Berberine Induces Pyroptosis by Promoting the NLRP3/ Caspase-1/ GSDMD Pathway in Papillary Thyroid Carcinoma
- +. These three authors contributed equally to this work.
- 1. Department of Endocrinology and Metabolism, Central Theatre General Hospital, China
- 2. Department of Geriatrics, Guangzhou Dongsheng Hospital, China
- 3. The First School of Clinical Medicine, Southern Medical University, China
- 4. School of Management, University College London, England
- 5. Department of Dermatology , First People’s Hospital of Wuxue, China
- 6. Department of Emergency, The 987th Hospital of the Joint Logistics Support Force, China
Abstract
Background: PTC (Papillary thyroid carcinoma) is a complex disease. We previously found that BBR (Berberine), an effective component of Chinese herbal medicine Coptis, has an inhibitory effect on PTC. The exact process is not understood, possibly linked to pyroptosis controlled by NLRP3 (Nod-like receptor protein 3). The purpose of our research was to investigate the effects of pyroptosis in BBR treatment of PTC.
Methods: PTC cell lines with different genetic backgrounds, such as BRAF(V600E) mutant (K1, BCPAP) and wild-type (TPC-1) PTC cells, were utilized in in vitro experiments. Pyroptosis was determined using in situ staining with Hoechst 33342 and PI. Levels of protein and mRNA were determined using western blotting and qRT-PCR. The IL-1β (interleukin-1β) and IL-18 levels were determined using ELISA kits. In order to measure ATP and LDH release, corresponding kits were used. The effectiveness of PTC cells in proliferation was assessed using a colony formation assay. BALB/c nude mice were utilized to create PTC xenografts.
Results: During in vitro experiments, BBR was found to suppress proliferation and promote pyroptosis in both BRAF(V600E) mutant and wild-type PTC cells. NLRP3, caspase-1, GSDMD (gasdermin D), L-1β and IL-18 were up-regulated by BBR. Moreover, NLRP3 activated caspase-1 to promote GSDMD expression. NLRP3 and GSDMD knockdown attenuated BBR mediated pyroptosis in PTC cells. Further, BBR activated the NLRP3/caspase-1/GSDMD pathway and induced pyroptosis. The formation of PTC transplantation tumor was significantly inhibited by BBR in vivo experiments.
Conclusions: As a result of our findings, BBR inhibits tumourigenesis in PTC cells of different genetic backgrounds by inducing pyroptosis, suggesting a promising therapeutic approach for tumours.
Keywords
• Berberine
• Papillary thyroid carcinoma
• Pyroptosis
• NLRP3
• GSDMD
Citation
Huang X, Lu Y, Li S, Chen W, Zheng Q, et al. (2026) Berberine Induces Pyroptosis by Promoting the NLRP3/Caspase-1/ GSDMD Pathway in Papillary Thyroid Carcinoma. JSM Biochem Mol Biol 10(1): 1040.
INTRODUCTION
The incidence of PTC worldwide has increased significantly, and the age-standardized incidence of PTC has increased rapidly compared with other subtypes [1,2]. Most of PTC is well differentiated, less invasive and has a good prognosis [3,4]. At present, the main treatment for PTC is surgery, which may be suspected of overtreatment [5]. Drug therapy, including TSH suppression therapy, molecular targeted therapy and neoadjuvant chemotherapy, can cause sever adverse reactions [6]. There is an urgent need for more effective and safe treatments for PTC. In contrast to apoptosis, pyroptosis is a lytic, inflammatory programmed cell death [7]. The key characteristics of this condition include cell swelling, breakdown, creation of openings in the cytomembrane, and release of internal cell components such as LDH, IL-1β, and IL-18 [8]. Dying cells can start pyroptosis in two primary methods: caspase-1/4/5/11 controls GSDMD (gasdermin D) -dependent pyroptosis, and caspase-3 controls GSDME (gasdermin E) -dependent pyroptosis [9]. The hinge region between GSDMD’s or GSDME’s N- and C-termini is cleaved by activated caspases [9]. In the plasma membrane, the N-terminal fragment oligomerizes and pores are formed, leading to higher osmotic pressure and ultimately causing cytolysis [9-11]. Furthermore, the creation of pores in the cellular membrane enables the outflow of internal substances, along with the liberation of fully developed IL-18 and IL-1β to enhance the inflammatory reaction [10,11]. Studies have shown that gasdermin-induced pyroptosis makes a distinct anti-cancer effect in specific cancer treatments [12,13]. Thus, therapeutic approaches to induce pyroptosis are promising treatments for PTC. The NLRP3 inflammasome, made up of NLRP3, ASC, pro-caspase-1, and NEK7, initiates pyroptosis by activating caspase-1 [14-16]. Studies have shown that NLRP3 inflammasome deficient mice had increased susceptibility to rectal cancer, and up-regulation of NLRP3 to increase the level of IL-18 had inhibited tumour formation [17]. Higher purity of thyroid cancer cells in surgically resected tissues was associated with lower expression levels of NLRP3.Patients with low levels of NLRP3 expression have a worse prognosis and higher rates of recurrence [18]. However, further study needs to be conducted to evaluate the crucial role that NLRP3 plays in PTC. Berberine (BBR), an isoquinoline alkaloid, is mainly found in Coptis. BBR is usually used in the clinical treatment of gastrointestinal infectious diseases without obvious drug side effects [19,20]. Studies have indicated that BBR could be a promising treatment and prevention for many types of cancers. In hepatocellular carcinoma HepG2 cells, BBR can induce pyroptosis by up-regulating Caspase-1, and also inhibit the viability, migration and invasion [21]. Besides, in breast cancer MDA-MB-231 cells, BBR decreased the growth and movement, while also enhancing LDH release and lowering the levels of NLRP3, Caspase-1, and IL-1β in cells in a dose-dependent fashion [22]. Moreover, BBR could promote ROS production to enhance the level of oxidative stress, which subsequently increased the early and late apoptosis of PTC cells and eventually inhibited the progression of PTC [23,24]. ROS is one of the important activators of NLRP3 inflammasome [25]. The potential connection between BBR and the anti cancer effects could involve NLRP3/Caspase-1 induced pyroptosis. Although BBR has effects on various tumor cells, its specific mechanism of action in thyroid cancer remains unclear. Therefore, BBR needs to be investigated for its effects on pyroptosis of PTC. An investigation of the effects and mechanisms of BBR on PTC was conducted using BRAF(V600E) mutant type and BRAF wild type cell lines. We experimentally demonstrated the application of BBR to the treatment of PTC, and the study benefits the clinical status of PTC patients.
