Epithelial to Mesenchymal Transition (EMT): From a Developmental Instructor to Metastasis Propeller
- 1. Department of Biochemistry, University of Hyderabad, India
Abstract
Epithelial to mesenchymal transition (EMT) is an essential physiological process in allmulticellular organisms. It plays pivotal role in the normal development of an organism but is equally important for tumor metastasis. For example, during mammary gland development, EMT plays critical role in mammary epithelial cellular migration and the establishment of the new tissue. Epithelial cells in terminal end buds (TEBs) show higher plasticity and induce EMT. The EMT in TEBs is regulated by various factors like epidermal growth factor (EGF), insulin like growth factor- 2 (IGF-2), Wnt-4 and hepatocyte growth Factor (HGF. Interestingly, cells at branching induce the expression of typical mesenchymal markers such as Vimentin and Snail. Besides its fundamental role in developmental process, a large body of data suggests that EMT is an important and integral process in breast tumor metastasis. Emerging studies revealed that EMT and partly tumor heterogeneity cause therapeutic resistance in breast cancer. Comprehending the complexity of the EMT may offer significant understandings that lead to the improvement of therapeutic targets for invasive cancer, and perhaps it can also be used as biomarkers pinpointing tumor subclasses with better likelihoods of relapse, metastasis and drug resistance leading to death. Herein we will review and provide critical comments on the role of EMT as a developmental instructor, metastasis propeller and master of therapeutic resistance using primarily breast as a model system.
Keywords
Tumor metastasis; Epithelial to mesenchymal transition; Breast cancer.
CITATION
Dwivedi A, Manavathi B (2017) Epithelial to Mesenchymal Transition (EMT): From a Developmental Instructor to Metastasis Propeller. JSM Biochem Mol Biol 4(3): 1030.
ABBREVIATIONS
EMT: Epithelial to Mesenchymal Transition; MET: Mesenchymal to Epithelial Transition; TEBs: Epithelial cells in Terminal End Buds; CTC: Circulatory Tumor Cells; CSCs: Cancer Stem Cells; HGF: Hepatocyte Growth Factor/Scatter Factor (HGF/SF); IGF-2: Insulin Like Growth Factor- 2; ESRP: Epithelial Splicing Regulatory Protein; δEF1: Delta-Crystallin Enhancer Binding Factor; SIP1: Smad Interacting Protein 1
INTRODUCTION
For all multi-cellular organisms, epithelial to mesenchyme transition (EMT) is a critical process and plays a central role in normal development as it involves in embryogenesis, wound healing, embryo implantation and placenta formation [1]. Furthermore, several developmental process including gastrulation, neural crest formation and organogenesis requires EMT. During embryonic development, neuro-ectodermal epithelial cells undergo EMT and give rise to highly migratory neural crest cells. As a consequence, they dissociate and become motile and migrate to different parts of embryo, where they undergo further differentiation into different cell types. Various signaling pathways such as TGFβ-SMAD, Wnt, growth factor *Corresponding author Bramanandam Manavathi, Department of Biochemistry, University of Hyderabad, Associate Professor, School of Life Sciences, Hyderabad-500046, India, Tel: 91 402313-4546; Email: Submitted: 10 August 2017 Accepted: 27 September 2017 Published: 29 September 2017 ISSN: 2333-7109 Copyright © 2017 Manavathi et al. OPEN ACCESS Keywords • Tumor metastasis • Epithelial to mesenchymal transition • Breast cancer signaling provides essential instructions to the cells to occur EMT [2]. For example, embryos deficient in Wnt3 cannot undergo EMT. Furthermore, deficiency of TGF-beta family proteins like Nodal and Vg1, which mediates Wnt action can leads to defects in mesoderm formation due to absence of EMT [3-5].
