Solitary Fibrous Tumor of the Neck: A Case Report
- 1. Department of Otorhinolaryngology-Head and Neck Surgery, Military Hospital Meknès, Morocco
ABSTRACT
Solitary fibrous tumors (SFTs) are rare spindle cell proliferations of mesenchymal origin whose etiology is still unknown. They are considered benign or low-grade malignant. In 20-30% of cases the behaviour of SFT is that of a locally aggressive and recurrent malignant tumor, with rare and late metastases.
Surgical removal allows pathological examination with immunohistochemical confirmation of the diagnosis. The similarity with various soft tissue spindle cell neoplasms makes the diagnosis of these mesenchymal tumors difficult.
Complete excision surgery appears to be the main prognostic factor. Complementary treatments have not proven to be effective. It is important to be aware of these tumors whose
evolution can be negative. Long-term follow-up should be recommended.
In this article, we report a case of SFT evolving in the nuchal region in a 40-year-old patient.
KEYWORDS
• Solitary fibrous tumor
• Neck
• Immunohistochemistry
• Myxoid
CITATION
Ouahidi Y, Belatik H, Hammoud ZC, Hmidi M, Touiheme N, et al. (2022) Solitary Fibrous Tumor of the Neck: A Case Report. Ann Otolaryngol Rhinol 9(1): 1281.
ABBREVIATIONS
STF: Solitary Fibrous Tumors
INTRODUCTION
SFTs are a rare entity of fibroblastic origin, mostly localized and benign. It was first described in 1931 by Klemperer and Rabin in the pleura. The majority of these tumors are benign in histology, but malignant forms do exist and can be lifethreatening because of their frequent recurrence and metastases. Progress in anatomopathological analysis and the appearance of immunohistochemical markers have led to the acceptance of the primary mesenchymal origin of these tumors.
Various authors have tried to define radiological characteristics for diagnostic purposes, or histological characteristics for prognostic purposes. However, the evolution of these tumors remains unpredictable, both locally and at a distance.
Numerous localizations have been reported. Cervical localization remains exceptional, characterized by its slow, often asymptomatic evolution.
We report a new observation of a benign soft tissue SFT with an exceptional location at the nuchal level.
CASE PRESENTATION
This is a 40-year-old man, with no notable pathological history, who presented to the ENT consultation for a left nuchal mass that had been evolving for 2 years, without signs of compression or weight loss.
Clinical examination revealed a left nuchal mass, firm, painless and fixed in relation to the deep plane, measuring 2.5 cm in long axis.
Cervical ultrasound shows a large, hypoechoic, discretely heterogeneous formation, deeply embedded in the surrounding muscular structures, without a separation line, vascularized by Doppler.
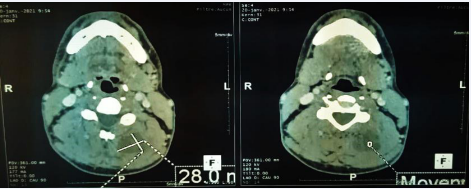
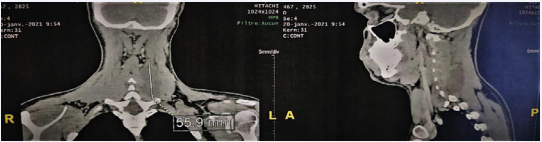
Cervical CT scan shows a low-density, richly vascularized left nuchal process reaching C3 above and C7 below, displacing the surrounding muscles, measuring 55.9 mm, without vertebral lysis or cervical adenopathy (Figure 1).
Figure 1: Axial slice CT scan of a soft tissue mass located in the left posterior cervical region.
(Figure 2)
Figure 2: CT scan in frontal and sagittal reconstruction of a soft tissue mass located in the left posterior cervical region.
The indication for surgery was established, and a monobloc excision by cervicotomy was performed, using a horizontal incision opposite the mass.
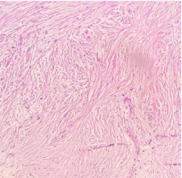
Histological analysis found a benign mesenchymal connective proliferation with partly myxoid spindle cells (Figure 3)
Figure 3: Tumor proliferation made of fusiform cells (HESx200).
