Aquatic Plants: Alternative Medium for Microalgae Growth
- 1. Lucia Helena Sipaúba-Tavares, Alexandra Maria Donadon Lusser Segali, Bruno Scardoelli-Truzzi, Aquaculture Center, Univ. Estadual Paulista-UNESP, Brazil
Abstract
The relevance of macrophyte and commercial fertilizer used as a culture medium for microalgae Ankistrodesmus gracilis and Haematococcus pluvialisis provided. Medium based on commercial fertilizer and macrophyte (E. crassipes) media produced a new medium formulation that proved to be efficient, at least in batch culture mode to enhance high density culture of the microalgae.
Citation
Sipaúba Tavares LH, Lusser Segali AMD, Scardoelli-Truzzi B (2015) Aquatic Plants: Alternative Medium for Microalgae Growth. Ann Aquac Res 2(1): 1009.
Keywords
• Ankistrodesmus gracilis
• Haematococcus pluvialis
• Eichhornia crassipes
• NPK (20-5-20)
• biochemical composition
ABBREVIATIONS
NPK: Inorganic Fertilizer; WC: Commercial Medium; M+NPK: Mixture of Macrophyte (Eichhornia crassipes) Extract and NPK Fertilizer (20-5-20); CHU12: Commercial Medium; TOC: Total Organic Carbon
INTRODUCTION
A culture medium is a tool for the maintenance and development of microalgae. The maximization of production and the minimization of costs are the most important issues to all involved in algae culture which may, be used with alternative medium such as inorganic fertilizer (NPK) and a mixture of macrophyte (Eichhorniacrassipes) extract and NPK fertilizer.
The inorganic fertilizer is simple, and may be used with alternative medium for microalgae, since they are widely available, they dissolve easily, they have a defined composition, high nitrogen and phosphorus rate, and they trigger moderate pH in the medium [1]. In aquaculture, the aquatic floating macrophyte, Eichhornia crassipes, retrieves inorganic nutrients from the water, mainly nitrogen and phosphorus, through roots and stores them in its biomass. Associated with inorganic fertilizers such as NPK, it results in an efficient alternative medium for Chlorophyceae algae [2].
Several algae species of the Chlorophyceae class have been reported to contain levels (25-50% dry weight) of neutral lipid. This fact suggests that this class of microalgae may represent a large pool of organisms from which the initial feed for zooplankton and fish larvae could be obtained [3]. Some of the most widely used in commercial freshwater culture are Haematococcus pluvialis, a unicellular green microalga, widely used as an additional pigment in aquaculture and a supplement item in the food industry [4], and Ankistrodesmus gracilis, which is highly appreciated by water organisms owing to its size, shape, thickness of cell walls and easy prey condition [5].
Current analysis investigates the effect of an alternative medium, NPK (20-5-20) fertilizer and NPK plus macrophyte (M+NPK) under growth rate and physiological parameters in batch culture mode (2-L). It also, verifies the use of the fertilizer (NPK) and the macrophyte (Eichhorniacrassipes) extract is a good tool for A. gracilis and H. pluvialis culture in the laboratory.
MATERIALS AND METHODS
The microalgae Haematococcus pluvialis (CMEA 227 C1) and Ankistrodesmus gracilis (005CH) were used to observe the effects of different media on their growth. Four culture media were used, namely, two commercial media (WC and CHU12) and two alternatives, one with the inorganic fertilizer NPK (20-5-20) [6] and other medium with a mixture of macrophyte (Eichhornia crassipes) extract and the NPK fertilizer (M+NPK). The latter consists of a mixture of macrophyte extract and NPK fertilizer (20-5-20). The mixture contains approximately 5 kg of E. crassipes that were ground and boiled in distilled water for one hour. The hot extract was filtered and autoclaved at 120° C, for 20 minutes. After autoclaving, a sample (70 mL) was collected, cooled and diluted with distilled water to 1.4 L. Additionally, 2.5 ml NPK and 100 ml inoculum of A. gracilis and H. pluvialis microalgae were added to obtain an approximate density of 3 x 105 cells.ml-1 and 0.26 x 105 cells.ml-1, respectively [2]. Vitamin B complex was added to the alternative media at the rate of 0.02 g.L-1 (7 mg - B1 ; 7 mg - B2 ; 5 mg - B6 ; 33 µg - B12) plus vitamin H (0.1 mg) [2,6]. Continuous illumination was applied and the alternatives and commercial media were used to grow the microalgae cultures in a 2-L container submitted to continuous aeration, at 23 ± 2° C. The samples were analyzed weekly (1, 7, 14, 21 and 28-days) during the study period to asses growth rate and other physiological parameters.
