Psoriasis-Associated IgA Nephropathy: Clinical Manifestations, Pathogenesis, and Therapeutic Approaches
- #. Dafeng He and Yaling Kong contributed equally to this work
- 1. Department of Nephrology, Northern Jiangsu People’s Hospital Affiliated to Yangzhou University, China
- 2. Xiamen Health and Medical Big Data Center (Xiamen Medicine Research Institute), Xiamen Key Laboratory of Natural Medicine Research and Development, China
Abstract
Psoriasis-associated IgA nephropathy (IgAN-pso) is a rare but clinically significant renal disorder characterized by complex interactions between immune dysregulation and cutaneous inflammation. This review describes the clinical presentations and histopathological characteristics of IgAN secondary to psoriasis, emphasizing the multifactorial mechanisms underlying its development and progression. Recent developments in understanding the immunological interplay between psoriatic lesions and renal IgA deposition have been analyzed, with particular attention given to the roles of aberrant mucosal immunity, genetic susceptibility, and systemic inflammatory mediators. Following the discussion of pathogenesis, this review examines current diagnostic challenges, particularly the differentiation between primary IgA nephropathy and secondary IgA nephropathy associated with psoriasis. It also evaluates therapeutic approaches that integrate dermatological and nephrological management, including the use of immunomodulatory agents. By synthesizing current clinical case studies with fundamental research findings, this study aims to offer clinicians an extensive understanding of disease identification and treatment strategies. Ultimately, the goal is to facilitate early diagnosis, optimize individualized therapeutic strategies, and improve long-term renal and dermatological outcomes for the patients.
Keywords
• Psoriasis, IgA Nephropathy, Secondary Nephropathy, Clinicopathology, Pathogenesis, Clinical Management
Citation
He D, Kong Y, Bi G (2025) Psoriasis-Associated IgA Nephropathy: Clinical Manifestations, Pathogenesis, and Therapeutic Approaches. Ann Clin Pathol 12(2): 1180.
INTRODUCTION
Psoriasis is a chronic immune-mediated inflammatory disorder characterized by abnormal proliferation of keratinocytes and systemic immune dysregulation, affecting approximately 2–4% of the global population [1]. Once considered a disease of erythematous scaly plaques, psoriasis is now recognized as a systemic inflammatory disorder that affects multiple organ systems beyond the skin—including the renal, cardiovascular, and metabolic systems—through sustained immune activation [2]. The pathogenesis of psoriasis involves genetic susceptibility, environmental factors, and abnormal immune activation, predominantly mediated through the IL-23/Th17 axis. Key cytokines implicated in this pathway include IL-17, IL-22, and TNF-α [3]. Disruption of the epithelial immune microenvironment (EIME) contributes to chronic inflammation and predisposes individuals to systemic complications [4]. This persistent inflammatory milieu, marked by elevated proinflammatory cytokines and oxidative stress, may ultimately lead to multi organ injury [5].
Renal involvement in psoriasis, although uncommon, has been increasingly reported, with IgA nephropathy (IgAN) representing a notable association [6]. As the most prevalent primary glomerulonephritis, IgAN is characterized by mesangial deposition of aberrantly galactose-deficient IgA1 (Gd-IgA1), leading to inflammation and progressive renal injury. Although IgAN has traditionally been regarded as a primary renal disorder, secondary forms arising from systemic inflammation, including psoriasis, have been documented. Epidemiological and clinical evidence further suggests an overlap between psoriasis and IgAN, though a direct causal relationship remains unclear [7,8]. Case reports have documented psoriasis patients who develop IgAN with severe manifestations, including rapidly progressive or crescentic glomerulonephritis, which complicate clinical management [9]. In our recent research, we have described the clinical and prognostic characteristics of a cohort of patients with psoriasis and biopsy-proven IgAN [10].
The pathophysiological link between psoriasis and IgAN involves systemic inflammation, which promotes aberrant IgA1 glycosylation and deposition of immune complexes [11]. Genetic studies (including those that use Mendelian randomization, which is a method that leverages genetic variants to assess causal relationships) have provided inconsistent evidence for a direct causal relationship [7,8,11]. Transcriptomic analyses further reveal shared inflammatory signatures, such as IL-1β and CXCL9, indicating potential crosstalk between skin and kidney inflammation [11]. Therapies targeting critical signaling pathways, such as IL-17 inhibitors, demonstrate effectiveness in treating both conditions [12]. However, certain immunomodulatory treatments, including anti TNFα agents, may exacerbate renal injury, underscoring the need for careful monitoring [13].
