Biogenic Derived Ag and Zno Nanoparticles Using Couroupita guianensis Aubl. Fruit Extract and its Antibacterial Potential
- 1. Department of Botany, Karnataka State Akkamahadevi Women’s University, Karnataka, India
- 2. Department of Botany, Bhaurao Kakatkar College, Karnataka, India
- 3. Department of Botany, Jagadamba Arts, and Science College, Hittinahalli LT, Karnataka, India
Abstract
In this study, we report the synthesis of Ag and ZnO Nanoparticles (NPs) produced from the fruit extract of C. guianensis. Silver (Ag) and Zinc Oxide (ZnO) have extensive applications in various areas of science and technology. These NPs were characterized using UV-Visible (UV-Vis) spectroscopy, Fourier Transform Infrared (FTIR) spectroscopy, X-Ray Diffraction (XRD) spectrum and Atomic Force Microscopy (AFM). The UV-Vis absorption spectra generated strong characteristic peaks of AgNPs (370nm) and ZnO-NPs (374nm). FTIR spectrum analysis was conducted to validate the use of phytochemicals in the reduction, capping and stabilization of Ag and ZnO-NPs. XRD spectra were examined to ensure phase purity, crystalline nature and size of the AgNPs (42nm) and ZnO NPs (26nm) respectively. Finally, the ultrastructural and nanomechanical properties of the NPs were studied via AFM analysis. Besides, this study determines antibacterial efficacy of Ag and ZnO-NPs fabricated from C. guianensis fruit extract against B. subtilis and E. coli bacterial strains. The B. subtilis had highest inhibition zone activity than E. coli and it was found to be AgNPs (12.02 ± 0.32 mm) and ZnO-NPs (11.18 ± 0.34 mm) respectively. This study demonstrated a significant potential for the use of these particles in biomedical applications due to their remarkable antibacterial activity.
Keywords
• Silver;
• Zinc Oxide Nanoparticles
• Couroupita guianensis
• Characterization
• Antibacterial Activity
CITATION
Daphedar AB, Hujaratti RB, Kaddipudi PJ, Kolar F. (2023) Biogenic Derived Ag and Zno Nanoparticles Using Couroupita guianensis Aubl. Fruit Extract and its Antibacterial Potential. Int J Plant Biol Res 11(1): 1136.
INTRODUCTION
Couroupita guianensis Aubl. belonging to the family Lecythidaceae, is well known for its therapeutic and ornamental values. It is a fast growing deciduous tree, widely cultivated in the tropical and subtropical regions of the world [1]. Almost all parts of this species namely roots, stems, leaves, flowers, fruits and seeds have been reported to possess various medicinal properties, it is used to cure cold, stomach ache, malaria, hypertension, tumours, pain, and inflammatory processes [2]. These properties of the species have been attributed to the presence of phenolics, flavonoids, terpenoids, tannins, alkaloids, couroupitine, indirubin, and isatin [3]. Plant extracts have captivated the attention of researchers because of their simplicity, low cost, and quick reaction time, as well as their capacity to reduce metal ions to metal nanoparticles [4]. Additionally, plant extracts comprises a wide variety of active compounds which assist in the reducing and stabilizing process and also act as templates for the synthesis of metallic nanoparticles [5]. The biosynthesizing precursors are not only safe to handle but the process easily renders itself to scaling up without the use of energy, high temperatures or toxic compounds thus providing an environmentally friendly alternate to physical and chemical methods [6].
The biosynthesized nanoparticles are idyllic candidates for medical applications, one of the most imperative property of such nanoparticles is antimicrobial activity. The adaptability of nanoparticles in rendering themselves to several applications is presently being discovered. Topmost in the list are silver and zinc oxide nanoparticles which find applications in medicine, sensors, renewable energies, environmental remediation, cosmetology, clothing, bio-therapeutic devices, surface disinfection and antimicrobial applications [6]. This could be attributed to their nanoscale size and high surface area to volume ratio which gives them enhanced biological, physical, and chemical properties as compared to their large scale counter parts [7]. Many green silver and zinc oxide nanoparticles have been reported for antibacterial activity [8-11]. Also supportive studies on plant derived silver and zinc oxide nanoparticles conclude that biogenic nanomaterials are biocompatible and an effective therapeutic agent against bacterial, fungal infections, and cancer treatment as well. The appearance of nanoparticles as new antimicrobial agents has boosted up the research for tackling these superbugs. As nanoparticles target the bacterial cell through multiple pathways, it becomes difficult for bacteria to escape from these magical agents, thus exhibiting antibacterial potential [12]. The fruits of cannonball tree are apparently rich in alkaloid, Couroupitine A (tryptanthrin), Couroupitine B (indirubin), malic acid, isocitric acid, stigmasterol, campesterol, hopane, rutin, quercetin, kaempherol, farmaricetin, luteolin and ursolic acid [3]. Thus, in the current study silver and zinc oxide nanoparticles were synthesized using fruit extracts of Couroupita guianens is as precursors and assessed for its antibacterial activity.