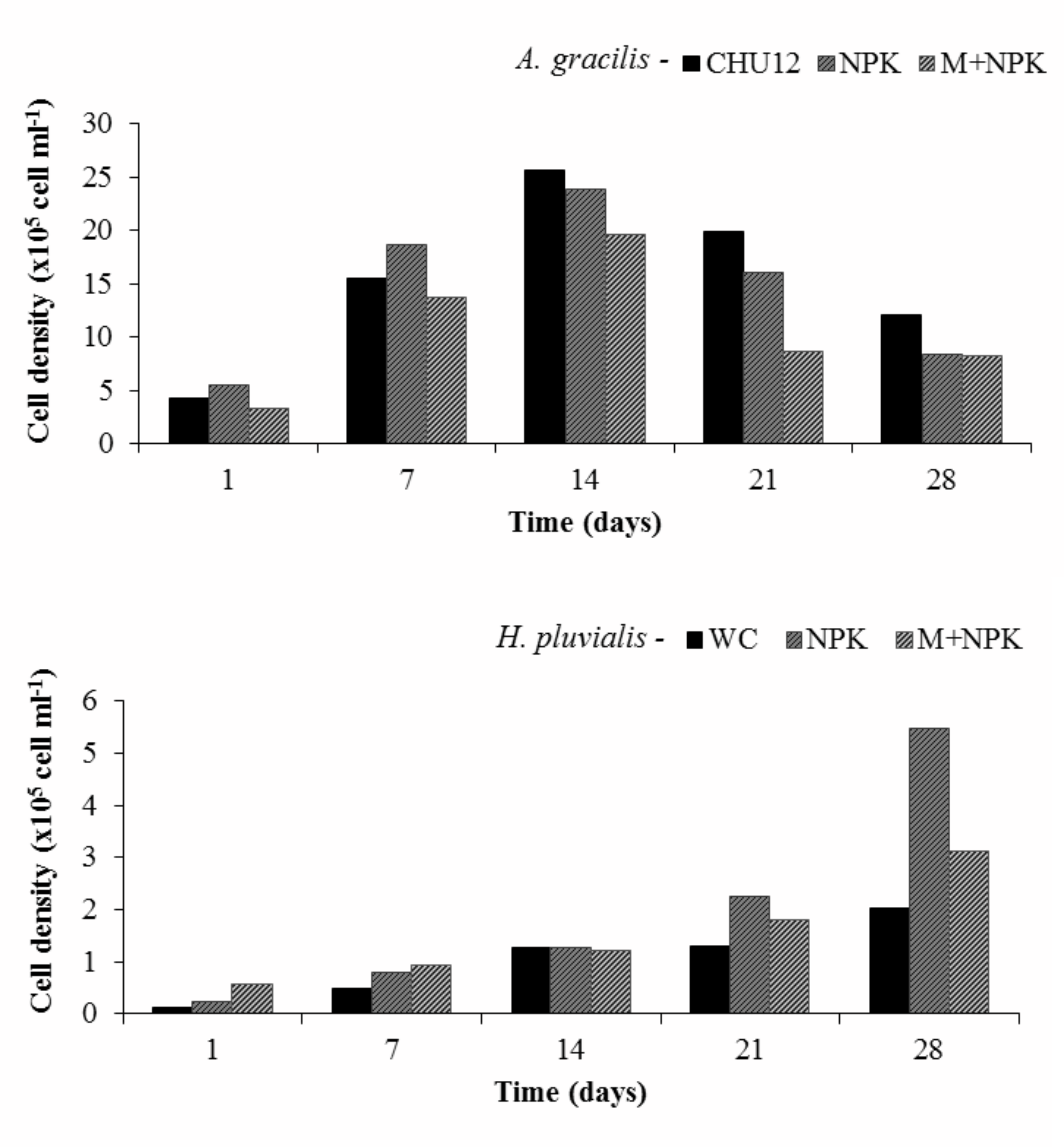
Figure 1 : Growth of Ankistrodesmus gracilis and Haematococcus pluvialis in different culture media during the study period.
Growth rate k (divisions per day) was calculated by the formula: k = (3.322/t2 -t1 x log N2 /N1 ) (t = time; N = number of cells; subscripts denote values at different times) [7]. Dry weight was determined by filtering the microalgae samples, dried at 60° C and weighed till constant weight [8]. The cell size of 50 specimens was determined with microscope Leica DFC 295 by image analysis system Las Core (LAS V3.8), with a 40X micrometric objective. Total organic carbon (TOC) was calculated by C = 0.1204.V1 .051 (C = carbon content in pg.cell-1; V = cell volume) using regression [9]. Chlorophyll-a was extracted with 90% alcohol and quantified at 663 and 750 nm [10]. The biomass was harvested weekly, centrifuged, and lyophilized for the analyses of proteins and lipids [11]. All experiments were carried out in triplicate. One-way ANOVA was applied between the media for simple verification.
