Acanthaster Can Go Faster: Rolling Motion in the Crown-ofThorns Starfish
- 1. UK
Abstract
We report the first account of rolling motion in the corallivorous crown-of-thorns starfish (COTS) Acanthaster spp., which is also the first report of this method of locomotion of any animal in water, and of an animal using this method of locomotion independently of escape from predation. In Weda Bay on the Indonesian island of Halmahera, we observed two adult COTS that were isolated on slopes of sand and sediment form their bodies into ball-like shapes and roll down the slope. Initiation of rolling was not connected to interactions with other animals such as predators or other COTS, but potentially plays a role in dispersal
Keywords
• Crown-of-thorns starfish
• COTS
• Sea star
• Acanthaster
• Rolling
• Locomotion
CITATION
Cranenburgh RM, Cranenburgh EC (2020) Acanthaster Can Go Faster: Rolling Motion in the Crown-of-Thorns Starfish. Ann Mar Biol Res 6(1): 1030.
ABBREVIATIONS
COTS: Crown-of-thorns Starfish
INTRODUCTION
In contrast to its ubiquity in human society, rolling motion in animals has only been observed in a handful of species, and then only on land with the sole function of rapid escape from predators. The dune spider Carparachne aureoflava, endemic to Namibia’s central Namib Desert, escapes from parasitoid pompilid wasps by initially running, then flipping on its side and wheeling down sand dunes, using its legs as the rim and spokes of the wheel [1]. The salamander Hydromantes platycephalus inhabits the steep slopes of California’s Sierra Nevada, where it coils its body and tucks in its limbs to roll a short distance as an anti-predator behavior [2]. A similar strategy is used by the caterpillars of the mother-of-pearl moth Pleurotya ruralis and carnation tortrix Cacoecimorpha pronubana [3,4], which coil into wheels to roll backwards on encountering a mechanical shock on a flat surface. The stomatopod Nannosquilla decemspinosa [5], of the Pacific coast of central America, and the larvae of the southeastern beach tiger beetle Cicindela dorsalis media of the Atlantic coast of the USA assume a head-to-tail coil and propel themselves by somersaulting and rolling when exposed or disturbed on a beach, the latter even being able to roll uphill when wind-assisted [6]. In all these examples, rolling is achieved by the animal adopting wheel-like structure with a defined rim, rather than a ball-like structure as observed in COTS. Animals that curl into balls for defense, such as hedgehogs (Erinaceinae), pill millipedes (Oniscomorpha) and the pill woodlouse (Armadillidium vulgare), may roll down a slope if an event stimulating this behavior occurs there, but this is incidental to the primary purpose of protecting vulnerable underparts of the body. Therefore, our observationadditionally represent the first report of rolling by deliberate tumbling in an animal, rather than by wheeled locomotion; the only other examples in nature being seed dispersal in plants of various species known as tumbleweeds (including the Russian thistles Salsola spp).
MATERIALS AND METHODS
Still photographs and video were taken using a Sony RX-100 camera in a Sea Frogs underwater housing.
RESULTS AND DISCUSSION
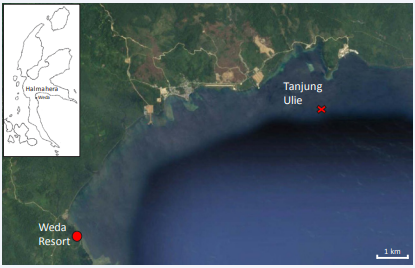
Weda Bay is on the south-east coast of Halmahera. The Bay is an inlet of the Halmahera Sea and comprises a sloping substrate of sand and sediment, fringed by coral reefs
(Figure 1)
Figure 1 The northern end of Weda Bay (Halmahera inset). The location of the rolling observed in COTS near Tanjung Ulie is indicated by a cross and located at 0° 27’ 36.73” N, 127° 58’ 32.02” E. (Google Maps
The sea temperature was 28°C and there was minimal current at the time of the observations. Scuba diving in the area takes place solely from the Weda Reef & Rainforest Resort, and the site in question is called ‘Tanjung Ulie’. On 3 January 2019 on a sandy slope at a depth of 10-12m, we serendipitously observed adult COTS on exposed sandy slopes raising their bodies by bringing their arms together, then maneuvering arms on the downslope side under their bodies, causing them to topple and roll down the slope while maintaining a tight ball-like shape with their arms (Figure 2).
