Computer-Based Cognitive Training Versus Paper and Pencil Training: Which is more Effective? A Randomized Controlled Trial in People with Mild Cognitive Impairment
- 1. Alzheimer Hellas, Petrou Sindica13, Greece
- 2. Department of Neurology, Aristotle University of Thessaloniki, Greece
Abstract
Objective: Traditional Cognitive Training with paper-pencil tasks (PPCT) and Computer-Based Cognitive Training (C-BCT) both are effective for people with Mild Cognitive Impairment (MCI). The aim of this study is to evaluate the efficacy of a C-BCT program versus a PPCT one.
Methods: One hundred and twenty four (n=124) people with amnesic & multiple domains MCI (aMCImd) diagnosis were randomly assigned in two groups, a PPCT group (n=65), and a C-BCT (n=59). The groups were matched at baseline in age, gender, education, cognitive and functional performance. Both groups attended 48 weekly 1-hour sessions of attention and executive function training for 12 months. Neuropsychological assessment was performed at baseline and 12 months later.
Results: At the follow up, the PPCT group was better than the C-BCT group in visual selective attention (p≤ 0.022). The C-BCT group showed improvement in working memory (p=0.042) and in speed of switching of attention (p=0.012), while the PPCT group showed improvement in general cognitive function (p=0.005), learning ability (p=0.000), delayed verbal recall (p=0.000), visual perception (p=0.013) and visual memory (p=0.000), verbal fluency (p=0.000), visual selective attention (p=0.021), speed of switching of attention (p=0.001), visual selective attention/multiple choices (p=0.010) and Activities of Daily Living (ADL) as well (p=0.001).
Conclusion: Both C-BCT and PPCT are beneficial for people with aMCImd concerning cognitive functions. However, the administration of a traditional PPCT program seems to affect a greater range of cognitive abilities and transfer the primary cognitive benefit in real life.
Keywords
• Traditional cognitive training
• Computer training
• MCI
CITATION
: Tsolaki M, Poptsi E, Aggogiatou C, Markou N, Zafeiropoulos S, et al. (2017) Computer-Based Cognitive Training Versus Paper and Pencil Training: Which is more Effective? A Randomized Controlled Trial in People with Mild Cognitive Impairment. JSM Alzheimer’s Dis Related Dementia 4(1): 1032.
ABBREVIATIONS
MCI: Mild Cognitive Impairment; aMCImd: amnestic and Multiple Domains MCI; CT: Cognitive Training; C-BCT: ComputerBased Cognitive Training; PPCT: Paper and Pencil Cognitive Training; ADL: Activities of Daily Living; ChEIs: Cholinesterase Inhibitors
INTRODUCTION
Mild Cognitive Impairment (MCI) is an intermediate stage of cognitive impairment, which may or may not lead to dementia [1]. Attention and executive function deficits are common in people with amnestic and multiple domains MCI (aMCImd) [2,3]. The deficits can interfere with Activities of Daily Living (ADL) [4], and may lead to increased risk of conversion to dementia [5].
Cognitive Training (CT), which is based on the idea of neuronal plasticity, is beneficial for people with MCI, since it seems to improve performance of the specific skills which are trained [6]. Moreover, CT not only optimizes the cognitive functioning of people with MCI, but it may also slow down cognitive decline as well as the onset of Major Neurocognitive Disorders [7]. Therefore, CT and, consequently, the cognitive improvement are potential to delay or minimize the conversion rate of MCI to Major Neurocognitive Disorders [8]. However, there is still a question; are all the programs of CT the same beneficial for people with MCI?
Traditional CT requires face-to-face contact and teaching of new learning strategies. It seems to be more effective when it is ecologically designed [9] and administered in groups [10]. On the other hand, research has shown that the use of new technologies such as in computer-based cognitive training (C-BCT) might provide positive effects on the cognitive function of MCI elderly [11]. Moreover, C-BCT is cost effective and it can be administered to populations that would otherwise not receive face to face interventions.
The aim of the current study was to evaluate the efficacy of a C-BCT program versus an ecologically designed program of traditional CT which was applied by paper and pencil (PPCT) in people with aMCImd. Moreover, the secondary aim was to evaluate which program has the potential to transfer the cognitive benefit in ADL.
