Cord Blood T Lymphocytes Homeostasis and Prenatal Inflammation: A Proposed Model of Poor Clinical Outcomes
- 1. Department of Pediatrics, Division of Neonatology, Morsani College of Medicine, University of South Florida, USA
Citation
Peña TQ, Luciano AA (2014) Cord Blood T Lymphocytes Homeostasis and Prenatal Inflammation: A Proposed Model of Poor Clinical Outcomes. Ann Pediatr Child Health 2(1): 1006.
Editorial
Nearly 12% of births in the U.S. are preterm, and prematurity is the leading cause of neonatal morbidity and mortality. While advances in medicine have increased the survival of premature infants, the rate of preterm birth in the U.S. has not decreased [1]. As survival has increased, the rates of neonatal systemic complications such as bronchopulmonary dysplasia (BPD), necrotizing enterocolitis (NEC) and sepsis have dramatically increased [2]. Understanding the causes of preterm labor and the pathophysiology of its systemic complications continue to be a challenge. Increasing evidence is demonstrating an association between neonatal inflammation, preterm labor, systemic complications and development of the neonatal immune system [3-8]. Recent findings have demonstrated fetal immunological consequences of intrauterine exposure to inflammation and its possible association with poor clinical outcomes [9-13]. Is it possible to identify preterm infants with a higher risk of developing systemic complications based on their cord blood immune signature? Our group has been asking this question and may have some promising answers.
The relationship between T lymphocyte development and intrauterine inflammation is still poorly understood. Adkins et al. used a murine model to demonstrate that T lymphocyte function can be modulated under certain conditions, and that the neonatal period is a unique developmental stage in which immune responses are highly plastic and dependent on the conditions of antigen exposure [14]. Previously, the function of the neonatal adaptive immune cells was considered to be immature. However, our understanding of the “immaturity” of neonatal adaptive immune cells has evolved, such that we now consider them to be competent and able to mount adult-level T cell responses under certain conditions [15-17]. The factors or conditions that affect T lymphocyte modulation in preterm infants are not clearly understood. Does inflammation play an important role?
Romero et al. were the first to describe a state of immune modulation secondary to fetal exposure to inflammation [18]. They described a fetal inflammatory response syndrome (FIRS) characterized by increased cord blood (CB) levels of IL-6 levels and fetal c-reactive protein (CRP), and activation of the innate immune system in preterm infants exposed to placental inflammation (chorioamnionitis). Although several investigators have assessed the role of innate immune activation in preterm FIRS,much less is known about the role of T lymphocyte activation in FIRS [19,20]. T lymphocyte activation is characterized by the presence of activation markers CD69, CD25, and HLA-DR on the surface of T lymphocytes. Furthermore, T lymphocyte activation may also lead to the development of memory T lymphocytes (CD45RO+ ). To clarify the role of fetal lymphocyte activation in preterm infants, our group assessed neonatal T lymphocyte activation and memory phenotype in CB by flow cytometric analysis. We described an activated T lymphocyte phenotype expressing elevated levels of CD25, HLA-DR and CD69 in CB of preterm infants and in CB of infants from pregnancies affected by chorioamnionitis [9].
Because prenatal inflammation is crucial for the development of FIRS, we investigated the possible role of chorioamnionitis in T lymphocyte activation. CB T lymphocytes from preterm infants exposed to both clinical and histological chorioamnionitis had an activated phenotype and expressed a marker of increased maturation (CD45RO+ ) [4]. Although the determinants of immune activation in the premature infant are not well understood, in some cases, it may be related to in utero exposure to inflammation from chorioamnionitis. Microbial products (endotoxin and bacterial DNA) in the amniotic fluid have been detected in the presence of chorioamnionitis and have been related to a systemic inflammatory response in the fetus [21,22]. To further investigate the interaction between inflammation and T lymphocyte activation/maturation, we performed in vitro studies involving Toll-like receptor (TLR) ligands. TLRs are transmembrane pattern recognition receptors that recognize bacterial products, and they are involved in the production of cytokines, chemokines and antimicrobial peptides. TLRs activate cells of the innate immune system and activate pathways that could be either pro- or anti-inflammatory [23]. In our in vitro studies, we used the ligands for TLR-4 (lipopolysaccharide [LPS]), TLR-5 (Flagellin), TLR2-6 (MALP-2) with pro-inflammatory properties and TLR-9 (CpG) with anti-inflammatory properties. Interestingly, in vitro exposure to TLR ligands induced expression of CD45RO in CB T lymphocytes. This effect was only present in CB T lymphocytes—adult naïve T lymphocytes did not increase expression of CD45RO after exposure to these ligands. These in vitro experiments demonstrated an important role for these TLRs in T lymphocyte development [10].
