Frutalin Recognizes Complement C3 Over-Expressed by Neoplasms
- 1. Department of Biochemistry and Molecular Biology, Federal University of Ceará, Brazil
- 2. Center of Experimental Biology, University of Fortaleza, Brazil
ABSTRACT
Frutalin (FTL) is a α-D-galactose-binding lectin obtained from the seeds of Artocarpusincisa L. It has presented several biomedical activities based on the recognition of the carbohydrate binding site. Bioinformatics techniques have sought to elucidate the molecular bases of interaction with biological receptors. Previous studies have demonstrated that Frutalin, in addition to promoting biological activities, is able to recognize differentially expressed glycoproteins in the blood in patients with neoplasms such as breast cancer and acute lymphoblastic leukemia (ALL). The aim of this work was to elucidate the molecular bases of interaction of Frutalin with Complement C3 Protein, a glycoprotein over-expressed in serum of patients with ALL and breast cancer. The possible interaction between FTL with the surface of the complement protein C3 was analyzed using molecular docking, with the Hex 8.0.0 platform. The 10 most energetic clusters were investigated for analysis of interaction energy, binding specificity, CRS-glycoprotein affinity, the amino acids involved and the attraction/repulsion forces. Molecular docking demonstrated high affinity to between FTL and C3 glycans, with high specificity and complexation energy, with 08 hydrogen bonds measuring up to 0.8 angstroms, with high reproducibility. It can be concluded that FTL is a lectin capable of promoting biological and biomedical activities due to the flexibility of its CRS, which allows the recognition of complex glycans present in glycoprotein’s involved in biological phenomena, which makes it a relevant biotechnological tool.
KEYWORDS
- Frutalin
- Breadfruit
- Molecular docking
- In silico
- Cancer
CITATION
Vieira Neto AE, de Oliveira Monteiro-Moreira AC, de Azevedo Moreira R (2018) Frutalin Recognizes Complement C3 Over-Expressed by Neoplasms. J Bioinform, Genomics, Proteomics 3(2): 1034.
ABBREVIATIONS
ALL: Acute Lymphoblastic Leukemia; BC: Breast Cancer; CBS: Carbohydrate Binding Site; FFT: Fast Fourier Transform; FTL: Frutalin
INTRODUCTION
Biomarkers in cancer research
Biomarkers are increasingly useful tools for cancer research. The search for new, specific and sensitive biomarkers continually aims to discover improvements in the diagnosis and confirmation of the neoplasms or that may have practical value in the evaluation of the treatment and in the behavior of the disease [1].
Acute lymphoblastic leukemia of Precursor Cells B (ALL-B) and Breast Cancer are examples of diseases that occur in worrying proportions and have to be more studied. Precursor Cell Lymphoblastic Leukemia-Lymphoma is the most common malignancy in childhood and breast cancer and the most common cancer among women worldwide after non-melanoma skin cancer. Featuring clinically and biologically heterogeneous diseases, early detection, reliable characterization, and accurate diagnosis are critical for reducing mortality [2].
Frutalin as biomedical tool
This work investigates the interaction of Complement C3, which is a differentially-expressed glycoprotein with changes in its glycosylation patterns in both diseases, with Frutalin (FTL), that is a plant lectin with affinity for α-D-galactose and α-D-mannose, obtained from the seeds of Artocarpusincisa L. FTL is a glycoprotein with approximately 66 kDa, which has a CBS in each monomer of its homotetramer. It is able to agglutinate erythrocytes since it is oligomerized under physiological conditions and promotes glycans recognition in two or more CBS simultaneously, agglutinating the red blood cells[3]. The three-dimensional structure of Frutalin has been elucidated by crystallographic methods [4] and has already shown to be compatible with other biological components[5,6]. FTL proved to be efficient in detecting the overexpression of Complement C3, which suggests an association with the development and characterization of cancer in LLA and breast cancer situations[2,7]. The aim of this study was elucidating the molecular bases of interaction of FTL with the C3 protein, in the recognition observed in previous studies.
