Are Mycobacterium Drugs Effective for Treatment Resistant Lyme Disease, TickBorne Co-Infections, and Autoimmune Disease?
- 1. Hudson Valley Healing Arts Center, USA
Abstract
Introduction: PTLDS/chronic Lyme disease may cause disabling symptoms with associated overlapping autoimmune manifestations, with few clinically effective published treatment options. We recently reported on the successful use of a mycobacterium drug, Dapsone, for those with PTLDS. We now report on the novel use of another mycobacterium drug, pyrazinamide, (PZA), in relieving resistant symptomatology secondary to Lyme disease and associated co-infections, while decreasing autoimmune manifestations with Behçet’s syndrome.
Method: Disabling multi-systemic/arthritic symptoms persisted in a Lyme patient with co-infections (Bartonella, tularemia) and overlapping rheumatoid arthritis/ Behçet’s disease, despite several rotations of classic antibiotic and DMARD regimens. Dapsone, a published treatment protocol used for Behçet’s syndrome, recently has been demonstrated to be effective in the treatment of PTLDS/chronic Lyme disease and co-infections. It was superior to prior treatment regimens in relieving some resistant chronic tick-borne/autoimmune manifestations; however, it did not effectively treat the skin lesions and ulcers secondary to Behçet’s disease, nor significantly affect the granuloma formation, joint swelling, and pain associated with Lyme, Bartonella, and RA. PZA, in combination with Plaquenil, minocycline and rifampin, relieved her resistant symptomatology secondary to Lyme and co-infections, her Behçet’s ulcers, as well as granulomatous skin changes. In addition, a quadruple intracellular combination of a tetracycline (doxycycline), combined with rifampin, Dapsone, and a quinolone (moxifloxacin) was effective in treating reactivation of her tularemia.
Conclusion: Further scientific studies are needed on the role of intracellular bacteria and mycobacterium drugs like Dapsone and pyrazinamide in the treatment of both chronic persistent bacterial infections and resistant autoimmune phenomena.
Keywords
• Lyme disease
• Bartonella
• Tularemia
• Behçet’s Disease/Syndrome
• Rheumatoid arthritis
• Dapsone
• Pyrazinamide
• Persister bacteria
Citation
Horowitz RI, Freeman PR (2016) Are Mycobacterium Drugs Effective for Treatment Resistant Lyme Disease, Tick-Borne Co-Infections, and Autoimmune Disease?. JSM Arthritis 1(2): 1008
ABBREVIATIONS
PTLDS: Post-Treatment Lyme Disease Syndrome; RA: Rheumatoid Arthritis; AI: Autoimmune Illness; PZA: Pyrazinamide, DMARDs: Disease-Modifying Anti Rheumatic Drugs; VEGF: Vascular Endothelial Growth Factor, MSIDS: Multiple Systemic Infectious Disease Syndrome
INTRODUCTION
Autoimmune diseases like rheumatoid arthritis and lupus are rising in incidence in the United States and environmental factors are being implicated [1-3]. Tick-borne diseases, such as Lyme disease are also increasing in number as per recent Centers for Disease Control (CDC) studies [4,5], and have been associated with autoimmune manifestations [6]. Up to 20-25% of patients may suffer the consequences of Post- Treatment Lyme Disease Syndrome (PTLDS) [7] after a tick bite, with or without associated autoimmune disease, leading to multiple symptoms, including disabling fatigue, arthritis, and neuropathy.
The National Science Foundation has identified Lyme disease as one of several emerging pandemic disease outbreaks that threaten global public health and world economies [8]. The CDC reported a significant increase in the number of Lyme cases in the US from 2005-2010, which led to revised estimates of over 300,000 new cases per year [5]. In August 2015, CDC researchers revised their estimates upwards once again, showing a 320% increase in Lyme cases in the past 20 years [9]. Birds are known to carry ticks and spread the infection across the US and Europe, accounting for part of the increased numbers [10].
Ticks now contain multiple bacterial, viral, and parasitic infections that can be simultaneously transmitted with Borrelia burgdorferi, the agent of Lyme disease. Co-infection is the rule, with ticks able to transmit multiple infections [11]. In one recent study, 45% of ticks were co-infected with up to five different pathogens, and up to eight pathogens were identified in the same tick, including multiple species of intracellular bacteria including: Borrelia, relapsing fever spirochetes, Anaplasma, Ehrlichia, spotted fever rickettsia, Bartonella spp. (henselae); ten species of Babesia, two Theileria spp. Wolbachia spp. Coxiella burnetii (Q-fever) and Midichloria mitochondrii, an associated symbiont [11]. Many of these infections are increasing in parts of the United States and worldwide [12], including tularemia, considered a rare infection years ago. It is a febrile, granulomatous, infectious zoonosis caused by Francisella tularensis, an aerobic, gram-negative, pleomorphic bacillus, which is found in ticks. F. tularensis is one of the most infectious bacterial species known [13]. Patients infected with Lyme disease and associated tick-borne co-infections like Babesia, Bartonella and tularemia are much sicker and often resistant to standard therapies, and may present with atypical clinical presentations [14].