Clinically, psoriasis-associated IgA nephropathy (IgAN pso) can present in a range of ways, from asymptomatic hematuria and/or proteinuria to nephrotic syndrome or rapidly progressive glomerulonephritis. Histological evaluation typically demonstrates mesangial IgA deposition, accompanied by glomerulosclerosis, crescents, or tubulointerstitial injury. Disease severity may correlate with renal dysfunction and the risk of ESRD, highlighting the importance of early detection and intervention [10]. Diagnosis of IgAN-pso requires a renal biopsy to distinguish it from other forms of nephropathy. Management strategies aim to control systemic inflammation using immunosuppressive therapies targeting psoriasis, alongside renal supportive care to maintain kidney function. Furthermore, innovative strategies, including regulatory T cell (Treg)-derived exosomes and mesenchymal stromal/ stem cells, show promise for dual tissue modulation and may offer improved safety profiles [14,15].
In summary, psoriasis and IgAN share immunopathological features, but the underlying mechanisms connecting the two conditions remain to be understood. This review aims to summarize the clinicopathological spectrum, elucidate the pathogenesis, and discuss current management strategies, thereby improving clinical recognition and therapeutic outcomes.
MAIN BODY
Clinical and pathological spectrum of IgAN-pso
Clinical manifestations: IgAN-pso involves both cutaneous and renal manifestations. The most common renal features include microscopic or macroscopic hematuria, proteinuria that may range from mild to nephrotic levels, and impaired renal function. The clinical presentation varies among psoriasis subtypes. Patients with erythrodermic psoriasis often exhibit hematuria, proteinuria, and lower limb edema, sometimes accompanied by severe renal impairment [16]. Those with generalized pustular psoriasis (GPP) may develop rapidly progressive renal dysfunction and biopsy-confirmed IgAN [17]. In cases of psoriatic arthritis (PsA), typical clinical features include joint pain, proteinuria, and hematuria, which often respond favorably to dual-targeted therapy [18].
The severity of psoriasis correlates with renal injury, as severe psoriasis is associated with reduced renal function, increased proteinuria, and a higher risk of progression to ESRD [10]. Studies on Mendelian randomization, however, have produced inconsistent results regarding the causal relationship between psoriasis and IgAN. Some analyses indicate no causal relationship, whereas others recognize overlapping inflammatory pathways that may connect the two conditions [7,8,11]. Overall, renal manifestations in IgAN-pso vary according to psoriasis subtype and disease activity, which underscores the importance of regular renal monitoring.
Pathological features: The pathological features of IgAN-pso are largely similar to those observed in primary IgAN, but they may also exhibit changes reflecting the systemic inflammation associated with psoriasis. On light microscopy, mesangial hypercellularity is typically observed, while immunofluorescence reveals dominant or co-dominant mesangial IgA deposits [19]. Crescent formation may occur, reflecting a disease course that is more aggressive, especially in cases of severe psoriasis [9]. In our recent study, crescentic lesions were observed in 46.2% of patients with IgAN-pso [10], which is higher than that observed in secondary IgAN related to ankylosing spondylitis (43.5%) or rheumatoid arthritis (34.6%) [20]. Gd-IgA1—a key player in primary IgAN—is also observed in IgAN-pso, though its staining intensity varies and may lack specificity, as similar staining patterns may occur in other conditions [19]. Electron microscopy typically confirms mesangial electron-dense deposits, supporting the diagnosis of IgAN.
Comparisons of pathological characteristics between IgAN-pso and primary IgAN were performed in our recent study. Unlike primary IgAN, IgAN-pso often exhibits enhanced inflammatory infiltrates or vascular alterations, reflecting systemic immune activation [10]. Furthermore, secondary IgAN caused by different underlying factors exhibits distinct pathological characteristics that correspond to their specific etiologies [Table 1]. Accurate differential diagnosis from infection-related glomerulonephritis is critical, especially in patients with superimposed infections [17]. Tubulointerstitial damage and markers of chronicity correlate with clinical severity. Immunohistochemical studies have identified inflammatory mediators, such as IL-1β and chemokines (e.g., CXCL9, CCL4), that may contribute to the pathogenesis of both psoriasis and IgAN [11].