MATERIALS AND METHODS
Cell culture
The K1 cell lines of thyroid cancer in humans (BNCC337851) were acquired from the BeNa Culture Collection, while BCPAP, TPC-1, and human normal thyroid epithelial cells (Nthyori31) were purchased from the Hasen biotechnology company. The media used to culture the cells was RPMI 1640 (BL303A, Biosharp). The cells were cultured at 37°C in a high-humidity environment with 5% CO2.
Cell transfection of siR004EA
For NLRP3 and GSDMD knockdown, siNLRP3, siGSDMD, and siNC were acquired from Hasenbio.K1, BCPAP, and TPC-1 cells underwent transient transfection as per the manufacturer’s guidelines, followed by incubation in a 37°C, 5% CO2, and humidified environment. The impact of silencing was examined using western blotting and qRT PCR 48 hours later. The downregulation sequences were as follows:
si-NLRP3: 5‘-3’UAAUUCCAACACCUGAAGCTT,
si-GSDMD: 5‘-3’UAAUUCCAACACCUGAAGCTT.
Cell proliferation
Cell proliferation was assessed using a colony formation assay. The cells in the exponential growth phase were made into cell suspensions and diluted. 5ml of cell suspension was added to the culture dish based on the quantities of 50, 100, and 200 cells in each dish, then incubated at 37? in 5% CO2 for a duration of 2 weeks. The culture was stopped when macroscopic clones started to form in the Petri dish, and it was stained with crystal violet (Solarbio, G1062) staining solution for ten minutes before being observed.
Cell apoptosis
Flow cytometry was used to detect cell apoptosis. Cell cultures were exposed to BBR (Shanghai yuan ye Bio Technology Co) at varying concentrations (0, 20, 40, and 80 uM) for a period of 48 hours. Afterwards, the cells were gathered, washed with PBS, and then the Annexin V-FITC/ PI Apoptosis Detection Kit (Kechuang Da, KCD-T2008) was applied following the manufacturer’s instructions. As a final step, analyses were conducted using a flow cytometer.
Cell viability
Cell viability was evaluated using MTT assay. Cells were exposed to varying levels of BBR (0, 20, 40, and 80μM) and a 5% CO2 environment was used to culture the cells at 37 °C. 10μL of MTT was added to each well after 24, 48, and 72 hours, and incubated for 3 hours each time. At 450 nm, the absorbance was measured.
ELISA
ELISA assay was utilized to assess the impact of BBR on the production of IL-18(MEIMIAN, MM-0139H1) and IL-1β (MEIMIAN, MM-0181H1). Supernatants were collected by centrifuging the cells. Next, the specimens were placed into 96-well trays and left to incubate for 2 hours at ambient temperature. Incubation was carried out in accordance with the manufacturer’s instructions using substrates, enzymes, and antibodies sequentially added to the sample wells. We then added a stop solution to each well, followed by prompt measurement of OD at 450 nm.
ATP release assay
An ATP assay kit was used to measure ATP release (Nanjing Jiancheng, A095-1-1). In 96-well plates, cells were seeded for 24 hours. BBR was then added to fresh medium for incubation with the transfected cells. We collected and analyzed the cell culture medium. Following that, ATP detection reagents were applied according to the manufacturer’s directions to the samples. ATP was determined by testing the sample’s luminescence. The ATP levels were calculated using the formula provided for ATP content calculation.
