Neurobehavioral Impairments and Disorders Induced by Developmental Exposure of Chlorpyrifos and Its Effective Treatments
- 1. Department of Zoology, Government College, India
Abstract
Chlorpyrifos (CPF) is one of the widely used organophosphate which is one of the few discovered chemicals to be the developmental neurotoxicant as it targets the critical period of developmental maturation of brain and targets the behavioral development. Many noncholinergic and cholinergic mechanisms are involved commencing the disrupted cell replication, axogenesis and the differentiation leading to neurobehavioral impairments with a number of developmental disabilities which are diagnosed in children at an alarming increasing rate. These persistent decrements in developmental abilities may well presage later development of neurodegenerative disease thus may lead to neurodegenerative diseases. All these above-mentioned mechanisms ultimately lead to enhanced oxidative stress. Therefore during the pregnancy, the natural antioxidants which are reported to have no side effects can be investigated as the treatment against CPF intoxication protecting the developing fetus brain. Apart from the above discussed antioxidants, certain Ayurvedic products having no side effects during the pregnancy and on the neural growth and development should be investigated and explored against CPF intoxications. There are certain such natural products discussed which can be the treatment against neurodegenerative effects in developing brain. Therefore, these natural products must be investigated for their therapeutic potential against CPF intoxication in developing brain leading to adverse neurobehavioral impairments and neurogenerative ailments.
Keywords
• Chlorpyrifos
• Neurodevelopment
• Toxicity
• Neurobehavioral
Citation
Sharma N (2025) Neurobehavioral Impairments and Disorders Induced by Developmental Exposure of Chlorpyrifos and Its Effective Treat ments. Ann Neurodegener Dis 9(1): 1043.
INTRODUCTION
The relation between the pesticide exposure, neurobehavioral and neurodevelopmental effects is an emerging area of great concern. This fact is evidently reported by the study of [1], where the endpoints examined included neurobehavioral, affective and neurodevelopmental outcomes amongst occupational (both adolescent and adult workers) and non-occupational populations (children) exposed to the neurotoxic pesticides. This creates the urge to explore more about the neurotoxic effects of pesticides across the lifespan. Of among 200 chemical neurotoxicants, many are developmental neurotoxicants [2]. Developmental neurotoxicity causes brain damage that is too often untreatable and frequently permanent. The consequence of such brain damage is impaired CNS function that lasts a lifetime and might result in reduced intelligence, as expressed in terms of lost IQ points, or disruption in behaviour. Disorders of neurobehavorial development affect 10–15% of all births [3]. Subclinical decrements in brain function are even more common than these neurobehavioral developmental disorders. All these disabilities can have severe consequences [4], they diminish quality of life, reduce academic achievement, and disturb behaviour, with profound consequences for the welfare and productivity of entire societies [5]. Developmental neurotoxicity studies have been reported by the epidemiological data showing that prenatal exposure to chemical neurotoxicants may be associated with an increased risk of pervasive developmental disorders, delays in cognitive development, and attention deficits. These developmental neurotoxicants have such an adverse effect on the fetal brain as it undergoes rapid growth and development, leaving them susceptible to long-term effects of these neurotoxic OPs. Many studies suggest the associations of developmental exposures to OPs and neurological deficits such as in IQ [6-8], increase in degenerative disorders like autism spectrum [9], attention deficit-hyperactivity [10,11], and pervasive developmental disorder [8-12]. Motor skill acquisition in infancy is another neurobehavioral end point that provides a foundation for downstream cognitive and socio-emotional development in childhood [13], and serve as an early benchmark of healthy neurological development [14]. Chlorpyrifos is one of the organophosphate insecticides which is the highest selling insecticide across the world [15]. Due to its lipophilic nature, chlorpyrifos can cross the blood brain, placental and lactational barriers which induces the developmental neurotoxicity resulting in long lasting neurobehavioral alterations [16]. CPF has the wide ranged effect on the critical period of developmental maturation and its target on brain and behavioral development is quite vulnerable for the embryonic development [17]. This has been evidently proved by the in vitro studies involving the dose administration of higher concentration levels resulted in the reduced neuronal growth exhibiting destruction of noncholinergic and cholinergic mechanisms commencing from the neurological disruptions such as cell replication, axogenesis and the differentiation [18]. These neurological disruptions during the sensitive developmental period in the fetal brain has been suggested to be associated with a number of developmental disabilities (learning disabilities, attention-deficit hyperactivity disorder, dyslexia, sensory deficits, mental retardation, and autism spectrum disorders) which are diagnosed in children at an alarming increasing rate [19-21]. These neurobehavioral disorders further can be broadly studied to be composed of a large group of behavioral impairments seen in association with neurodegenerative disease (e.g., stroke, multiple sclerosis, dementia, and neuro-oncological conditions), transient as well as permanent brain impairments (e.g., metabolic and toxic encephalopathies), injury (e.g., trauma, hypoxia, and/or ischemia) and motor skill development [22]. The persistent decrements in intelligence documented in children, adolescents, and young adults exposed in early life to neurotoxicants may well presage later development of neurodegenerative disease thus may lead to Parkinson’s Disease (PD), Autism spectrum disorders and Alzheimer’s disease as a result of developmental exposures to the neurotoxicants [23]. The mechanism of action of CPF as developmental neurotoxicant leading to neurobehavioral impairments including cholinergic, noncholinergic mechanisms and also as genotoxicant lead ultimately to imbalance of ROS and antioxidants in brain creating the oxidative stress. This oxidative stress in brain is evidently proved to be responsible for altered neurodevelopment leading to neurobehavioral alterations having long lasting effects as various impairments and disorders in later stages [24]. Therefore, to target this oxidative stress and imbalanced ROS it can be suggested that antioxidants can have modulatory effects as they can scavenge the free radicals [25]. So, natural antioxidant such as vitamin C, E, polyphenol compounds such as flavonoids, alkaloids, curcumin without any side effects in pregnancy can be suggested to prevent CPF induced neurodevelopmental toxicity thus ameliorating the neurobehavioral impairments and related disorders. This review sheds light on the role of antioxidants as the modulatory treatment against these neurodevelopmental alterations and outcomes induced via Chlorpyrifos induced neurobehavioral impairments and related disorders due its exposure in developmental period.
CHLORPYRIFOS AS A DEVELOPMENTAL NEUROTOXICANT
Chlorpyrifos is one of the widely used organophosphate pesticide, it represents a paradigmatic example of developmental neurotoxicant as it elicits developmental neurotoxicity at exposure levels below the threshold for systemic toxicity, such that adverse effects can occur in pregnant women and children Ricceri et al. [26], try changing general factual references to the latest years. Experimental studies in rodents indicate that pre- or postnatal exposure to chlorpyrifos affects various cellular processes (e.g. DNA replication, neuronal survival, glial cell proliferation), noncholinergic biochemical pathways (e.g., serotoninergic synaptic functions, the adenylate cyclase system), and causes various behavioral abnormalities (e.g. locomotor skills, cognitive performance) (Ricceri et al. [27], add more references). These findings, together with results of biomonitoring studies that indicate exposure of children, particularly in inner cities and in farming communities, to organophosphates [28], have led to regulatory restrictions on the residential use of certain OPs (e.g., diazinon, chlorpyrifos), and to heightened concern for their potential neurotoxic effects in children [29,30]. CPF has the wide ranged effect on the critical period of developmental maturation and its target on brain and behavioral development is quite vulnerable for the embryonic development [17]. This has been evidently proved by the in vitro studies involving the dose administration of higher concentration levels resulted in the reduced neuronal growth exhibiting destruction of noncholinergic and cholinergic mechanisms commencing from the disrupted cell replication, axogenesis and the differentiation [18], reported to have children with neurobehavioral impairments [8]. Some of the evidences clearly indicate this neurobehavioral impairment due to CPF intoxication such as Alvin et al. [31], demonstrated that the rats injected with CPF subcutaneously (dose range, 2.5 18 mg/kg b.wt.) for 30 days there was decrement in water maze hidden platform task and certain reflex activities like grasping, open field activity indicating the decline in neurobehavioral impairments. Few rodents studies so far have focused on the behavioral effects of CPF in early developmental phases such as in preweaning rats righting reflexes and cliff avoidance like reflex activities were found to be altered after repeated low level CPF exposure during late gestation and deficits in reflexes were observed in female pups after PND 1-4 exposure [32]. Therefore CPF neurotoxicity after prenatal and postnatal exposure and developing organisms appear more controversial as most of animal studies indicate that CPF exposure below threshold can lead to disruptive effects on CNS and neuro behavior [33,34].
