Sustainable and Optimised Processing of Tea (Camellia sinensis) and Tea Waste for Bioactive Phytochemicals and Their Applications Using Deep Eutectic Solvents as Alternate Green Solvents
- 1. Former Professor of AcSIR and Chief Scientist, CSIR-Central Food Technological Research Institute Mysore, India
Abstract
Tea is one of the widely consumed beverages and is methodically manufactured. This review describes the results of research studies using deep eutectic solvents (DES) / natural deep eutectic solvents (NADES), as alternate green solvents in the extraction, analysis, solubility enhancement, and application of bioactive constituents from tea. Methods to extract the maximum quantities of active constituents along with their mechanisms are discussed. The yield of the target compounds increased during extraction. Extraction times and quantities of solvents for processing are reduced. Selected modern techniques have been reported for exclusive adsorption of bioactive components. NADES are suitable for the extraction of bioactive compounds. Highly polymeric tea pigments could be isolated from black tea. Silver nanoparticles of selected NADES extracts of green tea exhibited anticancer potential. Lignin is isolated from tea residue waste and tea stem, which can reduce environmental pollution and utilize agricultural waste respectively. Tea enzymes are isolated with more purity, which are useful for industrial applications. Derived extracts can be used as additives in food, pharmaceutical and cosmetic products safely, as they are less toxic, than extracts of conventional organic solvents. Biofilms with high activity are reported using NADES extracts, which are useful for food industry. The use of DES / NADES is expected to increase in the near future. Pilot-scale extraction and evaluation of the economic feasibility are initiated. Researchers are using DES/ NADES as alternate green solvents during tea processing for various purposes. The application of DES/NADES in tea processing is improving.
Keywords
• Tea; Camellia sinensis; Theaceae; Deep eutectic solvents; Bioactive compounds; Catechins; Highly polymeric tea pigments; Polyphenols; Theaflavins; Thearubigins; Theabrownins; Flavonoids; Lignin; Celluloses; Mono and Polysaccharides; Alkaloids; Amino acids; Enzymes; Biofilms; Extraction; Analysis; Stability; Solubility.
Citation
Jaganmohanrao L (2026) Sustainable and Optimised Processing of Tea (Camellia sinensis) and Tea Waste for Bioactive Phytochemicals and Their Applications Using Deep Eutectic Solvents as Alternate Green Solvents. JSM Chem 12(1): 1067.
INTRODUCTION
Significance of phytochemicals from Tea (Camellia sinensis)
Tea is one of the most consumed beverages in the world, because of its flavor, taste and bioactivity. Teas are scientifically and systematically produced in the plantation industry. The global tea market value is approximately 50-54 billion USD, and the predicted compounded annual growth rate is 6-7%. Several varieties of tea are available currently worldwide. The selected famous varieties are green tea (unfermented), oolong tea (partially fermented), and black tea (fully fermented). In unfermented teas, enzymes are initially deactivated, and the original compounds (e.g., flavanols and catechins) of tea leaves are retained. Fully fermented teas means that the original compounds undergo oxidation during fermentation and fully oxidized compounds and their polymers are formed (e.g., theaflavins, thearubigins, theabrownins), later enzymes are deactivated. In partially fermented teas, enzymes are deactivated after partial fermentation. Hence, they contain both the original compounds of tea leaves and oxidized compounds. In addition, small quantities of dark tea, white tea (unfermented), yellow tea (partially fermented), and red tea (fully fermented) are also produced and consumed. Their flavor, taste and biological activity differs according to their chemical composition [1]. Tea is a rich source of biologically active ingredients, particularly polyphenols (e.g., flavonols, flavanols and theabrownins), alkaloids, methylxanthines (e.g., caffeine, theobromine and theophylline), polysaccharides, tannins, vitamins, proteins, amino acids (e.g., theanine), minerals, terpenoids, and caffeic acid [2]. Flavanols are epistructured catechins such as epicatechin (EC), epicatechin gallate (ECG), epigallocatechin (EGC) and epigallocatechingallate (EGCG- major), and nonepistructured catechins such as catechins (C), catechingallate (CG), gallocatechin (GC) and gallocatechin gallate (GCG). Catechins are the major antioxidants in tea. These possess the ability to scavenge free radicals and stimulate the detoxification activity of different enzymes, such as catalase, glutathione reductase and glutathione peroxidase. Because of the hydroxyl groups in the gallate moiety, EGCG and ECG are highly effective free-radical scavengers compared with many other standard antioxidants, such as ascorbic acid, tocopherol, and trolox. Catechins also retard extracellular matrix degradation induced by ultraviolet (UV) radiation and pollution. Catechins also directly affect the skin by activating collagen synthesis and inhibiting the production of matrix metalloproteinase enzymes [3]. The biological activities of tea are reported and described [4]. It is an effective antioxidant owing to its free radical scavenging and metal-chelating abilities. Hence, it is active against inflammation, clastogenesis, and several types of cancers (oral cancer, esophageal and gastric cancer, intestinal / colon / rectal cancer, lung cancer, prostate cancer, breast cancer, liver cancer, skin cancer, blood cancer and urinary tract cancer). Tea reduces DNA damage and mutagenesis due to oxidative stress or the presence of promutagens through antioxidant function, blocking activation pathways of mutagens, and suppressing the transcription of enzymes involved. Inhibition of low density lipoprotein (LDL) peroxidation, and suppression of fatty acid synthase suggests that tea may play a role in preventing cardiovascular diseases. In addition, black tea has beneficial effects on the gastrointestinal tract affecting motility, absorption and microflora. It is also antiviral owing to its enzyme-inhibiting and receptor-blocking properties. Catechins possess the anticancer, antihypertensive, anticoagulant, and antiulcer effects [4-6]. Endocrine effects (e.g., antidiabetic activity, antihyperlipidemic effects, antithyroid potential), inhibition of bone resorption, cardiovascular effects (e.g., antihypertensive effects, anticoagulant antiplatelet effects), gastrointestinal effects (e.g., antiulcer effects), bone growth (e.g., bone growth promotion, antiosteoporotic and antiosteopenic effect) and organ protective effects (e.g., age- related memory impairment and neuroprotective effects, cardioprotective effects, hepatoprotective effect, nephroprotective effects, anticataractogenic effects, in utero effects); CNS effects (e.g., antiepileptic effects, antiparkinson effects, antialzheimer effect, anti-anxiety effects); autoimmune state (e.g., autoimmune myocarditis, Sjogren’s syndrome, respiratory disorders, antiallergic effects and immunological insufficiencies). Green tea and white tea extracts exhibit antibacterial properties and antimicrobial effects [6,7]. Fresh tea leaves contain phenolic compounds (i.e., catechins and flavonoids), which can be oxidized during the fermentation under the enzymatic action of polyphenol oxidase and peroxidase to form tea pigments [i.e., theaflavins (TFs-simple polymers of catechins), thearubigins (TRs-polyhydroxy polymers formed by the oxidative coupling and other reactions of catechins, TFs, amino acids), and theabrownins (TBs-high polymers formed by the complexation of polysaccharides, amino acids, proteins)]. TRs and TBs are considered as highly polymeric tea pigments (HPTPs). Tea pigments possess natural colouring and preventive health care functions, and applied in food, medicine, and health care products. Theabrownins exhibits antihypercholesterolemia and antihyperlipidemic effects by producing hepatic chenodeoxycholic acid, activating the hepatic farnesoid X receptor, and inducing hepatic lipolysis. Theabrownins interfere with the circadian cycle, prevent fat and insulin resistance and also exhibit hypolipidemic activities. Theabrownins and polysaccharides boost the activity of antioxidant enzymes [8,9]. Polyphenol oxidase (PPO) is a copper-containing terminal oxidase found in natural organisms that catalyses the formation of unstable quinones using phenols. Theaflavins in black tea are formed from catechins in green tea, in vitro via PPO. Peroxidase (POD) is a porphyrin- containing metalloprotein that catalyzes the oxidation of theaflavins to thearubigins, owing to the hydroxylation of POD. Significant biological functions and health benefits of theaflavins, thearubigins include anti-inflammatory, hepatoprotective, antimutagenic, neuroprotective, and antioxidant activities, and anticancer properties. Tea pigments have extensive application in food, pharmaceutical, and chemical industries. Tea peroxidase is used in bioremediation, textile synthetic dye decolorization, polymer synthesis, paper and the pulp industry [10,11]. Lignin is a phenolic complex polymer and is hydrophobic and amorphous. It is comprised of phenylpropane units (e.g., sinapyl, p-coumaryl and coniferyl) with hydrophilic functional groups (i.e., carboxylic acids and hydroxyl groups) and possess a wide range of molecular weight. Lignin has antioxidant and antibacterial activities. Lignin is valuable in pharmaceutical industry as antioxidants, cosmetics, antimicrobial agents, prebiotics and drug carriers. Lignin applications include lignin based nanoparticles, flocculants, and luminescent materials, paints, dispersants, adhesives, and bio-oil. Lignin is also used to control environmental pollution as bioremediation of dyes and heavy metals, dust controlling, water retention agents, and soil stabilization [12,13].
Caffeic acid has beneficial effects on the human immune system. Caffeic acid possesses antioxidant, anticancer, antidiabetic, antihypertensive, antimicrobial, hepatoprotective, and antiviral activities. Caffeic acid reduces blood glucose levels by inhibiting the enzymes α-amylase and α-glucosidase in type-2 diabetes. Caffeic acid exhibited anticancer and anti-inflammatory activities by inhibiting various transcription factors [14]. Caffeine has the potential to treat cognitive disorders, including Alzheimer’s disease, Parkinson’s disease, depression, and schizophrenia [15]. Low doses of caffeine have positive effects on cognition, whereas higher doses improve exercise performance. Caffeine has ergogenic effects on endurance, muscle strength, and high intensity exercise. Tea polysaccharide combine with proteins to form acidic polysaccharides or acidic glycoproteins. Polysaccharides in tea are generally water-soluble. Combined with selenium, it can improve the antioxidant activity of polysaccharides. Tea polysaccharides contribute to antioxidant, immune regulation, anticancer, antidiabetic and antiobesity effects [8]. Conventional extraction methods (i.e., solvent extraction, Soxhlet extraction, hydro / steam distillation) and novel extraction technologies (e.g., ultrasonication assisted extraction, microwave assisted extraction, pressurized liquid extraction, subcritical water extraction, supercritical fluid extraction, and ionic liquid-based extraction) for the isolation of bioactive compounds from tea have been reported [2]. Conventional separation techniques (e.g., column chromatography and semi preparative HPLC) and supplementary modern techniques (e.g., high speed counter current chromatography, and supercritical fluid chromatography) have been used for the purification of bioactive compounds from tea [16]. Applications on a large scale are limited. Priority is being given to the development of highly efficient, safe and environmentally friendly large scale extraction and purification methods for industrial use [17]. However, extraction of tea, separation, purification and estimation of bioactive compounds from tea using green alternate deep eutectic solvents (DES) / natural deep eutectic solvents (NADES) have not been consolidated and discussed. This review aims to fill this gap.