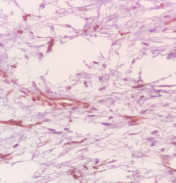
evoking several diagnostic hypotheses, with no histological evidence of malignancy. Several diagnoses can be evoked, mainly spindle cell tumors and pseudotumors including solitary fibrous tumor and hemangiopericytoma which may have the same immunohistochemical profile. Also Nodular fasciitis, which is a fibro-myofibroblastic pseudosarcomatous lesion rich in spindle cells, neurofibroma and spindle cell lipoma. The immunohistochemistry allows deciding. The resection limits were lesional. The immunohistochemical study showed diffuse and intense staining of the tumor cells by anti-CD34 antibody (Figure 4)
Figure 4: Positivity of the anti-CD34 antibodie.
and anti- actin antibody with an absence of staining by anti-desmin and PS100 antibody. It is therefore an SFT (Figure 5).
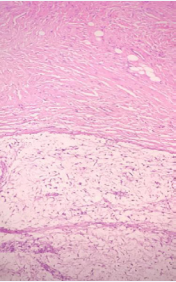
Figure 5: Microscopic examination of the solitary fibrous tumor shows the transition between the myxoid zone at the bottom and the fibrous fusocellular zone at the top.
A revision surgery was performed with extended excision of the margins. The postoperative course was simple. The clinical and radiological check-up at 3 and 6 months was normal, and the patient was followed up regularly without any notion of recurrence after 9 months.
DISCUSSION
SFT is a rare benign mesenchymal tumor most commonly found in the pleura [1]. It is a proliferation of collagen-producing cells. It was first described by Klemperer and Rabin in 1931 as a pleural fusocellular tumor [2]. It can occur at any age during adulthood and shows no predilection for gender [3]. The extrapleural location accounts for about 0.6% of soft tissue tumors [2].
This tumor affects all organs with, for the head and neck region, involvement of the orbit, salivary glands, external acoustic meatus, thyroid, tongue, infra temporal fossa, and sinuses. For this region, SFT is very rare and mostly benign [3].
In this case, the tumor affected the deep soft tissue of the nuchal region [3].
Clinically, the majority of patients are asymptomatic [3- 8]. They usually present as well- circumscribed, partially encapsulated neoplasms. They usually grow slowly but may reach a size greater than 100 mm [3], producing symptoms due to compression or with invasion of adjacent structures.
In the study by Stanisce et al. nearly two thirds of all cases presented with the complaint of a new mass and only 11% of all tumors were painful [4]. Hypoglycemia may be a presenting feature in 10% of cases and is thought to be related to the production of insulin-like growth factor by tumor cells [5].
Ultrasonography shows in most cases a solid echogenic mass with significant vascularity on Doppler [5]. On CT scan, the appearance is that of a well-limited, poly-lobed lesion with intense and homogeneous contrast [1]. However, the contrast may be heterogeneous due to areas of necrosis, myxoid degeneration, or intratumoral hemorrhage.
On MRI, the lesion is isointense to the muscle on T1-weighted sequences with intense gadolinium uptake [1]. The appearance is variable on T2-weighted sequences, depending on the different components of the tumor, in particular the amount of collagen and fibroblasts. Imaging can also be used to assess invasion of surrounding tissues.
Macroscopically, the tumor is often well circumscribed, encapsulated, and variable in size. On section, the tumor is often firm and multinodular. Myxoid, cystic or hemorrhagic changes may be observed [2].
Microscopy shows a light-coloured (pinkish-beige or grey) tumor with a fibrous centre, composed of spindle-shaped cells of heterogeneous arrangement. Mitoses are rare (less than 4 mitoses per 10 fields at high magnification). These tumors alternate between hypocellularity and hypercellularity, with thick bands of hyaline fibrosis. The cells may be round or spindle-shaped [5]. The cytoplasm is sparse and the cytoplasmic boundaries are poorly defined. The vascular network is highly developed and of hemangiopericyte type, made of thin-walled, branched vessels with perivascular hyalinization [2].
The histochemical immunoassay is a valuable aid to positive diagnosis. The majority of SFTs are positive for CD34 and bcl-2. They are usually negative for cytokeratins, smooth muscle actin, desmin, S-100 protein and c-kit [5].
In our patient, the anatomopathological examination concluded to a SFT without signs of malignancy [5].