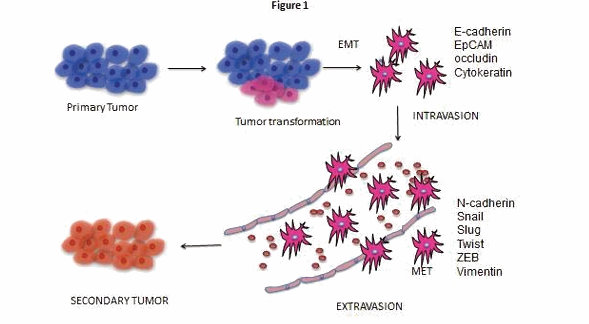
EMT is an essential physiological process in all multicellular organisms, which plays important role in the normal development of an organism but is equally important for tumor metastasis to occur. Metastasis comprises of a series of steps that includes epithelial to mesenchymal transition (EMT), loss of cell adhesion, cell migration, and new blood vessels formation, intravasation into blood or lymphatic vessels and extravasation (Figure 1).
Figure 1 A schematic presentation of the metastatic progression of a malignant tumor: The cancer progression starts with a primary tumor which is formed by cellular transformation at the site of origin (primary tumor). In metastatic cancer, few cells detach from the primary site of the tumor, intravasate and enter the blood vessels. By extravasation, these metastasis cancer cells enter the new site for colonization in the body to establish the secondary tumor.
These cells shows less epithelial features than mesenchymal and have self-renewal ability which is the indication of cellular heterogeneity of tumor which contains the sub-population of cancer cells which has cancer stem cells (CSCs) property and they can migrate from primary tumor site to distant secondary progression. In many cancers, the neoplastic cells exhibit EMT activation only when the tumor population is abundant with CSCs [6,7] on the other hand forced activation of EMT by means of chemical treatment increases the tumor initiating capacity and shows CSCs property [8,9].
Cellular heterogeneity of tumor gives advantage to cancer cell as cells with different morphology and molecular characteristic respond differently to chemotherapy and radiotherapy which lead to drug resistance [10]. EMT involvement in cancer progression and drug resistance has been shown mainly in in vitro model system, not much reports about its role in clinical trials till date. Recent development in technology gave deep insight regarding the molecular changes by gene expression profiling. Studies from gene profiling show numerous gene clusters and signaling molecules involved in EMT are related in cancer progression and drug resistance. Here, we will review the literature and provide critical comments on the role of EMT as a developmental instructor, metastasis propeller and master of therapeutic resistance using primarily breast as a model system.
EMT as a developmental instructor
The mammary gland is a unique organ and develops in three distinct stages. Initially, rudimentary mammary gland development takes place at embryonic stage, ductal elongation and branching at puberty and alveolar differentiation and tertiary ductal branching in pregnancy [11]. Most striking development of mammary gland takes place at puberty, where branching morphogenesis coupled with ductal elongation and rudimentary gland starts begins to elongate with the formation of terminal end buds (TEBs) [11]. During this stage, EMT plays critical role in cellular migration and the establishment of new tissue. Epithelial cells in TEBs show higher plasticity and induce EMT, and it is regulated by numerous extracellular factors like EGF/ErbB, IGF 2, wnt-4, hepatocyte Growth Factor/Scatter Factor (HGF/SF), MMPs and ovarian hormones like estrogen and progesterone [12,13]. Further, TEBs shown to secrete proteases like MMP3 and activate EMT by inducing Msx2/Cripto-1 pathway [13,14]. In addition, TEBs also express higher integrin and extracellular receptors as compared to mammary epithelium, since α2,α3 and β1 knockout mice shows significantly less branching [13].
Interestingly, cells at branching induce mesenchymal markers like vimentin, snail and expresses MMP3, the inducers of EMT [15]. Further, microarray analysis of genes in TEB identified known regulators of EMT like snail and twist are significantly up regulated when compared to mature ducts [16]. These studies support the involvement of EMT in mammary gland development. However, mammary epithelial cells of TEBs do not exhibit complete EMT, although they acquire multiple features of EMT.