Deep eutectic solvents and natural deep eutectic solvents
World population faces sustainable challenges, which are connected with health owing to environmental pollution. It needs to be controlled. Bioactive phytochemicals from foods provide solutions due to their health benefits. Various extraction and separation techniques are required for isolation of bioactive compounds. Conventional techniques are maceration, percolation, Soxhlet extraction, and solvent extraction. Disadvantages of these methods are longer extraction periods, high cost, and low yield. Modern techniques (i.e., enzyme-assisted extraction, ultrasound-assisted extraction, microwave-assisted extraction, subcritical fluid extraction, supercritical extraction, and high pressure-assisted extraction) are associated with shorter extraction times, low cost and high purity of target compounds. Various traditional solvents are used in these methods. There are several problems associated with both these solvent. These are harmful, flammable, toxicity, thermal instability, polarity, solubility, poor selectivity, their moisture content, disposal and concentration. Other concerns associated with the earlier methods are the recovery of bioactive components from the solvent, and transformation of the chemical structures of compounds during extraction due to various reactions (i.e., ionization, hydrolysis, oxidation etc.). Water is the most useful solvent in the pharmaceutical, agricultural, cosmetic, and food industries as physical and chemical characteristics of water satisfy most of the conditions required. But, water can extract polar and hydrophilic compounds only. Water-soluble impurities are another major problem. There is a demand for environmental responsive green solvents. Green solvents are essential for the extraction of bioactive compounds from plant sources. Value-added products related to the food, beverage, nutraceutical, pharmaceutical, and cosmetic industries need to be safe for human beings. Deep eutectic solvents (DES) are easily manufactured and found to be less harmful to organisms as these are less toxic than routine organic solvents. These solvents (homogenous liquids) are formed by linking a hydrogen bond acceptor (HBA) (e.g., quaternary ammonium) and a hydrogen bond donator (HBD) (e.g., urea, carboxylic acids or amine) at a quantified stoichiometric ratio through thermal / microwave heating with mixing / grinding / freeze drying [17-19]. Selected DESs are prepared using plant primary metabolites (e.g., carbohydrates, organic acids, alcohols and amino acids) and are called as natural deep eutectic solvents (NADES). Deep eutectic solvents are flexible / designable / tailored easily by changing the structure or ratio of parent components as per the need of applications, including extraction, isolation, separation, analysis of target compounds, and use as drug carrier. Complex hydrogen bonding exists between the components (HBD and HBA) of deep eutectic solvents [17,20]. This results in the depression of the melting point of the formed DES, compared to their parent compounds. DESs are non-flammable and have low vapor pressure. A review on the isolation of bioactive compounds (e.g., phenolic acid, flavonoids, tanshinone, keratin, tocols, terpenoids, carrageenans, xanthones, isoflavones, alpha mangostin, genistin and apigenin) using deep eutectic solvents has been published [20]. The extraction of bioactive compounds using DESs in terms of the quantity of sample, amount of solvent, extraction time and energy consumption is investigated and discussed [19]. The isolation of value-added compounds from agro-, forest- and food waste using DESs is illustrated [21]. Kalyniukova et al. [22], reviewed the extraction of bioactive compounds from medicinal and herbal plants using DESs. A systematic review of the extraction strategies of polyphenols from different plant sources using DES has been presented [23]. Lee et al. [24], presented a review of the isolation of organic and inorganic analytes from aqueous media using hydrophobic DESs. Wen et al. [25], discussed a study on the toxicity of cholinium-based deep eutectic solvents and found it to be minimal. These cholinium-based eutectic solvents have been reported to be decomposable. Various applications of NADES have been discussed [26]. In general NADESs have low toxicity and minimal conservational impact. Ferreira et al. [27], proposed a scheme to evaluate the toxicity of NADES based on earlier reports. NADES have the advantage of reproducing natural recovery methods for water insoluble primary and secondary metabolites in plants. NADES are used to isolate bioactive compounds from natural sources [28]. Researchers have investigated the physicochemical properties, functionalities, molecular interactions and solubilizing abilities of NADES, particularly hydrophilic NADES to explore their prospects [29]. The key parameters to develop extraction processes for isolation of bioactive compounds are NADES constituents and their molar ratio, density, viscosity, polarity, pH, stability and toxicity. NADES extracts can be included directly in food products, when used to separate and dissolve bioactive compounds (e.g., antioxidants, antibacterials and flavorants), including food additives. Owing to their advantages (i.e., easy preparation/synthesis, low volatility, high boiling points, water solubility/miscibility, extensive liquid range, high stability, sustainability, reusability, commercial viability, non-inflammability, biodegradability, renewable sources, eco-friendliness and insignificant toxicity compare to conventional solvents), DES/NADES are emerging as alternate green solvents in the processing of tea for bioactive constituents as well as their estimation.
SUSTAINABLE OPTIMIZED EXTRACTION, PRODUCTION, SEPARATION, ANALYTICAL PROCEDURES AND APPLICATIONS OF TEA CONSTITUENTS USING DEEP EUTECTIC SOLVENTS
In this review, details of sustainable extraction technologies, separation processes, and analytical methods of various teas using deep eutectic solvents are discussed, and presented. The stability of bioactive compounds increases with the use of deep eutectic solvents. It covered the extraction of tea using DES in combination with microwave irradiation (MA-DES), ultrasonication (UA DES), deep eutectic solvent-iron (II, III) oxide/molecularly imprinted polymers (DES-Fe3 O4 /MIPs), magnetic solid phase extraction (M-SPE), mechano-chemical extraction (MCE), deep eutectic solvent-solid liquid extraction (DES–SLE), magnetic molybdenum disulphide base deep eutectic solvent molecularly Imprinted Polymer (Fe3 O4 @ MoS2 @DES-MIP), molecularly imprinted resins cellulose nanofiber aerogels (MIRs@CNF AG), Magnetic agarose nanoparticles micro-solid phase extraction and deep eutectic solvents (MANPs-D- μSPE-DES), hollow fiber liquid microextraction (HF-LPME), and deep eutectic solvent pretreatment. The constituents of the DES extracts are analyzed using UV, FT-IR, HPLC-UV/DAD/ MS, Ultra-high-performance liquid chromatography with quadrupole-time-of-flight tandem mass spectrometry (UHPLC-Q-TOF- MS/MS), ultra-high-performance liquid chromatography with triple-quadrupole tandem mass spectrometry (UHPLC-QqQ-MS/MS), mass spectrometry selected ion monitoring (MS-SIM), X-ray diffraction (XRD), Dynamic light scattering (DLS), inductively coupled plasma-optical emission spectroscopy (ICP-OES), X-ray photoelectron spectroscopy (XPS) and thermogravimetric analyser (TG). The extraction mechanism is explained using a conductor-like screening model for the segment activity coefficient (COSMO-SAC), molecular dynamics simulations (MDS) and density functional theory (DFT). Solubility studies of bioactive compounds are discussed based on the conductor like screening model for real solvents dimerization, aggregation and reaction extension (COSMO-RS- DARE) models. The results and conclusions are provided in Table 1 along with details of the matrix, DES/NADESs (composition and molar ratio as well as water content), optimized conditions (i.e., extraction time, temperature, microwave energy and time, ultra-sonication energy and time details, stirring speeds and times, centrifugation speed and times etc.), and methods of extraction, determination and identification of target compounds. Outcome of the DES/ NADES processing of various teas, tea residue waste and tea agricultural waste is provided in add scheme figure and add support information.
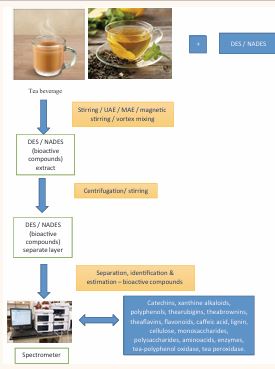
Extraction of bioactive compounds from tea (Fig.1)
Figure 1 EXTRACTION OF BIOACTIVE PHYTOCHEMICALS FROM TEA USING DES / NADES
Table 1: Sustainable and optimised procedures for bioactive molecules from tea and tea waste using deep eutectic solvents as alternate green solvents
|
No |
Matrix |
Compounds |
DES /NADES |
Optimized condition (HBA-HBD)* |
Method Determinat ion |
Yields / conclusions |
Reference |
|
1 |
Chinese green tea |
Catechin (C), epicatechin gallate (ECG), epigallocatec hin gallate (EGCG) |
HBDs Ethylene glycol, glycerol, 1,2-butanediol, 1,4-butanediol, 2,3-butanediol, 1,3-butanediol, 1,6-hexanediol HBA choline chloride |
Choline chloride- ethylene glycol (1:5) +30%water, heating method S/L- 0.0625 g/mL, time - 30 min, temp. - 60 °C. centrifugation, mixed with mobile phase of HPLC, filtration (0.2mm). |
HPLC |
Quantities (mg/g) C-3.629, ECG-35.25, EGCG - 114.2. |
[18] |
|
2 |
Green tea |
Catechins |
choline chloride, cactic acid |
Choline chloride- lactic acid extraction S/L-1g/35mL, microwave temp-66 °C, time-8 min. recovery-catechins, AB-8 macroporous resin, Elution water, aqueous ethanol (90%) |
MAE-DES |
Yield catechins- 154 mg/g, recovery -catechins: 75 to 86% |
[30] |
|
3 |
Green tea |
Alkaloids, theobromine, theophylline |
Choline chloride, ethylene glycol, glycerol, 1,4-butanediol, urea, formic acid, acetic acid, propionic acid. |
Choline chloride+Urea (1:2), Fe3O4/ MIPs FeCl2.4H2O-6.0g,+ FeCl3.6H2O- 15.6g)+ HCl -2.55mL,12M,+ water-50 mL, added to- NaOH-250mL,1.5M, stirring and washing, vacuum drying-24 h. DES-Fe3O4/MIPs, Fe3O4/MIPs (200mg)+DESs-2 mL(in isoprponol- 8ml), stirring@80°C-24h, polymerisation. green tea-1g+ ethyl alcohol-20mL, ultrasonication temp.- 50oC, time-1h, filter. added to M-SPE, washing solution- methanol-acetic acid (8:2). |
DES-Fe3O4 / MIPs, M-SPE. FT-IR, FE-SEM, HPLC. |
Quantities-mg/g theobromine-4.87, theophylline-5.07. recovery values theobromine-92%, theophylline-86%. |