SFT can sometimes be confused with hemangiopericytoma and only microscopic examination can differentiate them [6]. On the other hand, it should be noted that hemangiopericytoma is a more aggressive lesion [6] whereas SFTs are sometimes more diffuse and hypercellular [6]. Leiomyosarcoma and fibrosarcoma may also be considered in the differential diagnosis, but immunohistochemistry can be used to make a decision by showing a positive reaction to actin and desmin [6].
Stanisce et al. believe that nuclear atypia, more than 4 mitoses/10 HPF, and necrosis are likely to be associated with aggressive clinical behavior, but there is insufficient current evidence to indicate that they are predictive of recurrence by themselves [4]. In cases of uncertain malignant potential, Yokoi et al. suggested that p53 protein expression may be associated with aggressive tumor behavior [4].
Nevertheless, tumors with these histological signs of malignancy often show a benign clinical course and only 50% of these tumors show clinical signs of malignancy such as distant metastasis or local invasion [2].
The mainstay of successful treatment remains surgery with complete resection and clear margins [4]. There is no consensus regarding the anatomopathological margins at the time of surgical resection, however, it should be maximal (R0) [5]. The need for cervical lymph node dissection is not indicated [7].
Some studies have reported favorable results with postoperative chemotherapy and radiotherapy in cases of incomplete resection [7]. An in vitro study, testing the sensitivity of SFT with a mitotic index of 7/50 HPF with chemotherapy. The tumor was sensitive to 5- FU, adramycin, mitomycin-c, and doctaxel, which may open the door for probably the most aggressive, recurrent, unresectable, or incompletely resected SFTs [5].
SFT is generally a good prognosis tumor after surgical resection. The malignancy rate in the literature is 12-35% [2]. Among extra-thoracic SFTs, about 10% appear to be associated with local or distant recurrence. The most important factor in recurrence is infiltration of the surgical resection margins [2].
Although SFTs are generally benign, close clinical and radiographic follow-up is recommended because of their unpredictable metastatic and recurrent behaviors [7]. However, there is no clear consensus to date [5].
CONCLUSION
Solitary fibrous tumors of the neck are rare and often benign tumors with a favorable evolution. However, there are cases of malignant evolution that can be correlated with pejorative histological criteria.
The etiology is still unknown. Diagnosis is based essentially on histology coupled with immunostaining. Treatment is exclusively surgical and must be as carcinological as possible given the recurrent nature and unpredictable fate of this tumor.
The prognosis after excision is good, but the risk of local recurrence or metastasis requires prolonged surveillance. Management must be maximal and multidisciplinary.
ACKNOWLEDGEMENTS
Informed consent was obtained from all participants included in the study.
REFERENCES
- Wakisaka N, Kondo S, Murono S, Minato H, Furukawa M, Yoshizaki T. A solitary fibrous tumor arising in the parapharyngeal space, with MRI and FDG-PET findings. Auris Nasus Larynx. 2009; 36: 367-371.
- Harmouch T, Hamas N, Daoudi A, Amarti A. Une tumeur exceptionnelle du doigt: la tumeur fibreuse solitaire. Pan African Medical Journal. 2011; 10:49
- Zuniga MG, Orr BA, Chang SS, Tufano RP. Suboccipital Malignant Solitary Fibrous Tumor: Report of a Case. Ear Nose Throat J. 2012; 91: E25-E27.
- Stanisce L, Ahmad N, Levin K, Deckard N, Enriquez M, Brody J, et al. Solitary Fibrous Tumors in the Head and Neck: Comprehensive Review and Analysis. Head and Neck Pathol. 2020; 14: 516-524.
- Slimani O, Belghith C, Saoudi S, Tahar M, Ben Temim R, Mathlouthi N, et al. Tumeur fibreuse solitaire parautérine: à propos d’un cas. Pan Afr Med J. 2016; 25: 180.
- Dia Tine S, Tamba B, Diatta M, Gassama Barry BC, Niang P, Kounta A, et al. Tumeur fibreuse solitaire de la joue : à propos d’une observation. Med Buccale Chir Buccale. 2013; 19: 187-190.
- Shi W, Wei Z. Solitary fibrous tumor of the submandibular region. Oncology Letters. 2015; 9: 984-986.
- Goregaonkar R, Viswanathan S, Prathamesh P, Pankaj C, Kane SV. Solitary Fibrous Tumor Presenting as a Parapharyngeal Mass. International Journal of Head and Neck Surgery. 2010; 1: 29-33.