MATERIALS AND METHODS
Molecular docking
The interaction between Complement C3 and Frutalin (FTL) was analyzed using molecular docking. FTL and Complement C3 structures were available in the Protein Data Bank (codes 4WOG and 2A73, respectively).
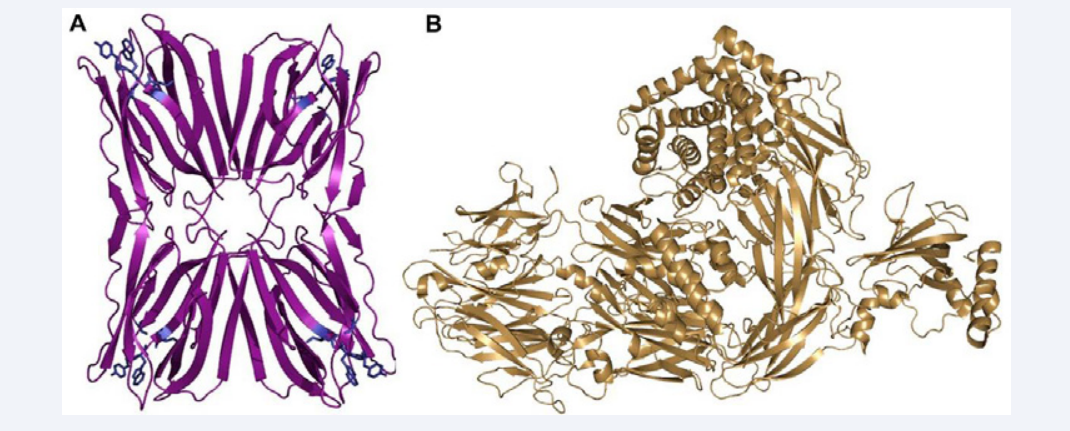
Frutalin and C3 structures (Figure 1) were solved by crystallographic methods [4,8] and have high resolution (1.6 and 3.3 angstroms, respectively).
Figure 1: Frutalin structure (purple) with one carbohydrate recognition site (blue) in each monomer (A); Complement C3 structure (B).
The C3 structure had its ligands removed manually, hydrogens and charges were added in both molecules, prior to perform molecular docking, and all chains were maintained in order to simulate the real interaction in physiological conditions (Figure 1).
The HEX software was used for molecular docking calculations [9], as it is able to predict interactions large molecules in intermediate processors (academic), besides having a platform compatible with didactic purposes. The parameters applied for the docking process were: Correlation type - Shape only; Calculation Device – GPU (graphic process units); FFT Mode (fast Fourier transform) - 3D fast life; Grid Dimension - 0.6; Receptor range - 180; Ligand Range - 180; Twist range - 360 and Distance range – 40; Solutions - 50000. Three-dimensional images of the interactions between ligands and the proteins were depicted using the PyMol software[10].
RESULTS AND DISCUSSION
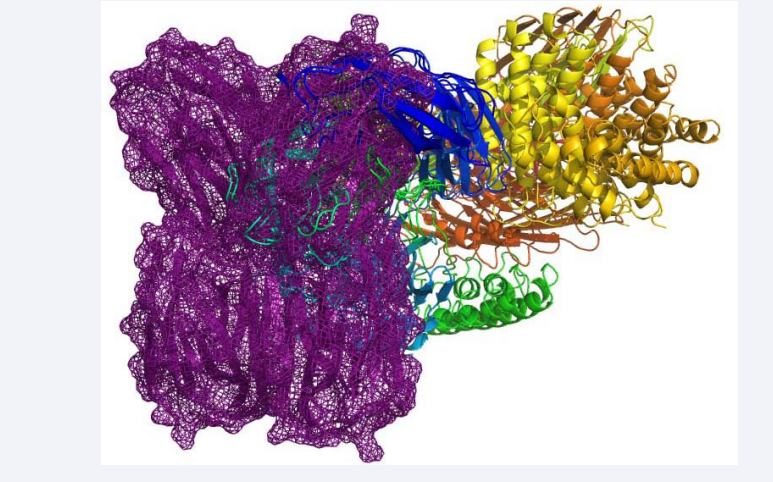
Molecular docking showed the interaction of FTL (receptor molecule) with Complement C3, after 50,000 possible fittings. In in vitro proteomic investigations[2,7] serum samples from patients with cancer were applied in chromatography column to interact with the FTL immobilized matrix. These results showed that the C3 molecule interacts with matrix, being considered a ligand while the FTL was considered receptor. The molecular bases of this interaction were elucidated through computational simulation. The 10 most stable clusters of this interaction were analyzed and initially presented high specificity, since they were overlapped in the same place, which indicates good energy stabilization of the formed complex, in that position (Figure 2).