Prenatal developmental toxicity
CPF exposure during the pregnancy period is vulnerable to the developing fetus and is area of major concern. The last trimester of pregnancy is the period of major window period for the neurogenesis and formation of other neuronal systems [35]. Major brain region formation occur in this period and it is considered as the period of intense activity of cerebrum and hippocampus thus attributing the developing fetus with the sensorimotor and physical development parameters [17]. The prenatal CPF exposure to developing fetus occurs through the placenta due to its lipophilic property [36]. Many investigatory studies have exposed the relation of dam exposure and toxicity effects in the neonate due to placental transfer by detecting the CPF levels in the umbilical cord and detecting its impact on the development of brain in children in later stages or during their growth period [11]. Exposure to mother to the toxic dose of CPF brings about increased fetal absorption, low birth weight along with visceral and skeletal abnormalities of the embryo [18]. CPF prenatal exposure invoked the negative effects on the development of locomotory, behavioral and cognitive abilities of infants. Prenatal exposure to organophosphate pesticides is negatively affecting the child neurobehavioral performance including abnormal reflexes in infants [8], mental and psycho motor development delay in toddlers [11,12], and lower intelligence levels and cognitive impairments in school children [6,7]. Several evidences have been reported to cause neurobehavioral impairments due to prenatal CPF intoxication such as in Table 1.
Table 1: The summary of experimental studies reported to have neurobehavioral effects in neonates due to CPF prenatal exposure.
|
Model System |
Dose and Route of Exposures in dams |
Time of Exposure |
Neurotoxic Effects |
References |
|
CD 1 mice |
Oral dose of 6 mg/kg b.wt. |
GD 14-17 |
Reduced motor behavior Hypereflexia (Hind limb grasping) Delay sensorimotor activities |
Venerosi et al., 2009 [32] |
|
ND 4 mice |
1 or 5 mg/kg/day in DMSO; subcutaneous |
GD 17-20 |
Foraging maze test results: Decreased spatial learning Decreased memory |
Haviland et al., 2010 [37] |
|
SD rat |
1 or 5 mg/kg/day in DMSO; subcutaneous |
GD 17-20 |
16 arm radial maze test results: Decreased spatial learning Decreased memory |
Levin et al., 2002 [38] |
|
ICR mice |
1 or 5 mg/kg/day in DMSO; subcutaneous |
GD 13-17 |
Memory impairment |
Chen et al., 2012 [35] |
|
Wistar rat |
0.01, 0.01, 10 mg/kg/day b.wt.; oral |
GD 14-20 |
Anoxigenic and anxiety like behavior |
Silva et al., 2017 [39] |
Many studies proved that the CPF exposure in early postnatal stages to result in the inhibition of acetylcholinesterase signifying it to be important component of development toxicity [15]. The development of major regulatory systems underlying behavior and physiology in neonates is primarily determined by the dam, the primary source of nutrition, grooming, and warmth required for immediate survival [40], thereby playing a crucial role in the postpartum development of the architecture of the brain. Chlorpyrifos can easily cross the blood brain barrier [41], therefore after passing down through lactation in neonates can easily target the brain developmental processes thus producing structural or functional changes that result in behavioral changes such as lower cognitive abilities, neuromotor and neurobehavioral activities [42-44]. These significant effects could be due to degradation of lipids and proteins after exposure of CPF in brain leading to generation of free radical species [45], creating the oxidative stress in brain. There are changes in antioxidant status leading to altered activities of cellular enzymes [46,47], exerting its effect on neurotransmitter signaling pathways [18]. The postnatal CPF intoxication and its resultant altered neurobehavioral effects are evidently reported by many experimental studies as in Table 1.
CHLORPYRIFOS INDUCED DEVELOPMENTAL NEUROBEHAVIORAL DEFICITS AND DISORDERS IN NEONATES
Pesticides alter the levels of neuroproteins that are important for normal brain development and thus neurobehavioral abnormalities are manifested as altered adult spontaneous behavior. The neurotoxic behavioral effects persist several months after the initial testing, indicating long-lasting or even persistent irreversible effects which confirms the long lasting changes in behavior when exposed during a critical period of brain development [48]. These neurobehavioral disorders further can be broadly studied to be composed of a large group of behavioral impairments seen in association with neurodegenerative disease (e.g., stroke, multiple sclerosis, dementia, and neuro-oncological conditions), transient as well as permanent brain impairments (eg., metabolic and toxic encephalopathies), and/or injury (e.g., trauma, hypoxia, and/or ischemia) [22]. There is growing concern that chronic or sub-chronic low-level exposure to OPs may affect neural patterning during embryonic development, and may contribute to various neurobehavioral disorders such as autism, anxiety, depression, and attention deficit hyperactivity disorder (ADHD) [11-49]. Various studies reported that the developmental exposures to CPF have drastic outcomes on the development of brain leading to the neuronal impairments at the exposures below the threshold for causing the systemic toxicity [50]. Further there are various experimental studies which showed that any stress occurring in early developmental stages have long term influences and changes in gene expressions in brain leading to behavioral impairments [51]. Many studies have been conducted to reveal such behavioral alterations in neonates due to Chlorpyrifos developmental exposure to dams such as in Table 2.
Table 2: The summary of experimental studies reported to have neurobehavioral effects in neonates due to CPF postnatal exposure.
|
Model System |
Dose and Route of Exposures in dams |
Time of Exposure |
Neurotoxic Effects |
References |
|
SD rats |
1-3 mg/kg b.wt., oral |
PND 11-14 |
Altered neurobehavioral responses |
Ricceri et al., 2006 [26] |
|
SD rats |
5 mg/kg b.wt., oral |
PND 10-16 |
Altered social interactions Behavioral alterations including anxiety |
Carr et al., 2011 [52] |
|
SD rats |
3 mg/kg/48hr. b.wt., oral |
PND 1-21 |
Reduced neuromotor activities |
Carr et al., 2001 [53] |
|
SD rats |
1 mg/kg b.wt., oral |
PND1-4 |
Reduced neurobehavioral and sensory reflexes |
Dam et al., 2000 [54] |
|
Wistar rats |
1 mg/kg b.wt., oral |
GD7-PND1 |
Decreased spatial learning and memory |
Gomez- Gimenez et al., 2017 [55] |
|
SD rats |
6 mg/kg b.wt., oral |
PND 1-21 |
Memory impairment, attention and learning deficit. |
Johnson et al., 2009 [56] |
- Anxiolytic effects of CPF using elevated plus- maze tests [57], open field tests which were included in thigmotaxic alterations induced due to prenatal exposure and post-natal exposure [58].
- Low frequency of behaviors of locomotion and rearing in open field tests are indicative of decreased locomotion and exploration level [59].
- The cognitive ability control and the impulsive attributes leading to highly aggressive behaviour [60].
- The alterations in sleep index or shift in sleeping time periods which is known to be the result of synaptic destruction leading to further, changes in sleep behaviors including a shift in timings [61].
- Motor agitation and hyperactivity signs. The model offspring of rats prenatally exposed to the single dose of CPF had difficulties solving the extrapolation escape test and showed poorer short and longterm memory performance. This confirmed that even pre-pregnancy chlorpyrifos exposure can cause neurobehavioral consequences in offspring [62].
