A Case of Squamous Cell Carcinoma with Adjacent Bronchopulmonary Cavitary Lesions: A Case Report and Literature Review
- 1. Department of Respiratory Medicine, the First Hospital of Jilin University, China
Abstract
Lung cancer accounts for 12% of all new cancer cases, 21% of all cancer deaths, and stands as the leading cause of death nationwide.
CITATION
Huang Y, Zhao Y (2024) A Case of Squamous Cell Carcinoma with Adjacent Bronchopulmonary Cavitary Lesions: A Case Report and Literature Review. Ann Clin Pathol 11(1): 1167.
INTRODUCTION
Lung cancer accounts for 12% of all new cancer cases, 21% of all cancer deaths, and stands as the leading cause of death nationwide [1]. Lung cavitation is generally defined as lung consolidation, mass, or node containing gas cavities in the lung. Cavitated lung lesions are uncommon on chest computed tomography scans. Cavitation is more common in infectious lung diseases such as pneumonia caused by Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenzae, Klebsiella pneumoniae, Mycobacterium tuberculosis (MTB), and invasive pulmonary aspergillosis. Other uncommon lung diseases like granulomatosis with polyangiitis (GPA), septic embolism, and rheumatoid nodules also necessitate vigilance in a differential diagnosis. Lung cancer with a solitary pulmonary cavity pattern is found in about 8% of all lung cancers and may pose a significant challenge for clinicians and radiologists [2]. Carcinomatous cavitation is a common cause of lung cavities, with the formation of lung cavities being most common in lung squamous cell carcinoma (SCC), often indicating a poor prognosis [3]. In a survey of 102 patients with lung cavities [4], 28 patients (27.4%) had cavitary lung cancer. Among these, 20 cases had secondary lung cancer (19.6%), and 8 cases were lung SCC (7.8%). Other causes of lung cavities include MTB infection, aspergillosis, and lung abscess. Pulmonary amyloidosis, eosinophilic GPA, sarcoidosis, and systemic lupus erythematosus rarely cause pulmonary cavities [5]. Here, we present a 77-year-old man with SCC and an adjacent bronchopulmonary cavity.
CASE PRESENTATION
A 77-year-old man presented with a 2-month history of cough, expectoration, right-sided chest pain, and dyspnea. He was admitted with a fever and hemoptysis for the last 3 days. On physical examination, his temperature was 36.6°C, heart rate 86 beats/min respiratory rate 20 breaths/min, and blood pressure 120/76 mmHg. No jugular vein irritability was observed. The chest appeared symmetrical; however, weak breath sounds were noted in the right lung, no rales was heard. The patient had a previous history of untreated diabetes mellitus and a heavy smoking history of 30 pack-years. There was no history of tuberculosis or drug use, and no family history of cancer. The patient reported a weight loss of approximately 5 kg within the last 3 months.
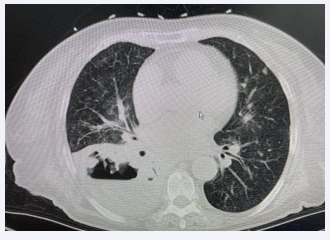
Laboratory tests revealed a white blood cell count of 12*109 /L, with a neutrophil percentage of 76%, and an elevated C-reactive protein level of 232 mg/L. The lung cancer marker cytokeratin 19 fragment (CYPRA21-1) was elevated at 6.44 ng/L (normal range 0–3.3 ng/ml). Sputum cultures and blood cultures yielded negative results for pathogenic microorganisms. CT scan of the chest revealed bronchial wall thickness in the upper and middle lobes of the right lung, along with bronchial changes in the middle and lower lobes of the right lung. Inflammation was observed in both lungs, particularly in the significant lower lobe of the right lung, featuring a combined lung abscess. Enlarged right hilar and mediastinal lymph nodes, as well as right-sided pleural effusion, were evident (Figure 1).
Figure 1: Lung CT imaging (soft tissue window).
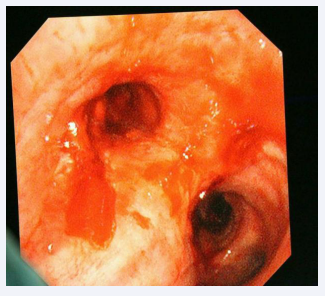
Bronchoscopic examination revealed significant thickening of the right middle and lower lobar bronchus, marked stenosis of the bronchial origin of the right middle lobe. Additionally, the opening of the bronchus in the lower lobe of the right lung remained open, and necrotic granulation tissue was visible, including a large cavity with obvious necrotic lung tissue surrounded by brown necrotic materials (Figure 2).
Figure 2: Bronchoscopy findings.