MATERIALS AND METHODS
Participants
The participants were recruited from the Day Care Center of Alzheimer Hellas “Saint Helen” in Thessaloniki (Greece) from September 2015 to September 2016. The study was carried out entirely at the day care center and was approved by the Scientific and Ethics Committee of Alzheimer Hellas. The participants signed an informed consent and were aware that they could withdraw their consent at any time. Inclusion criteria were ≥60 years of age, a diagnosis of aMCImd according to Petersen criteria [12] and spared language skills. The exclusion criteria were the diagnosis of Major Neurocognitive Disorder according to DSM5 [13], untreated depression/anxiety and severe behavioral problems as that could affect the results of the CT programs, other neurological disorders, pharmacological treatment with ChEIs and sensory deficits. The diagnosis was supported with neurological examination, neuropsychological/neuropsychiatric assessment, neuroimaging and blood tests. In order to minimize selection bias, the participants of the study were chosen at random by using computer-generated numbers. All the evaluators were blinded with respect to the group allocation. Each CT program included 48 weekly one hour sessions during 12 months, while there were booster sessions for the people who missed more than three sessions. The duration of 48 weeks was chosen, because it is proven that more training provides more benefit to people with MCI [14]. Moreover, since the study was carried out entirely at the Day Care Centre of Alzheimer Hellas “Saint Hellen”, it was essential not to disrupt the usual routine of the Centre.
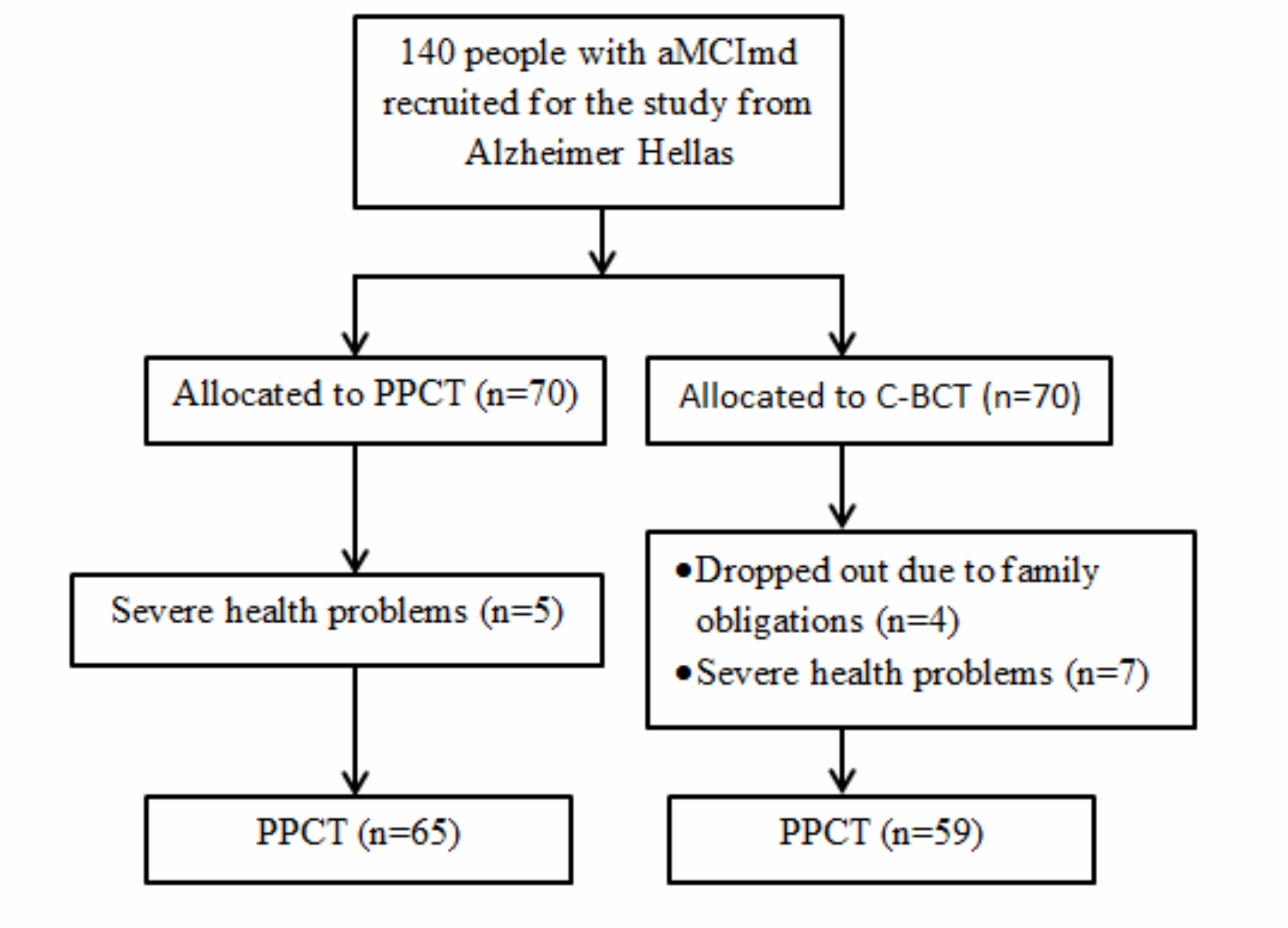
Figure 1 Flow chart of the study. Abbreviations: aMCImd: amnestic and Multiple Domains MCI; C-BCT: Computer-Based Cognitive Training; PPCT: Paper and Pencil Cognitive Training
Twelve (n=12) participants withdraw the study because they encountered severe health problems, while 4 dropped out due to family obligations. As a result, 124 people with aMCImd completed the study and were included in the statistical analysis (65 in PPCT group and 59 in C-BCT group) (Figure 1). Baseline characteristics of the participants and their cognitive and functional performance are presented in (Table 1).