- Many studies have been reported to investigate the gestational pesticide exposure leading to highly reduced levels in child IQ level [6,7], along with it there have been many investigations and cohort studies indicating the prenatal or postnatal exposure through different routes in the mother can effect the weight of the child. Further many studies also resulted in the fact that the reduction in birth weight also relates with the lower IQ as well poor cognitive functions which showed that the prenatal or postnatal exposure of chlorpyrifos to the dam can lead to the decrease birth weight accompanied with many neurodevelopment impairments in offspring [63,64].
- Attention problems associated with alertness, quality of alert responsiveness, cost of attention and other potential attention associated measures [65].
Disruptions in emotional, cognitive, and social behavior are common in neurodegenerative disease and many forms of psychopathology [66]. The major concerns related to developmental OP exposure are delayed effects following high level exposures as well as the impact of low level exposures during the lifespan which are suggested to be a risk factor for nervous system chronic diseases like Alzheimer’s, Parkinson’s, and Amylotrophic Lateral Sclerosis diseases [67]. Evidently CPF which is one of these organophosphate insecticide has been reported to cause various pathologies like excessive microglial activation and subsequent neuroinflammation leading to neuronal cell death which are involved in the pathogenesis and progression of several neurodegenerative diseases such as Parkinson’s disease [68], induces oxidative stress and neuronal damage causing Alzheimer’s disease (AD) [69]. Therefore, neurobehavioral impairments after prenatal and postnatal exposure of CPF in dams it is matter of great concern as it can further lead to pathologies and disorders in neonates and adults.
MECHANISMS INVOLVED IN CHLORPYRIFOS INDUCED NEUROBEHAVORIAL DISORDERS
Long-term low dose effects of chlorpyrifos exposure and its mechanism of action are linked to neurobehavioral diseases [70]. There are different pathways of mechanism of action of chlorpyrifos causing the neurological alterations leading to impairments in brain. Inhibition of enzyme acetylcholinesterase (AChE) is the major action of CPF neurotoxicity mechanism [71]. The serine hydrolase (AChE) is majorly present in the synaptic clefts of cholinergic signaling systems. Its main action in brain is to transmit the impulses by its hydrolysis at its serine and ester hydrolytic sites, the inhibitors block these sites thus inducing the reversible and irreversible inhibition. CPF capability to curb AChE, leading to hyper activation of cholinergic neurotransmitter systems [72], incomplete sentence. CPF alters the gene expression of neurotrophic factors [73], thus invoking the enhanced oxidative stress [74,75]. CPF is known to inhibit the antioxidative enzymes [76], leading to damage at DNA levels and further enhancing the Reactive Oxygen Species (ROS) resulting in elevated levels of oxidative stress. Acetylcholinesterase inhibition is yet another component in case of developmental toxicity where it causes alteration in neural development and induce the negative development when exposed to chlorpyrifos prenatally or postnatally [36]. CPF induces the developmental neurotoxicity which curbs the replication in cell cycle process neural cellular replication [77], which interfere with the differentiation of cell, interferes with cellular differentiation [78], invoking the higher levels of oxidative stress disrupting the neurotransmitter cell signaling systems which results in neurobehavioral impairments [75]..
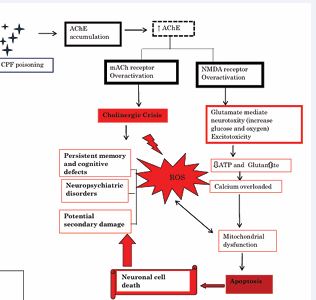
The course of action involving the AChE inhibition is accounted as it crosses the blood brain barrier [41]. As blood brain barrier provides protection to the brain from stress induced alterations and the other toxicant compounds that may enter the circulation plays significant role in stabilizing the constant environment for basic functioning of brain activities [79]. CPF destructs the blood brain barrier leading to inhibition of acetylcholinesterase activity generating reactive oxidative species in brain thus, increase in caspases activity indicates invoking to the beginning of higher apoptotic activities suggesting possible induction of apoptosis. The major transcription genes which are involved for neural maturation, signaling receptor synthesis, myelination, neurotransmitter receptor systems are also elicited by its activity [50-83]. CPF mainly targets the nuclear and cell signaling transcriptional gene factors such as cAMP leading to effect on their resultant actions such as modulations of G-proteins and various receptor systems related to cAMP pathways. These effects invoke other downstream processes such as apoptosis, oxidative stress and the excitotoxic cell death. These processes clearly indicate the genotoxicity exhibited by CPF. As per the studies CPF is suspected to be capable of inducing the DNA crosslinks as it contains two methoxy groups that act as alkylating agents, further it can induce the phosphorylation of DNA as the phosphorous moeity may act as the neucleophilic agents, cell cycle desruption have also been reported such as blocking of the checkpoints and transitions in the cell cycle thus, the DNA damage and the cell cycle alterations (including the upregulation mRNA genes such as p53) and apoptosis account for the genotoxicity [84]. The studies concluded that CPF induces the generation of these free radical species during the metabolism or due to disruptions in cell cycle leading to apoptosis or due cell deaths thus leading to alterations or chemical modifications in DNA bases and sugars. The basic mechanism of CPF leading to genotoxicity is its targeting on the mitochondrial membrane and its caspases thus leading to much increased apoptosis and generation ROS further causing much DNA damage and the genotoxicity [85]. Further inducing a clear excitotoxic neural death [86]. CPF mainly targets the neurotransmitter pathways in the developing brain [50-87], such as acetylcholine, dopamine, serotonin, endo-cannabinoid (noncholinergic) signaling pathways leading to neurodegeneration. CPF can undergo the mechanism of action by targeting the expression of NMDA receptor system [88], in brain leading to alteration in the gene expression of neurotrophic gene expression [86-89], thus invoking the oxidative stress [74,75]. The hyperactivation of NMDA receptor can trigger the neuronal injury which could be lethal reflecting the fact that it has greater ability to induce calcium influx. The over activation of these receptors induce glutamate mediated neurotoxicity called the glutamate induced excitotoxicity leading to excessive glucose and oxygen and thereby ATP levels decrease leading to increase in glutamate. Thus, accumulation of glutamate triggers the calcium influx further triggering various intracellular cascades and neural damage [90], as explained in Figure 1.
Figure 1 Action mechanics of CPF
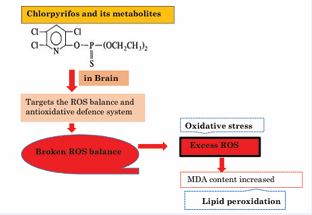
generation of ROS and induction of intracellular oxidative stress thereby disrupting normal cellular development and differentiation [92]. Chlorpyrifos has also been reported to also induce oxidative stress in different parts of the brain, liver through increased levels of reactive oxygen species (ROS), hydrogen peroxide (H2 O2 ), nitrate (NO3-) and nitrite (NO2-) [93]. Accumulation of ROS in all the region of the brain and other tissues may disturb the normal physiological function thus aggravating the toxicity symptoms of CPF. Several studies point to the production of ROS as a secondary means of toxicity [92]. These include hydroxyl, peroxyl radicals and hydrogen peroxide that target and inactivate biological macromolecules eventually damaging membranes and other tissues increasing the lipid peroxidation leading to excess oxidative stress as in Figure 2 [94].
These mechanisms of action can link chlorpyrifos to many neurobehavioral impairments and disorders mainly neurodevelopmental and neurodegenerative disorders as in Table 2.
Figure 2 Chlorpyrifos and its metabolites leading to oxidative stress in brain.
These mechanisms of action can link chlorpyrifos to many neurobehavioral impairments and disorders mainly neurodevelopmental and neurodegenerative disorders as in Table 2.