Patients with chronic symptoms after standard treatment for Lyme disease/PTLDS have been hypothesized to have multi factorial causes for their illness [15], including persistent infection with intracellular bacteria increasing inflammation, autoimmune phenomenon and/or tissue damage responsible for ongoing morbidity. We recently published on the effective use of Dapsone for Chronic Lyme disease/PTLDS [16], a mycobacterium drug known to have efficacy by addressing intracellular bacteria. Dapsone has also demonstrated efficacy in the treatment of Behcet’s syndrome. Behçet’s disease is a multi systemic, chronic, auto inflammatory disorder with a small vessel vasculitis, of unknown etiology [17]. Common symptoms include oral and genital ulcers, inflammation of the eye, skin lesions, and arthritis. In the case report presented here, we describe a patient with a history of treatment resistant Lyme disease, and associated intracellular co-infections including probable Babesiosis, Bartonella henselae, and Francisella tularensis, associated autoimmune disease, including seronegative rheumatoid arthritis and Behçet’s syndrome, who responded to a novel combination of multiple intracellular antibiotics used for other persister bacteria. The goal of this paper is to demonstrate the effect of pyrazinamide (PZA), another mycobacterium drug, in combination therapy as an effective treatment for Lyme disease, autoimmune phenomenon and associated intracellular co-infections.
CASE PRESENTATION
The patient is a 49-year-old white female with a past medical history significant for Lyme disease, multiple viral infections including HSV-2, HHV6 and EBV, fibromyalgia, and rheumatoid arthritis. She was diagnosed 19 years ago by a rheumatologist after complaining of chronic pain all over her body, described as being severe, bilateral and symmetric in nature. She had previously seen three rheumatologists, and been on multiple DMARD regimens including Enbrel (etanercept), Arava (leflunomide), Trexall (methotrexate), Remicade (infliximab), and prednisone. She failed all of the above regimens, including her present regimen of Imuran (azathioprine) 50 mg, 2 PO QD with Plaquenil (hydroxychloroquine) 200 mg, one PO BID, complaining of chronic severe pain and joint swelling. She was also on Ultram (tramadol) 50 mg, 1 to 2 PO TID, prn for her joint pain, with Cymbalta (duloxetine) 60 mg per day for her neuropathy and fibromyalgia symptoms. Despite these medications, and frequent intermittent use of prednisone, she still complained of severe pain in multiple joints, with neuropathic symptoms in her extremities. Other symptoms included persistent fatigue, occasional chills, flushing, and sweats, chest pain, cough, and disturbed sleep.
The patient had been seen by dermatology years ago, for multiple, red, painful skin nodules present over the flexor surfaces of her hands. A skin biopsy returned as positive for Winkelman’s granulomas [18,19], of unknown etiology. Churg-Strauss granulomatous vasculitis [20] was considered in the differential diagnosis, but she had no history of allergic rhinitis, asthma, or eosinophilia, ruling out the diagnosis. She also had large recurrent ulcers throughout her mouth, palate and tongue, in the genital area (anus, labia), as well as papulo-pustular lesions which would come and go on her extremities, consistent with Behçet’s syndrome [21]. She was using Imuran, colchicine, magic mouth wash (diphendyramine 12.5mg/5 ml; hydrocortisone 60 mg; Nystatin suspension 30 ml), and prednisone intermittently, without any clinical benefit.
Family history was positive for hypertension and heart disease, but no history of familial autoimmune illness. Social history was positive for having given up smoking several years ago (one pack per day for several years), with minimal caffeine and alcohol use. She lived in a known epicenter for tick-borne illness, but did not remember any tick bites or rashes consistent with an erythema migrans rash. She had a pet rabbit, but there was no known wild animal exposure. Review of systems was positive for chronic fatigue, sweats and chills, hot flashes with infrequent, heavy periods, unexplained hair loss and weight loss (several pounds), a recent cough that was nonproductive, intermittent chest pain, and joint pain throughout her body. She now complained of stiff, hot, red swollen joints, with associated bilateral edema of the lower extremities.
Physical examination revealed a well-developed, well-nourished white female in no apparent distress. She had a low-grade temperature of 99.7 degrees Fahrenheit, with a normal BP, pulse and respiratory rate. Initially, there were no orthostatic changes going from a sitting to standing position, although mild symptoms of dysautonomia developed over time, with a drop in her BP from 106/69 sitting to 89/71 standing, with an associated mild tachycardia. HEENT revealed multiple deep, large ulcerations on her inner lips, upper palate, and tongue, without any signs of eye inflammation. Her neck was supple with a normal thyroid, carotids were 2+ bilateral and symmetric with no bruits, and there was minimally enlarged axillary lymph node. Cardiovascular, pulmonary and abdominal examination was WNL; Lower extremities revealed 1+ edema, pulses were 2+ bilateral and symmetric, and her neurological exam was intact except for a slightly ataxic gait. Skin examination revealed rheumatoid changes of her hands and feet, with hot, swollen joints, with associated inflammation in the right knee. Granulomas were present bilaterally over the extensor surfaces of her fingers, as well as her elbows and forearms, painful to the touch.
