The Biological Role of Fetuin-B in Female Reproduction
- 1. Biointerface Laboratory, RWTH Aachen University, Germany
Abstract
Fetuin-B is a liver-derived serum protein, which is essential for female fertility. Mammalian oocytes are surrounded by a layer of extracellular matrix called zona pellucida (ZP). Fertilization triggers the proteolytic cleavage of ZP glycoproteins, rendering the ZP in a hardened state. ZP hardening prevents further sperm attachment and penetration, and protects the pre-implantation embryo. The protease ovastacin mediates definitive ZP hardening. Fetuin-B is a potent inhibitor of ovastacin preventing premature ZP hardening. Thus Fetuin-B keeps the ZP penetrable for sperm until fertilization. Fetuin-B deficient female mice are infertile, because their oocytes have a prematurely hardened ZP.
Keywords
Fetuin-B; Ovastacin; Zona pellucida; Infertility; In vitro fertilization.
Citation
Dietzel E, Floehr J, Jahnen-Dechent W (2016) The Biological Role of Fetuin-B in Female Reproduction. Ann Reprod Med Treat 1(1): 1003.
ABBREVIATIONS
ICSI: Intracytoplasmatic Sperm Injection; IVF: In vitro Fertilization; ZP: Zona Pellucida; ZP2: Zona Pellucida Protein 2
INTRODUCTION
Fetuin-B, a member of the Cystatin superfamily
The Fetuin proteins Fetuin-A (genetic symbol AHSG/FETUA) and Fetuin–B (FETUB), together with histidine-rich glycoprotein (HRG) and kininogen (KNG) are type 3 members of the cystatin (inhibitors of cysteine proteases) super family of proteins [1,2]. The type 3 family members are secreted, disulfide-bonded, multi-domain glycoproteins with more than one cystatin-like domain; AHSG, FETUB and HRG each contain two tandem cystatin domains, whereas KNG contains three cystatin domains. KNG displays cystatin-activity whereas the other type 3 family members appear not to be functional cystatins [3].The Fetuin-A and Fetuin-B proteins have a size of about 50-60 kDa and are predominantly expressed in the liver [4,5]. Both are secreted into the blood and thus reach all soft tissues. Fetuin-A was first isolated 1944 and was named “Fetuin” since it was most abundant in bovine fetal blood [6]. In the 1990ies several studies described a role of Fetuin in fertilization. We generated a Fetuin knockout mouse, which was completely fertile [7-10]. Three years after the generation of this Fetuin mouse, a second Fetuin protein, termed Fetuin-B was described. From the non, the original “Fetuin” was renamed Fetuin-A [5]. Previous to that date and mostly still today, commercial “Fetuin” preparations contain both, Fetuin-A and Fetuin-B. We recently showed that Fetuin-B, but not Fetuin-A is essential for fertilization [10,11].
Fetuin-B Gene Regulation
Mice have Fetuin-B serum concentrations of 156 ± 3 µg/ml (~3000 nM at 50 kDamol weight), while human serum contains 5 ± 1 µg/ml (100 nM) [4]. Recombinant mouse Fetuin-B inhibits ovastacin with an IC50 of 75 nM, and we assume a similar IC50 for human Fetuin-B and ovastacin [11]. Because the physiological concentration of Fetuin-B is closer to the IC50 in humans than in mice, the prevention of premature ZP hardening discovered in mice may be even more relevant to human reproductive biology.