Table 2: The summary of experimental studies reported to have neurobehavioral effects in neonates due to CPF postnatal exposure.
|
Model System |
Dose and Route of Exposures in dams |
Time of Exposure |
Neurotoxic Effects |
References |
|
SD rats |
1-3 mg/kg b.wt., oral |
PND 11-14 |
Altered neurobehavioral responses |
Ricceri et al., 2006 [26] |
|
SD rats |
5 mg/kg b.wt., oral |
PND 10-16 |
Altered social interactions Behavioral alterations including anxiety |
Carr et al., 2011 [52] |
|
SD rats |
3 mg/kg/48hr. b.wt., oral |
PND 1-21 |
Reduced neuromotor activities |
Carr et al., 2001 [53] |
|
SD rats |
1 mg/kg b.wt., oral |
PND1-4 |
Reduced neurobehavioral and sensory reflexes |
Dam et al., 2000 [54] |
|
Wistar rats |
1 mg/kg b.wt., oral |
GD7-PND1 |
Decreased spatial learning and memory |
Gomez- Gimenez et al., 2017 [55] |
|
SD rats |
6 mg/kg b.wt., oral |
PND 1-21 |
Memory impairment, attention and learning deficit. |
Johnson et al., 2009 [56] |
- Chronic organophosphate induced neuropsychiatric disorders (COPIND) occur without cholinergic symptoms and apparently are not dependent on AChE inhibition [95,96]. COPIND usually appears with a delay and persists for a long period possibly suggesting the permanent damage of the central nervous system [97-99]. The most common symptoms of COPIND include cognitive deficit (impairment in memory, concentration and learning, problems with attention, information processing, eye-hand coordination and reaction time), mood change (anxiety, depression, psychotic symptoms, emotional liability), chronic fatigue, autonomic dysfunction, peripheral neuropathy and extrapyramidal symptoms such as dystonia, resting tremor, bradikynesia, postural instability and rigidity of face muscles [100-110]. In children exposed to CPF during developmental period neurobehavioral impairments were observed [111]. Researches have suggested that mechanisms other than inhibition of AChE might also be involved. These alternative mechanisms may involve other protein targets (such as serine hydrolases, acyl peptide hydrolase) present in the nervous system leading to cognitive damage [95-113].
- Chlorpyrifos neurotoxicity correlate well with neurobehavioral deficits observed consequent to neurodegenerative diseases.
- Certain reports have evaluated the effects of the metabolite chlorpyrifos oxon (CPO) exposed in gestation, lactational and after weaning period on the development of Alzheimer’s Dementia later in life in the mouse model and confirmed that this process may be partially mediated by inflammation, oxidative stress, acetylcholinesterase (AChE) inhibition due to amyloid beta accumulation [114].
- Excessive microglial activation and subsequent neuroinflammation lead to neuronal cell death which are involved in the pathogenesis and progression of several neurodegenerative diseases such as Parkinson’s disease [68].
- Pesticides are composed of a parent product, inert ingredients, and in some cases agonists that enhance the functionality of the parent compound, and all of these ingredients may be degraded to metabolites that also distribute throughout the body. Consequently, chlorpyrifos and its metabolites might contribute to Autism Spectrum
- Disorder by manifesting various neurobehavioral impairments by inhibiting AChE, mitochondrial dysfunction and oxidative stress [115]. Further the other non-cholinergic pathways (GABAergic, glutamatergic, serotonergic and dopaminergic systems) [116].
-
These neurobehavioral impairments manifested neurodegenerative disorders are due to CPF exposure the apoptosis, excitotoxicity, mitochondrial dysfunction, inhibition of AChE and other noncholinergic pathways which lead ultimately to oxidative stress in brain and excessive production of ROS [117]. This increased oxidative stress produced due to developmental exposure of chlorpyrifos in children through their mother’s body is a matter of increased pathologies in children. Therefore, there is need of any natural compound which could be utilized as treatment in pregnant ladies and is an anti- oxidative, cross the placental, lactational and the blood brain barriers thus providing the protective and balanced environment of ROS in brain.
TREATMENT
Various mechanisms whether cholinergic and non- cholinergic are involved in the CPF neurotoxicity effecting the developing brain due to its prenatal or post-natal exposure. But all these mechanisms ultimately lead to enhanced oxidative stress. Therefore, during the pregnancy, the natural antioxidants without the side effects can be investigated as the treatment against CPF intoxication protecting the developing fetus brain. Antioxidants are compounds or agents that impede auto oxidation by interposing the formation of free radicals or by hindering propagation of free radicals such that they scavenge the species that instigate the peroxidations or decompose the free radical species. In case of CPF, they can easily neutralize the increased ROS generated by accepting or donating electron to eliminate the unpaired condition of the radical [118].
- Vitamins C and E are essential nutrients and considered the most important antioxidants obtained through the diet in citrus fruits, almonds, rooted vegetables etc. The antioxidant actions of vitamin E (the tocopherols and tocotrienols) lie in their ability to become incorporated into biological membranes to stablise and protect against lipid peroxidation [119], while the antioxidant properties of vitamin C (ascorbic acid) arise because vitamin C acts as an electron donor, thereby preventing other agents from becoming oxidised and quenching an overproduction of free radicals [120].
- Flavonoids are polyphenolic compounds and have a wide spectrum of biological activity and exhibit various properties as an antioxidant which is contributed by its molecular structure which can scavenge the free radicals such as it is anti-inflammatory in nature [121,122], anti-apoptotic, anti-cholinesterase activity which is one of the treatments for mild to moderate Alzheimer’s and Parkinson’s disease [123,124]. It is considered safe during pregnancy [125]. Due to its variety of defensive roles flavonoids can be the treatment for neuroprotection during developing period against CPF intoxication.
- Alkaloids constitute positive roles in ameliorating pathophysiology of neurobehaviour or neurological disorders by functioning as muscarine and adenosine receptors agonist, antioxidant, anti-amyloid and acetylcholinesterase and butyrylcholinesterase inhibitor, dopaminergic and nicotine agonists and NMDA antagonists [126]. These pathologies are basis of CPF induced intoxication during development period in children leading to neurodegeneration and consecutive behavioral impairments. Berberineisoneofsuchalkaloid extract which is of great therapeutic potential against neurodegenerative diseases and is known to neuroprotective [127]. It has been used in Chinese Ayurvedic medicines during pregnancy. Therefore, it can be explored against CPF intoxication inducing developmental neurotoxicity.
- Curcumin has an outstanding safety profile and a number of pleiotropic actions with potential for neuroprotective efficacy, including anti-inflammatory, antioxidant and anti-protein aggregating activities [128]. Due to its anti-inflammatory properties, it has been evidently found to boost brain power in babies [129]. Further these antioxidants and anti-inflammatory effects notonlybyblockingoxidativestressandneuroinflammation in neurotraumatic and neurodegenerative diseases by restoring cellular homeostasis and rebalancing redox equilibrium [130]. Thus because of its pluripotency, oral safety, long history of use, inexpensive cost, curcumine has potential against CPF induced neurodevelopmental disorders.
Apart from the above discussed antioxidants, certain Ayurvedic products having no side effects during the pregnancy and on the neural growth and development should be investigated and explored against CPF intoxications. There are certain such natural products discussed which can be used as a treatment against neurodegenerative effects in developing brain.
- Kushmanda: In ayurveda, Kushmanda Rasayna is used during pregnancy for nourishing the mother and developing a baby. It is likely to be safe to consume by lactating mothers. There are no adverse effects reported with use of Kushmanda in lactating mothers and breastfeeding babies. It has been reported to improve the memory, intellect and brain health. It is found to be effective in dullness in children due to undeveloped brain and seizures. It is found to be beneficial in autism and other neurological deficits in children, improves focus, attention, memory, speech and concentration. It has been reported to improve neurotransmission and repair of damaged neurons via enhanced regeneration of nerve synapses via changes in areas of brain critical to memory and cognitive abilities. Though its exact mechanism is not yet explored but it can be investigated effective against CPF intoxication.