Figure 2: Overlapping of the 3 most energetic clusters (colored) of C3, which suggests specific affinity for the FTL surface (purple) surrounding the CBS.
A protein has an intrinsic vibratory energy and when it binds with another molecule; its energy is stabilized by the complexation, which was also evidenced with the connection to Complement C3. The fit provided an energy stabilization of up to -26767.43 kcal/mol and proved to be efficient in all 10 most energetic clusters (Table 1). A similar result has been as have been found in other studies concerning the molecular interactions of the FTL in biomedical activities[5,6] (Table 1).
Table 1: Total energy of the interactions between FTL (receptor) and Complement C3 (ligand), after molecular docking simulation.
| Complex obtained by FTL-C3 interaction | Total energy obtained (kcal/mol) |
| Cluster 01: | -26767.43 |
| Cluster 02: | -26556.11 |
| Cluster 03: | -25255.56 |
| Cluster 04: | -24777.30 |
| Cluster 05: | -24430.97 |
| Cluster 06: | -23427.78 |
| Cluster 07: | -22620.34 |
| Cluster 08: | -22402.28 |
| Cluster 09: | -22321.76 |
| Cluster 10: | -21322.84 |
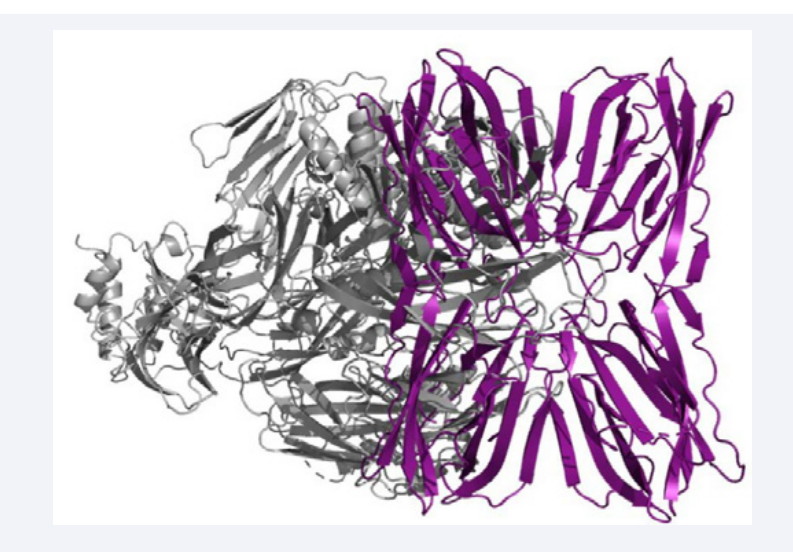
The high complexing energies observed in the 10 analyzed clusters indicate that there is high affinity of CBS for the ligand, and regarding the role of CBS in this recognition, it was important to investigate the surface of FTL and the location of its CBS in each monomer (Figure 3a).
Figure 3: Interaction of Complement C3 (gray) with FTL (purple), evidencing lectin recognition activity, interacting with two tetramer subunits simultaneously.
In fact, C3 is a large glycoprotein and interact with FTL with the involvement of two monomers. In general, plant lectins have their lectin activity measured by the ability to agglutinate erythrocytes, due to their ability to recognize glycans, agglutinating them. This agglutination occurs when the lectin binds to two or more erythrocytes, which recruits more than one CBS[11]. Based on this, FTL recognizes complement C3 with two monomers simultaneously. This interaction increases the stabilization and neutralizes the repulsions and the hydrophobic effects of the region that does not belong to the lectin activity, in Frutalin (Figure 3b).