RESULTS AND DISCUSSION
The need for nutritional sources which are safer than traditional animal products has renewed a great interest in plants and particularly in microalgae. Even when used in small amounts in livestock and aquaculture feeds, the algae have been credited with improving immune system, lipid metabolism, antiviral and antibacterial activity, improved gut function and stress resistance. Further, microalgae are also a source of protein, amino acids, fatty acids, vitamins, minerals and other biologically active photochemical [12].
Protein rates were high (between 28 and 48 % dry weight) and lipids contents were below 7% dry weight except to alternatives media (NPK and M+NPK) which was high (between 8 and 14 % dry weight) for A. gracilis (p0.05) was observed in the case of H. pluvialis between commercial medium and alternative media for protein and lipids. No difference (p>0.05) for lipids and proteins was observed during the study period except with regard to different culture media (p<0.05). Algal diets, rich in fatty acids, generally survived and grew better n many aquatic organisms [13].
Table 1: Summary of some physiological parameters of Ankistrodesmus gracilis and Haematococcus pluvialis cultured in CHUI2, WC, NPK and M+NPK media in a 2-L volume. The values are the mean of three replications and the variation (±) is the standard deviation.
|
Characteristics |
Ankistrodesmus gracilis
|
ANOVA |
Haematococcus pluvialis
|
ANOVA |
||||
|
CHU12 |
NPK |
M+NPK |
WC |
NPK |
M+NPK |
|||
|
Mean cell density (105.ml-1) |
16.51±6 |
16.24±6 |
12.68±5 |
p <0.05 |
1.12±1 |
1.45±1 |
1.61±1 |
p >0.05 |
|
Growth rate (k) |
0.19 |
0.22 |
0.24 |
- |
0.15 |
0.17 |
0.09 |
- |
|
Dry weight (pg.cell-1) |
3.52±2 |
5.12±3 |
4.43±2 |
p >0.05 |
1654±637 |
1660±820 |
1116±273 |
p>0.05 |
|
Mean cell size (µm) |
13.4±1 |
14.2±1 |
14.7±1 |
p >0.05 |
32.1±4 |
25.46±5 |
27.9±2 |
p <0.05 |
|
TOC (pg.cell-1) |
8.92±2 |
9.87±2 |
10.75±3 |
p >0.05 |
3994±1794 |
2047±1077 |
2289±791 |
p >0.05 |
|
Chlorophyll-a (mg.L-1) |
2172±1271 |
758±217 |
357±166 |
p <0.05 |
41±21 |
66±25 |
26±20 |
p <0.05 |
|
Lipids (% dry weight) |
2.8±1.4 |
12.6 |
9.8±1.3 |
p <0.05 |
3.8±1.2 |
4.7±1.7 |
4.7±2.8 |
p >0.05 |
|
Protein (%dry weight) |
31.7±4.8 |
41.2±3.4 |
37.7±7.6 |
p <0.05 |
43.1±6.8 |
39.9±4.7 |
38.8±6.5 |
p >0.05 |
-= not analyzed
The level of mean cell density for H. pluvialis and growth rate for A. gracilis were similar and some times higher in the alternative media (NPK and M+NPK) when compared with the commercial media (WC and CHU12). The microalga Ankistrodesmus gracilis had a density between 12.6 x 105 cells.ml-1and 16.5 x 105 cells. ml-1 in all culture media during the experimental period. This rate is adequate for larviculture since the number of algae cells recommended at the beginning of larval development is about 6,000 cells. ml-1. day-1 [14]. Although differences in the species composition may be attributed to genetics, differences are frequently due to culture conditions and laboratory methodology.
Microalgae culture in the media NPK and M+NPK had a similar productive response to cultures with commercial medium it may be hypothesized that the use of inorganic fertilizer and E. crassipes with NPK would provide similar or better results at lower costs. Cost of macrophytes was discarded and the plants were harvested at different sites of the university campus.
CONCLUSION
Macrophytes may be used as an important biotechnological tool with high potential for growth and maintenance of Chlorophyceae microalgae. Results demonstrated that physiological parameters such as growth rate, dry weight, mean cell size and TOC levels were higher in the alternative media for A. gracilis and similar between the media for H. pluvialis. The NPK (20-5-20) medium and macrophyte (E. crassipes) medium seem to be an adequate alternative to replace the conventional medium (WC and CHU12) due to good results in their growth and nutritional values. The need to improve the cultivation of microalgae as feed for fish larvae, pigment in aquaculture and also as supplementary additive in food industry may be accomplished by microalgae cultures in alternative culture media such as NPK (20-5-20) and macrophyte (E. crassipes).
ACKNOWLEDGEMENTS
The authors would like to thank FAPESP for its financial support (12/09844-4). Thanks are also due to Flávia Almeida B. Mora is for her laboratory assistance.