|
Table 1: Participant’s Demographic Characteristics M (SD) in Computer Training Group and Paper/Pencil Training Group according to t-test and Fisher’s exact test |
|||
|
Baseline characteristics M (SD) |
C-BCT |
PPCT |
p |
|
Age |
69.72 (6.35) |
67.66 (6.67) |
0.081 |
|
Gender Male/Female |
16/43 |
21/44 |
0.561 |
|
Education |
11.79 (4.72) |
11.80 (4.24) |
0.997 |
|
MMSE ‡ |
28.25 (1.42) |
27.72 (1.84) |
0.078 |
|
FUCAS † |
43.74 (1.96) |
44.00 (2.52) |
0.538 |
|
FRSSD† |
3.72 (1.96) |
3.95 (1.85) |
0.515 |
|
RAVLT1 ‡ |
5.35 (2.18) |
5.32 (2.00) |
0.931 |
|
RAVLT2 ‡ |
11.01 (2.60) |
10.90 (2.47) |
0.754 |
|
RAVLT3 ‡ |
8.33 (3.80) |
7.76 (3.53) |
0.389 |
|
ROCFT1‡ |
31.30 (4.30) |
29.78 (5.16) |
0.079 |
|
ROCFT2‡ |
15.45 (6.76) |
13.36 (6.93) |
0.091 |
|
FAS‡ |
10.84 (3.66) |
9.77 (2.59) |
0.061 |
|
TEA 1 ‡ |
28.29 (10.29) |
27.87 (10.58) |
0.825 |
|
TEA 2‡ |
45.45 (10.61) |
48.75 (11.99) |
0.113 |
|
TEA 4a‡ |
8.72 (1.22) |
8.72 (1.22) |
0.279 |
|
TEA 4b† |
6.35 (2.58) |
6.55 (3.25) |
0.708 |
|
TEA 6† |
5.00 (1.360) |
5.50 (2.20) |
0.140 |
|
Abbreviations: p = Statistical difference of performance; M (SD) = Mean (Standard Deviation);‡= More points/better performance; †= Less points/better performance; MMSE: Mini Mental State Examination; FUCAS: Functional Cognitive Assessment Scale; RAVLT1: Rey Auditory Verbal Learning Test-Digit spam; RAVLT2: Rey Auditory Verbal Learning Test-Learning ability; RAVLT3: Rey Auditory Verbal Learning Test-Delayed recall; ROCFT1: Rey Osterrieth Complex Figure test-Copy; ROCFT2: Rey Osterrieth Complex Figure test Delayed recall; FAS: Verbal Fluency Test; TEA 1: Test of Everyday Attention/Map searching in1 minute; TEA 2: Test of Everyday Attention/Map searching in 2 minutes; TEA 4a: Test of Everyday Attention/Switching of attention; TEA 4b: Test of Everyday Attention/Speed of switching of attention; TEA 6: Test of Everyday Attention /Multiple choices; FRSSD: Functional Rating Scale for Symptoms of Dementia |
|||
Cognitive interventions
a) The PPCT program: The primary aim of PPCT was to enhance abilities of attention and cognitive executive function, while the secondary was the enhancement of visual-verbal memory. The PPCT was administered in groups of 8 people, included teaching of memory strategies and comprised levels of difficulty. Paper and pencil tasks were ecological; they were yielding from Activities of Daily Living (ADL), such as the processing of a shopping list, search and detection of a range of elements in a telephone catalogue, or processing a restaurant menu. Each session consisted of 10 cognitive tasks. Participants had first to go through 3 tasks that required visual selective attention; afterwards, they proceeded with two tasks requiring updating of working memory and switching of attention, and later on, they practiced the shifting of visuo-spatial attention and the dual task. The same structure was followed during all the sessions.
b) The C-BCT program: The primary aim of the CT program was the enhancement of attention and executive function, while the secondary the enhancement of visual /verbal memory and visual–spatial abilities. The C-BCT used the “Complete brain workout” software (Oak Systems). From the above software 10 different tasks were chosen (Pyramids, Color ties, Linker, Matching pairs, Symbol block, Box Pacer, Color matching, Shape Match, Correct change, Number balance). Each one of the tasks mentioned above was presented in an alternative order. As for example, participants had first to go with a visual selective attention task, then with a working memory task, an abstract thinking task etc. However, the same hierarchy was consistent through all cognitive tasks, while there were five levels of difficulty. The program was administered in touch screens and none of the participants in the study had previous experience with computers. Therefore, the therapist was providing the participants with the appropriate instructions, while specific learning strategies were taught to the participants, so as they could complete successfully the tasks.