LDH release assay
LDH release was measured using a LDH Release Assay Kit (Beyotime, C0016). First, inocle the cells into a 96 well culture plate to ensure that the cell density does not exceed 80-90%. Then, wash the cells with PBS solution and replace them with fresh culture medium. According to the experimental requirements, the cells were divided into different groups, including the control group and the drug treatment group. Add drugs of different concentrations as needed and continue to cultivate. One hour before the scheduled time, add the LDH release reagent to the “maximum enzyme activity control well of the sample”, mix well and continue to culture. After the experiment, centrifuge the cell culture plate and take the supernatant for sample determination.
qRT-PCR
qRT-PCR was utilized to identify alterations in gene transcription levels within each group. Isolation of total RNA from cells and tissues was performed using the TRIzol reagent (Biosharp, BS258A) and Reverse transcription was performed with HiScript Q RT SuperMix for qPCR (Vazyme, R222). The 2×Q3 SYBR qPCR Master mix (Universal) kit (KCD-M1004) was used for quantitative PCR reactions. As a brief summary, the qPCR procedure involved 2 minutes at 95°C for pre-incubation and 45 cycles at 95°C for denaturation for 10 seconds and 60°C for annealing for 20 seconds. 2−ΔΔCT method was applied to raw data for processing.
Primer (5’to3’):
NLRP3-human-F: GATCTTCGCTGCGATCAACAG
NLRP3-human-R: CGTGCATTATCTGAACCCCAC
Caspase-1-human-F: AGCACGTTCCTGGTGTTCAT
Caspase-1-human-R: TTGGGCAGTTCTTGGTATTCA
GDSMD-human-F: GTGTGTCAACCTGTCTATCAAGG
GDSMD-human-R: CATGGCATCGTAGAAGTGGAAG
IL-18-human-F: CCCCGGACCATATTTATT
IL-18-human-R: GTTATCAGGAGGATTCATTTC
Human actin beta-RT-F: AGCGAGCATCCCCCAAAGTT
Human actin beta-RT-R: GGGCACGAAGGCTCATCATT
IL-1β-human-F: GAAGTACCTAAGCTCGCCAGTG
IL-1β-human-R: GGGCAGGGAACCAGCATCT
Western blotting
Each sample was treated with RIPA lysate (Biosharp, BL504) in an ice bath for 30 minutes, followed by centrifugation at 12000rmp for 20 minutes. After transferring the supernatant to a new EP tube, the protein content was tested using the BCA kit (Biosharp, BL521), followed by resolving protein samples through SDS-PAGE (Beyotime, P0015L), and transferring them onto PVDF (Millipore, IPVH00010) membranes. The blocking solution was supplemented with primary antibody, which was then diluted to the preferred concentration and left to incubate with the membrane overnight at 4 °C. We used anti-NRLP3 (Boster, BA3677), anti-GSDMD (Abcam, ab210070), and anti-Caspase-1 (Proteintech, 22915-1-AP) antibodies in this research. After that, secondary antibodies (biosharp, BL003A) were applied and incubated with the membrane for 1 h at 37 °C. Following this, ECL luminescent solution (Biosharp, BL520A) was introduced, and images were captured using an integrated chemiluminescence device (BioRad, ChemiDoc XRS+).
Double staining with Hoechst 33342 and PI
We evaluated the integrity of plasma membrane using Hoechst 33342 and PI (propidium iodide). Cultured cells were treated with Hoechst staining solution (Beyotime, C1025) at a concentration of 5μg/ml. Staining was performed in the dark for 10 minutes at 37 °C. Afterward, a solution of PI staining (Beyotime, ST511) was introduced at a concentration of 155μg/ml and the reaction was carried out in the dark for 10min at 4 ° C. Subsequently, the samples were examined using a fluorescence microscope and images were captured. The percentage of positive cells was quantitatively analyzed using Image J software. By using Image J software, we were able to analyze the percentage of positive cells.
PTC xenograft tumor model
We purchased 5- to 6-week-old male BALB/c nude mice from Beijing Vital River Laboratory Animals Technology Co., Ltd. They were then randomly placed in a specific pathogen-free animal laboratory grade environment. Approximately 5×105 K1 cells suspended in PBS were injected into the left armpit of each nude mouse for the xenograft tumor model. On the second day of tumor cell transplantation, we randomly divided the mice into groups receiving normal saline (control), BBR 50 mg/kg, BBR 100 mg/kg, and BBR 200 mg/kg. Intragastric administration of BBR was used for all mice. Tumor volume and formation were monitored for three days. The volume of the tumor (V) was determined using the formula V = 1/2 × L × W2(L, length; W, width). Following a period of three weeks, the euthanasia of all the mice took place. The tumor tissue was subsequently surgically removed, part of which was fixed in 10% formalin, and the other parts were flash frozen. Ethics approval for all animal experimentation protocols was obtained from the General Hospital of Central Theater Command.
Statistical analysis
The statistical information is displayed as the average plus or minus the standard deviation. Data was gathered from three separate trials, each performed three times. Two groups were compared using Student’s t-tests, while multiple groups were compared using one-way analysis of variance (ANOVA). Statistical significance was determined with a P value less than 0.05 (*P <0.05; **P <0.01; ***P <0.001; ****P <0.0001). For statistical analysis, GraphPad Prism 8 was used.
