Use of Hypochlorous Acid Solution as a Disinfectant in Laboratory Animal Facilities
- 1. Department of Clinical Laboratory Medicine, Teikyo University, Japan
Abstract
Hypochlorous acid solution was effective not only in preventing contamination of pathogens including opportunistic pathogens, but also in preventing infection without affecting serum biochemical variables. But bacterial microbiota may have changed due to drinking these solutions.
Keywords
• Microbiota
• Pseudomonas aeruginosa
• Rat
• Weak acid Hypochlorous Solution
Citation
Goto K (2015) Use of Hypochlorous Acid Solution as a Disinfectant in Laboratory Animal Facilities. Ann Clin Med Microbio 1(1): 1005.
INTRODUCTION
Recently, hypochlorous acid solution, a weak acid, has been used in a wide variety of settings, including agricultural fields, hospitals, food industry, day care centers, and animal facilities [1]. In this review, we introduce the use of weak hypochlorous acid solutions as disinfectants in laboratory animal facilities.
Hypochlorous acid solution is generated by the electrolysis of a sodium chloride solution. At the positive electrode, the anode, water (H2 O) is transformed into oxygen (O2 ) and hydrogen ions (H+ ), and chlorite ions (Cl- ) are transformed into chlorine (Cl2 ). Chlorine then reacts with water to produce hypochlorous acid (HClO) and hydrochloride (HCl) [2]. Weak acid hypochlorous solution is a chlorine-based disinfectant that is produced by mixing NaClO and HCl in water and adjusting it to a weak acidity of approximately pH 6 [3]. The most effective form of chlorine in weak acid hypochlorous solution is HClO. HClO has been reported to be effective against various microorganisms [4]. Ono et al. demonstrated in vitro that when Pseudomonas aeruginosa, Acinetobacterbaumannii, Staphylococcus aureus (MRSA), Enterococcus faecalis, Enterococcus faecium, and Enterococcus avium are treated with HClO solutions in the pH range of 5–8, within 15 seconds, the organisms are not detected in the cultures. In addition to these pathogens, mouse hepatitis virus, Sendai virus, lymphocytic choriomeningitis virus, Bordetellabronchiseptica, Pasteurellapneumotropica, and Corynebacteriumkutscheri could not be detected after treatment with 9 or 99 volumes of weak acid hypochlorous solution for 5 minutes at 25° C. They concluded that the solution has inactivation activity against laboratory rodent-specific viruses and bacteria when used in a sufficiently large volume or for a longer reaction time [3].
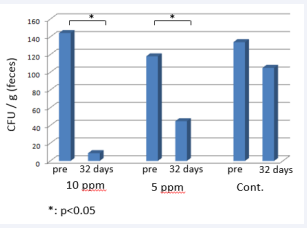
In contrast, the efficacy of HClO in solution is decreased, possibly owing to contact with organic materials in the stomach and intestines. Accordingly, when added to the drinking water provided to the animals, it has not been effective in eliminating pathogens [5]. As shown in Figure 1,
Figure 1: Number of P. aeruginosa in rat feces when 10 ppm or 5 ppm of weak acid hypochlorous solution or tap water was provided.
when 10 ppm or 5 ppm of weak acid hypochlorous solution was incorporated into the drinking water provided to P. aeruginosa infected rats for 32 days, the number of the bacteria in feces was significantly decreased. The infection could not, however, be completely eradicated. Takimoto et al. also suggested that incorporating the solution into the drinking water provided to animals is ineffective in preventing mouse norovirus infection and in eliminating mouse norovirus from already infected mice [6].
In an attempt to prevent P. aeruginosa infection in rats by incorporating hypochlorous solution into the drinking water, it was observed that the bacteria were not detected in feces from the 6 sentinel rats exposed to infected rats in a group that had received the hypochlorous solution up until 49 days of cohabitation [7]. These results suggest that the solution had sufficient preventative activity against P. aeruginosa infection.
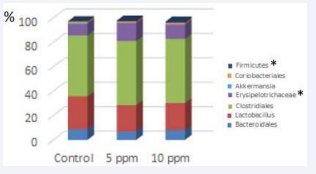
The effect of the solution on cecum microbiota in these rats is shown in Figure 2.
Figure 2: Microbiota of rats was analyzed using polymerase chain reaction amplification and terminal restriction fragment length polymorphism (T-RFLP) analysis. Ratio of T-RFLP peaks for Erysipelotrichaceae in rats who received 5 ppm solution was higher (X = 14.67, SD = 0.61) than that of the control group (X = 9.29, SD = 2.01; p < 0.01). For Firmicutes spp., the ratio of T-RFLP peaks for rats who received 10 ppm solution (X = 0.99, SD = 0.46) was higher than that of the control group (X = 0.18, SD = 0.36; p < 0.01). Results for other bacteria were similar across all groups. The hypochlorous acid solution is an effective tool for cleaning animal facilities but cannot completely eliminate pathogens from infected animals directly. The solution can, however, be used to prevent some bacterial infections such as those caused by P. aeruginosa. In these cases, hypochlorous acid solution can be incorporated into the drinking water; however, this may affect the cecum microbiota
The ratio of the terminal restriction fragment length polymorphism (T-RFLP) peaks did not differ across rats administered with 5 or 10 ppm solution as compared to that of the control group for any bacteria except Erysipelotrichaceae and Firmicutes.
CONFLICT OF INTEREST
These results suggest that the solution had sufficient preventative activity against P. aeruginosa infection when the solution was used as drinking water, however bacterial microbiota may have changed due to weak acid hypochlorous solution administration.