggested that the stimulated and pathway for induction of VEGF may differ in different cell, like the nuclear factor kappa B (NF-κB) [13]. VEGF can also impact endothelial cell expression of immunologically important molecules, decreasing expression of VCAM-1 (vascular cell adhesion molecule-1) increasing expression of FasL, leading to apoptosis [14]. VEGF is produced by the middle ear mucosa and its concentration increases in the exudative and middle ear mucosa in patients with otitis media. Sekiyama et al. showed the higher concentration of endotoxin and IL-8 in most middle ear effusion samples. In their study the concentration of endotoxin were significantly correlated with VEGF levels. In contrast, here was no correlation between IL-8 and VEGF levels or between endotoxin and IL-8. Their results suggest that VEGF is produced in the middle ear in response to stimulation with endotoxin and hypoxic conditions [15]. Bacterial infections increase the levels of both VEGF and its receptors. Elevated cytokine concentrations lead to increased vascular permeability and elevated secretion of exudative fluid. Moreover, they affect the infection of inflammatory cells of the mucous membrane, thus leading to neovascularization within the middle ear [16].
In our study, the mean concentration levels of VEGF-A and TGF-β in hypertrophic adenoid in children with adenoid hypertrophy and otitis media with effusion were significantly higher compared to patients with adenoid hypertrophy. High levels of this cytokine may indicate a bacterial pathogen as one of the causes of exudative otitis media in children [17].
Apoptosis in hypertrophic adenoid
Germinal center of adenoid plays an important role in cell death and cell proliferation [18]. The increase in size and more number of germinal centers was found in patients with hypertrophy adenoid compared to those without hypertrophy. Onal et al. determined that the increased adenoid apoptosis was associated with age. The significantly increased of apoptosis in hypertrophy adenoid compared to chronic adenoid. The authors felt that increased apoptosis with increasing age might account for the noted involution of lymphoid tissue as patients get older [19]. The number of lymphocytes T and B in adenoid and their cooperation in the immunologic and morphological involution depends on their proliferation and migration status. Apoptosis as the process that regulates lymphocyte homeostasis is to a large extent responsible for the immune function of the hypertrophic adenoid [20]. Tumor necrosis factor receptor superfamily member (Fas receptor, CD95) is a death receptor localized on the surface of cells, which triggers a signal transduction pathway leading to apoptosis. Fas- mediated apoptosis is depending on cytokine-2 (IL-2).The interaction between Fas and ligand FasL (FasL/ CD95L) regulates pathological and physiological processes. In the Fas-mediated apoptotic pathway, binding of FasL drives Fas clustering and binding of Fas to FADD. FADD recruits caspase-8 and caspase-10 form the death- inducing signaling complex (DISC) [21]. The antiapoptotic molecule Bcl-2 and its homolog Bcl-xL in lymphocytes accumulate greatly increased number of T and B cells [22]. These molecules control transport through the pores on the external mitochondrial membrane and prevent the exit of cytochrome c from mitochondria and apoptosis initiation. Germinal center cells do not display the expression of Bcl-2, but in the mantel zone cells express a high level of Bcl-2 [23].
Higher percentages of lymphocytes T (CD4+ and CD8+ ) and lymphocytes B (CD19+ ) showing the expression of death receptor Fas in hypertrophic adenoid in children with exudative otitis media compared to hypertrophy adenoid. Statistically significantly lower percentages of lymphocytes CD4+ Bcl-2+ , CD8+ Bcl-2+ , CD19+ Bcl-2+ was found in the group of children with exudative otitis media. The tendency of reduced percentages of lympocytes T and B lymphocytes with Bcl-2+ expression and elevated percentages of these cells with CD95+ expression may reflect local immunity disorders [24].