[37] |
|
4 |
Green tea |
Catechins |
Betaine, sucrose, D-sorbitol, D-maltose, D-glucose, xylitol, glycerol, citric acid, D-fructose, D-galactose, urea, maltitol. |
Betaine-glycerol-D- glucose (4:20:1)- 19% water, pH-7.16, S/L-100 mg/1.8 mL, ultrasonication time - 6.4 min. |
HPLC-UV |
Quantities EGCG- 102 mg/g, scale up-55fold. Storage EGCG in DES-60% (after 3 weeks). |
[17] |
|
5 |
Green tea |
Catechins, alkaloids, flavonoids |
Choline chloride, ethylene glycol, 1,2-butanediol, 1,3-butanediol, 1,4-butanediol, 2,3-butanediol, 1,6-hexanediol, malonic acid, lactic acid, malic acid, acetamine, methyl urea, urea |
Choline chloride -1,4-butanediol (1:1)+40%water, S/L- 0.05 g/mL, MCE speed - 4 m/s, time - 60 s |
MCE UPLC-HR- MS-SIM |
Yields (mg/g) EGC - 32.70, EC - 7.14, EGCG - 72.87, ECG - 12.58, theobromine - 0.37, caffeine - 22.38, kaempferol - 0.0031 |
[48] |
|
6 |
Chinese Dark Tea |
Caffeine |
Choline chloride, D-sorbitol, urea, oxalic acid, citric acid, L-(+)- diethyl tartrate, zinc chloride, lactic acid, propanetriol. |
Choline chloride + lactic acid (1:1) + 31 % water, S/L - 1:29, vortex, ultrasonication time -38 min, temp. – 58 oC, pH - 1.14 centrifugation- speed-10000 rpm, time-5min, supernatant filter-0.45μm, (HPLC). recovery-XAD-200, macroporus resin, adsorption extract—40mL, resin- 1.0g, temp.-25oC, time- 6 h, shaker speed-100rpm, equilibrium, filtration, desorption resin, ethanol-40mL, ultrasonication, temp.-25oC, time-6h, filtration, caffeine Solution. |
HPLC |
Yield caffeine - 27 mg/g. recovery – 95 to 96%, caffeine purity - 24% |
[39] |
|
7 |
Teas green teas (9) black teas (9) fruit teas (2) |
Catechin (C), epicatechin (EC), epigallocatec hin (EGC), epicatechin gallate (ECG), epigallocatec hin gallate (EGCG), gallocatechin (GC), gallocatechin gallate (GCG)) |
HBDs malic acid, citric acid, L-lactic acid HBA choline chloride, Girard’s reagent T (betaine hydrazide hydrochloride) |
Malic acid:GrT (2:1) + 30% water, SLE S/L - 1:10 mg/mL, stirring speed -1100 rpm time - 50 min, temp. – 50 oC. centrifugation speed - 2000 ×g time -5 min filtration UHPLC-UV |
DES–SLE– UHPLC– UV |
Quantities (mg/g) major compounds green tea EGCG - 27.7 to 63.1, black tea GC - 6.3 to 22.8, fruit teas EGC - 8.07 to 8.4. |
[31] |
|
8 |
Green tea |
Catechin, epicatechin, epigallocatec hin, epicatechin gallate, epigallocatec hin gallate. |
Fe3O4@MoS2@ DES- MIP adsorbent vinyl pyrrolidone (VP), malonic acid (MA), iron (III) chloride hexahydrate (FeCl3.6H2O), sodium acetate (NaOAc), molybdenum disulfide (MoS2), ethylene glycol dimethacrylate (EGDMA), methacryloxy propyltrimethoxy silane (MPS). |
VP:MA(1:1) Fe3O4@MoS2@DES -MIP sample solution-3 mL MIP-15mg, shaking time-120 min, temp.-40oC, solution MSPE, HPLC |
MSPE, HPLC |
Recovery (%) catechin- 85 to 88, epicatechin- 81 to 84, epigallocatechin- 80 to 84, epicatechin gallate – 80 to 84, epigallocatechin gallate – 90 to 99. green tea catechins - 55% |
[32] |
|
9 |
Green tea |
Epicatechin (EC), epigallocatec hin (EGC), epicatechingg allate (ECG), epigallocatec hin gallate (EGCG). |
HBA choline chloride HBDs glycerol ethylene glycol 1,2-propanediol glucose D-sorbitol sucrose1 xylitol citric acid lactic acid Malic acid Malonic acid Maleic acid |
choline chloride- glycerol (1:2)+40% water, S/L-1:36 centrifugation, vortex, ultrasonication, U.P.-461.5W, time -21min. centrifugation, speed- 12,000×g, time-10min, supernatant HPLC, TPC, AA. |
HPLC, TPC, FRAP, DPPH, ABTS |
TPC - 243 mgGAE/g, FRAP - 332 mmol Fe(II)/100 g DW, DPPH - 215 mmol trolox/100 g DW, ABTS - 99 mmol trolox/100 g DW. quantities - mg/g DW EGC - 24, EC - 9, EGCG - 94, ECG - 36 |
[33] |
|
10 |
Tea seed oil |
Phenolic compounds (free and bound) |
Choline chloride, glycerol, xylitol, ethylene glycol, malonic acid, propylene glycol. |
Free phenolics choline chloride
vortex, water bath, temp.-50°C, time- 1h vortex-1mn@15 min interval centrifugation@RT speed-3,500 rpm, time-15 min, DES phase bound phenolics Choline chloride - glycerol- ethylene glycol (1:1:1) choline chloride
centrifugation, speed-3,500 rpm time- 10 min. oil phase+DES-6g, repetition of steps, DES Phase. recovery DES phase, adsorption XAD-12 resin columnwash- water-200mL, elute-MeOH-Water (1:1)-200mL, eluent-evaporate, residue, dissolution, methanol-water (1:1), filteration, organic membane filtrate, analysis. |
UHPLC-Q- TOF- MS/MS, UHPLC- QqQ- MS/MS. |
25 compounds free phenolic compounds-83.91 μg/g, bound phenolic compounds-25.71 μg/g. bound phenolic compounds-25.71 μg/g. |
[42] |
|
11 |
Black tea |
Flavonoids morin, quercetin, kaempferol. |
Tetraethylammo nium chloride, lactic acid, borneol decanoic acid |
Dark tea-1g, water -50 mL, boiling- 30min. cool,infusion-sample MANPs-D- μSPE-DES preparation MANPsagarose powder (0.1 g)+HOAc(5%),+HCl 250 μL@ 85oC (2.5 h). FeCl2⋅4H2O- 0.67g,+ FeCl3⋅6H2O-1.7 g +in acidic mixture, mechanical stirring+ TPP-7 mL + NaOH(25%)-16 mL,stirring-1.5h, MANPs-strong magnet washed-water dried @65oC. DEStetraethylammonium chloride- lactic acid (1:3)MANPs-7.3 mg + sample solution (pH- 4.9) – 10 mLvorex-2.1 min, MANPs (flavonoids adsorbed),separated-magnet elutionMANPs with flavonoids +DES -100µL,ultrasonication- 5min,DES (flavonoids) HPLC-UV. |
HPLC-UV MANPs-D- μSPE- DES |
Quantities-mg/L LODs morin - 0.2,quercetin
- 0.9,kaempferol – 3.3. Black tea morin
<LOD. |
[45] |
|
12 |
Green tea |
Polyphenols |
HBA choline chloride, HBDs ethylene glycol, glycerol, glucose, oxalic acid, citric acid. |
choline chloride- ethylene glycol (1:2), S/L-1:40, magnetic stirring water bath temp.- 70oC, time -3h, centrifugation, speed-4,000×g time-25 min |
COSMO- SAC UV-Vis |
Total polyphenols- 20.12% |
[41] |
|
13 |
Green tea |
Caffeic acid |
Leucine, arginine, serine, lactic acid. |
Serine-lactic acid (1:4)+30%MeOH, green tea-5g, water-100 mL, time- 15 min. temp.-60oC, centrifugation, speed-4000rpm, time-10 min., filter- 0.45mm filtrate - sample HF length-8 cm (~0.22mL). DES filled in HF, dipped in sample. pH-3, salt-10% (w/v), time-6.8 min, temp.-25oC, stirring speed-840 rpm. DES extracts analyte, HPLC-UV |
HF-LPME, HPLC- UV |
Caffeic acid LOD- 0.3 ng/mL, LOQ-0.9 ng/ mL. EF - 418 to 438, recovery - 92 to 97%. green tea- 0.022mg/g |
[46] |
|
14 |
Kangzhuan dark tea |
Theabrownin s theabrownin fractions (TBFs) 6,7-dihydroxycou marin-6- glucoside, erucamide, caffeine, neohesperidin. |
HBA choline chloride, HBDs glycerol, ethylene glycol, sucrose, xylitol, citric acid, malic acid, gallic acid. |
Choline chloride- malic acid (1:2) + 30% water, S/L-1:20,ultrasonication U.P.-577W,time-25 min, centrifugtion speed-12000×g time-5min, supernatant, recovery,DES extract, filtration, concentration, ethylacetate extraction, aqueous phase, ethanol addition (80%), storage-8h, centrifugation speed- 4200×g, time-20min, precipitate, water dissolution, lyophilisation, theabrownins. |
UAE-DES DPPH, ABTS, FRAP. LC- MS/MS |
Yield theabrownins -12.59%, TBFS (60-40% methanol fraction), highest antioxidant activity DPPH - 45 μM AA /g, ABTS – 179 μMtrolox/g,FRAP- 371μM Fe(II)/g.49 compounds |
[44] |
|
15 |
Green Tea |
Green tea catechin extract (GTE)-DES- silver nanoparticles- anticancer activity, |
Urea, glycerol, betaine. |
DES based GTEs silver nanoparticles preparation green tea powder (225 mg),DES (glycerol:urea - 1:1+23%water)- 4.5mLvortex, ultrasonication, temp-RT,time- 45min centrifugation, speed-2898×g time-15min,ethanol dilution-10 fold,ultra-centrifugation- speed- 20,000×g time-1h,filteration-0.45mm, GTEs+2mMAgNO3 (1:1),stirring in dark-24h. DES-GTE-AgNPs |
UV-Vis, FT-IR, XRD, DLS, SEM, ICP-OES. |
DES based GTE silver nanoparticles – 235% more catechins. anticancer activity comparison, DES(glycerol:urea)- GTE silver nanoparticles – 60mg/mL doxorubicin -12.5mg/mL |
[53] |
|
16 |
Green tea (GT) |
Catechins chitosan films with DES extract |
Choline chloride, glycerol, lactic acid. |
Choline chloride +glycerol +lactic acid (1:1:1). NADES-10g,GT powder-0.5g, stirring,temp-80oC, time-15 min. filtration, filtrate centrifugationspeed- 21,000×g, temp-25 oC, time-30 min. supernatantNADES-GT extract. chitosan films |
HPLC- DAD, HPLC-MS, FT-IR, DPPH, FRAP. |
NADES GT extracts- antioxidant activity DPPH – 33 μM trolox eq,FRAP – 397 μMtrolox eq chitosan films-excellent properties- food use. |
[50] |
|
17 |
Green Tea |
Catechins epicatechin- EC, epigallocatec hin- EGC, epicatechinga llate-ECG, epigallocatec hingallate- EGCG |
Betaine, Urea, Malic Acid, Glycerol, Tartaric Acid, Sorbitol, Citric Acid, Sorbitol. |
Betaine-urea (1:2) +30%water (BU), S/L-1:20, ultrasonication, temp-50oC, time-30min, centrifugation, speed- 6000rpm time-10min, supernatant taken. |
HPLC, TPC, TFC, DPPH, FRAP, ABTS. |
GT-DES(BU) extract TPC – 134 mgGAE /g, TFC - 21mgRE/g, FRAP - 1.91 mmol Fe2+/g,DPPH IC50 - 0.09mg/mL,ABTS IC50 - 7.03mg/ mL.EC - 9.0 mg/g, EGC - 54.0 mg/g, ECG - 18.0 mg/g, EGCG - 29.2 mg/g. |
[34] |
|
18 |
Green tea |
Catechins |
Choline chloride, ethyl glycol, 1,4-butanediol, 1,2-propanediol |
Choline chloride + 1,4-butanediol (1:2)+50% water, S/L-1g/20mL, ultrasonication, temp.-50oC, time- 30 min, sentrifugation- 8000rpm, filteration, DES extract to MIRs@ CNFAG column washings methanol- 2ml, water-3mL,tea DES extract- 20mL,washing, methanol-Water (10:90)-5mL,elution,methanol-acetic acid (95:5)-5mL,eluent vacuum evaporation temp.-50oC catechin- purified. |
MIRs@ CNF AG, FT-IR, SEM, XRD, XPS, TG. |
Catechins adsorption capacity- 1stcycle~74 mg/g, 5thcycle~65 mg/g purity-86.75% |
[35] |
|
19 |
Anji white tea (green tea) |
Polysacchari des monosacchari descomposition fucose, rhamnose, arabinose, galactose, glucose, mannose, xylose, fructose, galacturonic acid, glucuronic acid. |
Choline chloride, 1,6-hexanediol, ethylene glycol, 1,4-butanediol. |
Choline chloride- 1,6-hexanediol (1:1)+30% water, S/L-5g:150mL, ultrasonication U.P.- 35kHz, 50W, temp – 25 oC, time – 40 min, centrifugation-speed - 9000×g, time - 20min temp.- 4 oC, filtrate+ethanol (95%, 4 fold). precipitation, precipitate, freeze drying, centrifugation, speed - 9000×g time - 20 min. temp.- 4 oC, drying – 40 oC, polysacchrides, dissolution-water, dialyzed, time – 2 h, temp. – 4 oC, lyophilisation, polysaccharides- purified, analysis |
FT-IR, ion exchange chromatography, antioxidant activity, DPPH, ABTS, FRAP, ORAC. |
Polysaccharides yield - 19.2%, hypoglycaemic activity - glucose uptake - 2.27times, Inhibition α-amylase - IC50- 0.36 mg/mL, α-glucosidase - IC50-0.18 mg/ mL. antioxidant activities of DES- UAE TPs are higher. |