RESULTS
NLRP3 and GSDMD were lowly expressed in PTC cell
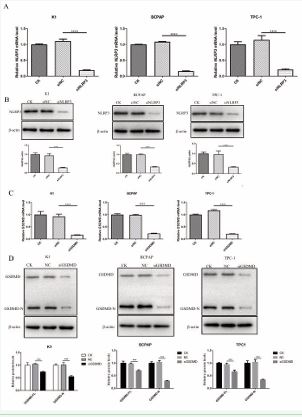
To investigate whether NLRP3 and GSDMD are differentially expressed in PTC cells and normal thyroid epithelial cells (Nthyori31), we respectively examined the protein levels of them in BRAF(V600E) mutant, BRAF wild-type PTC cell lines and Nthyori31 cells. We found that GSDMD and NLRP3 expression were lower in all types of PTC cells than in Nthyori31 cells, especially in the BRAF(V600E) mutant cell line. (Figure 1).
Figure 1 NLRP3 and GSDMD were lowly expressed in PTC cell. (A) Western blot analysis of NLRP3 and GSDMD expression in different PTC cell lines and immortalized thyroid epithelial cell lines (Nythori31). *P<0.05; **P<0.01; ***P<0.001.
PTC cell growth was inhibited by BBR in vitro
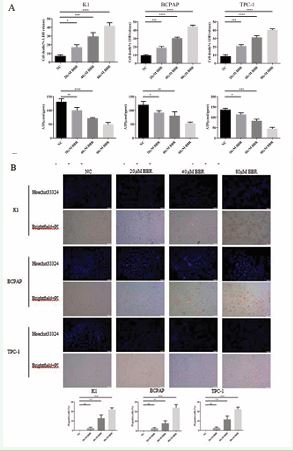
Our previous studies showed that BBR (Berberine), an effective component of Chinese herbal medicine Coptis, has an inhibitory effect on PTC. In order to evaluate the effect of BBR on the cytotoxicity of PTC cells and to screen for the optimal concentration and duration of action, we first examined the cell viability of PTC cells after BBR treatment at different concentrations and for different times. BBR treatment effectively suppressed PTC cells’ viability in a manner that is dependent on both dosage and time while having minimal impact on the viability of Nthyori31 (Figure 2A). As a result, 48 hours was chosen as the experimental treatment period. We next sought to verify whether BBR had an impact on the proliferation of PTC cells. Thus, we performed a colony formation assay with different concentrations of BBR. As the concentration of BBR increased, the colonies decreased significantly (Figure 2B). Then, apoptosis cells were measured using flow cytometry to determine whether BBR caused PTC cell death. It was obvious that PTC cells underwent more when BBR concentration was increased (Figure 2C). Overall, the findings indicated that BBR induces apoptosis and suppresses proliferation in PTC cells in a dose-dependent fashion.
BBR induced PTC cell pyroptosis
To further elucidate the mechanisms underlying the inhibitory effects of BBR on PTC, we examined pyroptosis related mRNAs and proteins to determine if BBR affected pyroptosis in PTC cells. Increased BBR concentrations led to an upregulation in the mRNA expression of NLRP3, Caspase-1 and GSDMD as shown by qRT-PCR analysis (Figure 3A). BBR was also observed to increase IL-1β and IL-18 mRNA expression (Figure 3B). Consistent with the qRT-PCR results, ELISA showed that IL-1β and IL-18 levels were significantly higher in the BBR-treated group than those in control group (Figure 3C). Western blot analysis showed an increase in the protein levels of NLRP3, Caspase-1, and GSDMD-N with higher concentrations of BBR. However, the impact of BBR on pro-caspase-1 and GSDMD expression was minimal (Figure 3D). In order to further evaluate the cellular damage, LDH and ATP levels in the supernatant of PTC cells were measured after administration of different concentrations of BBR. We found that BBR-treated groups showed a dose dependent increase in LDH secretion and a decrease in ATP level in the culture medium (Figure 4A). Increased LDH levels and decreased ATP levels are due to impaired cell integrity, efflux of cellular contents from disrupted cell membranes and reduced energy release. It prompted us to to monitor the integrity of PTC plasma membrane by in situ live cell staining with Hoechst 33342 and PI. PI positive rates were higher in the group treated with medium (40 μM) and high (80 μM) concentrations of BBR than those in control group (Figure 4B), which indicated that the integrity of the plasma membrane was compromised. The findings above indicate that BBR enhanced the levels of proteins and inflammation factors related to pyroptosis, resulting in disruption of the cytomembrane in a dose-dependent manner. This suggests that BBR promoted pyroptosis in PTC cells.