MATERIALS AND METHODS
Chemicals
Silver nitrate (AgNO3 ), zinc nitrate (ZnNO3 ) and other analytical reagents are purchased from Hi-media laboratories Pvt. Ltd. Mumbai, India. Nutrient agar media was also procured from Sigma Aldrich.
Collection and Extraction Process
The fruit of Couroupita guianensis (Figure 1)
Figure 1: a) Habit of C. guianensis, b) AgNPs c) Zn)-NPss.
was collected from the Botanical garden of Karnatak University, Campus, Dharwad, Karnataka, India. The collected diseased free fruits were washed with running tap-water to remove unwanted dirt’s on fruits. The fruit material chopped and converted into fine powder through electric grinder. The powder (20gm) was added to 80mL of deionized water and heated for 30 minutes at 60?C. The fruit extract (aqueous) was filtered through whatman No.1 filter paper and preserved in refrigerator at 4?C for further experimental studies.
Synthesis of Ag and ZnO-NPs
Silver (Ag) and zinc Oxide (ZnO) nanoparticles were synthesized in accordance with the previously established procedure [13]. For about 90ml of (1mM) silver nitrate is added to 250ml of Erlenmeyer flask containing 10ml of aqueous fruit extract. The reaction mixture becomes yellowish and later converted into dark brown color within 15 minutes indicating that the formation of silver nanoparticles (Figure 1). To synthesize zinc oxide nanoparticles, 85mL of (1mM) zinc nitrate is mixed with 15mL of fruit extract resulting the solution of reaction mixture was changed from light yellow to light brown in color which indicates the formation of ZnO nanoparticles by reduction of zinc ions (Figure 1).
Characterization of Ag and ZnO-NPs
The bioreduction and formation of Ag-ZnO nanoparticles were monitored visually by observing the color change. The aqueous extract of nanoparticles was determined absorbance spectrum by UV-Visible spectroscopy (Hitachi, U-3310) with 0.1nm resolution in the 200 to 800nm scale. The colloidal solutions were centrifuged (Remi R-8C) at 10,000 revolutions per minute for 10 minutes; the suspension was re-dispersed two to three times with distilled water and then left to dry in an oven until a fine granular powder was achieved. The fine powder of Ag-ZnO nanoparticles were subjected to FTIR (Nicolet, 5700) analysis wavelength ranges between 400 to 4000cm-1, to determine involvement of biomolecules for reduction, capping and stabilization of nanoparticles. The crystalline structure and phase purity of nanoparticles elucidate by X-ray diffractometer (RigaKu Smartlab SE) in the 2θ range between 30 to 80?C. Morphological structure and size of the nanoparticles were determined by AFM analysis (Nanosurf Easyscan 2).
Preparation of Bacterial Strains
Gram-positive Bacillus subtilis MTCC 736 (B. subtilis) and Gram-negative Escherichia coli MTCC 723, 1554 (E. coli) were obtained from the CSIR Laboratory, New Delhi and used as test organisms. Muller-Hinton nutrient broth media was then prepared to facilitate the growth of these bacterial strains. The bacteria were cultured and then grown at 37?C for 18 hours.
Preparation of Ag and ZnO Nanoparticles The crystalline powder of Ag and ZnO nanoparticles (0.01gm) were dissolved in 10ml of DMSO (Dimethyl Sulfoxide) solution and vortexed. The nanoparticles solution is kept at 4?C for 24 hrs until further analysis.
In-vitro Susceptibility
Test For in-vitro antibacterial testing, Ag and ZnO nanoparticles were utilized via a disc diffusion method against both Gram positive and Gram negative bacteria. The Muller-Hinton agar media was first prepared and poured into culture plates, before the bacterial strains were spread across the surfaces evenly using a cotton swab. Susceptibility testing was then conducted with 6mm sterile discs being used in the process. The discs were loaded with 5µL of C. guianensis fruit extract (used as a blank) and solution containing Ag-ZnO nanoparticles mediated fruit extract, respectively. The discs were then placed on culture plates and incubated for 24 hours at 37?C. After the incubation period, the zones of inhibition were observed and measured in millimeters (mm).