[47] |
|
20 |
Green tea |
Catechin (C), epigallocatec hin (EGC), epicatechin gallate (ECG), epigallocatec hin gallate (EGCG), kaempferol (KAM), theobromine (TBM), caffeine (CAF). |
Choline chloride, 1,2-butanediol, 1,3-butanediol, 1,4-butanediol, 2,3-butanediol. |
HBA:HBD-1:1 Choline chloride: 2,3-butanediol |
MDS, DFT, |
DES selected EGC. interaction energy of EGC & DESs : - 24 to -95 kJ/mol. |
[36] |
|
21 |
Dianhong black tea |
Polyphenols, flavnoids, alkaloids, catechins, proteins, amino acids, monosacchari des, theaflavins, thearubigins, theabrownins ,tannins. |
HBA choline chloride HBDs ethylene glycol, glycerol, citric acid, proline, malic acid, fructose, glucose, urea. |
DES choline chloride - ethylene glycol, tea powder (60- mesh) – 8 g, DES – 200 mL, ultrasonication temp. – 60 oC, time – 20 min. extract-cool temp.- 22 to 25 oC, centrifugation speed - 11963×g, time - 20min. supernatant, filter - 0.22 µm, analysis.D101 - macroporous resin, column DES extract – 40 mL loading time – 12 h, elution, water - 4.5 L (20 fold column volume), ethanol (95%) – 1 L, ethanol elute, concentration - under vacuum, final volume – 40 mL, filter - 0.22 µm, analysis. |
DES-UAE, UHPLC, LC-MS. |
LC-MS showed the presence of 50 compounds. major compounds quantities - mg/g epicatechin gallate - 8.75 theaflavin - 3,3′- digallate - 1.59, glucose - 4.97, theanine - 19.72, caffeine - 28.29. |
[49] |
|
22 |
Tea –two leaves and bud |
Polyphenol oxidase |
HBAs methyl trioctyl ammonium chloride, thymol, menthol. HBDs ethylene glycol, glycerol, hexanoic acid, octanoic acid decanoic acid, dodecanoic acid, tetradecanoic acid, oleic acid, camphor, thymol. |
Crude PPO preparation fresh tea leaves -10 g+ citric acid- Na2HPO4 buffer (0.1 M, pH 5.6, glycerol 10%) + vitamin C-0.2 g + EDTA-1mmol + PVPP - 4 g, homogenisation - 3 min. storage – 2 h temp. – 4 oC,filtration - gauze (4layers),filtrate, centrifugation, speed - 11000×g, time – 20 min. supernatantPPO (crude enzyme) storagetemp. – 4 oC. PPO purification DES thymol- dodecanoic acid (1:1) PPO crude extract- 20 mL, shaking, (NH4)2SO4 (41 %), DES:crude extract (v/v) - 0.5:1,mixed, extraction time - 75 min, pH - 5.6, temp. – 25 oC, TPP system, centrifugation, speed - 8000×g, time – 10 min. temp. – 4 oC, middle phase + citric acid-Na2HPO4 buffer (0.1M, pH5.6),tea PPO precipitate layer, dissolution,citric acid-Na2HPO4 buffer (0.1M, pH 5.6) – 5 mL, analysis. PPO purification DES thymol-dodecanoic acid (1:1) PPO crude extract- 20 mL, shaking, (NH4)2SO4 (41 %), DES:crude extract (v/v) - 0.5:1, mixed, extraction time - 75 min, pH - 5.6, temp. – 25 oC, TPP system, centrifugation, speed - 8000×g, time – 10 min. temp. – 4 oC, middle phase + citric acid-Na2HPO4 buffer (0.1M, pH5.6), tea PPO precipitate layer, dissolution, citric acid-Na2HPO4 buffer (0.1M, pH 5.6) – 5 mL, analysis. |
SDS- PAGE, Native PAGE. |
Polyphenol oxidase recovery – 78 % purity - 8.26 fold, enzyme specific activity - 52.82 U/ mg. TPP-upper DES phase recovered, reuse – 6 times. |
[10] |
|
23 |
Tea leaves, tea buds, tea stems, tea powder residue |
epigallocatec hin gallate, epicatechin gallate, theanine, caffeine. |
HBD Polypropylene glycol, PPG200, PPG400, polyethylene glycol, PEG200, PEG400, PEG600, PEG800, HBA citric acid |
DES: citric acid-PPG400 (1:1) + 30 % water, dried sample size – 60 mesh, S/L-10 mL/g, M.P.- 700 W,time - 15 min, temp.- 50 ?, centrifugation speed – 8000 rpm, time – 8 min, supernatant, dilution – 50 fold, acetonitrile – 70% analysis |
UPLC- QQQ- MS/ MS, DPPH, ABTS. |
Quantities – g/Kg epigallocatechin gallate - 15.58, epicatechin gallate - 12.85, theanine - 152.2, caffeine - 48.44. Fick’s 2nd law, MDS |
[50] |
|
24 |
White tea cake |
Polyphenols |
HBAs β-cyclodextrin (β-CD),hydroxypropyl- β-cyclodextrin (HP-β- CD),γ-cyclodextrin (γ-CD), HBDsglycerol, levulinic acid, L- lactic acid. |
Supra DES: HP-β-CD-lactic acid (1:5) + 40 % water, white tea cake powder – 150 mesh, S/L -1:40,U.P. – 500 W,frequency – 40KHz, time- 54 min, centrifugation, supernatant, analysis, processing. |
SEM, XRD, NMR, FT-IR, DPPH, ABTS |
Supra DES extract Tea polyphenols - 111.36 mg/g, ABTS – 82%, DPPH – 67% ethanol (60%) extract ABTS – 76% DPPH – 51% supra DES biofilm, ABTS - 1 to 2%,DPPH – 0.supra DES extract biofilms,ABTS – 38 to 54%, DPPH – 30 to 46%. |
[51] |
|
25 |
Black tea |
Highly polymeric tea pigments (HPTPs), thearubigins, theabrownins . |
Choline chloride, acetic acid, oxalic acid, propionic acid, succinic acid, malic acid, citric acid, ethylene glycol, glycerol, 1,4-butanediol, xylitol, sorbitol, xylose, glucose, urea. |
NADES: choline chloride-urea (1:2) + 30 % water, S/L - 0.4 g : 25 mL,temp. –85 oC, time – 18 min, centrifugation speed – 4500 rpm time – 10 min supernatant collected. Residue - repeat extraction, supernatant collected supernatants mixed – black tea pigment extract. Tea pigment quantity black tea extract – 2mL, oxalic acid solution – 2 mL, Water – 6mL, Ethanol – 10 mL, UV-Vis. black tea NADES extract, membrane, cutoff-3500 Da, NADES separation. black tea pigment extract, thearubigins, liquid- liquid extraction, ethyla acetate, n-butanol. theabrownins, remaining black tea extract, centrifugation, supernatant, residue. |
XRD, SEM, AFM, TG-DSC, Py-GC- MS, UV-vis, FT-IR, NMR, SERS, ABTS, DPPH, FRAP. |
Tea pigment yield – NADES - 40.3 %, water – 19.28 %. thearubugin fractions, theabrownin fractions |
[9] |
|
26 |
Tea waste |
Lignins |
choline chloride, oxalic acid, |
UV–Vis, FTIR, NMR, FESEM, XRD. |
Yield |
[12] |
|
|
Lignin – 37 % TPC – 106 mg GAE/g Antioxidant activity DPPH – 29 %. Antibacterial activity zone of inhibition Pseudomonas aeruginosa ~1.8 cm, Bacillus subtilis ~ 1.7 cm. |
|||||||
|
27 |
Tea stem |
Lignins |
HBA PEG 200, HBDs levulinic acid, oxalic acid, formic acid, lactic acid, |
PDES PEG200-oxalic acid (2:1) + 20 % water, tea stem - 1 g, PDES - 20 g, stirring, temp. – 140 oC, time – 4 h, cool – RT, vacuum filtration, solid (cellulose), wash, ethanol-water (1:1) - 150 mL, filtrate + washings evaporation- ethanol, water – 200 mL, lignin- precipitation freeze-drying, temp. – 4 oC time – 24 h. |
FTIR, XRD, NMR |
Isolation cellulose - 82 %, hemicellulose - 97 %, lignin – 71 %. |
[13] |
|
28 |
Tea two leaves and bud. |
tea peroxidase |
menthol, thymol, camphor. decanoic acid, hexanoic acid, octanoic acid, lactic acid, oleic acid, dodecanoic acid, |
HDES: menthol-hexanoic acid (1:1), Tea leaves – 1 g, Polyvinylpyrrolidon e – 0.5 g, sodium phosphate buffer – pH 7.0 homogenization – 3 min, ice bath – 3 h, (shaken-every 1 h), centrifugation, speed - 9000×g, temp. – 4 oC, time – 15 min. supernatant, tea POD extract. TPP system (NH4)2SO4 – 56 %, tea extract:HDES (1:1), stirring, ice bath – 1 h, pH - 6.0 TPP, organic phase, precipitate phase, aqueous phase, centrifugation, speed – 8000×g, temp. – 4 oC, time – 10 min. precipitate, dissolution, sodium phosphate buffer – 10 mL, pH -7.0, analysis protein conc., enzyme activity. |
TPP system, SEM |
Tea peroxidase recovery – 70 % purification - 8.25 fold, after five cycles recovery – 59 % purification – 6.55 fold, |
[11] |
Catechins: Catechins are extracted using NADES [choline chloride: ethylene glycol (1:5) + 30% water – selected] from green tea. Three extraction methods
*HBA-HBD: hydrogen bond acceptor-hydorgen bond donor (molar ratio) Abbreviations AA-scorbic acid,
ABTS-2,2’-azinobis(3-ethyl-benzothiazoline-6-sulfonic acid) radical, AFM-atomic force microscopy, AT-ambient temperature, CA-citric acid,
ChCl-choline chloride,
COSMO-RS DARE - conductor-like screening model for real solvents dimerization, aggregation and reaction extension COSMO-SAC -conductor-like screening model for segment activity coefficient
DES-deep eutectic solvent,
DES-MIP adsorbent - deep eutectic solvent-magnetically imprinted polymer adsorbent, DES- Fe3O4/MIPs - deep eutectic solvent-Fe3O4/molecularly imprinted polymers DES-MIRs@CNF AG - deep eutectic solvent (DES) based-molecularly imprinted resins cellulose nanofiber aerogels
DES-SLE-UHPLC-UV-deep eutectic solvent-solid liquid extraction-ultra high performance liquid chromatography -ultra violet, DFT-density functional theory, DLS-dynamic light scattering study,
DPPH-1,1-diphenyl-2-picrylhydrazyl radical, DSC-differential scanning calorimetry, DW-dry weight,
EDTA-ethylenediamine tetraacetic acid
Fe3O4@MoS2@DES-MIP- magnetic molybdenum disulfide base and deep eutectic solvent Molecularly Imprinted Polymer, FRAP-ferric reducing antioxidant power, HD-hydrodistillation,
HF-LPME- hollow fiber-liquid microextraction HPLC-high performance liquid chromatography, ICP-OES - inductively coupled plasma-optical emission spectroscopy LA-lactic acid,
LOD-limit of detection,
MANPs-D- μSPE-DES - magnetic agarose nanoparticles micro-solid phase extraction and deep eutectic solvents, MCE-mechano-chemical extraction, MDS-molecular dynamics simulations, MIP-magnetic Imprinted polymer
MP-microwave power,
MSPE-magnetic solid phase extraction M.temp-microwave Temperature, M.time- microwave extraction Time, NADES-natural deep eutectic solvent, (NH4)2SO4-di ammonium sulphate
NMR-nuclear magnetic resonance spectroscopy, PDES-polymerizable deep eutectic solvent, PEG-polyethyleneglycol, POD-peroxidase,
PPG-polypropyleneglycol, PPO-polyphenol oxidase, PVPP-polyvinylpyrolidone,
Py-GC-MS -pyrolyzer-gas chromatography-mass spectrometry, RE-rutin equivalents,
SEM-scanning electron microscopy, SERS-surface-enhanced raman scattering, SLE-solid-liquid extraction, S/L-solid to liquid ratio, Temp.-temperature,
TFC-total flavonoids content, TG-thermogravimetric analyzer. TPC-total phenolics content, TPP-triple / three phase partition, UAE-ultrasound-assisted Extraction, U.temp-ultrasonic bath Temperature, U.time-ultrasonic extraction time,
UHPLC-Q-TOF-MS/MS - ultra-high-performance liquid chromatography with quadrupole-time-of-flight tandem mass spectrometry, UHPLC-QqQ-MS/MS - ultra-high- performance liquid chromatography with triple-quadrupole tandem mass spectrometry,
UPLC-QQQ-MS/MS - ultra-high-performance liquid chromatography with triple-quadrupole tandem mass spectrometry, UNIQUAC-universal quasi-chemical, UP-ultrasound power,
UPLC-HR-MS-SIM-ultra performance liquid chromatography-high resolution-mass spectrometry-selected ion monitoring. UV Vis-ultraviolet and visible spectroscopy, VA-vortex-assisted,
XPS-X-ray photoelectron spectroscopy, XRD-X-ray diffraction.