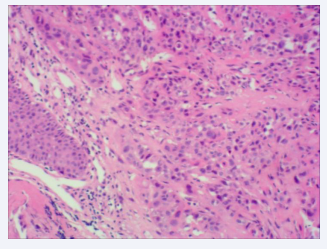
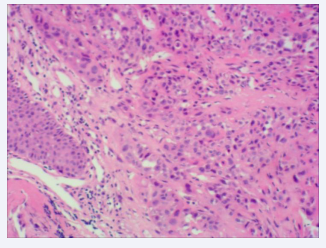
Biopsies were obtained during bronchoscopy at the opening of the middle and lower lobes, and bronchoalveolar lavage was performed on the lower lobe of the right lung. The culture and cytology of the bronchoalveolar lavage fluid were negative. A set of biopsy specimens, stained with hematoxylin and eosin (HE), indicated SCC in the right middle lobe and an inflammatory response in the lower lobe of the right lung (Figures 3,4).
Figure 3: HE staining of the tissue obtained from the right middle lobe of the right lung (×100).
Figure 4: HE staining of the tissue showing inframammary response (×100).
CT scans of the head and abdomen showed no significant abnormalities. Based on these findings, the patient was diagnosed with lung SCC (T2N2Mx), and the etiology of the lung cavity could not be determined. The patient did not qualify for surgery or immunotherapy. Unfortunately, systemic chemotherapy could not be administered due to a poor performance status (PS = 4) and renal insufficiency. Subsequently, hospice palliative care was administered afterward.
DISCUSSION
The pathological mechanisms of lung cavities are complex, involving purulent necrosis, caseous necrosis, ischemic necrosis, cystic expansion of lung structure, or lung tissue displacement caused by cystic structures. These mechanisms can result from various causes, including tumors, infectious diseases (such as MTB, lung abscess, aspergillosis), rheumatic diseases (such as Wegener granulomatosis), pulmonary embolism, and bronchiolitis obliterans [6]. Therefore, a thorough differential diagnosis of the pathological process and etiology of lung cavities is crucial to guide treatment decisions.
In this paper, we present a case of chest pain, hemoptysis, and fever initially diagnosed as a lung abscess. However, subsequent bronchoscopy confirmed SCC in the right middle lobe of the lung and cavitary lesions in the right lower lobe. It is noteworthy that there are few published reports on unique nonmalignant endobronchial lung spaces complicated with lung cancer. The diagnosis of this case was challenging, and the exact etiology of the pulmonary cavity could not be determined. Although not confirmed, we believe that the most likely etiology is malignancy or MTB, aspergillosis or bacterial infection.
Pulmonary cavities with a malignant etiology typically progress in three ways: (1) Swelling may lead to tumor rupture, causing cavitary necrosis. (2) Infection or rupture of the lung parenchyma distal to the tumor. (3) Secondary cancerous abscess produced by a bronchial embolus carrying microorganisms. There is evidence suggesting that the majority of empty lung cancers are of squamous cell origin. Due to the similarity of symptoms between empty lung cancer and other chronic respiratory tract diseases, diagnosing primary lung cancer in the early stage is challenging [7]. Patients with cavitary SCC (cSLC) may initially present with clinical manifestations of infection, leading to potential misdiagnosis. Therefore, we recommend maintaining vigilance for the possibility of malignancy in patients with lung cavities.
MTB infection is more likely to lead to cavity formation as it provides opportunities for organisms to reproduce. Additionally, patients with malignancy face a significantly increased risk of active MTB [8]. In a study of patients undergoing nonsmall cell lung cancer resection, concurrent MTB infection was found in 17 (1.3%) patients; 11 had SCC and 6 had adenocarcinoma [9]. Patients with lung cancer and active tuberculosis typically present characteristic clinical symptoms, including fever, fatigue, dyspnea, and hemoptysis. Bacteriologic analysis of the patient’s sputum and bronchoalveolar lavage fluid suggested negativity for MTB. However, due to the slow growth rate of MTB, even if tuberculosis culture is negative, it may not completely exclude the possibility of MTB infection.
Patients with lung cancer often exhibit immunodeficiency, increasing their susceptibility to fungal infections. Aspergillosis represents an uncommon pulmonary disease, encompassing invasive pulmonary aspergillosis, chronic necrotizing aspergillosis, and allergic bronchopulmonary aspergillosis. Some studies [10,11] have reported the coexistence of aspergillosis and lung cancer in immunocompromized patients who received high-dose chemotherapy. Distinguishing between malignant cavitary lesions and aspergillus is challenging due to overlapping chest CT findings. In this case, bacteriological analyses of sputum, bronchoalveolar lavage fluid, and tissue biopsies were negative for aspergillosis.
Patients with tumors are more prone to bacterial infections due to factors such as tumor obstruction, disruption of natural anatomical barriers (skin or mucosa), and treatment-related interventions (chemotherapy, radiotherapy, invasive tests, surgery). Obstructive pneumonia is common in lung cancer patients, with approximately 40%–50% initially presenting with obstructive pneumonia. Furthermore, around 10%–15% of obstructive pneumonia cases progress to serious complications such as lung abscess [12]. CT examinations often face challenges in distinguishing between lung abscesses and lung cavities with malignant etiology, potentially leading to misdiagnoses of adenocarcinoma as lung abscess in clinical practice. In the presented case, bacteriological analyses of sputum and bronchoalveolar lavage fluid were negative for bacterial infections. However, the inflammatory reaction observed in the bronchus of the lower lobe suggests that the lung cavity in this patient may be attributed to a lung abscess.