EMT as a metastasis propeller
Breast cancer is the most common cancer in women and majority of deaths from breast cancer are due to the metastasis not because of primary tumor. Mammary gland is the site for breast cancer development. It is well established that breast tumor metastasize to bone, liver, lung and brain. The process of metastasis comprises of a series of coordinated steps that includes epithelial to mesenchymal transition (EMT), loss of cell adhesion, cell migration, intravasation, travelling through circulation followed by extravasation and colonization [17]. Several studies demonstrated the potential role of EMT in breast cancer metastasis [18]. Numerous studies in breast cancer identified different signaling pathways like TGF-β and Wnt signaling participating in the induction of EMT [2]. It is really challenging to track the cancer cells undergoing EMT in the tissue. However, using WWP-MYC transgenic mice model provided strong evidence of EMT in breast cancer cell invasion and metastasis [19]. Another recent study revealed that mesenchymal markers like vimentin and snail, were enriched in circulatory tumor cells (CTC) in breast cancer patients [20]. Interestingly, they found that mesenchymal properties are associated with cluster of CTCs rather than single migratory cell. To further support this, collective cell migration was observed in breast cancer metastatic xenograft models [21]. Surprisingly, although main function of EMT to induce invasive phenotype, EMT also associates with tumor initiation and breast cancer stem cells. Now it is well accepted that the roles of cancer stem cells in tumor initiation and progression. Modulating the expression of EMT inducers promotes tumor initiation (cancer stem cells) and mammosphere formation [22]. Breast cancer stem cells which are characterized by CD44 high/CD24 low population express high levels of Snail, Twist, N-cadherin and low levels E-cadherin. These studies indicate that EMT not only involves in epithelial to mesenchymal transition but also plays critical role in cancer stem cell maintenance. Further studies are warranted to understand the role of EMT in cancer stem cells, tumor initiation and clinical relevance in breast cancer metastasis.
EMT is a process where epithelial cell has ability to transition into mesenchymal cells and again partially or fully restored back to the characteristic phenotype of the epithelial cells by a process called ‘mesenchymal to epithelial transition’ (MET). EMT is the major cause of invasion from primary tumor mass to the surrounding stroma, it is the first stage in a complex process of metastasis and formation of secondary tumor growth [23,24]. Mesenchymal to epithelial transition (MET), a reverse process of EMT, is associated with the stem like characteristic in development and cancer. In spite of their same clonal origin, the tumor cells vary in their morphology and also in molecular features. In almost all the cancers a very unique kind of cells are identified with stem cell like characteristic which shows high drug resistance and has enhanced potential for creating cellular heterogeneity as CSCs has ability of self-renewal and give rise to differentiated cell population as well [25,26]. Additionally, the theory of CSCs which claim that even the very minor population of CSCs has an ability of giving rise to new tumor and also the epigenetic modification within the cancer cells provides phenotypic variability resulting the tumor heterogeneity [27,28]. EMT plays role beyond the first step of carcinoma cell invasion as its activation by several mode like TGFβ treatment, Twist1 and Snail over expression is shown to be responsible for many properties of CSCs like high expression of CD44+ and reduced level of CD24-.These cells are more capable of suspension survival and tumor sphere formation and enhanced ability to generated tumor in mice [29,30].