|
Table 2: Between group differences at follow-up according to Univariate Analysis (ANOVA). |
||||
|
Cognitive/functional Performance M (SD) |
C-BT |
PPCT |
p |
F |
|
MMSE ‡ |
27.94 (1.61) |
28.41 (1.49) |
0.098 |
2.724 |
|
FUCAS † |
43.47 (1.84) |
43.46 (1.92) |
0.969 |
0.001 |
|
FRSSD† |
3.67 (1.68) |
3.20 (1.51) |
0.099 |
0.099 |
|
RAVLT1 ‡ |
5.89 (1.97) |
5.72 (2.28) |
0.650 |
0.207 |
|
RAVLT2 ‡ |
11.44 (2.40) |
11.87 (2.56) |
0.332 |
0.948 |
|
RAVLT3 ‡ |
8.94 (2.81) |
9.43 (3.33) |
0.425 |
0.641 |
|
ROCFT1‡ |
31.12 (4.36) |
31.48 (4.34) |
0.649 |
0.649 |
|
ROCFT2‡ |
15.38 (6.31) |
16.52 (7.25) |
0.354 |
0.354 |
|
FAS‡ |
11.71 (2.55) |
11.92 (3.14) |
0.061 |
0.676 |
|
TEA 1 ‡ |
26.94 (8.72) |
31.16 (10.97) |
0.022 |
5.370 |
|
TEA 2‡ |
45.14 (9.60) |
50.92 (13.12) |
0.007 |
7.430 |
|
TEA 4a‡ |
8.38 (2.16) |
8.60 (1.55) |
0.512 |
0.435 |
|
TEA 4b† |
5.23 (2.67) |
5.77 (2.19) |
0.225 |
0.225 |
|
TEA 6† |
4.95 (1.40) |
4.78 (1.73) |
0.569 |
0.327 |
|
Abbreviations: p = Statistical difference of performance; M (SD) = Mean (Standard Deviation);‡= More points/better performance; †= Less points/better performance; MMSE: Mini Mental State Examination; FUCAS: Functional Cognitive Assessment Scale; RAVLT1: Rey Auditory Verbal Learning Test-Digit spam; RAVLT2: Rey Auditory Verbal Learning Test-Learning ability; RAVLT3: Rey Auditory Verbal Learning Test-Delayed recall; ROCFT1: Rey Osterrieth Complex Figure test-Copy;ROCFT2: Rey Osterrieth Complex Figure test Delayed recall; FAS: Verbal Fluency Test; TEA 1: Test of Everyday Attention/Map searching in1 minute; TEA 2: Test of Everyday Attention/Map searching in2 minutes; TEA 4a: Test of Everyday Attention/Switching of attention; TEA 4b: Test of Everyday Attention/Speed of switching of attention; TEA 6: Test of Everyday Attention /Multiple choices; FRSSD: Functional Rating Scale for Symptoms of Dementia |
||||
Neuropsychological Assessment
The effectiveness of the intervention was assessed by a neuropsychological assessment performed at baseline, as well as 12 months later, after the completion of the 48 sessions. The neuropsychological assessment included a battery of psychometric tests consisted of a) Mini Mental State Examination [15,16] (MMSE), for the assessment of general cognitive function, b) Functional Cognitive Assessment Scale (FUCAS) for general functional performance [17], c) Functional Rating Scale for Symptoms of Dementia (FRSSD) for ADL (evaluates caregiver’s opinion about daily function of people with MCI) [18], d) Rey Auditory Verbal Learning test (RAVLT), for verbal memory [19], e) Rey Osterrieth Complex Figure test (ROCFT), for visual memory and visual constructive abilities [20] f) Verbal Fluency test (FAS) [21], and g) Test of everyday attention (TEA) [22] for attention deficits. The Geriatric Depression Scale (GDS) [23,24] was used in order to exclude patients with depression and the Neuropsychiatric Inventory (NPI) [25,26] for the exclusion of people with psychopathological symptoms. All participants were examined at the same time and place by the same psychologists who were not aware of the classification of the participants in the two training groups. We tried to avoid practice effects by using different test versions, wherever it was necessary, and namely the Taylor’s Complex Figure Test [27,28] for visual memory and visual constructive abilities, and California Auditory Verbal Learning [29] for verbal memory.