NLRP3/caspase-1/GSDMD axis was activated by BBR
To explore the relationship between NLRP3 and the increase of GSDMD-N, inflammatory factors and cell membrane disruption caused by BBR, we knocked down NLRP3 and GSDMD in PTC cells (Figure 5A-D) and detected the related changes. The BBR-induced increases of GSDMD, GSDMD-N, IL-1β, IL-18 and LDH, and the rise in the proportion of PI-positive cells were significantly attenuated when NLRP3 knockdown. The same weakening effect occurred when GSDMD knockdown (Figure 6A-D). It proved that the effect of BBR on pyroptosis is mediated by the regulation of NLRP3 and GSDMD. Subsequently, to further research whether the increase of GSDMD was due to NLRP3, we also examined the protein expression of pro caspase-1, caspase-1, GSDMD, and GSDMD-N when NLRP3 was knocked down. The results showed that the expression of pro-caspase-1, caspase-1, GSDMD, and GSDMD-N was reduced by NLRP3 knockdown in PTC cells (Figure.6E). Caspase-1 cleaves GSDMD, forming GSDMD-N fragments that are toxic to cell membranes and trigger pyroptosis [8]. Thus, the results indicated that NLRP3 activated Caspase-1 to promote GSDMD-N formation. As a result, BBR triggered.
Figure 2 BBR inhibited PTC cell growth in vitro. (A) MTT was used to test the inhibitory effect of BBR on K1, BCPAP and TPC-1 cell proliferation. Cells were treated with BBR as indicated. (B) Representative photographs of colony formation. Cells were treated with BBR as indicated for 48h. (C) Flow cytometry was used to detect the promoting effect of BBR on apoptosis of K1, BCPAP and TPC-1 cells. *P<0.05; **P<0.01; ***P<0.001. versus control group control (NC).
Figure 3 BBR induced PTC cell pyroptosis. (A) The mRNA expression of NLRP3, Caspase-1, and GSDMD in K1, BCPAP, and TPC-1 cells treated with different concentrations of BBR. (B) The mRNA expression of IL-1β and IL-18 in K1, BCPAP, and TPC-1 cells treated with different concentrations of BBR. (C) Expression levels of IL-1β and IL 18 in K1, BCPAP, and TPC-1 cells treated with different concentrations of BBR. (D) Protein expression of NLRP3, Pro-caspase-1, Caspase-1, GSDMD, and GSDMD-N in K1, BCPAP, and TPC-1 cells treated with different concentrations of BBR. *P<0.05; **P<0.01; ***P<0.001.
Figure 4 BBR induced PTC cell pyroptosis. (A) LDH and ATP levels in K1, BCPAP, and TPC-1 cells treated with different concentrations of BBR. (B) Representative images and quantitative results of PI positivity in K1, BCPAP and TPC-1 cells treated with different concentrations of BBR. Scale bars: 100 μm. *P<0.05; **P<0.01; ***P<0.001.
Figure 5 SiNLRP3 and siGSDMD mediated down-regulation of NLRP3 and GSDMD. (A) qRT-PCR analysis of NLRP3 expression after transfection with siNLRP3 or siNC in K1, BCPAP and TPC-1 cells. (B) Western blot analysis of NLRP3 expression after transfection with siNLRP3 or siNC in K1, BCPAP and TPC-1 cells. (C) qRT-PCR analysis of GSDMD expression after transfection with siGSDMD or siNC in K1, BCPAP and TPC-1 cells. (D) Western blot analysis of GSDMD expression after transfection with siGSDMD or siNC in K1, BCPAP and TPC-1 cells.
Figure 6 BBR induced PTC cell pyroptosis by activating the NLRP3/ caspase-1/GSDMD pathway. Protein expression of GSDMD and GSDMD-N in K1, BCPAP and TPC-1 cells with five different treatments (NC, BBR, BBR + siNC, BBR + siNLRP3, BBR + siGSDMD). (B) Levels of IL-1β and IL-18 in the supernatant of K1, BCPAP and TPC-1 cells with five different treatments (NC, BBR, BBR + siNC, BBR + siNLRP3, BBR + siGSDMD). (C) Levels of LDH in the supernatant of K1, BCPAP and TPC-1 cells with five different treatments (NC, BBR, BBR + siNC, BBR + siNLRP3, BBR + siGSDMD). (D) Representative images and quantitative results of PI positive rates in K1, BCPAP and TPC-1 cells with five different treatments (NC, BBR, BBR + siNC, BBR + siNLRP3, BBR + siGSDMD). Scale bars: 100 μm. (E) Expression of Pro-caspase-1, Caspase-1, GSDMD, and GSDMD-N in K1, BCPAP, and TPC-1 cells after NLRP3 silencing. *P<0.05; **P<0.01; ***P<0.001.
the NLRP3/caspase-1/GSDMD axis in PTC cells, and eventually led to the induction of pyroptosis.
BBR induced pyroptosis in PTC xenograft tumours
The in vitro findings that BBR promotes PTC pyroptosis prompted us to further explore whether BBR could also inhibit transplanted tumor through pyroptosis. Thus, we constructed a PTC xenograft tumor model by subcutaneous injection of K1 cells in the left armpit of BALB/c mice to directly confirm whether BBR affects the PTC formation and growth in vivo. Twenty-one days after PTC cells injection, tumours were dissected. The size of the tumor was notably smaller in BBR-treated mice compared to controls. As the concentration of BBR increased, the tumour volume became smaller (Figure 7A,B). The BBR treatment group exhibited a dose-dependent increase in the expression of GSDMD, caspase-1, and NLRP3 as indicated by Western Blotting analysis and Immunofluorescence (Figure 7C, E). As the dosage of BBR administered increased, the levels of IL-1β and IL-18 production and LDH release also increased (Figure 7D). As a result, BBR inhibits PTC xenograft tumour growth by promoting pyroptosis of PTC cells.