- Lemon balm (Melissa of????icinalis): The American Pregnancy Association list Lemon Balm as ‘likely safe’ for pregnant women when taken in these usual culinary amounts. Lemon balm is reported to be incredibly nutritive to the nervous system. It is calming and supports the mood while also increasing alertness. It helps to increase the cognitive speed. It is found to protect the aging of brain which indicates its anti-neurodegenerative property reference. Therefore, it can be an effective treatment against degenerative alterations due to CPF.
- Bacopa (Brahmi): Bacopa monnieri contains powerful compounds that may be antioxidant effects as it has been reported to neutralize free radicals and prevent oxidative stress thus inhibiting the neurodegenerative deficits such as Alzheimer’s, Parkinson’s and other diseases. It has myriad of effects due to its antioxidative nature such as anti-inflammatory, enhancing brain function, spacial learning and ability to retain information. Though no such studies have been investigated for its use in pregnancy but it is found to be safe according to Ayurveda in 3-4 weeks after pregnancy and is found safe during lactating period. Thus, it is an effective neuroprotective agent and it can be therapeutically explored for its properties against neurotoxic effects of CPF.
So these plant extracts are the natural sources which can be exploited for potential therapeutic use in pregnancy specifically against CPF induced developmental neurotoxicity and their consecutive disorders and behavioral alterations leading to prolonged long term effect and neurodegeneration.
CONCLUSION
Neurobehavioral impairments and disorders due to the developmental neurotoxicant exposure during the postnatal or prenatal period through mother to offspring is the great matter of concern. Chlorpyrifos is also one of these neurotoxicants which is largest selling organophosphate insecticide in the world and is creating great havoc in generations to generations. It had been evidently proved that it can easily pass the placental barrier and can pass down through lactation, thus entering the body of offspring crossing the blood brain barrier and leading to different pathologies through various mechanism of actions thus disrupting the signaling pathways both cholinergic and non- cholinergic. This leads to degeneracy of neuronal cells and cellular damages, apoptosis, excitotoxic pathways. Thus, creating oxidative stress in brain. This increased oxidative stress is manifested through various neurobehavioral impairments such as cognitive impairment, altered behavioral responses like anxiety, depression, reduced sleep cycle, low IQ level, reduced motor skills, attention deficits and they lead to lifelong prolonged disorders prevalent in childhood stage like Autism, Chronic organophosphate induced neuropsychiatric disorders (COPIND). Further due to degenerative processes going on in brain due to neurotoxicity many degenerative disorders like Parkinson’s and Alzheimer’s disease in later stages of life are found to occur evidently. So, there is great need of treatment against this CPF developmental neurotoxicity which would be safe in pregnancy, organic and without side effects. Therefore, many plant extracts which are antioxidants and are pleiotropic should be explored such as Vitamin C and E, flavonoids, alkaloids, polyphenols are such antioxidants add (gems of Ayurveda which have not yet been explored for their myriad of life saving properties) which can be utilized against CPF during pregnancy period and are neuroprotective thus curbing various neurobehavioral impairments and disorders.
Thus to summarize, there is a need of treatment against CPF intoxication as it is matter of global health concern and cause of many neurobehavioral disorders in children. Keeping in mind the safety of pregnant ladies and fetus or offspring natural antioxidant compounds can be the emerging research area for exploring much of their therapeutic potentials against chlorpyrifos induced neurobehavioral alterations and disorders in neonates and their long term effects.
SIGNIFICANCE OF STUDY
Neurobehavioral and neurodegenerative deficits due to Chlorpyrifos intoxication affecting the developing brain due to prenatal and postnatal exposure through dam is the major reason of concern globally. The mechanisms of neurotoxicity have been investigated so far but still no treatment without any side effects during pregnancy have been accounted which can curb the increased intoxicating effects in developing brain leading to neurobehavioral alterations and other neurodegenerative deficits in longterm. Therefore, natural antioxidants could be the treatment against CPF intoxication during the course of pregnancy. As per the study there are some other natural products I.e. Kushmanda, Lemon balm, Bacopa which can be of therapeutic potential against this CPF induced neurotoxicity leading to different neurobehavioral ailments and diseases.
REFERENCES
- London A, Benhar I, Schwartz M. The retina as a window to the brain-from eye research to CNS disorders. Nat Rev Neurol. 2013; 9: 44-53.
- Giordano G, Costa LG. Morphological Assessment of Neurite Outgrowth in Hippocampal Neuron-Astrocyte Co-Cultures. Current Protocols a Wiley Brand. 2012.
- Bloom N, Reenen JH. Why Do Management Practices Differ acrossFirms and Countries?. J Economic Perspectives. 2010; 24: 203-224.
- Bellinger AM, Reiken S, Carlson C, Mongillo M, Liu X, Rothman L, et al. Hypernitrosylated ryanodine receptor calcium release channels are leaky in dystrophic muscle. Nat Med. 2009; 15: 325-330.
- Gould EA, Higgs S. Impact of climate change and other factors on emerging arbovirus diseases. Trans R Soc Trop Med Hyg. 2009; 103: 109-121.
- Bouchard MF, Chevrier J, Harley KG, Kogut K, Vedar M, Calderon N, et al. Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environ Health Perspect. 2011;119: 1189-1195.
- Engel A, Shewmaker F, Edskes HK, Dyda F, Wickner RB. Amyloid of the Candida albicans Ure2p prion domain is infectious and has an in-register parallel β-sheet structure. Biochemistry. 2011; 50: 5971-5978.
- Rauh V, Arunajadai S, Horton M, Perera F, Hoepner L, Barr DB, et al. Seven-year neurodevelopmental scores and prenatal exposure to chlorpyrifos, a common agricultural pesticide. Environ Health Perspect. 2011; 119: 1196-1201.
- Shelton JF, Geraghty EM, Tancredi DJ, Delwiche LD, Schmidt RJ, Ritz B, et al. Neurodevelopmental disorders and prenatal residential proximity to agricultural pesticides: the CHARGE study. Environ Health Perspect. 2014; 122: 1103-1109.
- Marks LB, Yorke ED, Jackson A, Ten Haken RK, Constine LS, Eisbruch A, et al. Use of normal tissue complication probability models in the clinic. Int J Radiat Oncol Biol Phys. 2010; 76: S10-S9.
- Rauh VA, Garfinkel R, Perera FP, Andrews HF, Hoepner L, Barr DB, et al. Impact of prenatal chlorpyrifos exposure on neurodevelopment in the first 3 years of life among inner-city children. Pediatrics. 2006; 118: e1845-59.
- Eskenazi B, Marks AR, Bradman A, Harley K, Barr DB, Johnson C, et al. Organophosphate pesticide exposure and neurodevelopment in young Mexican-American children. Environ Health Perspect. 2007; 115: 792-798.
- Clearfield MW. The role of crawling and walking experience in infant spatial memory. J Exp Child Psychol. 2004; 89: 214–241.
- Noritz GH, Murphy NA; Neuromotor Screening Expert Panel. Motor delays: early identification and evaluation. Pediatrics. 2013; 131: e2016-27.
- Mansour SA, Mossa AT. Adverse effects of exposure to low doses ofchlorpyrifos in lactating rats. Toxicol Ind Health. 2011; 27: 213-224.
- Lowe MR, Butryn ML, Didie ER, Annunziato RA, Thomas JG, Crerand CE, et al. The Power of Food Scale. A new measure of the psychological influence of the food environment. Appetite. 2009; 53: 114-118.
- Flint-Garcia SA, Thornsberry JM, Buckler ES 4th. Structure of linkage disequilibrium in plants. Annu Rev Plant Biol. 2003; 54: 357-374.
- Neveen F, Michael S, Mayuram K. “The Consumer Online Purchase Decision: A Model of Consideration Set Formation and Buyer Conversion Rate Across Market Leaders and Market Followers”. ICIS 2003 Proceedings. 2003; 24.