Laboratory values revealed a negative Borrelia burgdorferi C6 ELISA, but positive CDC IgM Western blot [22], a low positive Borrelia hermsii IgG IFA (1:64 +, normal less than 1:64), possible prior exposure to tularemia, with a low level titer at 1:20 +, prior exposure to Mycoplasma pneumonia (IgG antibody, positive at 1.21, normal less than 0.90), a slightly elevated IgM level at 276 mg/dL (normal levels are below 271 mg/dL; she was subsequently ruled out for Waldenström’s macroglobulinemia); positive autoimmune antibodies, including a positive ssDNA IgG AB (80 u/mL, normal less than 69 u/mL), positive double-stranded DNA at 10 IU/ ml (normal levels less than 4), positive anti ganglioside antibodies (Anti-GM1, positive at 1:800; normal less than 1:800) consistent with an immune mediated neuropathy, and a positive anti-myelin antibody, with significantly elevated levels of inflammatory markers including an elevated CRP (12.3, normal less than 1 mg/dL), hs-CRP (49.4 mg/L, normal less than 1.0 mg/L), sedimentation rate (ESR) at 68 mm/h (normal less than 26 mm/h), with a mildly elevated platelet count at 431,000 (normal limits between 144 and 400,000/uL). She had borderline anemia (hemoglobin 12.6, normal greater than 13.2 g/dL; hematocrit 37.8, normal greater than 38.5) with normal iron/ferritin levels, consistent with inflammation and anemia of chronic disease. S. cerevisiae (ASCA) IgA and IgG levels were normal. A comprehensive metabolic panel was within normal limits, as was her urine micro albumin. ANA, rheumatoid factors, cyclic citrullinated peptides (CCP), Sjögren’s AB’s (SS-A, SS-B), CPK and complement studies were within normal limits (except for an isolated increase in CH50), as were blood levels of mercury, lead, and arsenic. Serum antigliadin antibodies and tissue transglutaminase levels (TTG) were also negative, ruling out celiac disease or gluten sensitivity as a cause of her skin lesions or neuropathy. HLA-DR4 by flow cytometry was not detected.
Other abnormal testing included mineral deficiencies, i.e., low iodine (35, normal between 50 to 109 mcg/L) and low zinc (57, normal levels between 68-161); other vitamin levels were normal (i.e., B12, folic acid and methylmalonic acid levels), but she was MTHFR positive with an elevated homocysteine level at 12, and history of sick euthyroid syndrome with normal T4 and TSH, with an isolated low T-3. She was in perimenopause with irregular menses, with an elevated LH, but normal FSH, progesterone and estradiol levels (ruling out significant estrogen deficiency as a cause of her night sweats). Tick-borne testing including Babesia microti and WA-1/duncani, Ehrlichia, Anaplasma, Rocky Mountain spotted fever, Q-fever and Brucella titers were negative, as was IgE food allergy testing. Adrenal testing, as well as viral titers and PCR’s were ordered for her chronic fatigue, and a Vitamin D level and DEXA scan was ordered to check her bone density, since she had intermittently been on prednisone for many years. HHV-6 titers were initially 1:320 (normal less than 1:10) with a negative PCR, and EBV VCA AB IgG was 4.99 (normal less than 0.91) with a negative PCR, consistent with prior exposure to both infections. Coxsackie A10 antibodies were low positive at 1:8 (normal less than 1:8), with prior exposure to HSV-2 (IgG greater than 8, normal less than 0.9 AI). DHEA/ cortisol testing revealed adequate cortisol levels. The Vitamin D level returned WNL, but the DEXA scan returned positive for severe osteoporosis.
A chest X-ray and CT angiography of the chest was done with contrast to evaluate her chest pain and cough. Mildly enlarged bilateral axillary lymph nodes were seen with a normal heart, pericardium, and lung parenchyma. X-rays bilaterally of her hands revealed degenerative changes in the bilateral radiocarpal and first carpometacarpal joints with scattered lucencies in the bilateral carpal bones and distal radius, consistent with osseous erosions related to inflammatory arthritis. Her X-ray of the left hip revealed severe osteoarthritis and early osteonecrosis of the left femoral head, related to frequent steroid use over the past several years.
The patient was initially started on Minocin (minocycline) 100 mg PO BID, Plaquenil (hydroxychloroquine) 200 mg, one PO BID (both a Lyme disease and DMARD regimen), Nystatin 500,000 units, two PO BID, with a sugar-free diet and high dose probiotics. Occult Babesia was suspected since the patient complained of frequent drenching night sweats and chills with associated “air hunger” (which are classic symptoms of babesiosis). Malarone (atovaquone 250 mg/proguanil hydrochloride 100 mg) [23] an anti-malarial drug, was therefore added to her protocol, and was temporarily helpful in decreasing her sweats and chills. She had already been ruled out for other causes of drenching sweats, such as menopause, hyperthyroidism, brucellosis, non-Hodgkin’s lymphoma or tuberculosis. She was on Imuran, an immunosuppressive drug, known to be a risk factor for cancer and/or reactivation of latent intracellular infections like tuberculosis [24].
Multiple rotations of intracellular drugs were needed in the next few months, as the patient continued to complain of extreme fatigue, night sweats and chills, severe migratory joint pain and neuropathy of her extremities (consistent with active Lyme disease), which interfered with sleep. These drug regimens initially included double and triple intracellular antibiotic regimens, such as a tetracycline with Zithromax (azithromycin) 250 mg PO BID and Malarone, 2 PO BID, or tetracyclines (doxycycline 100 mg PO BID or minocycline 100 mg PO BID) with rifampin 300 mg PO BID, Bactrim DS (sulfamethoxazole/ trimethoprim) one PO BID and Malarone, with herbal treatments for biofilms [25], and anti malarial herbs including liquid Cryptolepis sanguinolenta 20 drops PO BID, and capsules of Artemisia annua [26,27] 250 mg PO TID. There was only a partial response in symptoms, with a slight decrease in joint pain and swelling, but no change in her severe fatigue or poor sleep. She also continued to develop new oral ulcers on this regimen with painful granulomatous lesions of her extremities, with elevations in inflammatory markers, including rising sedimentation rates (ESR 63), and high levels of hs-CRP (35.3). She was rotated to a different regimen including Omnicef (cefdinir), with Plaquenil and Zithromax, but it had little effect on her symptoms. Low-dose naltrexone (4.5 mg HS) [28], in combination with Lyrica (pregabalin, 200 mg HS), Voltaren (diclofenac) 75 mg, one PO BID with meals), with Cytotec (misoprostol), 200 µg one PO BID, was therefore added to help with fibromyalgia symptoms, pain, neuropathy and inflammation (cutting back on tramadol use), while her rheumatologist increased her Imuran to 50 mg TID to try and decrease inflammation and cytokine production. Unfortunately, the patient was still not significantly better.