Statistical analysis
The statistical analysis was performed with the SPSS statistical software, version 23.0. Demographic, cognitive and functional characteristics at baseline were analyzed using a two tailed t test for independent samples or Fisher’s exact test, when it was necessary. Univariate analysis (ANOVA) was used in order to examine the between-group differences at the followup. The effect of cognitive training for both groups was analyzed with repeated measures analysis of variance (ANOVA). In order to calculate the magnitude of the cognitive training effects, we calculated standardized effect sizes for each neuropsychological measure.
|
Table 3: Mean and Standard Deviation of cognitive and functional performances of the CT and the P/PT groups at baseline and at follow-up according to repeated measures analysis of variance (ANOVA). |
||||||||
|
|
C-BCT |
|
PPCT |
|
||||
|
Cognitive/functional Performance M (SD) |
Baseline |
Follow-up |
p |
F |
Baseline |
Follow-up |
p |
F |
|
MMSE ‡ |
28.25 (1.42) |
27.94 (1.61) |
0.154 |
2.088 |
27.72 (1.84) |
28.41 (1.49) |
0.005 |
8.526 |
|
FUCAS † |
43.74 (1.96) |
43.47 (1.84) |
0.338 |
2.169 |
44.00 (2.52) |
43.46 (1.92) |
0.113 |
2.573 |
|
FRSSD† |
3.72 (1.96) |
3.67 (1.68) |
0.806 |
0.061 |
3.95 (1.85) |
3.20 (1.51) |
0.001 |
11.473 |
|
RAVLT1 ‡ |
5.35 (2.18) |
5.89 (1.97) |
0.042 |
4.327 |
5.32 (2.00) |
5.72 (2.28) |
0.175 |
1.882 |
|
RAVLT2 ‡ |
11.01 (2.60) |
11.44 (2.40) |
0.203 |
1.656 |
10.90 (2.47) |
11.87 (2.56) |
0.000 |
17.768 |
|
RAVLT3 ‡ |
8.33 (3.80) |
8.94 (2.81) |
0.071 |
3.388 |
7.76 (3.53) |
9.43 (3.33) |
0.000 |
29.865 |
|
ROCFT1‡ |
31.30 (4.30) |
31.12 (4.36) |
0.711 |
0.138 |
29.78 (5.16) |
31.48 (4.34) |
0.013 |
6.557 |
|
ROCFT2‡ |
15.45 (6.76) |
15.38 (6.31) |
0.916 |
0.011 |
13.36 (6.93) |
16.52 (7.25) |
0.000 |
26.791 |
|
FAS‡ |
10.84 (3.66) |
11.71 (2.55) |
0.061 |
3.666 |
9.77 (2.59) |
11.92 (3.14) |
0.000 |
52.118 |
|
TEA 1 ‡ |
28.29 (10.29) |
26.94 (8.72) |
0.227 |
1.493 |
27.87 (10.58) |
31.16 (10.97) |
0.021 |
5.582 |
|
TEA 2‡ |
45.45 (10.61) |
45.14 (9.60) |
0.924 |
0.009 |
48.75 (11.99) |
50.92 (13.12) |
0.071 |
3.371 |
|
TEA 4a‡ |
8.72 (1.22) |
8.38 (2.16) |
0.145 |
2.186 |
8.72 (1.22) |
8.60 (1.55) |
0.178 |
1.855 |
|
TEA 4b† |
6.35 (2.58) |
5.23 (2.67) |
0.012 |
6.814 |
6.55 (3.25) |
5.77 (2.19) |
0.001 |
12.522 |
|
TEA 6† |
5.00 (1.360) |
4.95 (1.40) |
0.713 |
0.137 |
5.50 (2.20) |
4.78 (1.73) |
0.010 |
7.085 |
|
Abbreviations: p = Statistical difference of performance; M (SD) = Mean (Standard Deviation); ‡= More points/better performance; †= Less points/better performance; MMSE: Mini Mental State Examination; FUCAS: Functional Cognitive Assessment Scale; RAVLT1: Rey Auditory Verbal Learning Test-Digit spam; RAVLT2: Rey Auditory Verbal Learning Test-Learning ability; RAVLT3: Rey Auditory Verbal Learning Test-Delayed recall; ROCFT1: Rey Osterrieth Complex Figure test-Copy; ROCFT2: Rey Osterrieth Complex Figure test Delayed recall; FAS: Verbal Fluency Test; TEA 1: Test of Everyday Attention/Map searching in1 minute; TEA 2: Test of Everyday Attention/Map searching in2 minutes; TEA 4a: Test of Everyday Attention/Switching of attention; TEA 4b: Test of Everyday Attention/Speed of switching of attention; TEA 6: Test of Everyday Attention /Multiple choices; FRSSD: Functional Rating Scale for Symptoms of Dementia |
||||||||
RESULTS
Baseline measurements showed that there were no statistically significant differences between the two groups in age (p= 0.081), gender (p= 0.561) or education (p= 0.997) nor in cognitive or functional performance (Table 1).