- Grandjean P, Landrigan PJ. Developmental neurotoxicity of industrialchemicals. Lancet. 2006; 368: 2167-2178.
- van den Hazel P, Zuurbier M, Babisch W, Bartonova A, Bistrup ML, Bolte G, et al. Today’s epidemics in children: possible relations to environmental pollution and suggested preventive measures. Acta Paediatr Suppl. 2006; 95: 18-25.
- Miodovnik A. Environmental neurotoxicants and developing brain.Mt Sinai J Med. 2011; 78: 58-77.
- Zasler ND, Ameis A, Riddick-Grisham SN. Life care planning aftertraumatic brain injury. Phys Med Rehabil Clin N Am. 2013; 24: 445-465.
- Landrigan PJ. Impacts of the Food Quality Protection Act on Children’sExposures to Pesticides. The Organic Center. 2005.
- Pangrazzi L, Balasco L, Bozzi Y. Oxidative Stress and Immune System Dysfunction in Autism Spectrum Disorders. Int J Mol Sci. 2020; 21: 3293.
- Lobo V, Patil A, Phatak A, Chandra N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn Rev. 2010; 4: 118-126.
- Ricceri L, Venerosi A, Capone F, Cometa MF, Lorenzini P, Fortuna S, et al. Developmental neurotoxicity of organophosphorous pesticides: fetal and neonatal exposure to chlorpyrifos alters sex-specific behaviors at adulthood in mice. Toxicol Sci. 2006; 93: 105-113.
- Ricceri L, Markina N, Valanzano A, Fortuna S, Cometa MF, Meneguz A, et al. Developmental exposure to chlorpyrifos alters reactivity to environmental and social cues in adolescent mice. Toxicol Appl Pharmacol. 2003; 191: 189-201.
- Lu T, Pan Y, Kao SY, Li C, Kohane I, Chan J, et al. Gene regulation andDNA damage in the ageing human brain. Nature. 2004; 429: 883-891.
- Eaton WW, Martins SS, Nestadt G, Bienvenu OJ, Clarke D, AlexandreP. The burden of mental disorders. Epidemiol Rev. 2008; 30: 1-14.
- Raffaele S, Farrer RA, Cano LM, Studholme DJ, MacLean D, Thines M, et al. Genome evolution following host jumps in the Irish potato famine pathogen lineage. Science. 2010; 330: 1540-1543.
- Teirstein AS, Machac J, Almeida O, Lu P, Padilla ML, Iannuzzi MC. Results of 188 whole-body fluorodeoxyglucose positron emission tomography scans in 137 patients with sarcoidosis. Chest. 2007; 132: 1949-53.
- Venerosi A, Ricceri L, Scattoni ML, Calamandrei G. Prenatal chlorpyrifos exposure alters motor behavior and ultrasonic vocalization in CD-1 mouse pups. Environ Health. 2009; 8: 12.
- Aldridge JW, Berridge KC, Rosen AR. Basal ganglia neural mechanismsof natural movement sequences. 2004; 82: 732-739.
- Venerosi A, Cutuli D, Colonnello V, Cardona D, Ricceri L, CalamandreiG. Neonatal exposure to chlorpyrifos affects maternal responses and maternal aggression of female mice in adulthood. Neurotoxicology and Teratology. 2008; 30: 468-474.
- Chen Q, Takada R, Takada S. Loss of Porcupine impairs convergent extension during gastrulation in zebrafish. J Cell Sci. 2012; 125: 2224- 2234.
- Whyatt RM, Barr DB. Measurement of organophosphate metabolites in postpartum meconium as a potential biomarker of prenatal exposure: a validation study. Environ Health Perspect. 2001; 109: 417-420.
- Haviland JB. Mu xa xtak’av: “He Doesn’t Answer”. J Linguistic Anthropol. 2010; 20: 195-213.
- Levin A, Chien-Fu Lin CF, James Chu CS. Unit root tests in panel data: asymptotic and finite-sample properties. J Econometrics. 2002; 108: 1-24.
- Silva A, Cavero S, Begley V, Solé C, Böttcher R, Chávez S, et al. Regulation of transcription elongation in response to osmostress. PLoS Genet. 2017; 13: e1007090.
- Huot RL, Brennan PA, Stowe ZN, Plotsky PM, Walker EF. Negative affect in offspring of depressed mothers is predicted by infant cortisol levels at 6 months and maternal depression during pregnancy, but no postpartum. New York Academy of Sciences. 2004; 234-236.
- Damani K. Parran, Angela Barker, Marion Ehrich, Effects of Thimerosal on NGF Signal Transduction and Cell Death in Neuroblastoma Cells, Toxicological Sciences. 2005; 86: 132-140,
- Costa RM, Cohen D, Nicolelis MA. Differential corticostriatal plasticity during fast and slow motor skill learning in mice. Curr Biol. 2004; 14: 1124-1134.
- Meerts I, Lilienthal H, Hoving S, van den Berg JH, Weijers BM, Åke Koeman, et al. Developmental Exposure to 4-hydroxy-2,3,3′,4′,5- pentachlorobiphenyl (4-OH-CB107): Long-Term Effects on Brain Development, Behavior, and Brain Stem Auditory Evoked Potentials in Rats. Toxicological Sciences. 2004; 82: 207-218.
- Vijverberg HP, van den Berg M. Re: Viberg H. Neurobehavioral derangements in adult mice receiving decabrominated diphenyl ether (PBDE 209) during a defined period of neonatal brain development. Toxicol Sci. 2003; 76: 112-120.
- Goel N, Kim H, Lao RP. An olfactory stimulus modifies nighttime sleep in young men and women. Chronobiol Int. 2005; 22: 889-904.
- López MV, Garcia A, Rodriguez L. Sustainable Development and Corporate Performance: A Study Based on the Dow Jones Sustainability Index. J Business Ethics. 2007; 75: 285-300.
- Aly AH, Debbab A, Proksch P. Fungal endophytes: unique plant inhabitants with great promises. Appl Microbiol Biotechnol. 2011; 90: 1829-1845.
- Lee L, Rodriguez J, Tsukiyama T. Chromatin remodeling factors Isw2 and Ino80 regulate checkpoint activity and chromatin structure in S phase. Genetics. 2015; 199: 1077-91.
- Bouchard MF, Bellinger DC, Wright RO, Weisskopf MG. Attention- deficit/hyperactivity disorder and urinary metabolites of organophosphate pesticides. Pediatrics. 2010; 125: e1270-1277.
- Slotkin TA. Cholinergic systems in brain development and disruption by neurotoxicants: nicotine, environmental tobacco smoke, organophosphates. Toxicol Appl Pharmacol. 2004; 198: 132-1351.
- Saunders M, Lewis P, Thornhill T. Research Methods for Business Students. 2012.
- Carr AJ, Hopkins WG, Gore CJ. Effects of acute alkalosis and acidosison performance: a meta-analysis. Sports Med. 2011; 41: 801-814.
- Carr AJ, Gibson B, Robinson PG. Measuring quality of life: Is quality of life determined by expectations or experience? BMJ. 2001; 322: 1240-1243.
- van Dam T, Larson K, Wahr J, Gross S, Francis O. Using GPS and gravity to infer ice mass changes in Greenland. Eos. Transactions American Geophysical Union. 2000. 81.
- Gómez-Giménez C, Ballestero D, Ferrer N, Rubio B, Izquierdo MT. Influence of crystal/particle size and gold content of a structured Au/C based sorbent on mercury capture. J Physics Chemistry of Solids. 2017; 110: 173-179.
- Johnson SA, Cubberley G, Bentley DL. Cotranscriptional recruitment of the mRNA export factor Yra1 by direct interaction with the 3’ end processing factor Pcf11. Mol Cell. 2009; 33: 215-226.
- Sánchez-Amate MC, Flores P, Sánchez-Santed F. Effects of chlorpyrifos in the plus-maze model of anxiety. Behav Pharmacol. 2001; 12: 285- 292.