(ultrasonication, stirring, heating) are used [18]. Heating results higher quantities of catechins. Heating extraction method is optimized. The optimum conditions standardized are the type of NADES (HBD and HBA of NADES, molar ratio, water content in NADES), solid to liquid ratio, extraction time, and temperature (Table 1, No.1). NADES are selected based on their low viscosity, low surface tension and polarity similar to the target compounds. Green tea samples are dried, ground, and sieved. Requisite quantities of the sample and NADES are taken. The mixture is subjected to extraction under optimized conditions of temperature and time to achieve higher extraction yields. Higher extraction efficiency in heating, is owing to increased diffusion, reduced viscosity and improved solubility. Then, the suspensions are cooled to room temperature and centrifuged. The supernatant is mixed with an equal volume of the mobile phase, filtered and subjected to HPLC-UV analysis. NADES extraction yields (catechin-3.629 mg/g, epicatechin gallate-35.25 mg/g and epigallocatechin gallate-114.2 mg/g) are higher than those of with other routine organic solvents. Li et al. [30], discussed the extraction of catechins from green tea leaves using microwave- assisted extraction (MAE) with NADES (choline chloride - lactic acid + 20% water). The optimized parameters are the NADES components (selection of HBD and HBA, molar ratio, and water content), solid to liquid ratio, microwave extraction time, and temperature (Table 1, No.2). NADES are preferred based on the polarity, viscosity, and solubility of target compounds. Lactic acid, as the HBD contributed to the higher extraction efficiency. Water is added to the NADES to obtain the required viscosity. Known quantities of the sample and NADES / solvent are mixed and extracted under the optimized conditions. The extracted solution is subjected to vacuum filtration and the filtrate is analyzed by HPLC for catechins. Temperature is a critical factor. An increase in temperature decreases the viscosity of the NADES, and its penetration into the sample improves. The mass transfer of target molecules into the NADES increases, because of the weakening of the intermolecular interactions in the sample. However an excess increase in temperature degrades the biomolecules. Catechins are recovered using a known quantity of AB-8 macro porous resin in a wet column. Initially, filtrate is loaded and eluted with water, followed by the required volume of 90 % aqueous ethanol. The ethanolic eluents are concentrated under vacuum, and the yields are calculated. Response surface methodology is used to achieve a higher extraction yield of catechins (154 mg/g). The recovery of catechins from NADES extract using AB-8 macro porous resin is in the range of 75 to 86 %. MAE requires shorter extraction periods and a lower quantity of solvent owing to its high extraction efficiency. Yields of target compounds (i.e., total catechins) in NADES extracts are higher, when compared to routine organic solvents. The ultrasonication-assisted extraction of catechins, mainly epigallocatechingallate, from green tea using ternary NADES [betaine-glycerol-D-glucose (4:20:1)-19% water] for use in cosmetic / pharmaceutical products is reported [17]. The optimized parameters included the type of NADES (HBDs and HBA of NADES, molar ratio, and water content in NADES), solid to liquid ratio, pH and ultrasonication extraction time (Table 1, No.4). NADES is selected based on the components that are allowed in cosmetics, neutral nature (pH near 7), high extraction efficiency, and low cost. The required quantities of the sample and solvent / NADES are mixed and vortexed. The mixture is subjected to extraction under optimized ultrasonication conditions for the required time at ambient temperature. The suspension is centrifuged under the preferred conditions and the supernatant is diluted and subjected to HPLC-UV analysis. Similarly, green tea is subjected to extraction using heating, stirring, and heating cum stirring. UAE provided higher extraction yields and maximum EGCG content in the extract. The yields are higher than those obtained using previously reported methods and routine organic solvents. SEM images of the green tea powder after extraction showed severe rupture and destruction of the physical structure of the sample. This facilitated the rapid exudation and dissolution of catechins by NADES. EGCG in NADES is stable up to 60% after three weeks at 60oC. NADES extract can be used directly in cosmetic and pharmaceutical applications of skin. A scale up of production studies is attempted. The time taken to achieve the same yield is longer. Economic considerations are also discussed. The extraction and estimation of seven catechins (catechin, epicatechin, epigallocatechin, epicatechin gallate, epigallocatechin gallate, gallocatechin, and gallocatechin gallate) from green, black, and fruit teas using DES [malic acid: betaine hydrazide hydrochloride or GrT (2:1) + 30 % water] is described [31]. The optimized extraction parameters included the type of DES (HBD and HBA of DES, molar ratio, water content in DES), solid to liquid ratio, extraction time, and temperature (Table 1, No.7). DES is preferred based on their acidity, stronger multi-interactions such as ionic/charge-charge and hydrogen bonding with targeted compounds as well as high extraction efficiency. Known quantities of the sample and DES (or ionic liquid / conventional solvents for comparison) are extracted under optimized conditions of stirring, and temperature for the required period. The suspension is then centrifuged under the necessary conditions. The supernatant is then diluted with methanol and filtered. The filtrate is then subjected to UHPLC- UV analysis. DES provided higher extraction yields of catechins than routine solvents. Major compounds of green teas, black teas and fruit teas are EGCG (27.7–63.1 mg/g), GC (6.3–22.8 mg/g), and EGC (8.07–8.4 mg/g) respectively. The authors claimed that the extracts could be used directly in applications, although yields are lower than those in earlier reports. DES [vinyl pyrrolidone: malonic acid (1:1 molar ratio)] based on magnetically imprinted polymers [Fe3 O4 @ MoS2 @DES-MIP- prepared from iron (III) chloride hexahydrate (FeCl3 .6H2 O), sodium acetate, molybdenum disulphide (MoS2 ), ethylene glycol dimethacrylate] is prepared to absorb the selected catechins (catechin, epicatechin, epigallocatechin, epicatechingallate, and epigallocatechin gallate) [32]. The target compounds are evaluated (Table 1 No.8). DES is selected based on its biocompatibility with catechins and its new functions. The DES functioned as a monomer. MIP is prepared using DES and target compounds (refer supporting information). Known quantities of the sample solution and MIP are shaken at the optimized temperature for the time required for adsorption. The adsorbed solution is separated using MSPE and analyzed using HPLC. The adsorption recoveries for various catechins are found to be in the range of 80 to 99 % as estimated by HPLC. The adsorption of EGCG from the green tea samples is 98%. MIP can be used up to five cycles. However, after five cycles adsorption recovery decreased to 55%. The extraction of catechins (epicatechin, epigallocatechin, epicatechingallate, and epigallocatechin gallate) from green tea using NADES [choline chloride:glycerol (1:2) + 40 % water - selected] is reported [33]. The optimized extraction parameters included the type of NADES (HBD and HBA of NADES, molar ratio, water content in NADES), solid to liquid ratio, and ultrasonication power and time (Table 1, No.9). NADES is selected based on its high extraction efficiency for total polyphenols, low toxicity, and corrosiveness. Requisite quantities of green tea powder and NADES are mixed and subjected to extraction by ultrasonication under optimized conditions for the required time. During ultrasonication, the total mixed solution is kept on ice to avoid degradation of tea polyphenols by temperatures. The crude tea extract is centrifuged under optimized conditions. The supernatant is recovered and subjected to evaluation of TPC, antioxidant activity, and high performance liquid chromatography (HPLC) analyses. Experiments are carried out using ethanol, UAE-ethanol and hot water as control. The addition of water to a certain level can reduce the viscosity of the NADES and preserve the supramolecular complexes and hydrogen bonding between HBD and HBA, which is favorable for high extraction efficiency. Appropriate ultrasound power can improve yields. Excess ultrasound power can cause enormous bubbling cavitation and hinder the transmission of ultrasound power. This could reduce the yield. The ultrasonication time also needs to be appropriate. More time may degrade the bioactive compounds and yields could decrease. Catechins are estimated using HPLC-UV. UAE-NADES achieved good extraction yields of catechins (EGC-24, EC-9, EGCG-94 ECG-36 mg/g on dry weight basis) when compared to other methods. The NADES extract obtained from green tea showed the highest total polyphenol content (TPC - 243 mgGAE/g) and antioxidant activities [FRAP - 332 mmol Fe(II), DPPH - 215 mmol trolox, ABTS - 99 mmol trolox per 100g material on dry weight basis], when compared to other conventional solvents and other methods. The authors mentioned that SEM analysis indicated that UAE NADES extraction led to a wobbly structure and surface erosion of the green tea leaves, which could result in greater penetration of the solvent into the plant material to provide good yields of the targeted compounds. Extraction of catechins (epicatechin, epigallocatechin, epicatechingallate, epigallocatechingallate) using NADES [betaine: urea-(1:2) + 30% water-selected] from green tea is reported [34]. The optimized parameters are the type of NADES (HBD and HBA), molar ratio, water content, ultrasonication temperature and time, centrifugation speed and time (Table 1, No.17). NADES is selected based on the beneficial effects of its components. Known quantities of sample and NADES are mixed and extracted using ultrasonication under the optimized conditions. The suspension is then centrifuged at the required speed and time. The supernatant is subjected to further analysis (HPLC and antioxidant activity). Extraction is also carried out using water and 50% ethanol for comparison. The addition of water to the NADES reduces its viscosity and increases its polarity. These properties improve the extraction efficiency. However, the addition of an excess amount of water disturbs the eutectic structure of the NADES and the extraction yields are reduced. Therefore, the addition of an appropriate amount of water is important. Green tea NADES (betaine: urea) extract showed the presence of high TPC (134 mg GAE / g) and TFC (21 mg RE / g), and exhibited higher antioxidant activities (FRAP: 1.91 mmol Fe2+ / g, DPPH: IC50 - 0.09 mg / mL, ABTS: IC50 - 7.03 mg / mL). Green tea NADES extracts showed a higher total catechins contents (110.01 to 115.92 mg / g) than other conventional solvents. However, the NADES (tartaric acid sorbitol) extract had the lowest total catechins content (60.46 mg / g) among the NADES extracts. The authors claimed that these NADES green tea extracts might be useful in the food, pharmaceutical, and cosmetic industries. Preparation of molecularly imprinted resins cellulose nanofiber aerogels (MIRs@CNF AG) for separation of catechins from the DES [choline chloride: 1,4-butanediol (1:2) + 50 % water] based extract is described [35]. The optimized parameters for tea extraction are NADES type (HBD and HBA of NADES, molar ratio, water content in NADES), solid to liquid ratio, ultrasonication temperature and time, and centrifugation conditions (Table 1, No.18). NADES is selected based on the high adsorption capacity of catechins on MIRs@CNF AG. Known quantities of green tea powder (80 mesh sieve) and NADES are subjected to extraction using ultrasonication under optimized conditions. The suspension is subjected to centrifugal filtration to obtain the tea NADES extract. MIRs@CNF AG (refer Supporting Information for preparation) is prepared and analyzed by FT-IR, SEM, XRD, XPS and TG. Lyophilized MIRs@CNF AG is used in syringe column to separate catechins. The solution is then washed with methanol and water. A known quantity of tea NADES extract is adsorbed onto the column and washed with 10 % methanol aqueous solution. Catechins are eluted using a methanol-acetic acid (95:5) solution. The eluent is evaporated under vacuum under the optimized conditions to obtain pure catechins. MIRs@CNFAG demonstrated underwater stability and compressibility. Hence, it is useful for the separation of catechin in aqueous environments. Hydrogen bonds, electrostatic attraction and π-π stacking forces are involved in the adsorption mechanism as per density functional theory. The catechins adsorption capacity in the first cycle is 74 mg / g (purity-86.75%). The absorption capacity is higher than that of individual MIRs as well as CNF AG. The MIRs@CNF AG column is reusable and the absorption capacity in the fifth cycle is marginally decreased to 65 mg / g. Interactions of NADESs (choline chloride combined with 1,2-butanediol, 1,3-butanediol, 1,4-butanediol and 2,3-butanediol) and target molecules from green tea (catechins, alkaloids and flavonoids) based on molecular dynamics simulations (MDS) and density functional theory (DFT) are reported to obtain a theoretical basis [36]. The MDS revealed the mechanisms of the extraction process. These interactions include hydrogen bonds, interaction energy, and radial distribution function. The interactions of the target compounds with the different NADESs varied, and some of the solvents are more soluble and showed good selectivity for certain compounds. These interactions facilitate the movement of bioactive molecules from the aqueous phase towards the NADES. The results showed that NADES solvents (choline chloride -2,3 butane diol), (choline chloride -1,3 butane diol and choline chloride -1,2 butane diol), and (choline chloride -1,4 butane diol) could extract bioactive compounds [epigallocatechin gallate (EGCG) and epigallocatechin (EGC)], [(epigallocatechin gallate (ECG) and EGC], and [catechin (CAT) and EGC] respectively (Table 1, No 20). The order of average total interaction energy between the target molecules and solvents is as follows EGC > ECG > EGCG > Kaempferol > Theobromine > Caffeine > CAT. The interaction energy of EGC with these solvents changed from - 24 to -95 kJ / mol. EGC showed more interactions and a strong affinity for NADES (choline chloride -2,3-butanediol). This NADES is suitable solvent for EGC extraction. This is due to the hydrogen bond interactions and electrostatic energy contributions between the EGC and NADES. These results demonstrate the potential applications of butanol derivatives in the tea industry.