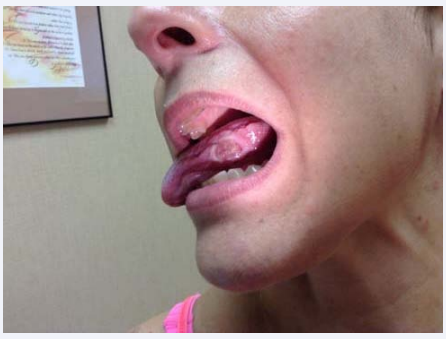
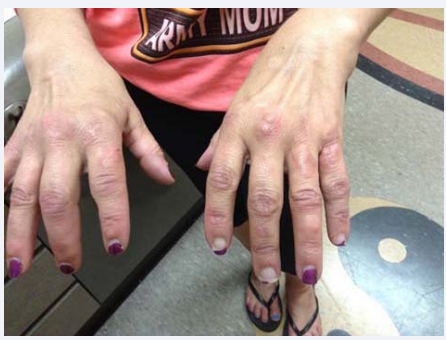
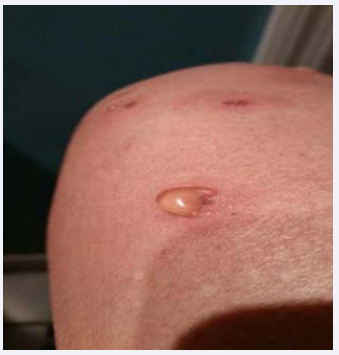
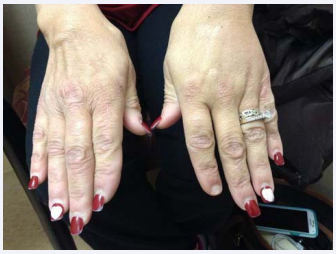
Due to the lack of significant progress, during her medical visit of December 2014, the patient was placed on a quinolone, Levaquin (levofloxacin), 500 mg PO QD in combination with her prior drug regimens. After 2 months on this regimen, her malarial symptoms diminished, with a decrease in inflammatory markers (ESR decreased from 50 to 38; CRP decreased from 39 to 1.6), but there was no major shift in her systemic symptoms, and none of these regimens significantly affected her mouth ulcers or painful granulomas of the extremities. Levaquin was therefore discontinued, and Dapsone, 100 mg PO QD, with leucovorin 5 mg per day, with extra folic acid was added to her minocycline, rifampin, and Malarone. Dapsone is known to be effective in treating Behçet’s syndrome [29]. When the patient returned 2 months later, her sweats had decreased to twice a month from once a day; there was no further air hunger; there were less frequent and severe neuropathic symptoms; and she had a slight decrease in her joint pain. Her clinical response was the best it had been in the past year (and was the best since starting DMARD regimens back in 1999). She still complained however of painful ulcers on the side of her tongue (Figure 1), as well as swollen joints and painful granulomas (Figure 2), and blistering lesions which appeared all over her body (Figure 3).
Figure 1 Large Behçet’s ulceration on the tongue.
Figure 2 Granulomas and swelling of the joints (before Minocycline, Rifampin and Pyrazinamide).
Figure 3 Papulo-pustular lesions.
Although the patient’s clinical response was improved from prior regimens, where her functional status improved from 30% to 50% of normal (with Borrelia hermsii titers turning negative), there was a sudden fourfold increase in her tularemia titers, going from 1:20 to 1:320, with Bartonella titers turning positive (Bartonella henselae, 1:64 +) with a simultaneous increase in vascular endothelial growth factor (VEGF) [30], a biochemical marker for active vasculitis (known to be associated with Bartonella). VEGF levels were previously undetectable, and were now 96 (normal less than 86 pg/ml). Her prior ESR which had temporarily decreased to 37, was now 69 mm/h, antiganglioside antibodies increased from 1:800 to 1:3200, and HHV-6 titers rose temporarily to 1:1280 (a fourfold rise). It appeared that the Dapsone, although helping to decrease her symptoms, was unmasking underlying dormant infections. We added Avelox (moxifloxacin) 400 mg PO QD to her doxycycline 100 mg PO BID, rifampin 300 mg PO BID, Plaquenil 200 mg PO BID, and Dapsone 100 mg PO QD (a four drug intracellular combination), and she was sent to an infectious disease (ID) physician for a second opinion regarding IV therapy (streptomycin, gentamicin), since she had already been on a triple intracellular combination of a tetracycline, rifampin and Levaquin, which has known efficacy against tularemia. She was immune compromised, and might require more aggressive therapy. The ID physician left the regimen in place, and felt that IV treatment was unnecessary, since tularemia titers subsequently decreased from 1:320 to 1:80 one month later, further decreasing to 1:40 and then 1:20 positive (baseline) titers over the next few months.