DISCUSSION
At present, the main treatment for PTC is surgery, which is often accompanied by a variety of complications including bleeding, wound infection, nerve injury, parathyroid injury, hypocalcemia, postoperative chylous leakage, postoperative dyspnea and asphyxia [26]. In addition, the existing drugs for the treatment of PTC often lead to a variety of side effects, such as cardiac insufficiency, gastrointestinal reactions, and bone marrow suppression. Thus, safe and effective drug treatment options are limited. Some traditional Chinese medicine has a good effect on the treatment of differentiated thyroid carcinoma, and the combined treatment strategy of traditional Chinese medicine has a broad application prospect. The presence of a BRAF(V600E) mutation in PTC is associated with increased risk of tumor invasiveness, lymph node metastasis, and recurrence, leading to a negative clinical outcome [27]. We selected two types of cell lines, BRAF(V600E) mutant type and wild type, to conduct experiments to explore the effect of BBR on these two types of PTC. Our study showed that BBR induces pyroptosis in PTC cells of different genetic backgrounds, confirming that NLRP3/Caspase-1/ GSDMD axis activation by BBR induces pyroptosis in PTC, providing a promising therapeutic strategy. Berberine, an isoquinoline alkaloid commonly present in Coptis and Phellodendron, is typically found in nature [28]. According to current evidence, BBR has great potential in cancer therapy [29,30]. BBR has been demonstrated to impact various crucial cellular functions, including cell growth, programmed cell death, halting of cell division, migration, and invasion [31,32]. Several conventional chemotherapeutic agents have been found to be more effective when BBR is added to them [33]. The antitumour effects of cisplatin and 5-fluorouracil on laryngeal carcinoma cells could be effectively promoted after pretreatment with BBR [34]. Furthermore, BBR exhibits significant therapeutic effects without causing obvious side effects even at relatively high doses, which has attracted much attention [30]. In spite of this, few studies have reported the efficacy of BBR in treating PTC, and the underlying molecular mechanism is unknown. To examine the effects of BBR on PTC, this study was conducted. Our findings indicate that BBR suppresses cell growth and induces cell death through apoptosis and pyroptosis in three PTC cell lines. We also tested this phenomenon in animal experiments. Xenograft nude mouse models treated with BBR showed significant inhibitory effects on PTC growth. Tumor cells undergo pyroptosis in vivo, which indicates that pyroptosis could be a significant factor in the formation and development of tumors [35,36]. Pyroptosis mediated by the gasdermin family increases the sensitivity of anaplastic thyroid cancer cells to apatinib and melittin [37]. Belonging to the gasdermin family, GSDMD undergoes cleavage by active caspase-1 to generate GSDMD-N, which in turn causes membrane puncture and is responsible for triggering inflammation and pyroptosis [9-11]. Pyroptosis was assessed by quantifying the proportion of PI-positive cells, LDH release, and levels of IL-1β and IL- [18]. Following treatment with BBR in PTC, there was a notable rise in the percentage of PI-positive cells, LDH release, and IL-1β and IL-18 production. GSDMD-N expression was also increased in BBR-treated PTC cells. However, these changes induced by BBR were effectively attenuated by knocking down either NLRP3 or GSDMD. Moreover, although NLRP3 and GSDMD were differentially expressed in BRAF-mutant and BRAF-wild type PTC cells, BBR had the same pyroptosis promoting effect in these cells. As a form of cell death, apoptosis is more common than pyroptosis. BBR stimulates PI3K/AKT/mTOR axis to suppress proliferation, induce apoptosis and promote autophagy, showing a tumor suppressive effect on anaplastic thyroid carcinoma [19]. Furthermore, our prior in vitro research validated that BBR suppressed the growth of PTC K1 cells stimulated by elevated glucose levels, accompanied by changes in PI3K/Akt/Nrf2 signaling pathway. [23]. BBR increased Nrf2 expression and translocation to the nucleus in K1 cells, resulting in elevated oxidative stress and ROS generation. It increases the early and late apoptosis of PTC K1 cells [24]. Our research also revealed that BBR suppressed cell proliferation and enhanced apoptosis.
Figure 7 BBR inhibited the growth of PTC xenograft tumours by inducing pyroptosis. (A) The subcutaneously transplanted tumours in nude mice after the injection of different treated cells (n =6). (B) The volumes of tumours in the nude mice. (C) Western blot analysis of NLRP3, pro-caspase-1, caspase-1, GSDMD, GSDMD-N expression in PTC tissue with or without BBR. (D) ELISA analysis of the pyroptosis-related inflammatory factors IL-1β, IL-18 and LDH in PTC tissue with or without BBR. (E) Immunofluorescence assay of NLRP3, caspase-1, GSDMD expression in PTC tissue with or without BBR. Scale bars: 100 μm. *P<0.05; **P<0.01; ***P<0.001; ****P<0.0001.