The clinical manifestations of cavitary lung disease are nonspecific and mainly include dyspnea, cough, hemoptysis, chest pain, and fever. Accurate diagnosis of lesions in the early stage of the tumor is crucial for patient prognosis. Chest X-ray and CT are often employed for detecting lung disease, with CT being particularly valuable for diagnosing cavitary lesions. The morphology, location, distribution, and other imaging findings of the cavity can provide indications of the underlying disease [13]. Specifically, distinguishing between malignant and nonmalignant etiology is pivotal in the diagnosis of cavitary lung disease.
Some evidence suggests that under CT, lung cancer-associated lung cavitary lesions typically exhibit a wall thickness greater than 4 mm, whereas the wall thickness of benign lesions such as cysts may be less than 4 mm [14]. However, caution should be taken in relying solely on wall thickness to differentiate the etiology of pulmonary cavities, as the wall thickness between malignant lung cavities and those associated with aspergillus and infectious etiologies can overlap. Therefore, a comprehensive approach involving pathological and microbiologic analyses is recommended for diagnosing lesions associated with lung cavities and guiding treatment decisions.
Due to the lack of specificity in the clinical manifestations of cavitary lung cancer and the subjective nature of CT scan interpretation, early misdiagnosis of cavitary lung cancer may occur. Distinguishing the etiology is crucial as it guides treatment and prognosis. In this case, the patient was diagnosed with lung SCC of the right middle lobe and cavitary changes in the lower lobe of the right lung. Sputum and bronchoalveolar lavage cultures, as well as exfoliated cells, were negative, with no MTB infection detected. Considering the combination of lung cavitation and inflammation with the patient’s symptoms and auxiliary examinations is essential. As tumors growing adjacent to the trachea, exhibit less common cavitary changes, clinicians should remain vigilant in recognizing such rare cases to avoid misdiagnosis and delayed treatment.
REFERENCES
- Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023; 73: 17-48.
- Parkar AP, Kandiah P. Differential Diagnosis of Cavitary Lung Lesions. J Belg Soc Radiol. 2016; 100: 100.
- Gafoor K, Patel S, Girvin F, Gupta N, Naidich D, Machnicki S, et al. Cavitary Lung Diseases: A Clinical-Radiologic Algorithmic Approach. Chest. 2018; 153: 1443-1465.
- Giacomelli IL, Barros M, Pacini GS, Altmayer S, Zanon M, Dias AB, et al. Multiple cavitary lung lesions on CT: imaging findings to differentiate between malignant and benign etiologies. J Bras Pneumol. 2019; 46: e20190024.
- Norman HE, Davis JM, Kadaria D. Difficult to Diagnose: An Unusual Cause of Cavitary Lung Lesion. Am J Case Rep. 2020; 21: e921274.
- Lal NR, Agarwal GR, Boruah DK. Role of Multidetector Computed Tomography in Differentiation of Benign and Malignant Cavitary Lung Lesions With a Histopathological Correlation: A Retrospective Cross-Sectional Study. Cureus. 2023; 15: e43005.
- Peter M Ellis, Rachel Vandermeer. Delays in the diagnosis of lung cancer. Journal of Thoracic Disease. 2011; 3: 183-8.
- Dobler CC, Cheung K, Nguyen J, Martin A. Risk of tuberculosis in patients with solid cancers and haematological malignancies: a systematic review and meta-analysis. Eur Respir J. 2017; 50: 1700157.
- Evman S, Baysungur V, Alpay L, Uskul B, Misirlioglu AK, Kanbur S, Dogruyol T. Management and Surgical Outcomes of Concurrent Tuberculosis and Lung Cancer. Thorac Cardiovasc Surg. 2017; 65: 542-545.
- Jiang S, Jiang L, Shan F, Zhang X, Song M. Two cases of endobronchial aspergilloma with lung cancer: a review the literature of endobronchial aspergilloma with underlying malignant lesions of the lung. Int J Clin Exp Med. 2015; 8: 17015-21.
- Dos Santos VM, da Trindade MC, de Souza DW, de Menezes AI, Oguma PM, Nascimento AL. A 76-year-old man with a right lung adenocarcinoma and invasive Aspergillosis. Mycopathologia. 2013; 176: 113-8.
- Rolston KV. Infections in Cancer Patients with Solid Tumors: A Review. Infect Dis Ther. 2017; 6: 69-83.
- Xue XY, Liu YX, Wang KF, Zang XF, Sun JP, Zhang MY, et al. Computed tomography for the diagnosis of solitary thin-walled cavity lung cancer. Clin Respir J. 2015; 9: 392-8.
- Ryu JH, Swensen SJ. Cystic and cavitary lung diseases: focal and diffuse. Mayo Clin Proc. 2003; 78: 744-52.