Alkaloids: Purification of green tea alkaloids (theobromine and theophylline) using NADES [choline chloride: urea (1:2)] based Fe3 O4 /MIPs as adsorbents for M-SPE is reported [37]. Fe3 O4 /molecularly imprinted polymers are prepared and NADES is added to Fe3 O4 / MIPs and polymerized to form NADES-Fe3 O4 /MIPs (refer supporting information for more details). NADES is selected based on its electrostatic and ion-exchange interactions with target molecules as well as its stronger recognition and high recoveries of target compounds. DES Fe3 O4 /MIPs are used as adsorbents in M-SPE. The requisite quantity of dried green tea is powdered and subjected to extraction with the required volume of ethanol under ultrasonication for 1 h at the required temperature. The suspension is filtered to obtain the green tea extract. Green tea extracts are passed through M-SPE. Then, the alkaloids are eluted from the M-SPE using a suitable solvent. The separation of alkaloids is achieved in good yield (Table 1, No.3). Developed method yielded theobromine (4.87 mg / g) and theophylline (5.07 mg / g) from green tea. The recovery values of theobromine and theophylline are 92.27 % and 87.51 % respectively. The developed method is very rapid. The above study is extended by the same authors [38], for the isolation of theophylline (5.82 mg / g, 92 %), theobromine (4.32 mg / g, 92 %), (+)-catechin hydrate (18.36 mg / g, 90 %), and caffeic acid (3.69 mg / g, 91 %) from green tea using a ternary DES based magnetic molecularly imprinted polymers for the dispersive magnetic solid-phase microextraction. The isolation of caffeine from Chinese dark tea has been reported using NADES [choline chloride:lactic acid (1:1) + 29% water-selected]. The optimum conditions are the type of NADES (HBD and HBA of NADES, molar ratio, and water content in NADES), solid to liquid ratio, vortex conditions, ultrasonication extraction time, and temperature, centrifugation speed, and time (Table 1, No.6). The NADES is selected based on the Kamlet-Taft polarity parameters (α, hydrogen-bond acidity, hydrogen bond accepting ability; β, hydrogen-bond basicity; π*, dipole/polarizability), stronger hydrogen bonding interactions between caffeine and NADES, hydrophobic interactions, electrostatic interactions, van der Waals forces, and higher extraction efficiency [39]. Requisite quantities of Chinese dark tea and NADES are initially subjected to extraction under optimized conditions using vortex shaking and later ultrasonication for the required period at certain temperatures. The mixture is then centrifuged at the required speed for the required period. The supernatant is then subjected to HPLC analysis. The caffeine quantity is reported. Caffeine is recovered from the NADES extract using resins. The required quantity of NADES extract is subjected to adsorption on known amount of macroporous resin (XAD-200) under optimized conditions. Resins containing the target compounds are separated by filtration after equilibrium. A known quantity of absolute ethanol is added to the resin and desorption is performed using ultrasonication under optimized conditions. The resins are separated via filtration after desorption. The caffeine quantity (27 mg/g) is calculated from the filtrate. Caffeine purity is limited (24%). Yield of caffeine with NADES is higher than routine solvents. The resin is re-used and tested for three cycles. Recovery is good. The developed method is used to achieve higher caffeine extraction yields from Chinese dark tea. The kinetic model of caffeine extraction is confirmed using Fick’s second law of diffusion. Caffeine interactions with NADES [HBA-choline chloride, HBDs glycerol, sorbitol, xylitol, glucose, sucrose, maltose and fructose with water] for solubility measurements have been reported [40]. The optimized parameters are the molar ratios of HBA, HBD and temperature. Caffeine solubility is determined experimentally. Excess caffeine and NADES / NADES-water are mixed. The mixed solutions are incubated for 24 h at different temperatures on an orbital shaker incubator. The samples are mixed at 60 rpm during the incubation. The solutions are centrifuged at 1000 rpm for 5 min. The supernatant is filtered through 0.22 µm. Filtrate is transferred to tubes containing methanol. The samples are diluted and measured spectrophotometrically for the determination of soluble caffeine. Theoretical solubility predictions are reported using the conductor-like screening model for real solvents (COSMO-RS). This depends on the quantum chemistry computations of a variety of solvation replicas. These are created based on statistical thermodynamics analysis of electrostatic surface interactions and the chemical potential of the components. The COSMO-RS DARE approach exhibited a perfect match between the experimental and computed solubility values. NADES [choline chloride-glycerol (1:2) +20% water] showed the highest (mole fractions-559.45×10-4) solubility for caffeine at 40oC, and HBA- choline chloride HBDs- sorbitol, xylitol and glucose (1:2, 1:1, 2:1-molar ratios; 0.8 NADES+0.2 water at 40oC) combinations are found to be effective for caffeine dissolution. The highly solubilizing character of NADES is due to intermolecular interactions (e.g., hydrogen bonding) of bioactive molecules with solvent constituents (e.g., choline chloride, sugars and water). The nitrogen centre of the imidazole ring of caffeine acts an acceptor, whereas the hydroxyl group of the second counterpart of the hydrogen-bonded complex acts as the hydrogen donor. Solubility computations performed using the COSMO-RS DARE approach for caffeine solubility values are mostly in qualitative agreement with the experimental data.
Polyphenols: Polyphenols are isolated from green tea using NADES [choline chloride: ethylene glycol (1:2)]. The optimized parameters included the type of NADES (HBD and HBA of NADES, molar ratio), solid to liquid ratio, and magnetic stirring temperature and time (Table 1, No.12). NADES is selected based on its low polarity and hydrophobicity similar to that of tea polyphenols [41]. Requisite quantities of tea powder and NADES are mixed well and subjected to extraction by magnetic stirring at an optimized temperature for the required time. The solutions are centrifuged under the necessary conditions. The supernatant is collected for further analysis. Polyphenols are estimated using UV-Vis spectroscopy. The method achieved good extraction yields of polyphenols (20.12%). The authors discussed the COSMO-SAC methodology as an effective screening tool for determining the extractability of tea polyphenols and solvent selection for bioactive components extraction. As per the COSMO SAC methodology, -profiles, activity coefficients, and interaction energies of solvents are considered for their extraction performance. The COSMO-SAC appears to be useful as a screening technique of the solvent for the extraction of tea polyphenols. Yield of polyphenols is higher with NADES, when compared to routine solvents. Free and bound phenolic compounds from tea seed oil are isolated using NADES [free phenolic compounds: choline chloride:glycerol (1:1); bound phenolic compounds: choline chloride:glycerol:ethylene glycol (1:1:1) and choline chloride:propylene glycol (1:1). The optimized parameters are the type of NADES (HBD and HBA of NADES, molar ratio), and extraction temperature (Table 1, No.10). NADES for free phenolic compounds are selected based on their neutral pH, polarity similar to that of phenolic compounds, suitable viscosity and steric hindrance. NADES with glycerol as the HBD showed higher efficiency for bound phenolics than for methanol. However, the other selected NADESs also showed higher extraction efficiencies than the earlier one. The temperature is also a key factor in the extraction process. Free phenolics are separated from tea seed oil - Requisite quantities of tea seed oil and n-hexane are mixed using a vortex. The required quantity of NADES is added to the mixture and vortexed thoroughly under the optimized conditions of temperature and time in a water bath. The mixture is centrifuged at the required speed and time. The NADES fraction containing free phenolic compounds is collected and taken for further work. Bound phenolic compounds are also isolated using NADES: Tea seed oil layer without free phenolic compounds is separated. A known quantity of tea seed oil residue (without free phenolic compounds) is subjected to alkaline hydrolysis under the required conditions and neutralized with acid. The bound phenolic compounds are then hydrolyzed. The mixture is centrifuged and the oil phase is separated from the aqueous phase. Phenolic compounds are separated from the oil phase using NADES, as per the above procedure. Bound phenolic compounds in the NADES phase are used for further work-up. The recovery of phenolic compounds is carried out using the XAD-12 macro porous resin. NADES extracts with phenolic compounds are adsorbed onto the XAD-12 resin column. It is initially washed with deionized water. The adsorbed phenolic compounds are eluted using methanol-water (1:1). The methanol eluents are evaporated to dryness. The residue is re-dissolved in methanol-water (1:1) and filtered through an organic membrane for further analysis. Twenty-five compounds are identified from the above extracts using UHPLC-Q-TOF-MS/MS and quantified using UHPLC-QqQ-MS/MS [42]. Quantities of free phenolic compounds (83.91 μg / g) and bound phenolic compounds (25.71 μg / g) obtained using NADES are higher than that of normal organic solvents (free-55.59 and bound- 13.31 μg / g respectively). NADES [choline chloride-glycerol-ethylene glycol (1:1:1) extractedthe highest quantity (35.44 μg / g) of bound phenolic compounds. Both the free and bound phenolic compounds extracted using NADES exhibited good antioxidant capacities. Bound phenolic compounds displayed different antioxidant performances according to structure-activity relationship (SAR) and chemo metric analyses. The selected phenolic compounds changed SAR and meaningfully supports the antioxidant capacities after binding [43].