increases the early and late apoptosis of PTC K1 cells [24]. Our research also revealed that BBR suppressed cell proliferation and enhanced apoptosis. NLRP3 is a part of the inflammasome, which can mediate pyroptosis by promoting the maturation of caspase-1 [14-16]. Our results showed that BBR upregulated the expression of NLRP3 and caspase-1 and subsequently increased the expression of GSDMD at both mRNA and protein levels. Mechanistically, we confirmed that NLRP3 knockdown inhibited caspase-1 activation and GSDMD-N maturation, showing that NLRP3 can trigger caspase-1 to facilitate the creation of GSDMD-N. As a result of our study, the NLRP3/caspase-1/GSDMD axis played a role in the pyroptosis of PTC cells, suggesting a novel mechanism of BBR-mediated cell death in PTC. A few improvements could be made to this study. There is still a need to investigate whether BBR impacts cell migration and invasion. It is also necessary to investigate the upstream regulators of NLRP3 inflammasome activation by BBR. Furthermore, there is a need for further exploration and evaluation of the clinical effectiveness of BBR for PTC. Additionally, it may be worthwhile to consider combining BBR with other treatment options. As a result of our findings, BBR induces pyroptosis in PTC cells, making it a potential therapeutic agent for PTC. Mechanistically, BBR mediates pyroptosis in PTC cells by activating NLRP3/caspase-1/GSDMD axis. This research provides experimental evidence that BBR can be applied to the treatment of PTC, which will improve its clinical efficacy.
Data Sharing Statement
Data from this article will be shared upon reasonable request from the corresponding author.
Funding
This work was supported by Hubei Provincial Administration of Traditional Chinese Medicine [grant numbers ZY2023F048]; Nature Science Foundation of Hubei Province [grant numbers 2017CFB797].
Statement of Ethics
Every animal study followed ARRIVE recommendations and was carried out in compliance with the National Research Council’s guidelines for the treatment and utilization of laboratory animals. This project was approved by the Ethics Committee for Medical Laboratory Animal Welfare, General Hospital of Central Theater Command, China [grant numbers 20230026].
Credit Authorship Contribution Statement
Xiaoyan Huang and Yanhong Lu was responsible for conceptualizing, supervising, validating, visualizing, and writing the original draft. Shicheng Li was responsible for data management and formal analysis. Wei Chen is responsible for software and methodology. Qiaojun Zheng and Yiming Hou were in charge of resources, investigation. Jiayang Tang: Methodology. Shengwei Zhang: Formal analysis. Jing Dong and Ling Yue contributed to conceptualization, supervision, and both the original draft and review and editing of the writing.
Declaration of Competing Interest
The writers assert that they possess no recognized conflicting monetary concerns or personal associations that may have seemed to impact the research documented in this article.
REFERENCES
- Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66: 115-132.
- Zheng R, Zhang S, Zeng H, Wang S, Sun K, Chen R, et al. Cancer incidence and mortality in China, 2016. J Natl Cancer Cent. 2022; 2: 1-9.
- Mao Y, Xing M. Recent incidences and differential trends of thyroid cancer in the USA. Endocr Relat Cancer. 2016; 23: 313-322.
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71: 209-249.
- McLeod DSA, Zhang L, Durante C, Cooper DS. Contemporary Debates in Adult Papillary Thyroid Cancer Management. Endocr Rev. 2019; 40: 1481-1499.
- Ye L, Yue L. Progress of clinical medication for differentiated thyroid carcinoma. Journal of Medical Research & Combat Trauma Care. 2018; 31: 544-549.
- Tang R, Xu J, Zhang B, Liu J, Liang C, Hua J, et al. Ferroptosis, necroptosis, and pyroptosis in anticancer immunity. J Hematol Oncol. 2020; 13: 110.
- Li M, Jiang P, Yang Y, Xiong L, Wei S, Wang J, et al. The role of pyroptosis and gasdermin family in tumor progression and immune microenvironment. Exp Hematol Oncol. 2023; 12: 103.
- Loveless R, Bloomquist R, Teng Y. Pyroptosis at the forefront of anticancer immunity. J Exp Clin Cancer Res. 2021; 40: 264.
- Ding J, Wang K, Liu W, She Y, Sun Q, Shi J, et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature. 2016; 535: 111-116.
- Wang C, Ruan J. Mechanistic Insights into Gasdermin Pore Formation and Regulation in Pyroptosis. J Mol Biol. 2022; 434: 167297.
- Hu Y, Wen Q, Cai Y, Liu Y, Ma W, Li Q, et al. Alantolactone induces concurrent apoptosis and GSDME-dependent pyroptosis of anaplastic thyroid cancer through ROS mitochondria-dependent caspase pathway. Phytomedicine. 2023; 108: 154528.
- Guo H, Ma R, Zhang Y, Yin K, Du G, Yin F, et al. Ibuprofen inhibitsanaplastic thyroid cells in vivo and in vitro by triggering NLRP3-ASC- GSDMD-dependent pyroptosis. Inflammopharmacology. 2024; 32:733-745.
- Sharma D, Kanneganti TD. The cell biology of inflammasomes: Mechanisms of inflammasome activation and regulation. J Cell Biol. 2016; 213: 617-629.