Figure 2 Rolling motion in A. cf. solaris (a) Illustration of the initiation of rolling in A. cf. solaris: (a1) the normal flat morphology. (a2) the tube feet on all arms walk in the direction of the mouth, resulting in the body rising. (a3) the body forms a pronounced hump supported by the distal ends of the arms curling outwards, and when the arms are vertical, the downslope arms are tucked under the body. (a4) the body tumbles in the direction of the tucked arms and begins to roll. (b1-4) Sequential stills from the video of the rolling of A. cf. solaris taken at quarter-turn intervals; the video can be viewed at: https://stock.adobe.com/uk/search/video?serie_id=324635868&asset_id=324635868.
The stills in figure 2b are from the video recording in electronic supplementary material of the rolling motion; the initiation of rolling was seen but not captured. Locomotion by rolling was witnessed in two different COTS on separate occasions over a one-hour period. Rolling continued until they impacted with a mass of coral or another structure, at which point they would extend their arms and use their tube feet to regain their normal stance. The recorded rolling time was 23.5s and the speed was estimated as 0.1 ms-1 with 8 revolutions per minute. There were no predators of COTS seen in the area such as the giant tritonCharonia tritonis, and no other animals had interacted with the COTS before or during the rolling. Rolling behavior has been seen previously in Weda Bay, during control of COTS outbreaks where the population density was high, both without disturbance and when handled by divers (Rob Sinke and Demos Sikome, personal communication), but had not previously been seen in isolated individuals as described here.
COTS are perhaps the most extensively researched and reported of all echinoderms, with observations spanning 250 years and including numerous recent studies on their movement, yet to our knowledge this is the first report of deliberate locomotion by rolling – indeed the first report for any animal in an aquatic environment. When walking using tube feet, larger A. solaris typically reach 33 cm per minute (0.0055 ms-1) on sand which is twice that on coral [7]. Our estimation of 0.1 ms-1 by rolling, albeit from a single recording and clearly dependent on the gradient of the slope, represents an 18-fold increase in speed compared to walking on sand, with a presumed reduction in energy expended. It has been reported that COTS placed on sand move rapidly towards nearby physical structures and that they spend more time moving in the absence of prey [7]. It is possible that the rolling locomotion enables COTS to reach a suitable food source with minimal time and effort, in addition to reducing competition with other COTS where the population density is high. In the specific example of Weda Bay, COTS outbreaks have occurred in recent years on the northern reefs where they are abundant and have been controlled by removal and lethal injection, yet they are absent from the extensive reefs of pristine coral off the western shore. The rolling that has been observed during outbreak control efforts may in some cases be linked to COTS curling into defensive balls with their spines outermost, and rolling where this happens to be on a coral outcrop or a slope, but it appears to primarily be a response to overcrowding by moving more rapidly and efficiently to new feeding areas. The curling behavior has not been reported elsewhere to our knowledge in response to attack by the giant triton, which COTS instead attempt to outrun
Olfactory signals are known to be important in COTS, with those from the giant triton reported to cause rapid movement away from the source, whereas COTS move towards seawater conditioned with aggregation factors released from other COTS in a Y-maze assay [8]. However, vision was shown to be more important than chemoreception in experiments in a semi-controlled environment using untreated and blinded COTS – vision helped them to locate reef structures at least 5 m away, whereas chemoreception was only effective at very short distances [9]. COTS have a single compound eye at the base of the distal-most tube foot of each arm containing approximately 250 ommatidia and able to form true images, with peak sensitivity at 470 nm and an oval visual field measuring approximately 100° horizontally and 30° vertically, combined with the lowest temporal resolution reported of any animal eye (a 0.6-0.7 Hz flicker fusion frequency); thus vision is attuned for detecting large static objects against a blue background [10]. The relative contribution of vision and chemoreception to the initiation of the rolling behavior remains to be elucidated.to keep the sensitive tube feet away from nematocysts [11], and is similar to the posture on sand before the arms on one side are tucked in to initiate the roll. When the distal tube feet contact the substrate during rolling, it is not clear if they are used to push in the direction of travel - further study would be required to confirm whether the rolling is passive or partially assisted. In areas where the substrate is flat rather than sloping, it is possible that rolling is assisted by current.