Table 1: Comparison of clinicopathological features between secondary IgAN associated with psoriasis, ankylosing spondylitis, and rheumatoid arthritis.
|
|
IgAN-RA (n=26) |
IgAN-AS (n=46) |
IgAN-Pso (n=89) |
P |
|
Crescents |
9(34.6) |
19(41.3) |
41(46.1) |
0.525(χ2, 1.41) |
|
C0 |
17 (65.4) |
27 (58.7) |
48(53.9) |
-- |
|
C1 |
6 (23.1) |
18 (39.1) |
37(41.6) |
-- |
|
C2 |
3 (11.5) |
1 (2.2) |
4(4.5) |
-- |
|
GS, % |
11.3 (24.4)a,b |
8.85 (25.2)b |
20(23.4)a |
0.005 |
|
GS ≥ 50% |
2 (7.7) |
2 (4.3) |
13(14.6) |
0.182f |
|
S, % |
6.5 (10.9) |
1.1 (7.85) |
6.30(13.3) |
0.142 |
|
S-alone |
18(69.2) |
23(50) |
56(62.9) |
0.22(χ2, 3.16) |
|
S-adhesion |
20(76.9) |
23(50) |
56(62.9) |
0.076(χ2, 5.26) |
|
T lesions |
|
|
|
0.226f |
|
T0 |
17 (65.4) |
37 (80.4) |
57(64.0) |
-- |
|
T1 |
7 (26.9) |
7 (15.2) |
27(30.3) |
-- |
|
T2 |
2 (7.7) |
2 (4.3) |
5(5.6) |
-- |
|
ATIL |
15 (57.7) |
16 (34.8) |
42(47.2) |
0.147(χ2, 3.79) |
|
Necrosis |
2 (7.7) |
5 (10.9) |
12(13.5) |
0.842f |
|
Artery lesions |
24 (92.3) |
40 (87.0) |
68(76.4) |
0.128(χ2, 4.19) |
|
ILAL |
17 (65.4)a |
16 (34.8)b |
28(31.5)b |
0.006(χ2, 10.1) |
|
AO/T |
2 (7.7) |
5 (10.9) |
11(12.4) |
0.941f |
|
IC deposits on IF |
|
|
|
|
|
Along the loops |
7 (?26.9) |
5 (?10.9) |
16(18.0) |
0.231f |
|
IgG |
5 (19.2) |
3 (6.5) |
19(21.3) |
0.253f |
|
IgM |
17 (65.4)a |
14 (30.5)b |
36(40.4)a,b |
0.025f |
|
Fibrin |
1 (3.8) |
2 (4.4) |
6(6.9) |
0.898f |
IgAN-RA: IgAN associated with Rheumatoid Arthritis; IgAN-AS: IgAN Associated With Ankylosing Spondylitis; IgAN-pso: IgAN associated with psoriasis, GS: glomerular sclerosis; S: segmental sclerosis; T: Renal Tubular Atrophy/Interstitial Fibrosis; ATIL: Acute Tubular Injury Lesion (including acute tubular necrosis and detachment of the brush border in renal tubules); ILAL: Interlobar Artery Lesion (including interlobular artery hyaline degeneration, intimal thickening, vascular thrombosis, or occlusion); AT/VO: arterial thrombosis or vascular occlusion; IC deposits on IF: immune complex deposits identified by immunofluorescence.
Statistical analysis: The statistical tests used were Student's t-test (for normally distributed continuous data), Mann-Whitney U-test (for non-normally distributed continuous data), chi-square test (for categorical data with expected frequency >5), and Fisher's exact test (for categorical data with expected frequency <5). Bonferroni-corrected pairwise comparisons defined statistical significance at P < 0.05 (chi-square) or α = 0.0167 (Fisher's exact). Shared letters (a, b, c) in the table denote no significant difference.
In summary, the pathology of IgAN-pso is characterized by classic mesangial IgA deposition and hypercellularity. These features are frequently accompanied by crescents and interstitial inflammation, collectively reflecting intricate cutaneo-renal immune crosstalk.
Imaging and Auxiliary Examinations: Imaging and laboratory investigations are essential for the evaluation and monitoring of psoriasis patients with suspected or confirmed IgAN. Renal ultrasonography is frequently employed to assess kidney size, parenchymal echogenicity, and to exclude obstructive causes, though it lacks specificity for diagnosing IgAN. Biochemical analyses of blood and urine are essential for detecting hematuria, quantifying proteinuria, and assessing renal function using serum creatinine and estimated glomerular filtration rate (eGFR). Elevated inflammatory markers, including C-reactive protein (CRP) and immunoglobulin levels, often accompany active disease and may serve as predictors of renal outcomes [10].
Novel diagnostic tools may facilitate the early detection and monitoring of IgAN-pso. For example, fluorescence-optical imaging (FOI) can detect subclinical musculoskeletal inflammation in psoriasis, potentially identifying patients with systemic involvement, including simultaneous renal involvement [21]. Beyond renal evaluation, systemic effects of psoriasis and IgAN have been assessed using complementary imaging modalities. Optical coherence tomography angiography (OCTA), for instance, can detect retinal microvascular alterations in psoriasis patients, which may reflect similar pathological changes in the renal microvasculature [22]. Additionally, laser speckle contrast imaging (LSCI) has been explored to evaluate skin perfusion in psoriatic lesions, potentially providing indirect insight into systemic inflammation [23].