Other investigators have demonstrated that these TLR ligands play an important role in neonatal immunity [24,25]. LPS has been widely studied and has been used in many animal models to mimic a state of chorioamnionitis and preterm labor [26,27]. LPS, an endotoxin produced by Gram negative bacteria, has been implicated as a factor that triggers preterm labor in humans. In plasma, LPS binds LPS-binding protein (LBP) forming a complex that stimulates macrophages by binding CD14; CD14 can then be shed from monocytes and macrophages into the plasma (soluble CD14 [sCD14]). Limited information is available about the presence of LPS in fetal circulation or preterm CB after preterm labor. Our group recently demonstrated that LPS and sCD14 were significantly higher in CB of preterm versus term infants. In addition, there was a positive correlation between CRP and LPS levels, indicating an association between inflammation and LPS in utero. Histological chorioamnionitis (HC) also played a role since higher LPS levels were found in placentas from pregnancies complicated by preterm labor [28].
Is the migration of these T lymphocytes also affected? The traffic of T lymphocytes from blood to tissues occurs via a series of interactions between lymphocytes and endothelial cells. T lymphocytes typically traffic through secondary lymphoid organs in search of peptides bound to major histocompatibility complex molecules expressed by antigen-presenting cells. After exposure to antigen triggers the T lymphocyte, they gain distinct homing characteristics. Homing receptors that are important for T-cell homeostasis include α4 β7 and CCR4. The homing molecule α4 β7 promotes T lymphocyte entry into intestinal sites, and CCR4 supports T lymphocyte chemotaxis and entry into nongastrointestinal sites through interaction with its ligand induced by inflammation [29]. Our group demonstrated a distinct homing receptor pattern in preterm infant CB T lymphocytes. The high proportion of α4 + β7 + cells in CB of term infants may reflect the critical role of intestinal homing—and perhaps the importance of exposure to microbial elements in the gut—in neonatal T-cell development. We showed that in vitro exposure of CB cells to microbial TLR ligands alters the expression of these homing receptors: expression of α4β7 is decreased and expression CCR4 is increased. Our finding of fewer α4 + β7 + T cells in preterm infant CB may reflect an earlier (premature) maturation of these cells in response to premature exposure to microbial elements. A consequence of this exposure may be increased migration of these cells to nongastrointestinal sites (e.g., lungs, skin, or brain) that may underlie the systemic inflammatory organ dysfunction often seen in premature infants.
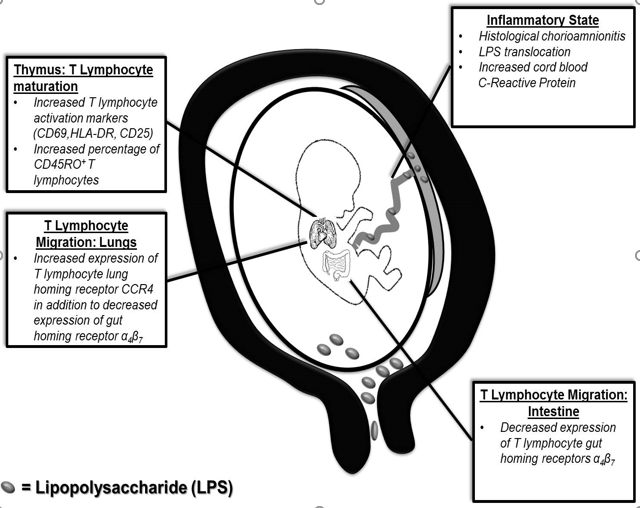
In our proposed model (Figure 1),
Figure 1 Proposed mechanism of increased T lymphocyte activation and maturation in utero. Lipopolysaccharide (LPS) crosses to the fetal side either by translocation of the placenta or via ascending infection. An inflammatory state is present in utero demonstrated by the presence of chorioamnionitis and elevation of CRP. Change in T lymphocytes homeostasis seen (activation and maturation). These changes could be associated to clinical outcomes.
an inflammatory state during preterm labor (characterized by HC, LPS translocation and increased CRP) leads to changes in T lymphocyte phenotype (activation and maturation) and migration. Understanding the neonatal immune system will continue to challenge clinicians. As mentioned above, many factors play a role in neonatal mortality and morbidities due to infection/inflammation. Promising preliminary data from our group demonstrated that infants who developed NEC had a lower percentage of T lymphocytes expressing α4 β7 . While NEC is clearly multifactorial, mucosal protection could be compromised if T lymphocyte migration to the gut is affected in utero. Alterations in mucosal immunity could predispose premature infants to uncontrolled inflammation and infection in the gut, thus predisposing to them to the development of NEC. In addition, premature infants that developed late onset sepsis demonstrated an increased proportion of T lymphocytes expressing CD45RO and PD-1. CD45RO+ cells have been exposed to antigens and should have a more rapid immune response as they are re-introduced to antigen. Could these cells be in an “immune tolerance” state? PD-1 and its ligands negatively regulate immune responses decreasing the activity of these T lymphocytes. These could explained the increasing risk for late onset sepsis on these infants. As we expand our understanding of this exciting field of immunology, we will improve our approach to investigations of infants exposed to inflammation in utero. These new and promising CB immune findings could help the clinicians to identify infants at higher risk of developing systemic complications and to improve their outcomes.
Acknowledgements
The authors would like to thank Doris Wiener and Jane Carver for the editing help; and Eleanor Molloy and Larry Dishaw of the Neonatal Immunity and Clinical Outcomes International Research Group for their advice.