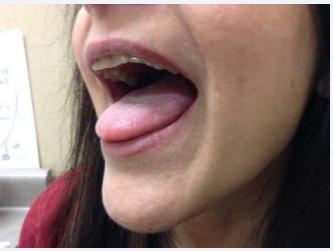
By December 2015, the patient had made continuous progress with some systemic symptoms, and was off the Avelox, but still had symptomatic joint pain interfering with sleep. She still had ongoing severe dermatological manifestations of blood-filled blisters, oral/genital ulcerations, and increased granulomas appearing everywhere except her chest and back. She was placed on a dairy free, gluten free diet, since the patient reported increased dermatological symptoms when she was off her diet during her honeymoon (despite negative celiac testing [31] and IgE allergy profiles), along with a trial of 2000 mg of IV glutathione, to assist as an antioxidant/anti-inflammatory/detoxification support. This was partially effective in helping decrease pain, swelling and fatigue, but was inadequate to relieve her worst symptoms. Due to her recalcitrant course, and prior positive response to Dapsone (an intracellular mycobacterium drug used for leprosy and Behçet’s) [32], we discussed starting her on pyrazinamide, (PZA) [33], another intracellular mycobacterium drug, with Plaquenil, minocycline and rifampin, since no regimen had been effective in clearing the ulcers and painful granuloma formation. The rheumatologist wanted to place her on Rituxan (rituximab) for her resistant RA and granulomatosis with poly angiitis, but since she previously reactivated intracellular infections while on an immunosuppressive agent, we chose to first try 500 mg of PZA, 2 PO QD (based on her body weight), combined with rifampin and minocycline as her last option. We monitored her liver function every two weeks and provided liver support with alpha lipoic acid 600 mg PO BID and milk thistle 250 mg PO BID. To our surprise, during the 2nd month on the regimen, she had a significant clinical improvement. She reported being up to 80% of normal functioning, where she started at 20% to 30% during her initial consultation, with improvement in her most resistant dermatological symptoms. The oral ulcers had finally resolved (see Figure 4), the granulomas were breaking up on her skin (see Figure 5), and according to the patient, it was the best medication protocol she had tried in the past 20 years.
Figure 4 Clearing of tongue ulcers.
Figure 5 Decreased joint pain/granulomas.
The patient’s liver functions remained WNL during two months on the protocol, and since she was continuously improving, we left her on the PZA for one additional month. Her sweats, chills, fatigue, shortness of breath, swollen glands, joint pain, stiffness, and poor balance improved, and she was taken off PZA during her office visit of March, 2016.
The patient underwent a left hip replacement for osteonecrosis of the femoral head, and was placed on Plaquenil 200 mg PO BID, Minocin 100 mg PO BID, Rifampin 300 mg PO BID, Bactrim DS one PO BID and Nystatin 500,000 U PO BID, with high dose probiotics post-op. She was anemic and zinc levels were low at 61 ug/dl post-op (normal greater than 68 ug/dl), so she was prescribed a multi mineral supplement with iron and zinc. Within seven to ten days off the pyrazinamide she did have a return of papulo-pustular lesions in between her toes, plantar surfaces of her feet, legs, buttocks and face, with a minor return of tongue ulcerations (but still greatly improved). However, the lesions were healing by the time of her next office visit, and her systemic symptoms were otherwise stable and improved. Her ESR had decreased to 45 mm/h, and we were able to lower her Imuran dose. The mycobacterial combination drug protocols were the most effective for her resistant Lyme and autoimmune symptoms, with PZA being the most efficacious for the dermatological manifestations of Behçet’s and arthritic/ granulomatous changes.
DISCUSSION
The rising incidence of multiple tick-borne infections, such as Lyme, other Borrelia species, Babesia, Bartonella [34], and tularemia [35,36], requires a high index of suspicion in any patient with an unexplained chronic fatiguing, musculoskeletal illness. Unfortunately, the insensitivity of the present two-tiered testing for Lyme and other Borrelia species (like B. miyamotoi), using an ELISA followed by a Western blot, as well as the inability of the newer C6 Lyme ELISA [37,38] to pick up all positive cases, can lead to inaccurate diagnoses [39]. This patient had a negative C6 ELISA and a CDC positive Western blot indicating prior infection. There are multiple species of Borrelia in the United States, and the lack of a gold standard for their diagnosis makes producing accurate statistics difficult. Some pathogenic strains belonging to the B. burgdorferi sensu lato complex have a worldwide distribution, yet they are rarely considered or tested for [40]. The same testing issues exists for Babesia and co-infections like Bartonella, where standard lab testing lacks sufficient sensitivity to pick up all pathogenic species, leading to inaccurate diagnoses [41,42]. Several of these bacteria like Bartonella are stealth intracellular/intravascular pathogens, avoiding immune surveillance. That was initially the case for this patient, although later in her clinical course, she reactivated several intracellular infections while on immunosuppressive therapy.
Multiple bacterial and parasitic tick-borne infections in the same host may not only be difficult to detect, but can significantly increase comorbidity. Human babesiosis, for example, has multiple species [43] which may be transmitted at the same time as a tick bite causing Lyme disease [44], increasing the duration and severity of symptoms, [45, 46] but do not always cause classic malarial type presentations, such as frank hemolytic anemia, and/or thrombocytopenia and elevations in liver functions. It should be suspected in splenectomized and otherwise immune compromised individuals with severe febrile illnesses, but can also be found in those with unexplained drenching night sweats, day sweats and chills with respiratory complications [47,48]. Babesia is able to establish a chronic infection in the animal population [49], and has been found to persist in humans after standard antibiotic therapies [50-52].
Tularemia can also present with one of several different clinical presentations and persist in the human host. Although the patient did not have classic clinical manifestations of tularemia, i.e., the ulceroglandular, oculoglandular, typhoidal or pneumonic forms [53], it is highly likely that the combination of immune suppressive drugs and multiple overlapping infections like Bartonella altered her clinical presentation.