In addition to imaging techniques, circulating levels of Gd-IgA1 and related autoantibodies are emerging as potential biomarkers for IgAN activity. However, their clinical utility in IgAN-pso remains to be fully validated [19]. Together, these imaging and laboratory tests enable early detection, assessment of disease activity, and prognostic evaluation in patients with IgAN-pso.
Pathophysiological mechanisms of IgAN-pso
Dysregulated immunity: from skin inflammation to renal IgA deposition: The immunopathogenesis of IgAN-pso involves dysregulated immune responses that drive abnormal IgA production and mesangial deposition. As a chronic immune-mediated disorder, psoriasis is characterized by systemic inflammation mediated by T helper 17 (Th17) cells and key cytokines, such as IL 17 and IL-23 [24,25]. These mediators not only sustain cutaneous inflammation but also promote the synthesis of Gd-IgA1, a key mediator in IgAN pathogenesis. Gd IgA1 forms circulating immune complexes that deposit in the mesangium, triggering complement through the alternative and lectin pathways and inducing glomerular injury [13,26,27].
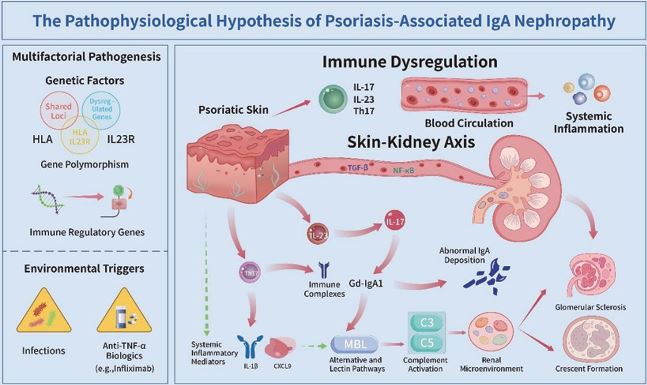
Elevated Th17-associated cytokines and enhanced mesangial IgA deposition in patients with IgAN-pso reflect systemic immune-mediated overproduction and renal deposition of IgA [16,28]. These processes are further amplified by Gd-IgA1-containing immune complexes, which activate complement through the alternative and lectin pathways, thereby exacerbating glomerular injury [29]. Psoriasis-driven systemic inflammation further promotes the formation of immune complexes and renal accumulation, establishing a mechanistic link between cutaneous and renal pathology. Moreover, the clinical efficacy of IL-23/IL-17-targeted biologics (e.g., secukinumab) in improving both cutaneous and renal pathology confirms the central role of this axis in disease pathogenesis [12,13]. Thus, Th17/IL-23-mediated immune dysregulation in psoriasis facilitates aberrant IgA synthesis, immune complex deposition, and complement activation, which together propel the development of secondary IgAN [Figure 1].
Figure 1 The pathophysiological hypothesis of psoriasis-associated IgA nephropathy (IgAN-pso). In IgAN-pso, psoriatic skin inflammation activates the Th17/IL-23 axis, driving systemic immune dysregulation that promotes aberrant Gd-IgA1 synthesis. This leads to immune complex deposition in the renal mesangium, complement activation, and subsequent glomerular inflammation and damage. Genetic susceptibility and environmental triggers modulate this process, with the skin-kidney axis facilitating organ crosstalk through cytokines and immune cell migration.
The skin-kidney axis: Systemic inflammation and organ-crosstalk: The skin–kidney axis describes the pathophysiological interplay through which psoriatic skin inflammation contributes to renal injury, particularly in secondary IgAN. The Th17/IL-23 pathway plays a central role in mediating this immune response, contributing to both cutaneous and systemic inflammation. This systemic inflammation causes the release of proinflammatory cytokines and chemokines into the bloodstream, which in turn disrupts the renal microenvironment and promotes glomerular injury [16,30].
Emerging studies identify IL-23 and IL-17 as key mediators that connect skin and kidney inflammation [27]. Keratinocyte-derived IL-23 stimulates Th17 differentiation and IL-17 production, promoting systemic immune activation and aberrant IgA synthesis [31]. Moreover, systemic inflammatory mediators originating from psoriatic skin lesions enhance the deposition and activate the complement cascade within renal tissue. These systemic cytokines also facilitate the infiltration of macrophages and T lymphocytes into the glomeruli and interstitium, thus further exacerbating renal injury [32].