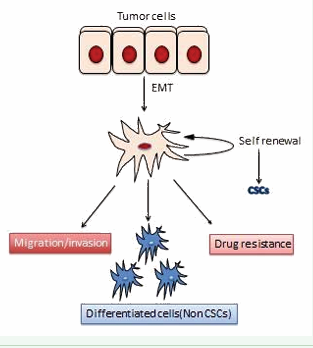
Advanced development in technology has provided a deep insight in detection and treatment of cancer, which has greatly improved the survival of the patient even though it is a challenge due to relapse and chemo resistance because of which many patient dies. Cancer is heterogeneous population of cells containing stem like cells (CSCs) which show high reoccurrence and drug resistance [31]. One of the reasons behind this resistance is due to higher expression of anti-apoptotic proteins, increased level of ABC transporter and also transmembrane transport proteins which facilitate the drug efflux [32]. Recent studies have shown that chemotherapy and radiotherapy can eradicate the bulk of non CSCs population than CSCs [33]. By analyzing the gene expression profiles of tumor samples and the patient survival from whom the samples are obtained a strong connection has been found between EMT related proteins and therapeutic resistance [33]. In many cases of breast cancer which shows high drug resistance has increased expression of EMT-associated genes [34]. Additionally in other studies where the correlation between gene expressions profiles and response to EGFR or PI3K inhibitor in clinical trials and also in many cell line from patient shows seventy six (76) EMT signature genes responsible for clinical resistance to these inhibitory chemicals [35], implying the inherent association of the expression of EMT genes with the development of drug resistance. Together all this observation makes very strong notion about the role of EMT in the development of drug resistance towards several therapeutics in breast cancer and it also demonstrate the functional relation between EMT activation and CSCs phenotype [33] (Figure 2). Considering the inherent role of EMT and CSCs in the development of therapeutic drug resistance, future awaits targeting EMT and CSCs as possible cure for breast cancer.
Figure 2: Schematic diagram representing the fate of a detached cancer cell. The disseminated cancer cell which has anoikis resistance can undergo self-renewal to maintain its population, can confer drug resistance, exhibits migratory/invasive properties or it can differentiate into non-cancer stem cell populations..
Molecular mechanisms of EMT activation
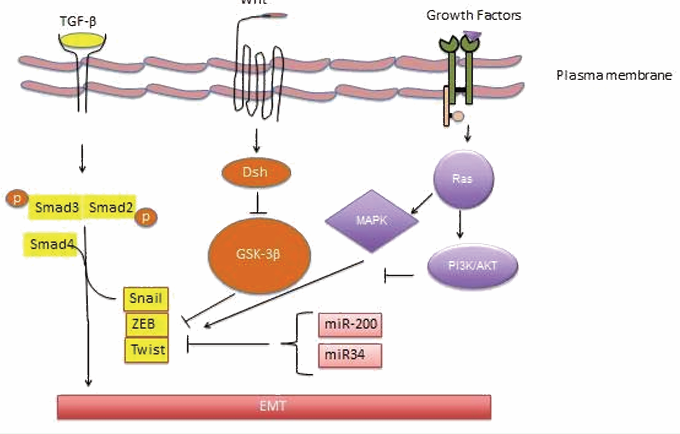
EMT induction takes place by means of several extracellular stimuli and resultant intracellular signaling pathways. Among all, TGFβ–SMAD signaling plays central role in countless well-known pathways of EMT [36,37]. In addition, other signaling pathways like Wnt, growth factor–receptor tyrosine kinase, and ECM integrin signaling pathways, also appear to play important role in the activation of EMT in different combinations [38] (Figure 3).
Figure 3: Major signaling pathways involved in EMT. TGF-β is the primary regulator of EMT. TGF binding to the receptors leads to the activation of Smad2 and Smad3 which then interact with Smad4 to form Smad complex. This Smad complex is then translocated into nucleus where they assist and collaborate with transcriptional factors like Snail, ZEB and Twist to control the expression of TGF-β target genes which include down regulation of epithelial markers and upregulation of mesenchymal markers. Further it also work together with other signal factors like Wnt and growth factors to regulate EMT. miRNAs, miR-200 family and miR-34also regulate EMT by controlling the expression of EMT-TFs.
Together all these various signal transduction pathways unite and lead to the activation of a small set of transcription factors that organizes the gene expression changes related to EMT. Together these transcription factors are known as ‘EMT-inducing transcription factors’ (EMT-TFs) which further categorized into three different protein families- the Snail (Snail and Slug, it is also known as SNAI1 and SNAI2, respectively), ZEB (including ZEB1 and ZEB2), and helix–loop–helix (including TWIST1, TWIST2, and TCF3) families [39]. In addition, other transcription factors like PRRX1, YAP1/TAZ, and SOX4 have also been known to play critical roles in EMT. The EMTTFs control the expression of other genes together which are common targets as well as they regulate their own expression also [39]. For instance, the upstream regulator of many EMT-TFs likes SLUG, TWIST1, and ZEB1 is Snail that promotes their expression. On the contrary, TCF3 appeared to function as a downstream molecule whose expression is regulated by many other EMT-TFs, like Snail, Slug, and ZEB1 [40,41].