Patients with Bartonella have a broad range of clinical manifestations resembling other diseases, the result of over 30 different species/subspecies; at least 18 are implicated in human infections [54]. Bartonella are Gram-negative bacteria that are known to be transmitted by not only ticks, but also fleas, lice, mites, and biting flies. These include B. henselae (ticks), the agent of cat scratch fever, B. quintana, the agent of trench fever (lice), and B. bacilliformis, i.e., Carrion’s disease, transmitted by sand flies. New Bartonella species like B. koehlerae have recently been identified, and antibodies against Bartonella henselae, B. koehlerae, or B. vinsonii subsp. berkhoffii was found to be high in Lyme disease (46.6%), arthralgia/arthritis (20.6%), chronic fatigue (19.6%), and fibromyalgia (6.1%) implying a possible causal role [55]. Bartonella species are also known to cause rheumatologic manifestations resembling RA and Lupus, with associated myositis, osteomyelitis and systemic vasculitis. Immunocompromised patients are at risk for severe, life threatening illness [56]. No single treatment has been shown to be effective for all Bartonella-associated disease.
Published research demonstrates that the effects of co-infections in humans is diverse and complex [57], and there is a lack of scientific consensus as to why many patients go on to develop chronic symptoms despite standard courses of treatment for Lyme and co-infections. One hypothesis regarding PTLDS, is that persistent infection with Borrelia burgdorferi, apart from autoimmune phenomenon and tissue damage, is underlying chronic symptomatology. Evidence of the persistence of Lyme disease comes from xenodiagnostics studies [58] and evidence that bio film colonies provide a protective niche for Borrelia species [59,60]. Research also demonstrates that Borrelia can form persister cells [61-64] while able to avoid immune surveillance through changing outer surface proteins through gene recombination [65], and avoid complement mediated killing [66], hiding in the intracellular compartment [67,68]. Other persister bacteria exist as intracellular infections like TB, leprosy, and Q-fever, and treatment failures may be due to using intracellular drugs not known to be effective in treating persister bacteria, like mycobacterial infections. Bartonella, an intracellular infection, is known to persist with bacteremia, potentially spanning decades in duration, even in immune competent hosts [69]. Some of these bacteria, such as Lyme and Bartonella, also cause autoimmune manifestations, increasing arthritic complications in rheumatologic diseases [70,71], while causing a vasculitis (Borrelia, Bartonella) [72,73], and associated granuloma formation (Bartonella, tularemia) [74,75].
Many pathogenic bacteria are able to persist, driving inflammatory processes and chronic disease, and are found in the intracellular compartment [76]. One example is Mycoplasmas, which like Lyme disease [77], have been shown to interact non-specifically with B-lymphocytes resulting in the modulation of immunity promoting autoimmune reactions and rheumatoid diseases [78,79], and are able to increase pro-inflammatory cytokines including IL-1, 2, and 6. Bartonella species also can cause arthritis with polyclonal B-cell activation, commonly found in patients with rheumatologic or chronic inflammatory diseases [55]. Our patient had evidence of exposure to Lyme disease, relapsing fever Borrelia (B. hermsii), Mycoplasma spp., tularemia and Bartonella, with probable Babesiosis. These infections may interact and increase inflammation, leading to joint pain and elevated inflammatory markers (CRP and ESR) in a patient with an autoimmune illness. This patient also had low zinc levels, which can increase the production of inflammatory cytokines and impair the ability to fight chronic infections [80] but it was not until the underlying infections were treated with PZA, that the inflammation and Behçet’s ulcerations started to resolve.
Bartonella have been associated with chronic bacteremia and/or chronic endocarditis, and one of the classic manifestations is bacillary angiomatosis [81,82]. The cutaneous lesions of bacillary angiomatosis are papules or subcutaneous nodules with ulcerations, where Bartonella species cause a systemic vasculitis secondary to increased oxidative stress affecting blood vessels. The main mechanism of oxidative stress-induced angiogenesis involves hypoxia-inducible factor/vascular endothelial growth factor (VEGF) signaling [83], which was seen in this patient, who had evidence of increased oxidative stress (increased homocysteine, ESR, CRP) and elevated levels of VEGF. The granulomas, papules and ulcers present eventually responded to antibacterial therapy.
Bartonella is also able to cause inflammation in the eye, including uveitis and retinitis, with a broad range of multi systemic manifestations, including various rheumatological, neurological [84], cardiovascular, and gastrointestinal manifestations [85]. These are some of the same clinical manifestations seen in Behçet’s syndrome, which results in a vasculitis with ulcerations, with associated cardio-pulmonary disease (pericarditis, pleuritis, cough), ophthalmological problems (uveitis), G.I. problems (nausea, pain, diarrhea), arthritic and neurological problems (chronic meningoencephalitis, balance problems) with accompanying systemic manifestations of severe fatigue [21]. Is it possible that Bartonella species are in part responsible for Behçet’s disease? The patient’s clinical response to multiple intracellular medications, especially pyrazinamide, in combination with a tetracycline and rifampin, implies one or more intracellular infections as the primary etiology.