Clinically, improvement in both skin and kidney manifestations following IL-23/IL-17-targeted therapy supports a functional link between psoriasis and renal involvement [10,12]. Furthermore, the skin-kidney axis may involve additional molecular signaling pathways, particularly those mediated by chemokine-receptor interactions. Emerging evidence from single-cell transcriptomic analyses indicates that chemokine mediated immune cell trafficking between the skin and kidneys contributes to this crosstalk. Understanding these mechanisms lays the groundwork for the development of dual-organ therapeutic strategies in IgAN-pso.
In conclusion, the skin-kidney axis integrates psoriatic skin inflammation and changes in renal microenvironment via systemic inflammation and immune cell migration [Figure 1].
Strategies for clinical management
Diagnostic Workflow and Differential Diagnosis: In patients with psoriasis who develop kidney symptoms, a structured diagnostic approach is crucial to identify the specific renal pathology and to distinguish IgAN from other kidney disorders. The initial evaluation should include a detailed medical history that separately addresses the progression of renal symptoms, the severity of psoriasis, and any previous exposure to nephrotoxic agents or systemic therapies. This should be followed by a thorough physical examination to identify potential systemic complications.
Essential laboratory evaluations include urinalysis to detect hematuria or proteinuria, measurement of serum creatinine, estimation of eGFR, and targeted serological tests for autoimmune markers and infectious agents to rule out other causes. Because psoriasis is an independent risk factor for chronic kidney disease (CKD) and ESRD, routine monitoring of renal function is strongly recommended, especially in patients with moderate to severe psoriasis [6].
Renal biopsy remains the gold standard for evaluating suspected glomerular disease. It confirms the diagnosis of IgAN and distinguishes primary IgAN from secondary forms. Although immunostaining for Gd-IgA1 provides supportive evidence, it is not specific to primary IgAN, as Gd-IgA1 deposition may also occur in secondary IgAN or staphylococcal infection-associated glomerulonephritis (SAGN) [19]. Therefore, clinicopathological correlation is essential for accurate diagnosis. Furthermore, the differential diagnoses should carefully consider drug induced nephropathies, especially in patients receiving immunosuppressive agents or biologics.
Multidisciplinary collaboration among dermatologists, nephrologists, and pathologists is essential for integrating clinical findings, laboratory results, and histopathological data. This coordinated approach supports accurate diagnosis and facilitates timely interventions, helping prevent progressive renal injury. A structured diagnostic workflow that incorporates clinical evaluation, ancillary testing, renal biopsy, and input from multiple specialties provides the foundation for effective management of renal complications in patients with psoriasis [33].
Treatment principles and drug selection: The management of IgAN-pso requires a balanced strategy that addresses systemic inflammation while preserving renal function. Therapeutic decisions should target both dermatological and renal manifestations, emphasizing immunomodulatory strategies that avoid exacerbating renal injury.
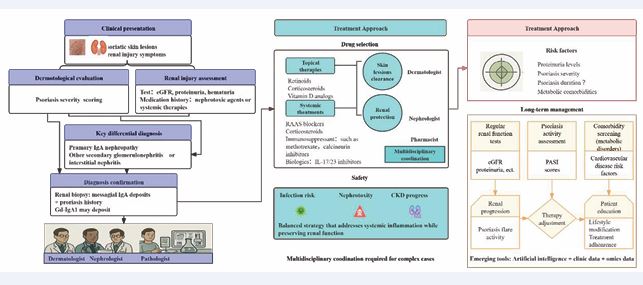
Conventional immunosuppressants, including systemic corticosteroids and calcineurin inhibitors (e.g., cyclosporine), can be effective for both conditions. However, their nephrotoxic potential requires careful dosing and close monitoring. Biologic agents, such as IL-17 inhibitors, have emerged as promising therapies, improving psoriatic lesions and reducing systemic inflammation, which may also benefit renal outcomes. However, their safety in patients with IgAN remains to be fully established, particularly regarding the deposition of immune complexes and infection risk [33]. Targeting shared immunological pathways, especially T-cell mediated mechanisms, offers a rational approach for managing patients with overlapping disease processes. Personalized management of IgAN-pso should consider disease severity, renal function, comorbidities, as well as goals and tolerance of patient-specific treatments. Regular monitoring should include assessment of cutaneous lesions, renal parameters (eGFR, proteinuria), and drug-related adverse effects. Emerging approaches, such as metabolomic profiling, may help identify individual metabolic variations and guide dose optimization to enhance efficacy while minimizing toxicity [34]. Multidisciplinary collaboration between dermatologists and nephrologists is essential to develop tailored therapeutic strategies and facilitate the timely management of complications. In summary, effective management of IgAN-pso integrates immunomodulatory and nephroprotective interventions, combining both conventional and novel therapies in alignment with each patient’s specific needs [Figure 2].