At the follow-up, the analysis of variance showed that the PPCT group was better than the C-BCT group in visual selective attention in one minute (TEA) (p= 0.022, F= 5.370), and visual selective attention in two minutes (TEA) (p= 0.007, F= 7.430) (Table 2).
According to the repeated measures analysis of variance between baseline and follow-up, the C-BCT group showed improvement in working memory (p= 0.042, F= 4.327) and speed of switching of attention (p= 0.012, F= 6.814) while the rest of the cognitive and functional abilities were preserved. The PPCT group showed improvement in general cognitive function (p= 0.005, F= 8.526), ADL (p= 0.001, F= 11.473), learning ability (p= 0.000, F= 17.768), delayed recall (p= 0.000, F= 29.865), complex figure copy (p= 0.013, F= 6.557) and delayed recall (p= 0.000, F= 26.791), verbal fluency (p= 0.000, F= 12.118), visual selective attention (p= 0.021, F= 5.582), speed of switching of attention (p= 0.001, F= 12.522) and visual selective attention/multiple choices (p= 0.010, F= 7.085) (Table 3).
DISCUSSION
The effectiveness of traditional CT in people with aMCImd as well as the effectiveness of C-BCT has been well demonstrated [30-35]. However, there aren’t many comparatives studies, to provide evidence whether the traditional CT is more effective than the C-BCT in MCI and vice versa. The aim of the study was to compare the effectiveness of two CT programs, which although they had the same primary target, (the enhancement of executive function and attention) they used a different mean of training.
To provide evidence about which CT program was more beneficial, we investigated the effectiveness of PPCT vs C-BCT in people with aMCImd at the end of the interventions. According to the results, at the follow-up, the PPCT had better performance than the C-BCT in visual selective attention. The rest of the cognitive functions didn`t show any difference. As far as we know there aren’t randomized controlled studies to compare effectiveness of traditional PPCT vs C-BCT in people with MCI. However, the study by Man, Chung, & Lee in 2012 [36], investigated the effectiveness of a virtual reality computer based memory training program, versus the same program administered by paper and pencil, in people with questionable dementia. The results have shown that although both groups improved memory performance, the virtual reality computer memory training, improved more cognitive abilities than the paper and pencil group, which is in contrast to our results. However, they targeted memory performance while our target was the improvement of attention and executive function. Furthermore they used a virtual reality computer program, which plays an important role in computer’s effectiveness. However, their study has several limitations, such as small sample size and methodological gaps.
In our study the repeated measures in each group have shown that both groups improved their cognitive performance. The C-BCT group, at the end of the study, preserved stable their cognitive and functional abilities and showed improvement in executive function. Our results are in agreement with other studies that support that C-BCT is effective for people with MCI [37-39]. This result makes sense, since there is a strong association between functional performance and executive cognitive function [40].
Eventually, the traditional PPCT enhanced the cognitive abilities which were trained and the cognitive benefit was transferred in daily living. This is the ultimate goal in cognitive rehabilitation [41]. As our results indicates, the Functional Rating Scale for dementia (FRSSD), a test which measures totally caregiver’s opinion, was improved. This result is very important, since the successfulness of CT is needed to be confirmed not only from participant’s results, but also by caregiver’s satisfaction. Even though there are several studies that confirm the effectiveness of CT in MCI, in a broad range of cognitive abilities [42,43], there is little research to demonstrate significant improvements in ADL [31,44]. The study by Brum in 2009 [44] also indicated that after the CT the experimental group improved both in cognitive abilities (attention-time orientation) and ADL (shopping skills-dealing with finances). The use of ecologically valid activities in his cognitive training, as indeed in our PPCT group is probably the reason of this result. Our C-BCT didn’t show significant improvement in ADL. In agreement with our study, Coyle, Traynor, & Solowij, in 2015 [45], in their review, indicated that despite the fact that C-BCT programs can improve attention, executive function, visual or verbal memory, they can’t demonstrate significant improvements in ADL. Maybe new C-BCT with ecological tasks, or even better virtual reality C-BCT programs would be more effective to transfer the impact of training in real life.
Limitation
A limitation of our study is that the tasks which have been used in CT programs was not ecological valid. Despite the fact that both C-BCT and PPCT were practicing executive function and attention abilities (which was the main target of our study) only the PPCT used ecological tasks. Therefore, it sounds obvious that only the participants who underwent the paper and PPCT could be improved in ADL. The next step of our study is to compare the effectiveness of an ecological designed PPCT versus a relative ecological C-BCT in order to evaluate the different effectiveness of each program in people with MCI.
Another limitation of our study is that we didn’t use selfreport questionnaires so as to provide evidence about the potential changes of training in the quality of life of people with MCI. Maybe this kind of questionnaires would show a more informative view on the self-perceived efficacy of the training.