The role of metalloproteinase in hypertrophic adenoid
Matrix metalloproteinases (MMP) are zinc-dependent endopeptidases which have a key role in the degradation of extracellular matrix. (MMP) and their inhibitors (TIMP) are produced during the inflammatory process by lymphocytes, neutrophils, macrophages, mast cells and eosinophils. The coordinate expressions of MMP and TIMP are involved, tissue remodeling cell transmigration during inflammatory disease, proliferation and apoptosis [25]. The literature shows that level of MMPs and TIMPs in relation to inflammatory and hypertrophic process in adenoids and the resulting otitis media with effusion. Guedez et al. demonstrated that TIMP-1 expressed by isolated germinal center B cells, but antiapoptotic mechanism is also not mediated by Bcl-2 nut likely inducing Bcl-xL [26]. Another author showed diffuse dissemination of MMPs activity in adenoid tissue. The level of MMP-7 and MMP-9 was higher of hypertrophied tissue, but no differences were observed in the level of TIMP-1 and MMP-2. The concentrations of MMP-8 and MMP-9 and TIMP1 in children with adenoid hypertrophy and secretory otitis media reached statistically significantly higher concentrations than those in children belonging to the reference group [27]. This result suggest that MMPs promotes of the extracellular matrix responded to inflammatory changes in adenoid tissue.
CONCLUSION
The different mediators of inflammation like cytokines, VEGF-A and TGF-β involved in the pathogenesis of otitis media with effusion in children. Incresed levels of pro-inflammatory cytokines (Th-1) suggest that abnormalities in the immune system triggering chronic, uncontrolled, inflammation may be a driver of disease. Elevated MMPs and TIMPs levels may induce an increased remodeling of the extracellular matrix. Indicate enhanced apoptosis of activated lymphocytes T and B, can lead to disturbances in local immune response involving the middle ear. Accurate recognition of inflammatory markers activity may become, a viable preventative or treatment in children with otitis media with effusion.
REFERENCES
- Silvia Regina Amorim Pereira, Silvia Fuerte Bakor, Luc Louis Maurice Weckx. Adenotonsillectomy in facial growing patients: spontaneous dental effects. Braz J Otorhinolaryngol. 2011; 77: 600-604.
- S Murat, I Akif, I Abdulkadir, A Hale, A Ibrahim, P Ercan, O Semih. Risk factors for otitis media with effusion in children with adenoid hypertrophy. Acta Otorhinolaryngol Ital. 2020; 40: 133-137.
- CC Goodnow, CG Vinuesa, KL Randall, F Mackay, R Brink. Control systems and decision making for antibody production. Nat Immunol. 2010; 11: 681-688.
- B León, FE Lund. Compartmentalization of dendritic cell and T-cell interactions in the lymph node: anatomy of T-cell fate decisions. Immunol Rev. 2019; 289: 84-100.
- Michal Kotowski, Artur Niedzielski, Grazyna Niedzielska, Patrycja Lachowska-Kotowska. Dendritic cells and lymphocyte subpopulations of the adenoid in the pathogenesis of otitis media with effusion. Int J Pediatr Otorhinolaryngol. 2011; 75: 265-269.
- K Pazdro-Zastawny, T Zato?ski. The effect of middle ear effusion on the inner ear condition in children. Adv Clin Exp Med. 2020; 29: 325- 330.
- Romain Kania, Pierre Vironneau, Huong Dang, Béatrice Bercot, Emmanuelle Cambau, Benjamin Verillaud, et al. Bacterial biofilm in adenoids of children with chronic otitis media. Part I: a case control study of prevalence of biofilms in adenoids, risk factors and middle ear biofilms. Acta Otolaryngol. 2019; 139: 345-350.
- MM Todorovi?, E Zvrko. Immunoregulatory cytokines and chronic tonsillitis. Bosn J Basic Med Sci. 2013; 13: 230-236.
- JM Bernstein, M Ballow, S Xiang, KO’Neil. Th1/Th2 cytokine profiles in the nasopharyngeal lymphoid tissues of children with recurrent otitis media. Ann Otol Rhinol Laryngol. 1998; 107: 22-27.
- Beata Zielnik-Jurkiewicz, Wanda Stankiewicz-Szymczak. Pro- inflammatory interleukins in middle ear effusions from atopic and non-atopic children with chronic otitis media with effusion. Eur Arch Otorhinolaryngol. 2016; 273: 1369-1378.