Figure 2 Comprehensive Management Flowchart for Psoriasis-Associated IgA Nephropathy (IgAN-pso). Gd-IgA1: galactose-deficient IgA1; RAAS: The Renin-Angiotensin-Aldosterone System; CKD: chronic kidney disease; eGFR: estimated glomerular filtration rate; PASI scores: the psoriasis area and severity index scores.
Prognostic Evaluation and Follow-up Management: The prognosis of IgAN-pso is influenced by disease severity, therapeutic response, and comorbidities. Key indicators, such as proteinuria, hematuria, and changes in renal function, provide essential information for assessing renal outcomes. Meanwhile, psoriasis severity reflects systemic inflammation. Effective control of systemic inflammation is associated with improved renal prognosis, highlighting the importance of coordinated management of both conditions [33]. Comorbidities such as cardiovascular disease and metabolic syndrome, which are common in patients with psoriasis, may adversely affect prognosis and should be managed alongside both psoriasis and IgAN.
Long-term follow-up should include regular clinical evaluation, renal function testing, urinalysis, and dermatologic examination. Close monitoring is especially necessary during periods of active disease or treatment adjustment to promptly detect changes in renal function or psoriatic activity, enabling timely treatment adjustment to prevent irreversible organ damage [Figure 2]. Patient education on symptom recognition and adherence to treatment is equally essential. In a cohort of 87 patients of IgAN-Pso, 14.9% (14/87) progressed to ESRD or experienced a decline in eGFR greater than 50% over a median follow-up of 34.8 months [10]. This outcome is worse than the long-term renal prognosis observed in secondary IgAN associated with ankylosing spondylitis, and increased inflammation was identified as a risk factor for ESRD in IgAN-pso patients [35]. Future studies should focus on identifying and validating novel biomarkers for early detection of renal involvement and disease recurrence. The development of multimodal predictive models that integrate clinical, laboratory, and molecular data is essential for assessing long-term treatment responses and prognosis. AI offers potential for personalized risk stratification and follow-up by analyzing complex clinical and imaging data [36]. Additionally, further research into the shared immunopathogenic mechanisms between psoriasis and IgAN may uncover new therapeutic targets.
Future directions
Although advances have been made in understanding and managing IgAN-pso, significant knowledge gaps and clinical challenges remain. The immunopathological mechanisms linking psoriasis and secondary IgAN— particularly abnormal IgA deposition and the skin-kidney axis, which describes the interplay between cutaneous and renal immune responses—require further investigation. Well-designed clinical trials are needed to validate diagnostic biomarkers, such as Gd-IgA1, with a focus on their ability to differentiate secondary from primary IgAN. Additionally, the efficacy and safety of biologic therapies in this patient population need to be evaluated in prospective studies.
Interdisciplinary collaboration between dermatologists and nephrologists is crucial. Integrated care models that incorporate shared decision-making, coordinated diagnostics, and unified treatment plans can significantly improve patient outcomes. The establishment of joint clinics or standardized care pathways may further enhance clinical coordination and management efficiency. Furthermore, AI and machine learning hold promise for supporting diagnosis, predicting disease progression, and personalizing treatment. However, challenges related to data quality, model interpretability, and ethical considerations—such as patient privacy and algorithmic bias—must be addressed to enable their reliable implementation [36].
Future research should focus on elucidating the shared immunological and genetic mechanisms underlying psoriasis and IgAN. By integrating multi-omics technologies with multidisciplinary approaches, translational studies are essential to bridge current knowledge gaps. These efforts will facilitate the development of targeted strategies to improve diagnosis, therapeutic interventions, and long term outcomes in affected patients.
CONCLUSION
IgAN-pso is an immune-mediated disorder characterized by diverse clinical manifestations. The diagnosis of IgAN-pso requires comprehensive clinical and pathological assessments. The pathogenesis involves a complex interaction between immune dysregulation, genetic susceptibility, and the skin–kidney axis, a bidirectional mechanism linking cutaneous and renal immune responses. Recent studies have revealed novel mechanisms connecting cutaneous and renal diseases, underscoring the clinical and pathological heterogeneity among patients and the existence of distinct phenotypes.
From the perspective of clinical management, an individualized and balanced treatment approach is required, tailored to the severity of both psoriatic and renal involvement. Multidisciplinary collaboration among dermatologists, nephrologists, immunologists, and pathologists is essential to optimize patient outcomes. Long-term follow-up is recommended for the
risk of disease progression and the systemic effects of immunosuppressive and biologic therapies on both organs. The integration of novel biomarkers and advanced imaging techniques may further enhance diagnostic precision and support treatment strategies that are more adaptive and patient-responsive.