- Levine RV, Norenzayan, A, Philbrick, K. Cross-Cultural Differences inHelping Strangers. J Cross-Cultural Psychol. 2001; 32: 543-560.
- Conto CA, Akseer S, Dreesen T, Kamei A, Mizunoya S, Rigole A. Potential effects of COVID-19 school closures on foundational skills and Country responses for mitigating learning loss. Int J Educational Dev. 2021; 87: 102434.
- Laviola G, Macrì S, Morley-Fletcher S, Adriani W. Risk-taking behavior in adolescent mice: psychobiological determinants and early epigenetic influence. Neurosci Biobehav Rev. 2003; 27: 19-31.
- Feinberg I, Campbell IG. Sleep EEG changes during adolescence: an index of a fundamental brain reorganization. Brain Cogn. 2010; 72: 56-65.
- Grabovska S, Salyha Y. ADHD-like behaviour in the offspring of female rats exposed to low chlorpyrifos doses before pregnancy. Arh Hig Rada Toksikol. 2015; 66: 121-127.
- Perera TD, Luber B, Nobler MS, Prudic J, Anderson C, Sackeim HA. Seizure expression during electroconvulsive therapy: relationships with clinical outcome and cognitive side effects. Neuropsychopharmacology. 2004; 29: 813-825.
- Whyatt RM, Rauh V, Barr DB, Camann DE, Andrews HF, Garfinkel R, et al. Prenatal insecticide exposures and birth weight and length among an urban minority cohort. Environ Health Perspect. 2004; 112: 1125- 1132.
- Jurewicz J, Radwan M, Wielgomas B, Dziewirska E, Karwacka A, Klimowska A, et al Human Semen Quality, Sperm DNA Damage, and the Level of Reproductive Hormones in Relation to Urinary Concentrations of Parabens. J Occup Environ Med. 2017; 59: 1034- 1040.
- Levenson JS, Willis GM, Prescott DS. Adverse Childhood Experiences in the Lives of Male Sex Offenders: Implications for Trauma-Informed Care. Sex Abuse. 2016; 28: 340-359.
- Sánchez-Santed F, Colomina MT, Herrero Hernández E. Organophosphate pesticide exposure and neurodegeneration. Cortex. 2016; 74: 417-426.
- Koo TK, Li MY. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J Chiropr Med. 2016; 15: 155-63.
- Salazar MR, Carbajal HA, Espeche WG, Dulbecco CA, Aizpurúa M, Marillet AG, et al. Relationships among insulin resistance, obesity, diagnosis of the metabolic syndrome and cardio-metabolic risk. Diab Vasc Dis Res. 2011; 8: 109-116.
- Amani L. Mycotoxines et champignons mycotoxinogènes dans les grains de sorgho commercialisé en Tunisie, Incidence et profils écophysiologiques. Thèse de doctorat, Institut Supérieur de Biotechnologie de Monastir, Monastir. 2016; 224.
- Casida JE, Nomura DK, Vose SC, Fujioka K. Organophosphate- sensitive lipases modulate brain lysophospholipids, ether lipids and endocannabinoids. Chemico-Biological Interactions. 2008; 175: 355-364.
- Lallement R, Capitanio L, Ruiz-Dern L, Danielski C, Babusiaux C, Vergely J, et al. Three-dimensional maps of interstellar dust in the Local Arm: using Gaia, 2MASS, and APOGEE-DR14. 2018.
- Slotkin TA, Miller DB, Fumagalli F, McCook EC, Zhang J, Bissette G, et al. Modeling geriatric depression in animals: biochemical and behavioral effects of olfactory bulbectomy in young versus aged rats.1999.
- Kovacic P. Mechanism of organophosphates (nerve gases and pesticides) and antidotes: electron transfer and oxidative stress. Curr Med Chem. 2003; 10: 2705-2709.
- Saulsbury FT. Successful Treatment of Prolonged Henoch-Schönlein Purpura with Colchicine. Clinical Pediatrics. 2009; 48: 866-868.
- Hedayati SS, Minhajuddin AT, Afshar M, Toto RD, Trivedi MH, Rush AJ. Association between major depressive episodes in patients with chronic kidney disease and initiation of dialysis, hospitalization, or death. JAMA. 2010; 303: 1946-1953.
- Schoups A, Vogels R, Qian N, Orban G. Practising orientation identification improves orientation coding in V1 neurons. Nature. 2001; 412: 549-553.
- Crumpton TL, Seidler FJ, Slotkin TA. Generation of reactive oxygen species by xanthine derivatives in MDA-MB-231 human breast cancer cells. 2001; 66: 143-146.
- Daniel Ravid, Jerod White, David L Tomczak, Ahleah F. Miles, TaraS. Behrend. A Meta-Analysis of the Effects of Electronic Performance Monitoring on Work Outcomes. Personnel Psychol. 2022.
- Landrigan PL, Fuller R. Pollution, health and development: the need for a new paradigm. Reviews on Environmental Health. 2016.
- Slotkin TA, Epps TA, Stenger ML, Sawyer KJ, Seidler FJ. Cholinergic receptors in heart and brainstem of rats exposed to nicotine during development: implications for hypoxia tolerance and perinatal mortality. Brain Res Dev Brain Res. 1999; 113: 1-12.
- Buchan J, O’May F. Determining skill mix: practical guidelines for managers and healthcare professionals, Human Resources Development Journal. 2000; 4: 111-118.
- Weiss IC, Pryce CR, Jongen-Rêlo AL, Nanz-Bahr NI, Feldon J. Effect of social isolation on stress-related behavioural and neuroendocrine state in the rat. Behav Brain Res. 2004; 152: 279-295.
- Chauhan S, Kumar S, Jain A, Ponpuak M, Mudd MH, Kimura T, et al. TRIMs and Galectins Globally Cooperate and TRIM16 and Galectin-3 Co-direct Autophagy in Endomembrane Damage Homeostasis. Dev Cell. 2016; 39: 13-27.
- Gupta RA, Shah N, Wang KC, Kim J, Horlings HM, Wong DJ, et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature. 2010; 464:1071-1076.
- Slotkin TA, Seidler FJ. Comparative developmental neurotoxicity of organophosphates in vivo: transcriptional responses of pathways for brain cell development, cell signaling, cytotoxicity and neurotransmitter systems. Brain Res Bull. 2007; 72: 232-274.
- Slotkin RK, Martienssen R. Transposable elements and the epigeneticregulation of the genome. Nat Rev Genet. 2007; 8: 272-285.
- Gültekin Gülümser, Baran Gülen. A Study of the Self-Concepts of 9–14 Year-Old Children with Acute and Chronic Diseases. Social Behavior and Personality: an international J. 2007; 35: 329-338.
- Slotkin JR, Mislow JM, Day AL, Proctor MR. Pediatric disk disease.Neurosurg Clin N Am. 2007; 18: 659-667.
- Ida C. Zündorf, Hans-Otto Karnath, Jörg Lewald. Male advantage insound localization at cocktail parties. Cortex. 2011; 47: 741-749.
- Slotkin TA, MacKillop EA, Ryde IT, Seidler FJ. Ameliorating the developmental neurotoxicity of chlorpyrifos: a mechanisms-based approach in PC12 cells. Environ Health Perspect. 2007; 115: 1306-1313.
- Bebe FN, Panemangalore M. Exposure to low doses of endosulfan and chlorpyrifos modifies endogenous antioxidants in tissues of rats. J Environ Sci Health B. 2003; 38: 349-363.
- Mehta A, Clarke JT, Giugliani R, Elliott P, Linhart A, Beck M, et al. Natural course of Fabry disease: changing pattern of causes of death in FOS - Fabry Outcome Survey. J Med Genet. 2009; 46: 548-552.
- Meister D. Twenty-First-Century Challenges to Ergonomics. Ergonomics in Design: The Quarterly of Human Factors Applications. 1998; 6: 33-34.