Theabrownins and thearubigins: Theabrownins are extracted from Kangzhuan dark tea using NADES [choline chloride: malic acid (1:2) + 30% water] with ultrasonication [44]. The optimized extraction parameters are the NADES type (HBD and HBA of NADES, molar ratio, water content in NADES), solid to liquid ratio, ultrasonication power, and extraction time (Table 1, No.14). NADES is selected based on its low pH, molecular interactions, highly protective effect against thermal degradation of extracts, and high extraction efficiency towards theabrownins. Known quantities of tea powder and NADES are placed in a beaker kept in an ice water bath and subjected to UAE under optimized conditions of ultrasonication power and time. The total solution is centrifuged under the required conditions and the supernatant (NADES extract of theabrownins) is collected for further analysis. NADES extracts of theabrownins are mixed, filtered and concentrated. The concentrate is then extracted using ethyl acetate. The aqueous phase is then separated. Anhydrous ethanol is added to the aqueous phase until the ethanol content reached to 80%. The solution is then allowed to stand for 8 h. The solution is centrifuged and the precipitate of theabrownins is collected. The theabrownins are completely dissolved in water and freeze-dried. The lyophilized theabrownins powder is stored at 4oC for further analysis. This method resulted in higher extraction yields of theabrownins (12.59 %). Theabrownins are fractionated into fractions (TBFs) using silica. TBF (eluted with 60 - 40 % methanol) showed the highest antioxidant activity (DPPH - 45 μM AA/g, ABTS – 179 μM trolox/g, FRAP - 371 μM Fe (II)/g). LC-MS/MS analysis of the TBFs showed the presence of forty-nine compounds. The major compounds are 6,7 dihydroxycoumarin-6- glucoside, erucamide, caffeine and neohesperidin. Highly polymeric tea pigments (HPTPs) are extracted from black tea using NADES [choline chloride:urea (1:2) + 30% water]. HPTPs are separated from NADES extract using membrane (molecular weight cut-off 500–1000 Da). Optimised parameters of the extraction are the NADES type (HBD and HBA of NADES, molar ratio, water content in NADES), solid to liquid ratio, extraction time and temperature (Table 1, No.25). NADES is selected based on influence of hydrogen bonding network, high conductivity, and low viscosity. Known quantities of black tea powder and NADES are mixed and soaked in requisite quantity of boiling water for required period. Solution is centrifuged under preferred conditions of speed and time. Supernatant is collected. Residue is extracted second time under the same conditions. Both supernatants are combined collected as black tea extract. Requisite quantities of black tea extract, saturated oxalic acid solution, and ethanol combined. UV absorbance at 380 nm observed and tea pigment is calculated. Extraction of black tea with water is carried out for comparison. Structural and morphological information of HPTPs is analysed using different chromatographic and spectroscopic techniques (Py-GC-MS, UV-Vis, FT-IR, NMR, XRD, SEM, AFM, TG-DSC). Antioxidant activity (DPPH, ABTS, FRAP) of HPTPs is evaluated. Total tea pigment in NADES tea extract is separated using dialysis membrane (cutoff-3500Da) from NADES [9]. Thearubigins fractions are isolated using liquid-liquid extraction (i.e., ethyl acetate, n-butanol). Theabrownins are isolated by centrifugation of remaining aqueous portion (i.e., supernatant and residue). Total polyphenols, polysaccharides, flavonoids, aminoacids, protein and caffeine are evaluated in all these fractions. Higher quantities of HPTPs are reported from fractions of NADES extract, when compared to water extract. Fractions from NADES extract exhibited higher antioxidant activities. Functional groups are similar in both water and NADES extracts. However, intensities are higher in NADES extract. Pigments in NADES extract exhibited higher degree of polymerization. Fractions of TRs and TBs from black tea NADES extract could be useful in food, medicine, and cosmetics industries owing to their beneficial effects.
Flavonoids: Isolation and quantification of flavonoids (morin, quercetin, kaempferol) is reported using magnetic dispersive micro-solid phase extraction based on magnetic agarose nanoparticles and deep eutectic solvents [tetraethyl ammonium chloride:lactic acid (1:3) - selected]. Flavonoids are isolated from black tea at microgram per litre and analyzed using HPLC-UV [45]. LODs are found to be low for flavonoids in this method when compared to other reported methods. The optimized variable parameters included the type of DES (HBD and HBA, molar ratio), solid to liquid ratio, amount of adsorbent, volume of desorption solvent, amount of salt, extraction time, pH of the sample solution, and desorption time (Table1, No. 11). DES is selected based on the adsorption and desorption capacities of the target analytes through hydrogen bonding and dipole-dipole interactions. Known quantities of dark tea sample are boiled in water for the required period and cooled. The sample solution is infused. Known quantities of agarose and an acidic solution [acetic acid (5%) + hydrochloric acid) are treated at the optimized temperature for the required time. Measured quantities of FeCl2 ⋅4H2 O and FeCl3 ⋅6H2 O are dissolved in the above solution using mechanical stirring. Known quantities of TPP and sodium hydroxide (25%) are added and the mixture is stirred for the required time. The formed MANPs are separated using a magnet, washed with water, and dried at the required temperature. Known quantities of MANPs and the sample solution (pH-4.9 with hydrochloric acid) are mixed using a vortex for the required time. MANPs with flavonoids are separated using a magnet. Flavonoids are eluted using the requisite quantity of DES from MANPs using ultrasonication for the required time. DES phase is separated and subjected to analysis using HPLC-UV. The adsorption mechanism involves hydrogen bonding between the polar groups on the MANPs and the phenolic hydroxyl groups in the flavonoids. Moreover, π-type hydrogen bond have been reported between the hydroxyl functional groups of MANPs and the conjugated aromatic rings of flavonoids. Limit of detections (LODs) and quantifications (LOQs) of flavonoids are in the range of 0.2–1.1 μg/L and 0.66–3.63 μg/L respectively. These values are very low, compared to those of the other methods. Authors used this method with black tea samples, where the quantities of selected flavonoids are very low (quercetin - 4.6 µg/L) or not detectable (Morin, Kaempferol). This method is also used for other foods.
Caffeic acid: Caffeic acid is separated from green tea extracts using a hollow fiber membrane (HF- LPME) filled with NADES [serine:lactic acid (1:4) + 30% methanol - selected]. The optimized parameters are type of the NADES (HBD and HBA, molar ratio, solvent content), pH, salt concentration, extraction time and temperature, and stirring speed (Table 1, No.13). The preferred NADES is based on its hydrophobicity, high affinity for analyte molecules, stability on the wall- pores and lumen of the hollow fiber during extraction, and easy preparation without purification steps [46]. Known quantities of green tea and boiling water are infused at an optimized temperature for the required time. The solution is centrifuged for the required time at the optimum speed. The supernatant is filtered and the filtrate is sampled. A hollow fiber membrane of a specified length is washed in acetone under sonication, and dried. The prepared HF is dipped into the NADES to fill the parts and wall pores. The HF external parts are then washed with water. The HF lumen is filled with the NADES using a suitable syringe. The HF ends are closed using suitable needle tips. A U-shaped HF filled with NADES is placed in known quantity of the sample solution (pH-3.0) on a magnetic stirrer at an optimized speed for the required period to extract the analyte. NADES extracted the caffeic acid from the sample. Hydrogen bonding between the functional groups of NADES and caffeic acid and the π-type hydrogen bond between the polar groups of NADES and the conjugated aromatic rings in caffeic acid are key factors for efficient extractability. The high viscosity of NADES is reduced by the addition of methanol (30%) which allowed mobility. The NADES extracted analytes are analyzed using HPLC UV. Lower LOD (0.3 ng/mL) and LOQ (0.9 ng/mL) as well as higher recovery (92-97%) and enrichment factors (418 438) have been reported for caffeic acid. The unspiked green tea sample is analyzed and caffeic acid is found to be 0.022 μg/g. The HF-LPME method is found to be more efficient than previously reported methods for caffeic acid extraction.
Polysaccharides: Polysaccharides are isolated from Anji white tea (green tea) using NADES [choline chloride:1,6-hexanediol (1:1) + 30 % water-selected]. Optimized parameters include the type of NADES (HBD and HBA of NADES, molar ratio and water content), solid to liquid ratio, ultrasound power, ultrasound extraction time and temperature, and centrifugation speed, time and temperature (Table 1, No.19) to obtain the maximum quantity of polysaccharides [47]. NADES is selected based on its high extraction rate and efficiency towards polysaccharides along with the antioxidant activity. The required quantities of tea sample and NADES are combined and subjected to extraction using ultrasonication under optimized conditions. The solution is centrifuged at the required speed, temperature and time. The filtrate is combined with the required quantity of ethanol to precipitate polysaccharides. The mixture is filtered, and the precipitate is freeze-dried and centrifuged under the optimized conditions. The obtained product is then dried at a suitable temperature. The solid is redissolved in water and dialyzed at preferred parameters of time and temperature. Tea polysaccharides (TPs) solution is lyophilised to obtain the purified TPs. The TPs are stored under optimum conditions. The yield of NADES extracted tea polysaccharides (19.2%) is higher than that of hot water-extracted tea polysaccharides (5.5%). The molecular weights of TPs in the NADES-UAE extraction are lower than those in the hot water extraction. The cavitation effects due to ultrasound treatment broke the glycosidic chain of the high-MW polysaccharides and the MWs of TPs from the NADES-UAE extract are reduced. The low-MW TPs exhibited higher antioxidant activities. The total carbohydrate content of the NADES extracted polysaccharides (73 %) is higher than that of hot water extracted polysaccharides (65%). The NADES extracted polysaccharides contained higher quantities of arabinose. The extracted polysaccharides contain monosaccharides (fucose, rhamnose, arabinose, galactose, glucose, mannose, xylose, fructose, galacturonic acid, and glucuronic acid). NADES extracted polysaccharides exhibited higher hypoglycaemic activity in L6 cells and higher inhibitory activities against α-amylase (IC50 - 0.36 mg/mL) and α-glucosidase (IC50 - 0.18 mg/mL) than hot water extracted polysaccharides. TPs from this NADES extract exhibited higher antioxidant activity than TPs from the hot water extract as well as from other DESs. All of the above results indicated that the NADES extracted TPs possess higher functional activities and potential applications. Lignins: Lignins are isolated from tea residue waste using NADES [choline chloride:oxalic acid (1:1)] pretreatment with magnetic stirring and extraction with solvent [acetone:water (1:1)] under stirring [12]. Optimised conditions are the type of NADES (HBD and HBA of NADES, molar ratio), solid to liquid ratio, magnetic stirring speed, time, and temperature, stirring extraction time for mass transfer, and centrifugation speed, time and temperature (Table 1, No.26). NADES is selected based on its acidic strength, intermolecular hydrogen bonding network disruption capability, and higher extraction efficiency towards lignin. Requisite quantities of tea residue waste and NADES are mixed and incubated under preferred conditions of time and temperature using magnetic stirring at required speed for pretreatment. Known quantity of solvent mixture (acetone-water) is added and stirred for required period to extract lignin. The mixture is centrifuged under required conditions of speed and time. Supernatant is collected and acetone is removed under vacuum evaporation. Aqueous NADES extract (containing lignin) is centrifuged under preferred parameters of speed and time to separate the precipitated lignin. Separated lignin is washed with water and dried under required conditions of temperature and time. Dried lignin (yield~12 to 13%) is stored for further processing. NADES is recovered and reused (5 to 8 cycles) for lignin isolation. Lignin is analysed by UV-Vis, FT-IR, NMR, FESEM, and XRD for its characteristics. Lignin is evaluated for TPC (106 mgGAE / g), antioxidant activity (DPPH scavenging activity – 29%), antimicrobial activity (zones of inhibition: Pseudomonas aeruginosa ~1.8 cm; Bacillus subtilis ~ 1.7 cm), and methoxyl (6.82 %) and carboxyl acid (2.30 ± 0.96 mmol/g) groups. Lignin yield is in the range of 10 to 15 % from tea waste. Purity of lignin is in the range of 70 – 77%. Valorisation of tea residue waste is presented for production of lignin. Yield and purity of lignin from tea waste residue needs to be improved. This technique is helpful in the direction of efficient management of food waste. Lignins, cellulose and hemicellulose are isolated from tea stem powder using PDES [PEG200: oxalic acid (2:1) + 20% water] pretreatment [13]. Optimised conditions are the type of PDES (HBD and HBA of PDES, molar ratio, water content), solid to liquid ratio, stirring, time, and temperature. PDES is selected based on Kamlet Taft solvatochromic parameters, appropriate acidity, low viscosity, its efficiency to break down tea stem and separation into lignocellulose, cellulose and lignin, Requisite quantities of tea stem powder and PDES are mixed and stirred. Mixture is treated under preferred conditions of temperature and time. Mixture is cooled and filtered under vacuum. Solid fraction is washed with required quantity of ethanol-water (1:1). Cellulose-rich solid fraction is dried to constant weight at necessary temperature. Filtrate and washings are combined and ethanol is removed under vacuum. Required quantity of water is added to the remaining solution to precipitate lignin. The precipitated lignin is separated and freeze dried under required conditions of time and temperature. Treated, untreated tea stem and lignin samples are analysed. Hemicellulose (97%) and lignin (71%) are separated. Cellulose (82%) is retained and converted to levulinic acid (38 to 52%). Lignin average molecular weight is reduced to 1283 g/mol owing to PDES intervention as per density functional theory. Nanoparticle size of lignin is also reduced to D50 212nm. PDES treatment increased the value of agricultural waste (i.e., tea stem).