- Chunyan Feng, Qicheng Zhang, Guangquan Zhou, Jing Zhang, Yanshu Zhang. Roles of T follicular helper cells in the pathogenesis of adenoidal hypertrophy combined with secretory otitis media. Medicine. 2018; 97: e0211.
- Beata Zelazowska-Rutkowska, Elzbieta Ilendo, Bozena Skotnicka, Jolanta Wysocka, Edwina Kasprzycka. Production of cytokines by mononuclear cells of hypertrophic adenoids in children with otitis media with effusion. Folia Histochem Cytobiol. 2012; 50: 586-589.
- B Berse, JA Hunt, RJ Diegel, P Morganelli, K Yeo, F Brown, RA Fava. Hypoxia augments cytokine (transforming growth factor-beta (TGF- beta) and IL-1)-induced vascular endothelial growth factor secretion by human synovial fibroblasts. Clin Exp Immunol. 1999; 115: 176-182.
- Rajendra S Apte, Daniel S Chen, Napoleone Ferrara. VEGF in signaling and disease: beyond discovery and development. Cell. 2019; 176: 1248-1264.
- Kohsuke Sekiyama, Jun-ichiro Ohori, Shoji Matsune, Yuichi Kurono. The role of vascular endothelial growth factor in pediatric otitis media with effusion. Auris Nasus Larynx. 2011; 38: 319-324.
- NR London, RK Gurgel. The role of vascular endothelial growth factor
and vascular stability in diseases of the ear. Laryngoscope. 2014; 124: 340-346.
- Beata Zelazowska-Rutkowska, Bozena Skotnicka, Bogdan Cylwik. Vascular endothelial growth factor and transforming growth factor β in hypertrophic adenoids in children suffering from otitis media with effusion. Cytokine. 2020; 133: 155125.
- Yang Zhang, Laura Garcia-Ibanez, Kai-Michael Toellner. Regulation ofgerminal center B-cell differentiation. Immunol Rev. 2016; 270: 8-19.
- Merih Önal, Taner Y?lmaz, Elif Bilgiç, Sevda Fatma Müftüo?lu, O?uz Ku?çu, R?za Önder Günayd?n. Apoptosis in chronic tonsillitis and tonsillar hypertrophy. Int J Pediatr Otorhinolaryngol. 2015; 79: 191-195.
- Jeffrey C Rathmell, Craig B Thompson. Pathways of apoptosis in lymphocyte development, homeostasis, and disease. Cell. 2002; 109: S97-107.
- A Yamada, R Arakaki, M Saito, Y Kudo, N Ishimaru. Dual role of Fas/FasL-mediated signal in peripheral immune Tolerance. Front Immunol. 2017; 5: 403-408.
- ML Hartman, M Czyz. BCL-w: apoptotic and non-apoptotic role inhealth and disease. Cell Death Dis. 2020; 11: 260-263.
- Tomás Kucera, Hana Pácová, David Veselý, Jaromír Astl, Jindrich Martínek. Apoptosis and cell proliferation in chronic tonsillitis and oropharyngeal carcinoma: role of nitric oxide and cytokines. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2004; 148: 225-227.
- B ?elazowska-Rutkowska, J Wysocka, B Skotnicka. Chosen factors of T and B cell apoptosis in hypertrophic adenoid In children with otitis media with effusion. Int J Pediatr Otorhinolaryngol. 74 2010; 74: 698-700.
- Magnus S Ågren, Ulrich Auf dem Keller. Matrix metalloproteinases: How much can they Do? Int J Mol Sci. 2020; 21: 2678.
- L Guedez, L Courtemanch, M Stetler-Stevenson. Tissue inhibitor of metalloproteinase (TIMP)-1 induces differentiation and an antiapoptotic phenotype in germinal center B cells. Blood. 1998; 92: 1342-1349.
- Beata Zelazowska-Rutkowska, Bozena Skotnicka, Elzbieta Hassmann- Poznanska, Bogdan Cylwik. Selected cytokines in hypertrophic adenoids in children suffering from otitis media with effusion. Int J Pediatr Otorhinolaryngol. 2020; 128: 109700.