CONCLUSION
Our study indicated that C-BCT and PPCT are both beneficial for aMCImd. The administration of a traditional PPCT program may be more effective in the transfer of cognitive benefit in ADL. The use of ecologically designed CT programs has the potential to be successful [7], since these programs are related to ADL [38]. However more studies are needed in order to evaluate which type of training is more effective.
ACKNOWLEDGEMENTS
This study was conducted in the Alzheimer’s Day Care Center “Saint Helen”, which is funded by the Department of Mental Health of the Greek Ministry of Health.
REFERENCES
- Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L. Mild cognitive impairment: a concept in evolution. J Intern Med. 2014; 275: 214-228.
- Johns EK, Phillips NA, Belleville S, Goupil D, Babins L, Kelner N, et al. The profile of executive functioning in amnestic mild cognitive impairment: disproportionate deficits in inhibitory control. J Int Neuropsychol Soc. 2012; 18: 541-555.
- Clément F, Gauthier S, Belleville S. Executive functions in mild cognitive impairment: emergence and breakdown of neural plasticity. Cortex. 2013; 49: 1268-1279.
- Burton CL, Strauss E, Hultsch DF, Hunter MA. Cognitive functioning and everyday problem solving in older adults. Clin Neuropsychol. 2006; 20: 432-452.
- Saunders NL, Summers MJ. Longitudinal deficits to attention, executive, and working memory in subtypes of mild cognitive impairment. Neuropsychology. 2011; 25: 237-248.
- Choi J, Twamley EW. Cognitive rehabilitation therapies for Alzheimer's disease: a review of methods to improve treatment engagement and self-efficacy. Neuropsychol Rev. 2013; 23: 48-62.
- Belleville S. Cognitive training for persons with mild cognitive impairment. Int Psychogeriatr. 2008; 20: 57-66.
- Mowszowski L, Batchelor J, Naismith SL. Early intervention for cognitive decline: can cognitive training be used as a selective prevention technique? Int Psychogeriatr. 2010; 22: 537-548.
- Moreau D, Conway AR. The case for an ecological approach to cognitive training. Trends Cogn Sci. 2014; 18: 334-336.
- Desai AK, Grossberg GT, Chibnall JT. Healthy brain aging: a road map. Clin Geriatr Med. 2010; 26: 1-16.
- Klimova B, Maresova P. Computer-Based Training Programs for Older People with Mild Cognitive Impairment and/or Dementia. Front Hum Neurosci. 2017; 11: 262.
- Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004; 256: 183-194.
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5®). American Psychiatric Pub. 2013.
- Poptsi E, Kounti F, Agogiatou C, Bakoglidou E, Soumpourou A, Zafeiropoulos S, et al. Use it more and keep it alive: a Longitudinal Randomized Controlled Trial in people with Mild Cognitive Impairment. Hell J Nucl Med Suppl. 2017; 20: 233-243
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975; 12: 189-198.
- Fountoulakis KN, Tsolaki M, Chantzi H, Kazis A. Mini mental state examination (MMSE): a validation study in Greece. Am J Alzheimers Dis Other Demen. 2000; 15: 342-345.
- Kounti F, Tsolaki M, Kiosseoglou G. Functional Cognitive Assessment Scale (FUCAS): a new scale to assess executive cognitive function in daily life activities in patients with dementia and mild cognitive impairment. Human Psychopharmacol Clin Exp. 2006; 21: 305-311.
- Hutton JT. Alzheimer’s Disease in Conn’s Current Therapy. 1990; 778-781.
- Rey A. L'Examen Clinique en Psychologie. Paris: Press Universitaire de France. 1958.
- Rey A. L’examen psychologique dans les cas d’encéphalopathie traumatique. Arch de Psychol. Arch de Psychol. 1941; 28: 286-340.
- Kosmidis MH, Vlahou CH, Panagiotaki P, Kiosseoglou G. The verbal fluency task in the Greek population: Normative data, and clustering and switching strategies. J Int Neuropsychol Soc. 2004; 10: 164-172.
- Robertson IH, Ward T, Ridgeway V, Nimmo-Smith I. The structure of normal human attention: The Test of Everyday Attention. J Int Neuropsychol Soc. 1996; 2: 525-534.
- Yesavage JA, Sheikh JI. 9/Geriatric depression scale (GDS) recent evidence and development of a shorter version. Clin gerontol. 1986; 5: 165-173.
- Fountoulakis KN, Tsolaki M, Iacovides A, Yesavage J, O'Hara R, Kazis A, et al. The validation of the short form of the Geriatric Depression Scale (GDS) in Greece. Aging (Milano). 1999; 11: 367-372.