Statistical Analysis
The data were expressed in One-way ANOVAs using IBM statistical software SPSS version 20. The triplicate data were analyzed by Duncan’s multiple range tests and p ? 0.05 value was considered significant.
RESULTS AND DISCUSSION
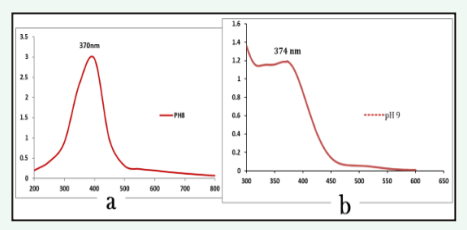
The synthesis of Ag-ZnO nanoparticles using fruit extract of C. guianensis was confirmed by the colour change of the reaction mixture from colorless to dark brown which can be visually detected. This colour change inferred the formation of nanoparticles, due to the excitation of surface plasmon resonance, which was further confirmed by recording the UV-visible absorption spectrum. The maximum absorbance peak was observed at 370nm and 374nm for silver and zinc oxide nanoparticles respectively (Figure 2).
Figure 2: UV-vis absorption Spectrscopy analysis a) AgNPs b) ZnO-NPs synthesized from fruit extract of Couroupita guianensis.
The SPR pattern is dependent on the characteristics of the specific metal particles, their size and shape, as well as the dielectric properties of the medium used for the synthesis and the inter nanoparticle coupling interactions [14]. The obtained results are in consistent with the previous verdicts where silver oxide and zinc oxide nanoparticles synthesized using Persicaria hydropiper and Deverra tortuosa displayed UV-vis absorbance maximum at this wavelength [15,16].
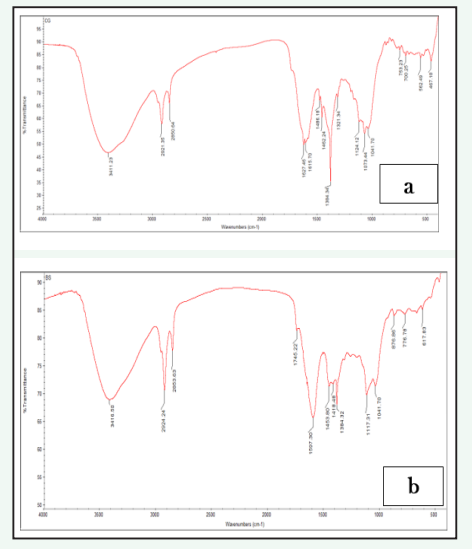
FTIR spectrum is a potent analytical tool used to identify biomolecules and detect functional groups involved in the synthesis of silver nanoparticles (Figure 3).
Figure 3: FTIR spectrum analysis a) AgNPs b) Zinc oxide synthesized using fruit extracts of Couroupita guianensis.
The spectrum showed different absorptions peaks at 3411.23, 2921.35, 2850.64, 1627.46, 1462.29, 1384.34, and 753.23 cm-1. The intense band 3411.23cm-1 is associated to stretching of (OH) hydroxyl group and vibration of phenol and alcoholic group [17]. The peak 2921.35 and 2850.64cm-1 corresponds to the (C-H) stretching of aromatic compounds [18]. The band at 1627.46cm-1 was assigned to stretching of carbonyl (C-O) group. The small peak 1462.29cm-1 was assigned to C-O- stretching vibration mode of phytochemicals like water soluble components of phenolic. The band 1384.34cm-1 was corresponds to C=C stretching of aromatic amines [19]. The weaker band 1073.44cm-1 arose due to the C-N stretching vibrations of amines. The band 753.23 is assigned to the bending vibration in the S-H moiety bonded to the CH2 group. These well-known phytochemical groups like proteins, amino acids, carboxyl group, aromatic amines, and phenolics can bind and stabilize the AgNPs.