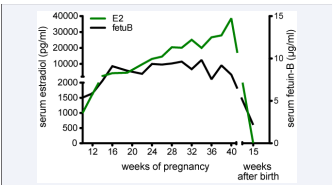
We showed that the serum Fetuin-B concentration was higher in female mice than in males suggesting Fetuin-B regulation by sex steroids [4]. Recently, we reported similar serum Fetuin-B concentrations in men and women of 3.6 ± 1.2 µg/ml and 3.6 ± 0.7 µg/ml, respectively [12]. Hormonal contraception with synthetic estrogen ethinyl estradiol is more than doubled serum Fetuin-B suggesting an estrogen-mediated regulation of hepatic Fetuin-B expression. Our earlier study did not control hormonal contraception in the females, which may have inadvertently caused the discrepancy in serum Fetuin-B baseline levels in these two studies. Hormonal changes during female menstrual cycles did not lead to Fetuin-B variations in women [12]. Keeping in mind that an oocyte needs more than 5 cycles to mature [13,14], a constant serum Fetuin-B concentration during the menstrual cycle might be necessary to inhibit spurious release of ovastacin during oocyte maturation and thus allow fertilization of the next ovulated oocyte. Serum Fetuin-B was associated with fertilization rate in human IVF patients. In this pilot study serum Fetuin-B was a better predictor of IVF rate than high serum estradiol [12]. IVF cycles with successful fertilization showed increasing Fetuin-B serum concentrations upon controlled ovarian stimulation, while it was constant during IVF cycles when fertilization failed. Fetuin-A serum levels were elevated in IVF patients as well, but were not associated with the reproductive potential [15]. Thus serum Fetuin-B, but not Fetuin-A, may be used as a predictive marker for the fertilization rate in IVF. Fetuin-B levels attained during IVF stimulation may help to decide whether oocytes should be fertilized by IVF or by intra cytoplasmatic sperm injection (ICSI) to overcome the ZP as a barrier. Physiological high serum estradiol attained during pregnancy was associated with high serum Fetuin-B as well Figure (1).
Figure 1 Association of serum Fetuin-B and endogenous estradiol during pregnancy. Serum estradiol (E2, green) and serum Fetuin-B (Fetuin -B, black) of one woman is depicted during and after the pregnancy.
An estrogen receptor-binding site is present between the genetic loci of Fetuin-A and Fetuin-B on human chromosome 3 [16], about 8000 bp up stream of the Fetuin-B gene. Thus a direct influence on the Fetuin-B synthesis is unlikely. Accordingly, Fetuin-B expression was affected by exceedingly high endogenous estradiol only [12]. In summary, estrogen may not lead to a direct stimulation of Fetuin-B expression. Instead signaling cross-talk and the activation of transcription factors other than the estrogen receptor may stimulate Fetuin-B expression. Studies in cultured human hepatoma cells indeed demonstrated that Fetuin-B expression is induced by the farnesoid X receptor [17]. Gene reporter studies involving estrogen and farnesoid X receptor agonists will tell if serum Fetuin-B can be elevated, and if this rescues female infertility.
Role of Fetuin-B in female fertility
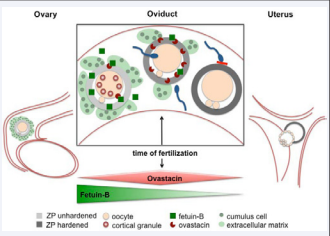
Oocytes, the female gametes, start developing during fetal life. Developing oocytes are surrounded by nourishing follicle cells, collectively forming the follicle. The zona pellucida (ZP) is an extracellular matrix of oocytes. The ZP forms in secondary follicles, and consists of four glycoproteins, zona pellucida protein ZP1-4 [18]in humans. In mice the ZP only consists of ZP1-3, and ZP4 is a pseudo gene. With ongoing follicle development, the antrum is formed, a fluid filled follicular cavity. The follicular fluid comprises a mixture of serum-derived components and follicle cell derived factors. At the time of ovulation, the follicle wall ruptures and the oocyte is discharged into the oviduct long with surrounding cumulus cells and follicular fluid. Pre-fertilization oocyte development usually proceeds through meiosis and arrests at metaphase 2. Already during this period, a small number of cortical granules, which are located beneath the oolemma, can be exocytosed [19,20]. Cortical granules contain, among others, the metal loprotease ovastacin [21-24]. Ovastacin cleaves ZP2 protein, which in its native state