Similarly the expression and function of EMT-TFs are also controlled by other regulatory mechanisms like miRNAs other than signaling molecules [42]. The family of miR-200 is well known to regulate the EMT program than any other miRNA family. It has been shown that they down regulate the expression of ZEB1 and ZEB2 proteins; interestingly, the expression of miR200 is repressed, in return, by ZEB1 and ZEB2, creating a double-negative feedback loop [43,44]. Likewise, miR-34 family reduces the expression of Snail, which is also known to be down regulated by Snail, establishing one more double-negative regulatory feedback loop between EMT-TFs and miRNAs [45,46]. The EMT-TFs are also regulated at post-translational level. Glycogen synthase kinase 3 beta (GSK3β) phosphorylates Snail at two consecutive serine-rich motifs, which is then targeted for ubiquitination followed by proteasomal degradation [46]. On the other hand, ERK1/2, p38, and JNK phosphorylates TWIST1 at Ser68 which protects it from ubiquitin-mediated degradation [47]. Thus, there are several mechanisms at different levels to control the EMT-TFs, which collectively regulate the activation of EMT program. Although much is known about the transcriptional regulation of ETM but very little information is available on alternative splicing of numerous genes that are also interconnected with EMT progression [48]. Alternative splicing can create and sustain important characteristic of different cell and tissue types [49,50]. For instance, EMT–reliant splicing variations also been reported in human breast tumors [51]. A relevant example of specific splicing factors which are known to modulate EMT is given by ESRP1 and ESRP2, epithelial restricted splicing regulators [52,53]. It has been shown that Snail, a transcription repressor, binds to the ESRP1 promoter at E-boxes and down regulates its target gene expression [54]. Loss of ESRP1 induced the Rac1b expression, resulting in Snail induced EMT progression [55], consequently disturbing actin organization. Recent findings revealed that TGF-β induces the expression of δEF1 and SIP1 which in turn binds to the promoter of ESRP2 and represses its expression resulting in more alternative splicing events during EMT [56]. On the contrary, loss of expression of ESRP2 caused a reduction in cell-cell connection by inducing the expression of EMT-related transcription factors like δEF1 and SIP1, but not SNAIL, SLUG or TWIST [57], implying that ESRP1 and ESRP2 might stop EMT by diverse mechanisms. Overall, the current findings suggest that alternative splicing plays important role in EMT and tumor development via altering alternative isoform expression of genes essential for epithelial and mesenchymal cell morphology and motility [51].
CONCLUSIONS AND FUTURE DIRECTIONS
The emerging studies revealed that EMT is critical for mammary gland development as well as breast cancer metastasis. Breast cancer is a heterogeneous disease. One of the heterogeneity in breast tumors is EMT and cells with stem cell like properties. It is now widely accepted that presence of EMT and cancer stem cells are involved in resistance to make ineffective to cancer therapies. Since breast cancer is heterogeneous in nature, future cancer target therapies undoubtedly need to consider the heterogeneity of cancer cells to increase the patient survival. So, better understanding of processes and factors involves in EMT in cancer progression will play important role in developing of new therapies for not only to bulk tumors but also for cells with stem cell like properties which involves in metastasis, relapse and drug resistance of the tumor. Further, studying the EMT in in vivo is challenging because of lack of animal models and it’s very transient and reversible nature. Recent studies revealed that cancer cells migrate collectively rather than single cells. It will be interesting to study how these cells coordinate each other in the process of EMT and cell migration. Although huge challenges ahead in targeting EMT in cure of cancer, recent advances in imaging and advancements in our molecular understanding of EMT provide subtle leads in this direction.