An FTIR spectroscopy analysis was conducted to identify the functional groups associated with the formation of ZnO nanoparticles (Figure 3). The obtained spectrum peak 3416.50cm 1could be due to the OH stretching or protein (N-H) amide A [20]. The intense band 2924.24cm-1 corresponds to the C-H stretching of alkyl groups [21]. The weak band 2853.63cm-1 indicates the presence of H-C=O:C-H stretching of aldehydes. The observed peaks 1745.42cm-1 were assigned to stretching of esters. The intense peak 1597.30cm-1 was attributed to the N-H bending of amides. The spectrum bands occurs in between 1300 to 1600cm-1 were corresponds to carboxyl groups C=O, C-N and C-H [22]. The absorption peak 1041.70cm-1 may be due to the presence of C-O-C asymmetric stretching of ester group. The peaks appear in the range between 600 to 400cm-1 may be assigned to the stretching Zn-O bands [23]. The evidence suggests that the formation of ZnO nanoparticles is a result of the interaction between phenolic compounds.
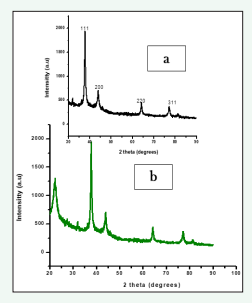
The purity and crystalline structure of the synthesized AgNPs was confirmed by X-ray crystallography analysis (Figure 4).
Figure 4: XRD spectrum analysis a) AgNPs b) ZnO nanoparticles synthesized by fruit extract C. guianensis.
The XRD pattern of AgNPs synthesized from fruit extract of C. guianensis shows different bragg’s peaks at 2θ angle such as 37.74?, 44.90?, 64.32?, and 77.31? which indexed lattice planes (111), (200), (220) and (311) respectively. These assigned high peaks in XRD analysis indicated active silver composition with the indexing [24]. The resultant data matched with the data base JCPDS, file No: 04-0783. The calculated FWHM intense peak (111) showed average size of the AgNPs which was calculated by using Debye Sherrer’s equation and it was found to be 42nm. Our result is strongly supported with the result of [24] synthesized AgNPs by leaf extract Allophylus serratus.
XRD spectrum analysis of ZnO nanoparticles was determined using fruit mediated extract of C. guianensis. The XRD pattern shows different diffraction peaks at 32.14?, 34.98?, 46.61?, 65.10? attributes to (111), (200), (220) and (311) miller indices of ZnO nanoparticles respectively (Figure 4). The small and narrow peak of XRD peaks represents growth of crystal nuclei and nucleation. The observed crystalline structure of ZnO nanoparticles matched with JCPDS file No. 36-1451. The average size of ZnO nanoparticles was estimated to be 26nm, computed from the Debye Scherrer equation and the Full Width Half Maximum (FWHM) highest intense peak. Recent reports have shown similar results in biogenic ZnO nanoparticles synthesized from Elaeagnus angustifolia L. leaf extract [25].
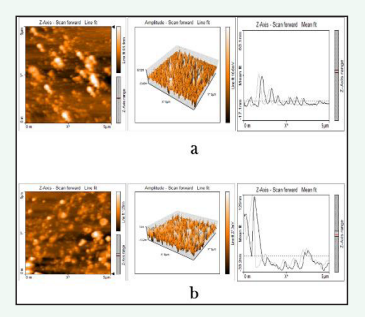
Atomic Force Microscopy (AFM) determines the size, shape and length of the AgNPs. AFM image showed size of the AgNPs was found in the range between 20 to 50nm (Figure 5).
Figure 5: AgNPs synthesized by aqueous fruit extract of C. guinancis, a) Two-dimensional, b) Three- dimensional, c) Particle size distribution of silver nanoparticles.
The three dimensional image of the AgNPs represents height of nanoparticles is 11.2nm and 100nm width. These particles are poydispersed and spherical in shape. Similar size and shape of the AgNPs was reported by [26] using Trichosanthes tricuspidata plant extract. The obtained ZnO-NPs from fruit extract of C. guinancis were subjected to AFM analysis. It is technique used to determine size, and shape of the nanoparticles. (Figure 7) represents the particle size which was found to be 30 to 75nm. The 3D image (Figure 5) represents length (15.3nm) and width 100nm. These nanoparticles are monodispersed and spherical in shape.
As shown in (Figure 6,7),
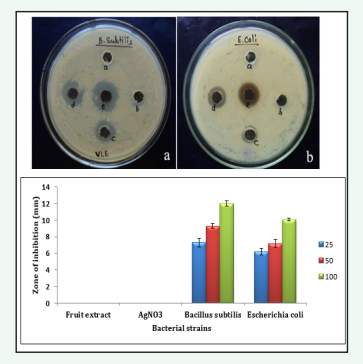
Figure 6: Well diffusion method on different concentrations of AgNPs against B. subtilis and E. coli strains a) a. Fruit extract (negative control), b. AgNO3 (negative control), c. 25µg/ml, d. 50µg/ml e. 100µg/ml respectively. Stastically the results were found to be p < 0.05.