- Cummings JL, Mega M, Gray K, Rosenberg-Thompson S, Carusi DA, Gornbein J. The Neuropsychiatric Inventory comprehensive assessment of psychopathology in dementia. Neurology. 1994; 44: 2308-2314.
- Politis AM, Mayer LS, Passa M, Maillis A, Lyketsos CG. Validity and reliability of the newly translated Hellenic Neuropsychiatric Inventory (H-NPI) applied to Greek outpatients with Alzheimer's disease: a study of disturbing behaviors among referrals to a memory clinic. Int J Geriatr Psychiatry 2004; 19: 203-208.
- Taylor LB. Localisation of cerebral lesions by psychological testing. Clin Neurosurg. 1969; 16: 269-287.
- Taylor LB. Psychological assessment of neurosurgical patients. Func Neurosurg. 1979; 165-180.
- Delis D, Kramer J, Kaplan E, Ober B. California Verbal Learning Test. San Antonio. The Psychological Corporation. 1987.
- Barnes DE, Yaffe K, Belfor N, Jagust WJ, DeCarli C, Reed BR, et al. Computer-based cognitive training for mild cognitive impairment: results from a pilot randomized, controlled trial. Alzheimer Dis Assoc Disord. 2009; 23: 205-210.
- Tsolaki M, Kounti F, Agogiatou C, Poptsi E, Bakoglidou E, Zafeiropoulou M , et al. Effectiveness of nonpharmacological approaches in patients with mild cognitive impairment. Neurodegener Dis. 2011; 8: 138-145.
- Boripuntakul S, Kothan S, Methapatara P, Munkhetvit P, Sungkarat S. Short-term effects of cognitive training program for individuals with amnestic mild cognitive impairment: a pilot study. Phys Occup Ther Geriatr. 2012; 30: 138-149.
- Herrera C, Chambon C, Michel BF, Paban V, Alescio-Lautier B. Positive effects of computer-based cognitive training in adults with mild cognitive impairment. Neuropsychologia. 2012; 50: 1871-1881.
- Lim MH, Liu KP, Cheung GS, Kuo MC, Li R, Tong CY. Effectiveness of a multifaceted cognitive training programme for people with mild cognitive impairment: a one-group pre-and posttest design. HKJOT. 2012; 22: 3-8.
- Rodakowski J, Saghafi E, Butters MA, Skidmore ER. Non-pharmacological interventions for adults with mild cognitive impairment and early stage dementia: An updated scoping review. Mol Aspects Med. 2015; 43:38-53.
- Man DW, Chung JC, Lee GY. Evaluation of a virtual reality-based memory training programme for Hong Kong Chinese older adults with questionable dementia: a pilot study. Int J Geriatr Psychiatry. 2012; 27: 513-520.
- Smith GE, Housen P, Yaffe K, Ruff R, Kennison RF, Mahncke HW, et al. A cognitive training program based on principles of brain plasticity: Results from the Improvement in Memory with Plasticity-based Adaptive Cognitive Training (IMPACT) Study. J Am Geriatr Soc. 2009; 57: 594-603.
- Gaitán A, Garolera M, Cerulla N, Chico G, Rodriguez-Querol M, Canela-Soler J. Efficacy of an adjunctive computer-based cognitive training program in amnestic mild cognitive impairment and Alzheimer's disease: a single-blind, randomized clinical trial. Int J Geriatr Psychiatry. 2013; 28: 91-99.
- Lee TM, Chan FH, Chu LW, Kwok TCY, Lam LCW, Tam HMK. Auditory-based cognitive training programme for attention and memory in older people at risk of progressive cognitive decline: a randomised controlled trial. Hong Kong Med J. 2017; 23: 12-15.
- Ferreira NV, Cunha PJ, da Costa DI, dos Santos F, Costa FO, Consolim-Colombo F. Association between functional performance and executive cognitive functions in an elderly population including patients with low ankle-brachial index. Clin Interv Aging. 2015; 10: 839-847.
- Belleville S, Boller B, del Val LP. Cognitive Training in Mild Cognitive Impairment. In Cognitive Training. Springer International Publishing. 2016; 187-197.
- Belleville S. Cognitive training for persons with mild cognitive impairment. Int Psychogeriatr. 2008; 20: 57-66.
- Carretti B, Borella E, Fostinelli S, Zavagnin M. Benefits of training working memory in amnestic mild cognitive impairment: specific and transfer effects. Int Psychogeriatr. 2013; 25: 617-626.
- Brum PS, Forlenza OV, Yassuda MS. Cognitive training in older adults with Mild Cognitive Impairment: impact on cognitive and functional performance. Dement Neuropsychol. 2009; 3: 124-131.
- Coyle H, Traynor V, Solowij N. Computerized and virtual reality cognitive training for individuals at high risk of cognitive decline: systematic review of the literature. Am J Geriatr Psychiatry. 2015; 23: 335-359.