Behçet’s disease was named in 1937 after the Turkish dermatologist who first described the classic symptom complex, but it was previously referred to as “Silk Road Disease”, since an increased prevalence was seen in the areas surrounding the old silk trading routes in the Middle East and Central Asia [86]. The higher prevalence of Behçet’s in those areas implies either genetic (HLA B-51) and/or environmental precipitating factors. Bartonella species, such as B. quintana are highly endemic in those geographic areas; B. quintana induces the same lesions seen in bacillary angiomatosis; and parasites that are endemic to the Middle East [87] could hypothetically be associated factors with Bartonella in the etiology of Behcet’s disease, since leishmaniasis, caused by protozoan parasites and spread by the bite of certain sand flies (like Bartonella) can present with cutaneous, mucocutaneous, or visceral forms, all of which all involve ulcer formation, which can involve the skin, mouth, and nose [88,89]. Dapsone, effective in Behçet’s disease, has both antibacterial and anti-parasitic properties. We recently demonstrated that Dapsone has clinical efficacy against Borrelia burgdorferi (a bacteria) as well as Babesia species, which are intraerythrocytic parasites [16]. Multiple overlapping infections may therefore be responsible for Behçet’s disease, which is presently of unknown etiology. Advanced microbiological techniques were unavailable years ago to determine if an infectious cause was present. Sulfa drugs have been used with benefit in those with other granulomatous illness like Wegener’s granulomatosis [90]. Dapsone, which inhibits bacterial synthesis of dihydrofolic acid, via competition with PABA (like other sulfonamide antibiotics), can be helpful in some patients with Behçet’s syndrome [17], again implying an infectious etiology, and similarly, Bartonella species which cause granulomas, are usually responsive to sulfa antibiotics.
Bacterial infections are one known cause of autoimmune disease. Lyme disease, a multi systemic illness like Bartonella, can be associated with a vasculitis, and autoimmune manifestations [91-101]. Rheumatoid factor correlates with antibody titers against Borrelia species, such as Borrelia garinii (a European geno species of Borrelia) [102] 57% of patients with RA in some studies show reactivity to multiple Borrelial antigens [93], and Lyme arthritis, a disease caused by a bacterium, can mimic RA [94]. Inflammatory cytokine production with elevations in TNF-alpha, IL-1, IL-6, as well as elevations in chemokines (CXCL-9, CXCL-10, CCL-19) [103,104] with autoantibody production, especially with molecular mimicry against neural tissue [105], are also a known feature of the infection. Overlapping bacterial infections can also increase inflammation in an autoimmune illness. Low dose naltrexone, which has been used in bacterial infections (Lyme), fibromyalgia and other autoimmune diseases [28], such as Crohn’s disease [106] and MS [107], may also have been of benefit in this patient in controlling cytokine production. It should be considered as an immune modulator in those with AI disease. Immunosuppressive therapies like Imuran and prednisone, although necessary and lifesaving in some autoimmune illnesses, may cause reactivation of underlying infections, with other potential side effects, like osteonecrosis of the hip, seen in this patient.
Animal and human data suggest that bacterial organisms also play some role in the initiation and propagation of autoimmune disease like RA, as well as other forms of arthritis, such as osteoarthritis. Bacterial cell wall components, i.e., lipo polysaccharides (LPS) appears to up regulate inflammation in cartilage (chondrocytes) [108], and LPS appears to trigger the NF-kappa B pathway of inflammation through interactions with toll-like R4. FSTL-1 is also a protein/immune modulator, induced by an infection with Borrelia burgdorferi, with an established role in autoimmune arthritis [109], and depending on the geno species of Borrelia [110] we are infected with, and our own HLA subtype, (HLA-DR 2, 4) [111,112], there are varied inflammatory manifestations.
Associated viral infections and environmental triggers may also play a role in autoimmune (AI) disease. CMV antibodies have been found more frequently in patients with RA, varicella virus was recently shown to trigger giant cell arteritis [113,114], and MS patients may have reactivation of EBV infections as an underlying trigger [115]. Our patient reactivated HHV-6 and had evidence of prior exposure to EBV. BPA, an endocrine disruptor, also may provoke autoimmune reactions, causing molecular mimicry [116], and small particle pollution has recently been shown to be a strong environmental risk factor for RA, JIA, Lupus, and scleroderma [3]. Environmental toxins increase IL-17, which is a pro-inflammatory cytokine seen with Lyme and other AI illness [117]. One of the disabling manifestations of Lyme and associated co-infections (such as Bartonella) is severe neuropathy, which can be worsened with environmental toxins, such as mercury, lead, arsenic, and BPA. Our patient’s neuropathy improved with both Dapsone and pyrazinamide and inflammatory manifestations were also improved with IV glutathione. The MSIDS model of chronic disease, described by the first author [118], is a multi factorial model for chronic illness, where several overlapping factors, including chronic infections and environmental toxins, can increase disease manifestations. The interaction of multiple bacteria (Borrelia, Bartonella, and tularemia), parasites (Babesia), viruses (HHV-6, Coxsackie), environmental and genetic/epigenetic factors can influence both the appearance and severity of inflammation, chronic disease and autoimmune illness [119].
The four-fold rise in tularemia titers, with simultaneous evidence of other relapsing infections (new Bartonella titers with an elevated VEGF, and elevations in HHV-6 titers) with a positive clinical response to a tetracycline, rifampin, Dapsone and a quinolone (Avelox) with tularemia titers returning to baseline, argues for multiple latent infections. Lyme, Bartonella and other tick-borne diseases are known to relapse in immune suppressed individuals [120,121]. Our patient was on Imuran, an immunosuppressive agent, and had multiple tick-borne infections (some of which are known to also have an immunosuppressive effect) [122,123], causing particularly severe clinical manifestations.