Future research should emphasize translational studies to clarify disease mechanisms and identify new therapeutic targets. Rigorous clinical trials evaluating biologic and targeted immunotherapies are essential to improve efficacy and reduce adverse effects. The development of reliable prognostic tools will support accurate risk stratification and personalized management, ultimately enhancing quality of life and long-term outcomes. Combining insights from immunology, genetics, and clinical medicine is essential for deepening our understanding of this complex disease and improving its treatment.
FUNDING
This work is supported by Yangzhou City Basic Research Program (Joint Special Project)-Health and Wellness Category (Grant number 2025-2-01).
REFERENCES
- Li L, Lu J, Liu J, Wu J, Zhang X, Meng Y, et al. Immune cells in the epithelial immune microenvironment of psoriasis: emerging therapeutic targets. Front Immunol. 2024; 14: 1340677.
- Gisondi P, Bellinato F, Girolomoni G, Albanesi C. Pathogenesis of Chronic Plaque Psoriasis and Its Intersection With Cardio-Metabolic Comorbidities. Front Pharmacol. 2020; 11: 117.
- Vi?i? M, Kaštelan M, Brajac I, Sotošek V, Massari LP. Current Concepts of Psoriasis Immunopathogenesis. Int J Mol Sci. 2021; 22: 11574.
- Dainichi T, Iwata M. Inflammatory loops in the epithelial-immune microenvironment of the skin and skin appendages in chronic inflammatory diseases. Front Immunol. 2023; 14: 1274270.
- Sorokin AV, Remaley AT, Mehta NN. Oxidized Lipids and Lipoprotein Dysfunction in Psoriasis. J Psoriasis Psoriatic Arthritis. 2020; 5: 139- 146.
- Grandinetti V, Baraldi O, Comai G, Corradetti V, Aiello V, Bini C, et al. Renal dysfunction in psoriatic patients. G Ital Nefrol. 2020; 37: 2020- vol1.
- Li C, Wang B, Han T, Xu Y, Li X. Causal relationship between inflammatory skin diseases and immunoglobulin A nephropathy: A Mendelian randomization and enrichment analysis study. Skin Res Technol. 2024; 30: e13915.
- Cao W, Xiong J. Causal associations and potential mechanisms between inflammatory skin diseases and IgA nephropathy: a bi- directional Mendelian randomization study. Front Genet. 2024; 15: 1402302.
- Garces CC, Hernandez Garcilazo N, Sharma A, Nader G. Severe psoriasis presenting with rapidly progressive (crescentic) IgA- predominant glomerulonephritis. BMJ Case Rep. 2021; 14: e242627.
- He D, Bi G, Mou H, Wang R, Lu C, Tang Z. Clinicopathological features and renal outcomes in patients with IgA nephropathy secondary to psoriasis. Clin Exp Med. 2025; 25: 207.
- Chen Y, Huang M, You Z, Sa R, Zhao L, Ku C, et al. Unveiling the genetic link and pathogenesis between psoriasis and IgA nephropathy based on Mendelian randomization and transcriptome data analyses. Arch Dermatol Res. 2024; 316: 717.
- Ochi M, Toyama T, Ando M, Sato K, Kamikawa Y, Sagara A, et al. A case of secondary IgA nephropathy accompanied by psoriasis treated with secukinumab. CEN Case Rep. 2019; 8: 200-204.
- Segawa Y, Ishida R, Kanehisa F, Nakai K, Morimoto M, Seno M, et al. IgA nephropathy in a patient receiving infliximab for generalized pustular psoriasis. BMC Nephrol. 2020; 21: 366.
- Ou M, Cao J, Luo R, Zhu B, Miao R, Yu L, et al. Drug-loaded microneedle patches containing regulatory T cell-derived exosomes for psoriasis treatment. Acta Biomater. 2025; 198: 452-466.
- 15.Dashti M, Mohammadi M, Dehnavi S, Sadeghi M. Immunomodulatory interactions between mesenchymal stromal/ stem cells and immune cells in psoriasis: therapeutic potential and challenges. Stem Cell Res Ther. 2025; 16: 241.
- Zhang L, Xue S, Yu J, Si H, Xu Y, Li J, et al. IgA nephropathy associated with erythrodermic psoriasis: A case report. Medicine (Baltimore). 2019; 98: e15433.
- Klimko A, Toma GA, Ion L, Mehedinti AM, Andreiana I. A Case Report of Generalized Pustular Psoriasis Associated With IgA Nephropathy. Cureus. 2020; 12: e10090.
- Xue H, Ci X, Luo M, Wu L, Du X, Li L, et al. Tofacitinib combined with leflunomide for treatment of psoriatic arthritis with IgA nephropathy: a case report with literature review. Clin Rheumatol. 2022; 41: 2225- 2231.