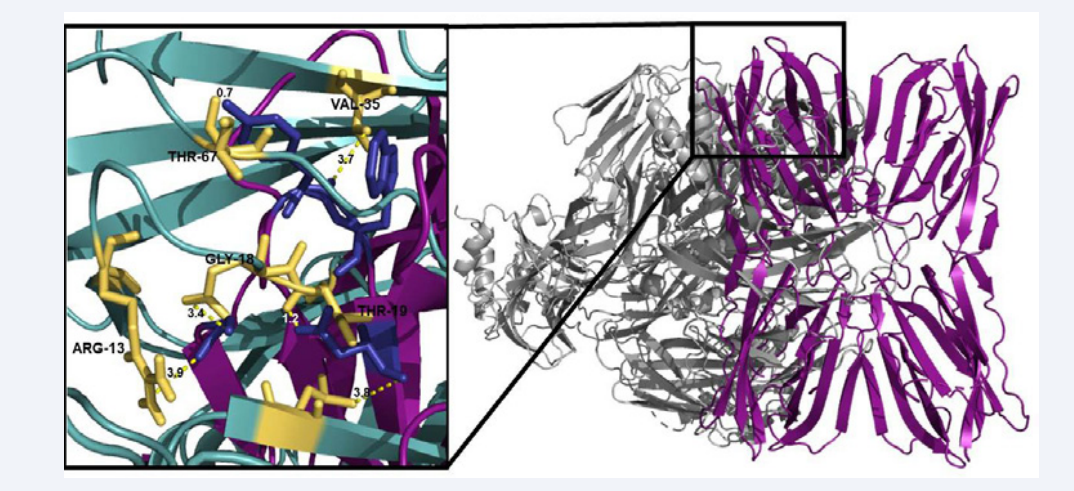
According to previously information about FTL amino acid residues[6] that promote lectin activity (Gly25, Tyr146, Trp147 and Asp149), the interaction of these residues with the amino acids of Complement C3 was measured. As a result, was observed the action of 6 amino acid residues (please specify these 6 residues), which made bonds of up to 0.8 angstroms and could stabilize the region and promote the binding between the two molecules (Figure 4). The results corroborate with in vitro findings [2,7] (Figure 4).
Figure 4: Carbohydrate recognition site of FTL (blue) showing hydrogen bonds with 6 amino acidresidues (yellow) of Complement C3 (0.8 – 3.9 angstroms).
In order to assess the affinity of the FTL for glycoprotein’s, oligosaccharides and glycans with higher molecular size, it can be suggested that the size and flexibility of the CBS promotes greater recognition by stabilizing in the middle of so many bonds. Therefore, the binding energy against a monosaccharide is much lower than that observed in biological activities already reported[12], in which FTL showed a high recognition activity in silico. It is known that plant lectins have been studied because they have specificity and affinity to carbohydrates in several biological processes[13]. Based on this, this work strengthens the suggestion of investigating plant lectins with high specificity to glycans characteristic of specific diseases, seeking biotechnological potential.
CONCLUSION
The molecular docking results reflect a specific interaction between Frutalin and Complement C3 protein. Over expressed in cancer added to its specificity, stability in physiological conditions and its affinity to glycans make it a useful tool in research Concerning cancer and biomarkers. FTL is a plant lectin capable of promoting biomedical activities due to the flexibility of its CBS, which allows the recognition of glycoproteins involved in ALL and breast cancer.
ACKNOWLEDGEMENTS
University of Fortaleza (UNIFOR), Experimental Biology Center (NUBEX), National Council for Scientific and Technological Development (CNPq), National Council for the Improvement of Higher Education (CAPES), Fundação Cearense de Amparo á Pesquisa (FUNCAP) and Agency for Financing Studies and Projects (FINEP), for all support.