CONCLUSION
Our observations reveal a novel method of locomotion in one of the most intensively studied and ecologically important marine organisms, they add to the number of species that roll as an alternative method of locomotion, and represent the first account of this behavior independent of escape from attack by predators or from the risk of predation. We anticipate that these observations will result in further research to determine the incidence of COTS rolling motion throughout the Indo-Pacific region, encompassing the Acanthaster species complex, and will allow a re-evaluation of the models used to estimate the spread of COTS and resultant impact on coral reef ecosystems during periodic outbreaks
ACKNOWLEDGEMENTS
We would like to thank Rob Sinke and Demos Sikome of Weda Reef & Rainforest Resort for dive guiding and additional observations of rolling motion in COTS.
REFERENCES
- Henschel JR. Spiders wheel to escape. S Afr J Sci. 1990: 86: 151-152.
- García-París M, Deban SM. A novel antipredator mechanism in salamanders: rolling escape in Hydromantes platycephalus. J Herpetol. 1995; 29: 149-151.
- Brackenbury J. Caterpillar kinematics. Nature 1997; 390: 453.
- Brackenbury J. Fast locomotion in caterpillars. J. Insect Physiol. 1999; 45: 525-533.
- Full R, Earls K, Wong M, Caldwell R. Locomotion like a wheel? Nature 1993; 365: 495.
- Harvey A, Zukoff S. Wind-powered wheel locomotion, initiated by leaping somersaults, in larvae of the southeastern beach tiger beetle (Cicindela dorsalis media). PLoS ONE. 2011; 6: e17746.
- De’ath G, Moran PJ. Factors affecting the behaviour of crown-of-thorns starfish (Acanthaster planci L.) on the Great Barrier Reef: 2: Feeding preferences. J Exp Mar Biol Ecol. 1998; 220: 107-126.
- Uthicke S, Doyle J, Duggan S, Yasuda N, McKinnon AD. Outbreak of coral-eating crown-of-thorns creates continuous cloud of larvae over 320 km of the Great Barrier Reef. Sci Rep. 2015; 5: 16885.
- Allen JD, Richardson EL, Deaker Dione. Larval cloning in the crown-of-thorns sea star, a keystone coral predator. Mar Ecol Prog Ser. 2019; 609: 271-276.
- Volger C, Benzie J, Lessios H, Barber P, Wörheide G. A threat to coral reefs multiplied? Four species of crown-of-thorns starfish. Biol Lett. 2008; 4: 696-699.
- Haszprunar G, Volger C, Wörheide G. Persistent gaps of knowledge for naming and distinguishing multiple species of crown-of-thorns-seastar in the Acanthaster planci species complex. Diversity. 2017; 9: 22.
- Pratchett MS, Cowan ZL, Nadler LE, Caballes CF, Hoey AS, Messmer V, et al. Body size and substrate type modulate movement by the western Pacific crown-of-thorns starfish, Acanthaster solaris. PLoS ONE. 2017; 12: e0180805.
- Hall MR, Kocot KM, Baughman KW, Fernandez-Valverde SL, Gauthier MEA, Hatleberg WL, et al. The crown-of-thorns starfish genome as a guide for biocontrol of this coral reef pest. Nature. 2017; 544: 231-238.
- Sigl R, Steibl S, Laforsch C. The role of vision for navigation in the crown-of-thorns seastar, Acanthaster planci. Sci Rep. 2016; 6: 30834.
- Petie R, Hall MR, Hyldahl M, Garm A. Visual orientation by the crown-of-thorns starfish (Acanthaster planci). Coral Reefs 2016; 35: 1139-1150.
- Barnes DJ. Locomotion response of Acanthaster planci to various species of coral. Nature. 1970; 228: 342-344.