- Moossavi M, Parsamanesh N, Bahrami A, Atkin SL, Sahebkar A. Role of the NLRP3 inflammasome in cancer. Mol Cancer. 2018; 17: 158.
- Sharma BR, Kanneganti TD. NLRP3 inflammasome in cancer and metabolic diseases. Nat Immunol. 2021; 22: 550-559.
- Zaki MH, Vogel P, Body-Malapel M, Lamkanfi M, Kanneganti TD. IL-production downstream of the Nlrp3 inflammasome confers protection against colorectal tumor formation. J Immunol. 2010; 185: 4912-4920.
- Bai J. The Expression and Significance of NLRP3 in Papillary Thyroid Carcinoma. 2021.
- Shi XZ, Zhao S, Wang Y, Wang MY, Su SW, Wu YZ, et al. Antitumor Activity of Berberine by Activating Autophagy and Apoptosis in CAL- 62 and BHT-101 Anaplastic Thyroid Carcinoma Cell Lines. Drug Des Devel Ther. 2023; 17:1889-1906.
- Qi F, Zhao L, Zhou A, Zhang B, Li A, Wang Z, et al. The advantages of using traditional Chinese medicine as an adjunctive therapy in the whole course of cancer treatment instead of only terminal stage of cancer. Biosci Trends. 2015; 9: 16-34.
- Chu Q, Jiang Y, Zhang W, Xu C, Du W, Tuguzbaeva G, et al. Pyroptosis is involved in the pathogenesis of human hepatocellular carcinoma. Oncotarget. 2016; 7: 84658-84665.
- Yao M, Fan X, Yuan B, Takagi N, Liu S, Han X, et al. Berberine inhibits NLRP3 Inflammasome pathway in human triple-negative breast cancer MDA-MB-231 cell. BMC Complement Altern Med. 2019; 19: 216.
- Ni J, Wang F, Yue L, Xiang GD, Zhao LS, Wang Y, et al. The effects and mechanisms of berberine on proliferation of papillary thyroid cancer K1 cells induced by high glucose]. Zhonghua Nei Ke Za Zhi. 2017; 56: 507-511. Chinese.
- Zhu z. Effect and Mechanism of Berberine in Papillary Thyroid Cancer. 2021.
- Swanson KV, Deng M, Ting JP. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol. 2019; 19: 477-489.
- Nikhil Soni, Bapuji Shrawan Gedam, Akhtar M. Thyroidectomy: post- operative complications and management. Int Surg J. 2019; 6: 1659- 1663.
- Ye Z, Xia X, Xu P, Liu W, Wang S, Fan Y, et al. The Prognostic Implication of the BRAF V600E Mutation in Papillary Thyroid Cancer in a Chinese Population. Int J Endocrinol. 2022; 2022: 6562149.
- Song D, Hao J, Fan D. Biological properties and clinical applications of berberine. Front Med. 2020; 14: 564-582.
- Vlavcheski F, O’Neill EJ, Gagacev F, Tsiani E. Effects of Berberine against Pancreatitis and Pancreatic Cancer. Molecules. 2022; 27: 8630.
- Xiong RG, Huang SY, Wu SX, Zhou DD, Yang ZJ, Saimaiti A, et al. Anticancer Effects and Mechanisms of Berberine from Medicinal Herbs: An Update Review. Molecules. 2022; 27: 4523.
- Sun Q, He M, Zhang M. Research progress on anti-tumor effect of berberine. Chinese Traditional and Herbal Drugs. 2021; 52: 603-612.
- Yang XH, Zhang LJ, Luo MJ, Luo S, Gong YY, Chen T. Research progress in mechanism of berberine’s antitumor action. Zhongguo Zhong Yao Za Zhi. 2021; 46: 2449-2455.
- Liu D, Meng X, Wu D, Qiu Z, Luo H. A Natural Isoquinoline Alkaloid with Antitumor Activity: Studies of the Biological Activities of Berberine. Front Pharmacol. 2019; 10: 9.
- Gao X, Wang J, Li M, Wang J, Lv J, Zhang L, et al. Berberine attenuates XRCC1-mediated base excision repair and sensitizes breast cancer cells to the chemotherapeutic drugs. J Cell Mol Med. 2019; 23: 6797- 6804.
- Yan Z, Da Q, Li Z, Lin Q, Yi J, Su Y, et al. Inhibition of NEK7 Suppressed Hepatocellular Carcinoma Progression by Mediating Cancer Cell Pyroptosis. Front Oncol. 2022; 12: 812655.
- Li Y, Wang W, Li A, Huang W, Chen S, Han F, et al. Dihydroartemisinin induces pyroptosis by promoting the AIM2/caspase-3/DFNA5 axis in breast cancer cells. Chem Biol Interact. 2021; 340: 109434.
- Zhao Q, Feng H, Yang Z, Liang J, Jin Z, Chen L, et al. The central role of a two-way positive feedback pathway in molecular targeted therapies- mediated pyroptosis in anaplastic thyroid cancer. Clin Transl Med. 2022; 12: e727.