Tularemia is a particularly recalcitrant and relapsing disease if not caught early in the course of the illness. Treatment options include doxycycline, [124] quinolones (ciprofloxacin, levofloxacin), and chloramphenicol, with IM or IV streptomycin or gentamicin for severe resistant disease (which are potentially nephrotoxic and ototoxic). Although some research supports the use of fluoroquinolones to treat tularemia, clinical experience and in vitro data regarding the efficacy of quinolones are limited, and relapse rates of up to 50% have been reported with these agents [125,126]. The patient had a positive clinical response to Plaquenil with a four drug intracellular antibiotic combination, i.e., Avelox (a fourth generation quinolone), doxycycline, rifampin, and Dapsone, despite being on immunosuppressive medication (Imuran). Whether this novel combination of antibiotics could be an effective treatment for tularemia requires further investigation. Our prior experience with Dapsone [16], is that it was efficacious in the treatment of Lyme disease and Babesiosis, when combined with other intracellular antibiotics. The combination of Dapsone with a quinolone, tetracycline and rifampin, appears to have similarly increased the drug’s efficacy for tularemia in this patient. Plaquenil, which was part of the protocol, also seems to be an important component in helping those suffering with Lyme, intracellular co-infections, and autoimmune manifestations. It has an effect in both Lyme [127] and lupus [128], while alkalizing the intracellular compartment, increasing the efficacy of other intracellular antibiotics. It was first proven to be effective in Q-fever, another intracellular persister bacterium [129].
Pyrazinamide may be another option in our antibiotic arsenal for persistent tick-borne infections and autoimmune manifestations. The patient in our case report had failed a cephalosporin (Omnicef), tetracyclines (doxycycline, minocycline), rifampin, Bactrim, and quinolones (Levaquin) for her tick-borne co-infections, while inadequately responding to multiple DMARD regimens, as well as Dapsone for her Behçet’s and vasculitis. Despite meeting the established international criteria for Behçet’s syndrome (oral apthtous ulcers, which were present at least three times in 12 months, genital ulcers, and skin lesions which were papulo-pustular in nature) [21] and taking established therapies for Behçet’s, it was not until pyrazinamide was prescribed that she had a positive clinical response.
Pyrazinamide’s known biological mechanism of action in infections includes disruption of M. tuberculosis membrane transport and energetics by pyrazinoic acid, with both bacteriostatic and bacteriocidal effects [130]. It is a key sterilizing drug in first-line tuberculosis (TB) regimens and exerts its activity during the first two months of treatment in human infections [131]. Should PZA be considered in other difficult to treat, chronic infections leading to high rates of morbidity and mortality? PZA is a critical drug for treating persistent TB infections, by killing persisters (Yin) as in the Yin–Yang model of persisters and latent infections described in 2014 by Zhang [61]. In a growing population of bacteria, there is a small population of non-growing or slowly growing persisters, which may revert to a small number of growing bacteria (reverters). Combinations of multiple intracellular and extracellular antibiotics, which address both actively growing bacteria and non-growing, or slowly growing persisters, while addressing biofilms and other known bacterial forms, may be necessary to fully eradicate underlying pathogens. Due to the potential side effects of PZA, including possible liver toxicity at high doses [132], and the potential for resistance [133], we need further clinical prospective research on the efficacy of PZA and other persister drugs in severe chronic rheumatologic and autoimmune disease. Although our patient had no elevation in LFT’s on PZA, we recommend further investigation on the benefits of agents that can protect the liver, like milk thistle and alpha lipoic acid [134,135].
Conclusion: This case report on the use of mycobacterium drugs (Dapsone, PZA) in Lyme, associated co-infections, and Behçet’s syndrome implies that there may be a broader utility for these drugs in persistent intracellular infections with autoimmune manifestations. The Dapsone and PZA protocols were the most effective for resistant Lyme and autoimmune symptoms, with PZA being the most efficacious for the dermatological manifestations of Behçet’s and arthritic/granulomatous changes. Although this is a single case report on the use of PZA, it models and extends our previous study in which Dapsone was clinically effective in a cohort of 100 individuals with Lyme and associated tick-borne infections. Large prospective studies need to be undertaken to evaluate the efficacy and safety of mycobacterium drugs in multiple chronic infectious diseases.
ACKNOWLEDGEMENTS
We thank our patient for allowing us to share her case and Sonja Siderias, LPN for her expert research assistance.
REFERENCES
1. Selmi C. The worldwide gradient of autoimmune conditions. Autoimmun Rev. 2010; 9: A247-A250.
3. Pfau JC, Serve KM, Noonan CW. Autoimmunity and asbestos exposure. Autoimmune Dis. 2014; 782045.
4. Centers for Disease Control Lyme disease Data and Statistics.
8. National Science Foundation Special Report: Ecology of Emerging Infectious Diseases.
13. Tularemia: News & Perspectives Medscape.
15. Horowitz RI. Why can’t I get better? Solving the mystery of Lyme and chronic disease. 2013.
32. WHO model prescribing information: Drugs used in leprosy. 1998.
35. Education Key to Stop Tick-Borne Illnesses (White Clothes Help Too). 2016
36. Colorado health officials: Tularemia cases on record-breaking pace. Jesse Paul. 2015.
43. Centers for Disease Control Laboratory diagnosis of babesiosis: Babesia species.
46. Mylonakis E. When to suspect and how to monitor babesiosis. Am Fam Physician. 2001; 63: 1969-1975.
54. Birtles RJ. Bartonellae as elegant hemotropic parasites. Ann NY Acad Sci. 2005; 1063: 270-279.
64. Lewis K. Persister cells. Ann Rev Micro. 2010; 64: 357-372.
75. Asano S. Granulomatous lymphadenitis. J Clin Exp Hematopathol. 2012; 52: 1-16.
83. Kim Y-W, Byzova TV. Oxidative stress in angiogenesis and vascular disease. Blood. 2014; 123: 625-631.
87. Rand Monograph: Parasitic diseases.
88. Leishmaniasis Fact sheet N°375. World Health Organization. 2016.
103. Steere AC, Glickstein L. Elucidation of Lyme arthritis. Nat Rev Immunol. 2004; 4: 143-152.
125. WHO. A brief guide to emerging infectious diseases and zoonoses. 2014.