mediates gamete recognition in mice and humans [25]. Cleavage of ZP2 by ovastacin leads to structural modifications in the ZP, resulting in a block to sperm binding [22]. This process is called ZP hardening. Blood-derived hepatic Fetuin-B plays a critical role Figure (2) in regulating ZP hardening, because it is a highly specific inhibitor of the ovastacin protease. Liver is the main source of circulating Fetuin-B protein, yet small amounts of Fetuin-B mRNA may also be produced in the ovary by hitherto unspecified cell types [4]. Rat cumulus cells were shown to express Fetuin-A mRNA, but Fetuin-Bwas notanalyzed [26]. In any case, ovarian Fetuin-B is dispensible for fertility, since Fetuin-B knockout ovaries produced healthy offspring when transplanted into ovariectomized wild type females producing only hepatic Fetuin-B [11]. Fetuin-B protein readily passes the blood-follicle barrier and thus is present in follicular fluid [12]. It is likely that Fetuin-B is stored in the extracellular matrix of the cumulus cells, as was shown for other serum-derived factors [27]. ZP-associated Fetuin-B inhibits any ovastacin protease activity released before or around ovulation, and thus prevents premature ZP hardening [11]. Thus Fetuin-B keeps the oocyte penetrable for fertilization. Fetuin-B deficiency in mice leads to the complete and irreversible female infertility. The next relative to Fetuin-B, Fetuin-A, shares26 % amino acid sequence similarity, yet is dispensible for fertilization, at least in mice [10,28]. Mouse and human Fetuin-B share61 % sequence identity [4], and thus Fetuin-B may play a role in human fertility as well, namely that Fetuin-B deficiency could be associated with female infertility. Ovulation discharges the follicular fluid and the cumulus-oocyte complex into the oviduct, which mediates further transport into the uterus Figure (2).
Figure 2 Fetuin-B is essential for fertilization. The oocyte develops in the ovary. After ovulation cumulus cells and follicular fluid-derived Fetuin-B protein surround the oocyte. Fetuin-B inhibits spuriously released ovastacin, and prevents premature ZP hardening. Fetuin-B thus maintains female fertility. At the time of fertilization, cortical granules are burst-released, and the high amount of ovastacin liberated overwhelms the Fetuin-B inhibitory potential, which leads to physiological ZP hardening. Further sperm binding and penetration is blocked. The hardened ZP protects the pre-implantation embryo during its travel down the oviduct into the uterus. Embryo hatching occurs immediately before the embryo implants into the endometrium. Fetuin-B concentration surrounding the oocyte is initially high enough to inhibit spurious ovastacin activity, but diminishes quickly along the route the oocyte and pre-implantation embryo take to meet sperm and implantation site. Thus fetuin-B concentration matches the need required at the respective sites of the reproductive tract.
Initially the oocyte will be surrounded by enough Fetuin-B protein to inhibit the activity of ovastacin, which is spuriously released from cortical granules. Fetuin-B was also found secreted by the oviduct proper [29]. The relative contribution to ovastacin inhibition of follicular fluid Fetuin-B vs. oviduct secreted Fetuin-B, remains to be determined [30]. Physiologically in mice, forty hours post ovulation the ZP hardens definitively, probably due to continuous dilution of Fetuin-B below the minimum required concentration for ovastacin inhibition [31]. Thus apart from complete Fetuin-B deficiency in knockout mice, partial deficiency leading to low serum Fetuin-B concentrations should eventually also lead to premature ZP hardening and reduced fertility. Premature ZP hardening is a common complication in In vitro fertilization (IVF) decreasing the fertilization rate. To improve IVF rate, serum additives like bovine Fetuin, fetal calf serum or human serum are commonly added to oocyte culture media. The serum additives of media are ill defined and have also infection risks, since they contain materials of animal or human origin. Therefore fully defined IVF media are highly desirable. We showed that the active ingredient in common IVF media additives was