- Cassol CA, Bott C, Nadasdy GM, Alberton V, Malvar A, Nagaraja HN, et al. Immunostaining for galactose-deficient immunoglobulin A is not specific for primary immunoglobulin A nephropathy. Nephrol Dial Transplant. 2020; 35: 2123-2129.
- He D, Wang R, Liang S, Liang D, Xu F, Zeng C, et al. Comparison of secondary IgA nephropathy in patients with ankylosing spondylitis and rheumatoid arthritis. Mod Rheumatol. 2020; 30: 648-656.
- Koehm M, Ohrndorf S, Foldenauer AC, Rossmanith T, Backhaus M, Werner SG, et al. Fluorescence-optical imaging as a promising easy-to-use imaging biomarker to increase early psoriatic arthritis detection in patients with psoriasis: a cross-sectional cohort study with follow-up. RMD Open. 2022; 8: e002682.
- Castellino N, Longo A, Fallico M, Russo A, Bonfiglio V, Cennamo G, et al. Retinal Vascular Assessment in Psoriasis: A Multicenter Study. Front Neurosci. 2021; 15: 629401.
- Schaap MJ, Chizari A, Knop T, Groenewoud HMM, van Erp PEJ, de Jong EMGJ, et al. Perfusion measured by laser speckle contrast imaging as a predictor for expansion of psoriasis lesions. Skin Res Technol. 2022; 28: 104-110.
- AbuHilal M, Walsh S, Shear N. The Role of IL-17 in the Pathogenesis of Psoriasis and Update on IL-17 Inhibitors for the Treatment of Plaque Psoriasis. J Cutan Med Surg. 2016; 20: 509-516.
- Sharma A, Upadhyay DK, Gupta GD, Narang RK, Rai VK. IL-23/Th17 Axis: A Potential Therapeutic Target of Psoriasis. Curr Drug Res Rev. 2022; 14: 24-36.
- Chang S, Li XK. The Role of Immune Modulation in Pathogenesis of IgA Nephropathy. Front Med (Lausanne). 2020; 7: 92.
- Liu T, Li S, Ying S, Tang S, Ding Y, Li Y, et al. The IL-23/IL-17 Pathway in Inflammatory Skin Diseases: From Bench to Bedside. Front Immunol. 2020; 11: 594735.
- Vaz AS, Penteado R, Cordinhã C, Carmo C, Gomes C. IgA vasculitis(Henoch-Schönlein purpura) nephritis and psoriasis in a child: is there a relationship?. J Bras Nefrol. 2021; 43: 603-607.
- Kaartinen K, Safa A, Kotha S, Ratti G, Meri S. Complement dysregulationin glomerulonephritis. Semin Immunol. 2019; 45: 101331.
- Rahman MF, Kurlovs AH, Vodnala M, Meibalan E, Means TK, Nouri N, et al. Immune disease dialogue of chemokine-based cell communications as revealed by single-cell RNA sequencing meta- analysis. Preprint. bioRxiv. 2024; 2024.07.17.603936.
- Jia Q, Hu J, Wang X, Deng Y, Zhang J, Li H. Malassezia globosa Induces Differentiation of Pathogenic Th17 Cells by Inducing IL-23 Secretion by Keratinocytes. Mycopathologia. 2024; 189: 85.
- Akiyama M, Shimomura K, Yoshimoto H, Sako M, Kodama M, Abe K, et al. Crohn’s disease may promote inflammation in IgA nephropathy: a case-control study of patients undergoing kidney biopsy. Virchows Arch. 2022; 481: 553-563.
- Yin T, Zhang T, Ma L. A Novel Immune-Related Three-Gene Signature and Immune Infiltration Insights in Psoriasis and Chronic Kidney Disease. Clin Cosmet Investig Dermatol. 2025; 18: 267-286.
- Li SS, Liu Y, Li H, Wang LP, Xue LF, Yin GS, et al. Identification of psoriasis vulgaris biomarkers in human plasma by non-targeted metabolomics based on UPLC-Q-TOF/MS. Eur Rev Med Pharmacol Sci. 2019; 23: 3940-3950.
- He D, Wang R, Liang S, Liang D, Xu F, Zeng C, et al. Spectrums and Prognosis of Kidney Disease in Patients with Ankylosing Spondylitis. Kidney Dis (Basel). 2020; 6: 444-452.
- Bilgin E. Current application, possibilities, and challenges of artificial intelligence in the management of rheumatoid arthritis, axial spondyloarthritis, and psoriatic arthritis. Ther Adv Musculoskelet Dis. 2025; 17: 1759720X251343579.