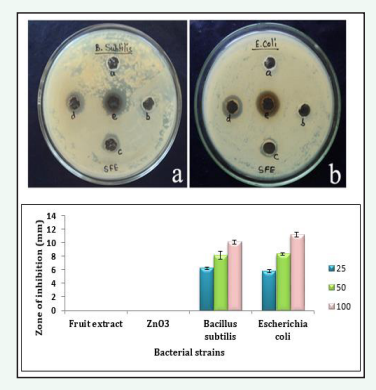
Figure 7: Well diffusion method on different concentrations of ZnO-Nps against B. subtilis and E. coli strains a) a. Fruit extract (negative control), b. ZnNO3 (negative control), c. 25µg/ml, d. 50µg/ml e. 100µg/ml respectively. Stastically the results were found to be p < 0.05.
the antibacterial activity of Ag and ZnO-NPs produced from fruit extract of C. guinancis evaluated against Gram positive (Bacillus subtilis) and Gram negative bacteria (Escherichia coli). The well diffusion method can be used to determine the size (in millimeters) of the inhibition zones for bacterial colonies. The suspension of Ag and ZnO NPs were treated separately with different concentrations viz. 25, 50, 100µg/ml and fruit extract, AgNO3 as negative control. The diameter of the inhibition zone for Bacillus subtilis and Escherichia coli were respectively measured at 7.3 ± 0.5, 9.31 ± 0.3, 12.02 ± 0.32mm and 6.2 ± 0.4, 7.16 ± 0.48, 10.06 ± 0.16mm while the corresponding values for ZnO-NPs were 6.23 ± 0.17, 8.15 ± 0.6, 10.08 ± 0.31mm and 5.8 ± 0.25, 8.3 ± 0.18, 11.18 ± 0.34mm respectively; showing that an increasing concentration of silver and ZnO-NPs produced a greater inhibition zone in Bacillus subtilis and Escherichia coli respectively. The results of the study revealed that AgNPs demonstrated the strongest zone of inhibition activity when compared to ZnO-NPs. Similar results was reported by [27] using green synthesized Ag and ZnO nanocomposite produced from leaf extract of Rumex Crispus.
Previous studies have revealed that colloidal solutions of AgNPs are highly effective against S. aureus compared to E. coli [28]. Furthermore, metallic nanoparticles have shown promising results in combating antibacterial agents. The mechanism behind the antibacterial efficacy of Ag and ZnO-NPs is thought to be mainly due to interacting with the surface membrane of bacteria, ultimately leading to cytoplasmic leakage and rupture [29,30]. The Ag and Zn ions have been shown to initiate the generation of ROS (Reactive Oxygen Species), disrupt ATP synthesis, denature protein in the cells, destabilize ribosome’s, affect mitochondrial function, imbalance metabolic activity and damage bacterial DNA, leading to cell death [31-33]. It has also been reported that when the bacterial surface membrane comes in contact with Ag and ZnO-NPs, the bacterial cell wall changes its shape and becomes inactive, leading to bacterial cellular dysfunction and eventually cell death or necrosis [34].
CONCLUSION
The biosynthesis of Ag and ZnO-NPs from the fruit extract of Couroupita guianensis is a green approach that has numerous benefits, including its simplicity, efficiency, affordability, economic sustainability and ecological friendliness. The optical, ultrastructural, and morphological properties of Ag and ZnO nanoparticles were characterized. The antibacterial efficacy of both Ag and ZnO nanoparticles was evaluated against Gram positive (B. subtilis) and Gram negative (E. coli) bacterial strains and was found to be dose dependent. This study demonstrated a significant potential for the use of these particles in biomedical applications due to their remarkable antibacterial activity.
REFERENCES
1. Sumathi S, Anuradha R. Couroupita guianensis Aubl: An updated review of its phytochemistry and pharmacology. Asian Pharmacy Pharmacol. 2017; 3(1): 1-8.
2. Kumar CS, Naresh G, Sudheer V, Veldi N, Anurag AE. A Short Review on Therapeutic Uses Of Couroupita Guianensis Aubl. Int. Res. J Pharm Appl Sci. 2011; 1: 105-108.