Fetuin-B protein [11]. For a successful fertilization to occur, sperm must penetrate the ZP and fuse with the oocyte. Sperm-oocyte fusion triggers the completion of oocyte meiosis, as well as a sudden burst of exocytosis of cortical granules containing ovastacin. At this point the ovastacin concentration overwhelms the inhibitory capacity of Fetuin-B Figure (2) leading to fertilization-triggered ZP hardening [11]. The structural modifications in the ZP block further sperm binding, which was assumed to be necessary to block fertilization by additional sperm (polyspermy). Contrary to this belief, oocytes, which cannot undergo ZP hardening for various reasons, do not show polyspermy, but undergo early developmental abortion instead [22,32,33]. Thus a block to polyspermy does not depend on ZP hardening, but ZP hardening is essential to protecting the pre-implantation embryo. In sufficient fertilization-triggered ZP hardening lead to embryo lysis before implantation [29]. Embryo hatching usually happens at the blastocyst stage immediately prior to embryo implantation in the uterus Figure (2). The reversal of ZP hardening during hatching remains to be studied. Theoretically, a irreversibly hardened ZP should hinder blastocyst hatching and thus prevent implantation. In assisted reproduction, ZP hatching is facilitated by laser-mediated ZP piercing to increase implantation rate. The extended culture of embryo In vitro, up until the blastocyst stage seems to promote irreversible ZP is hardening. Vice versa an unhardened ZP could lead to early hatching and embryo loss. Studying the role of Fetuin-B in ZP hatching may help to address these points in the future.
Role of Fetuin-B outside reproductive biology
Fetuin-B is also expressed in males, despite the fact that they will never require control of ZP hardening. Therefore, Fetuin-B likely has other roles in biology outside female fertility. Up until now, Fetuin-B is the only known natural inhibitor of astacin proteases including meprin, a protein family involved in many diverse biologies including tissue homeostasis and cancer[34]. Little is known about the role of ovastacin / Fetuin-B in tumor biology. Somatic cell-to-germline dysregulation is a hallmark of many human tumors [35]. Ovastacin follows that rule in that ovary and uterus tumors expressed ovastacin mRNA, while ovastacin expression is confined to growing oocytes in healthy subjects [36,37]. Tumor cell growth arrest could be induced by targeted tumor therapy using ovastacin antibody-toxin conjugates [37]. Manipulating Fetuin-B as an ovastacin inhibitor should clarify the role of ovastacin protease in tumor biology. Published research showed that over expression of Fetuin-B in skin squamous carcinoma cells led to suppression of tumor growth in nude mice [38]. Our own Fetuin-B deficient mice show no increased tendency in spontaneous tumor genesis [11,39]. Whether Fetuin-B plays a role in other cancers must be studied employing respective tumor models.
DISCUSSION & CONCLUSION
This short review focused on the role of Fetuin-B in female fertility. Fetuin-B obviously participates in a novel proteolytic network of ZP proteins as substrate, ovastacin as executing protease, and Fetuin-B as a protease inhibitor that is reminiscent of proteolytic networks operating in e.g. blood clotting and complement cascades, and many other biological pathways. Future studies will show if the regulation of this proteolytic network is involved in certain forms of idiopathic infertility, and if the network can be exploited for non-hormonal contraception. Whether or not Fetuin-B also plays a role outside reproductive biology e.g. in cancer, has to be determined.
ACKNOWLEDGEMENTS
The research was supported by a grant from Deutsche Forschungsgemeinschaft and by the START program of the Medical Faculty of RWTH Aachen University.
REFERENCES
3. Abrahamson M, Alvarez-Fernandez M, Nathanson CM. Cystatins. Biochem Soc Symp. 2003; 179-199.
6. Pedersen KO. Fetuin, a new globulin isolated from serum. Nature 1944; 154:575.
18.Wassarman PM. Zona pellucida glycoproteins. J Biol Chem. 2008; 283: 24285-24289.
39.Wessling J. Gene deletion and functional analysis of Fetuin-B. Diss RWTH Aachen. 2007.