- Ray DE, Richards PG. The potential for toxic effects of chronic, low-dose exposure to organophosphates. Toxicol Lett. 2001; 120: 343-351.
- Kharb S, Singh V, Ghalaut PS, Sharma A, Singh GP. Glutathione levelsin health and sickness. Indian J Med Sci. 2000; 54: 52-54.
- Levy D, Anderson KM, Savage DD, Kannel WB, Christiansen JC, Castelli WP. Echocardiographically detected left ventricular hypertrophy: prevalence and risk factors. The Framingham Heart Study. Ann Intern Med. 1988; 108: 7-13.
- da Silva Meirelles L, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006; 119: 2204-2213.
- Tan SC, Yiap BC. DNA, RNA, and protein extraction: the past and the present. J Biomed Biotechnol. 2009; 2009: 574398.
- Milka Brdar-Jokanovi?, Milan Ugrinovi?, Dejan Cviki?, Nenad Pavlovi?, Jasmina Zdravkovi?, Sla?an Adži?, et al. Onion Yield and Yield Contributing Characters as Affected by Organic Fertilizers. Field Veg. Crop Res. 2011; 48: 341-346.
- Davies R, Ahmed G, Freer T. Psychiatric aspects of chronic exposure to organophosphates: diagnosis and management. Advances in Psychiatric Treatment. 2000; 6: 356-361.
- Davies SC, Cronin E, Gill M, Greengross P, Hickman M, NormandC. Screening for sickle cell disease and thalassaemia: a systematic review with supplementary research. Health Technol Assess. 2000a; 4: 1 99.
- Karoussis IK, Salvi GE, Heitz-Mayfield LJ, Brägger U, Hämmerle CH, Lang NP. Long-term implant prognosis in patients with and without a history of chronic periodontitis: a 10-year prospective cohort study of the ITI Dental Implant System. Clin Oral Implants Res. 2003; 14: 329-339.
- Kamel F, Hoppin JA. Association of pesticide exposure with neurologic dysfunction and disease. Environ Health Perspect. 2004; 112: 950-958.
- London K, Bruck M, Ceci SJ, Shuman DW. Disclosure of Child Sexual Abuse: What Does the Research Tell Us About the Ways That Children Tell? Psychology, Public Policy, and Law. 2005; 11: 194-226.
- Roldán-Tapia L, Parrón T, Sánchez-Santed F. Neuropsychological effects of long-term exposure to organophosphate pesticides. Neurotoxicol Teratol. 2005; 27: 259-266.
- Ross JM, Stewart JB, Hagström E, Brené S, Mourier A, Coppotelli G, et al. Germline mitochondrial DNA mutations aggravate ageing and can impair brain development. Nature. 2013; 501: 412-415.
- Venkategowda PM, Mahendrakar K, Rao SM, Mutkule DP, Shirodkar CG, Yogesh H. Laryngeal air column width ratio in predicting post extubation stridor. Indian J Crit Care Med. 2015; 19: 170-173.
- Ishfaq Ahmed, Ihsan Mabood Qazi, Suraiya Jamal. Assessment of proximate compositions and functional properties of blends of broken rice and wheat flours. Sarhad J Agriculture. 2016; 32: 142- 150.
- Taghavian F, Vaezi Gh, Abdollahi M, Malekirad AA. Comparative toxicological study between exposed and non-exposed farmers to organophosphorus pesticides. Cell J. 2016; 18: 89-96.
- Ruckart PZ, Ettinger AS, Hanna-Attisha M, Jones N, Davis SI, Breysse PN. The Flint Water Crisis: A Coordinated Public Health Emergency Response and Recovery Initiative. J Public Health Manag Pract. 2019; 25: S84-S90.
- Joan Esteban, Debraj Ray. Conflict and Distribution. J Economic Theory. 1999; 87: 379-415.
- Floria Pancetti, Cristina Olmos, Alexies Dagnino-Subiabre, Carlos Rozas, Bernardo Morales. Noncholinesterase Effects Induced by Organophosphate Pesticides and their Relationship to Cognitive Processes: Implication for the Action of Acylpeptide Hydrolase. J Toxicol Environmental Health, Part B: Critical Rev. 2007; 10: 623- 630.
- Wong-Yu IS, Mak MK. Multi-dimensional balance training programme improves balance and gait performance in people with Parkinson’s disease: A pragmatic randomized controlled trial with 12-month follow-up. Parkinsonism Relat Disord. 2015; 21: 615-621.
- Anthony M. Shelton, Steven E. Naranjo, Jörg Romeis, Richard L. Hellmich. Errors in Logic and Statistics Plague a Meta-Analysis (Response to Andow and Lövei 2012). Source: Environmental Entomology. 2012; 41: 1047-1049.
- Quek J, Pua YH, Clark RA, Bryant AL. Effects of thoracic kyphosis and forward head posture on cervical range of motion in older adults. Man Ther. 2013; 18: 65-71.
- Oruç B. Edge Detection and Depth Estimation Using a Tilt Angle Map from Gravity Gradient Data of the Kozakl?-Central Anatolia Region, Turkey. Pure Appl. Geophys. 2011; 168: 1769-1780.
- Yu PB, Deng DY, Lai CS, Hong CC, Cuny GD, Bouxsein ML, et al. BMP type I receptor inhibition reduces heterotopic [corrected] ossification. Nat Med. 2008; 14: 1363-1369.
- Packer M, Coats AJ, Fowler MB, Katus HA, Krum H, Mohacsi P, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001; 344: 1651-1658.
- Miller RG, Mitchell JD, Moore DH. Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND). Cochrane Database Syst Rev. 2012; 2012: CD001447.
- Morales-Cano D, Menendez C, Moreno E, Moral-Sanz J, Barreira B, Galindo P, et al. The Flavonoid Quercetin Reverses Pulmonary Hypertension in Rats. PLoS ONE. 2014; 9: e114492.
- Romero V, Akpinar H, Assimos DG. Kidney stones: a global picture of prevalence, incidence, and associated risk factors. Rev Urol. 2010; 12: e86-e96.
- Caton SJ, Rana OF, Batchelor BG. Distributed image processing over an adaptive Campus Grid. Concurrency and Computation: Practice and Experience. 2009; 21: 321-336.
- Adrover JM, Nicolás-Ávila JA, Hidalgo A. Aging: A Temporal Dimension for Neutrophils. Trends Immunol. 2016; 37: 334-345.
- Emerenciano M, Gaxiola G, Cuzon G. Biofloc technology (BFT): a review for aquaculture application and animal food industry. Biomass now-cultivation and utilization. 2013; 12: 301-328.
- Syed Talib Hussain, Shen Lei, Tayyaba Akram, Muhammad Jamal Haider, Syed Hadi Hussain, Muhammad Ali. Kurt Lewin’s change model: A critical review of the role of leadership and employee involvement in organizational change. J Innovation Knowledge. 2018; 3: 123-127.
- Neag MA, Mocan A, Echeverría J, Pop RM, Bocsan CI, Cri?an G, et al. Berberine: Botanical Occurrence, Traditional Uses, Extraction Methods, and Relevance in Cardiovascular, Metabolic, Hepatic, and Renal Disorders. Front Pharmacol. 2018; 9: 557.
- Cole TJ, Flegal KM, Nicholls D, Jackson AA. Body mass index cut offs to define thinness in children and adolescents: international survey. BMJ. 2007; 335: 194.
- Sonja Kay, Carlo Rega, Gerardo Moreno, Michael den Herder, JoãoH.N. Palma, Robert Borek, et al. Agroforestry creates carbon sinks whilst enhancing the environment in agricultural landscapes in Europe. Land Use Policy. 2019; 83: 581-593.
- Farooqui ZA, Chauhan A. Neuroendocrine Tumors in Pediatrics. Glob Pediatr Health. 2019; 6: 2333794X19862712.