Multiple classes of compounds: Simultaneous extraction of multiple classes of compounds (catechins, alkaloids, and flavonoids) from green teas using mechanochemical extraction (MCE) with NADES [choline chloride:1,4-butanediol (1:1) + 40% water] is described [48]. The optimized parameters are the type of NADES (HBD and HBA, molar ratio and water content), solid to liquid ratio, MCE speed, and extraction time (Table 1, No.5). Target compounds are identified and quantified using UPLC- MS-SIM. The application of different NADESs [i.e., choline chloride-ethylene glycol for extraction of catechins, theaflavins, monosaccharides, and choline chloride glycerol for separation of free amino acids] has been reported for the maximum extraction of various compounds from Dianhong Congou black tea. The D-101 macro porous resin is used for the purification of target compounds from NADES extracts. HPLC, and LC-MS are used for the qualitative and quantitative analyses. The optimized parameters are the type of NADES (HBD and HBA of NADES, molar ratio, and water content), solid to liquid ratio, ultrasonication temperature and time, centrifugation speed, and time (Table 1, No. 21). DESs are selected based on choline chloride as HBA and HBDs are preferred for their efficiency in the extraction of target compounds. Requisite quantities of tea powder (60-mesh maintained at 4oC) and NADES are mixed and subjected to extraction using ultrasonication at an optimized temperature for the required time. The solution is centrifuged at the preferred speed for a suitable period. The supernatant is collected and used for further analyses. A known volume of NADES extract adsorbed on D101-macroporous column for required time. Initially, it is eluted with the required quantity of water to remove the NADES. The adsorbed target compounds are eluted using a known quantity of ethanol (95%) and concentrated under vacuum. LC-MS analysis revealed the presence of fifty compounds in these extracts. LC-MS also showed that the extracts of NADESs (choline chloride-citric acid, choline chloride-proline, and choline chloride-ethylene glycol) contained flavonoid glycosides, theasinensins, and hydrolyzable tannins. The NADES (choline chloride-ethylene glycol) and NADES (choline chloride-glycerol) exhibited higher recovery rates. Extract from NADES (choline chloride-ethylene glycol) displayed high antioxidant and α-glucosidase inhibitory effects due to the presence of hydrolyzable tannins (e.g., galloyl-HHDP glucose), flavonoid glycosides, and theasinensins. Hence, NADES (choline chloride-ethylene glycol) is found to be superior for the extraction of most bioactive compounds from Dianhong Congou black tea [49]. Major compounds (quantities) reported are epicatechin gallate (8.75 mg/g), theaflavin-3,3′-digallate (1.59 mg/g), glucose (4.97 mg/g), theanine (19.72 mg/g) and caffeine (28.29 mg/g). Efficient extraction of bioactive phytochemicals (epigallocatechin gallate, epicatechin gallate, theanine, caffeine) from tea residue (leaves, buds, stems) using DES [citric acid-PPG400 (1:1) + 30% water – selected] with microwave assistance is reported [50] Figure 1. The optimized conditions are the type of DES (HBD and HBA of DES, molar ratio, and water content), solid to liquid ratio, microwave power and time, centrifugation speed, and time (Table 1, No. 23). DES is selected based on its proper polarity and great efficiency for extraction of all four target compounds. Known quantities of tea waste powder and DES are mixed and subjected to extraction using preferred microwave power and temperature for required time. Extraction mixture is subjected centrifugation at necessary speed for requisite time. Supernatant is diluted with known volume of acetonitrile (70%) and taken for analysis and further separation to individual compounds. Kinetic study indicated that the extraction followed Fick’s second law. Controlled experiments are carried out using conventional solvents (i.e., water and methanol) for comparison. Total yield of compounds (229.07 g/Kg) in DES extract is high, when compared to conventional solvents (70 to 90 g/Kg). DPPH and ABTS scavenging antioxidant activities of DES extract are higher than water extract and comparable to methanol extract. Low polar catechins are separated using ethyl acetate liquid-liquid extraction from DES extract. Required quantity of potassium hydroxide is added to remaining DES extract to break the bonds between citric acid and PPG 400 to obtain aqueous two phase system. Caffeine is enriched in PPG 400 phase and theanine retained in potassium citrate phase. Quantities (g/Kg) of the individual compounds isolated are epigallocatechin gallate (15.58), epicatechin gallate (12.85), caffeine (48.44) and theanine (152.20). Selective separation of all the four target compounds is explained using solute solvent interactions, molecular dynamics simulations. Selected DES exhibited superior performance to extract theanine, when compared to earlier studies. Electrostatic interactions and hydrogen bondings between citric acid and theanine played a key role. Theanine and caffeine recovered from the respective phases using resins.
Tea enzymes: Tea polyphenol oxidase (PPO) is isolated and purified from the tender leaves and buds of tea plant using DES [thymol:dodecanoic acid (1:1)] via three-phase partitioning (TPP). Known quantities of fresh tea leaves, citric acid-disodium hydrogen phosphate buffer (with glycerol, pH 5.6), vitamin C, ethylene diamine tetra acetic acid (EDTA), and polyvinylpyrrolidone (PVPP) are mixed and homogenized for necessary period. The homogenized mixture is maintained at the preferred temperature for the required time and filtered through four layers of gauze. The filtrate is centrifuged at the optimum speed for the required period. The supernatant contained crude PPO. Crude PPO is collected, stored at 4oC and used in further experiments at the same temperature. Optimized parameters for crude PPO purification are ammonium sulphate concentration, DES-to- crude extract ratio, extraction time and pH (Table 1, No.22). DES is selected based on its ability for purification fold with respect to enzyme specific activity (Selected DES-enzyme specific activity - 52.82 U/mg). A known quantity of crude PPO is shaken and dissolved in diammonium sulphate (41%) solution. The required volume of DES is mixed with this solution and stored at the optimal temperature and time to form the TPP system. The TPP system is centrifuged under optimized conditions of speed, time, and temperature. The middle phase is collected and mixed with Citric acid-di sodium hydrogen phosphate buffer (0.1M, pH-5.6), to obtain the PPO precipitate. The PPO precipitate is dissolved in citric acid-di sodium hydrogen phosphate buffer (0.1M, pH5.6). The standing / extraction time and pH of the system are critical for TPP system formation. The enzyme activity and protein concentration are measured. The tea polyphenol oxidase recovery (78 %) and purification folds (8.26) are estimated. SDS-PAGE and Native PAGE are used to analyse the PPO [10]. The upper DES phase is recovered from the TPP system and re- used six times. This method can be used for the large scale production of PPO. Tea peroxidase (POD) is isolated and purified from the tender leaves and buds of tea plant using NADES [menthol:hexanoic acid (1:1)] via triple phase partitioning (TPP). Known quantities of tea leaves, polyvinylpyrrolidone, pre-cooled sodium phosphate buffer (pH 7.0) are homogenized in a ratio of 1:0.5:2 (g/g/ mL) for required time. The homogenate is immersed in an ice bath for preferred time with shaking every one hour. Then, homogenate is centrifuged under preferred parameters of speed, time and temperature. The supernatant contains tea peroxidase crude enzyme [11]. The optimized parameters for the purification of tea peroxidase enzyme are concentration of di ammonium sulphate, DES-to-crude extract ratio, extraction time and pH, centrifugation speed time and temperature (Table 1, No.28). HDES is selected based on its, low viscosity, low conductivity, internal hydrogen bonding network, highest recovery, and purification fold towards tea peroxidase enzyme. Requisite quantity of di ammonium sulphate is added to known quantity of crude peroxidase enzyme. Then, required quantity of HDES is added and stirred. Total mixture is maintained in an ice bath to form a three phase system (i.e., upper organic phase, lower aqueous phase and middle phase with precipitated enzyme) for required time. The mixture is centrifuged at preferred conditions of speed, time and temperature. Middle phase is separated and dissolved in known volume of sodium phosphate buffer of required pH. Protein concentration and enzyme activity are evaluated. Tea peroxidase recovery is 70 % and purification fold is 8.25. HDES is recycled for five times for purification of the tea peroxidase enzyme. Recovery and purification fold are 59 % and 6.55 respectively in the fifth cycle. Tea peroxidase isolated using HDES is more stable and pure, when compared to the tea peroxidase produced using t-butanol. SEM studies indicated the formation of peroxidase enzyme-HDES complexes. Further HDES can be recycled. Authors claim applicability of the HDES TPP system on industrial scale.
Miscellaneous applications of DES / NADES based green tea extracts: Preparation of chitosan films using green tea ternary NADES [choline chloride:glycerol:lactic acid (1:1:1)] extracts enriched with antioxidant (DPPH, FRAP evaluations) catechins (epicatechin, epigallocatechin, epicatechin gallate, and epigallocatechin gallate) is reported [51]. NADES is selected based on its eco-friendly nature and is used as an additive to prepare chitosan films. The NADES and green tea powder are stirred under optimized conditions of temperature and time (Table 1, No.16). The extract is filtered and the filtrate is centrifuged at the preferred speed and time. The supernatant is green tea NADES extract. The green tea NADES extract showed good antioxidant properties (DPPH – 33 μM trolox equivalents and FRAP – 397 μM trolox equivalents). Green tea NADES extracts are used for the preparation of chitosan films (refer supporting information). Green tea ternary NADES extracts formed transparent, uniform, and homogeneous chitosan films (without cracks or pores) with high water- barrier properties and high resistance. Green tea polyphenolic compounds provided different surface structures, and the surface of the biofilms became rougher with an increase in green tea polyphenol content. Direct incorporation of green tea NADES extract (10 %) into chitosan films is a good alternative to use of synthetic antioxidants in chitosan films. These films may be useful in the food and allied industries. Tea polyphenols from white tea cakes are extracted using supra DESs [Hydroxypropyl-β- cyclodextrin (HP β-CD: L-lactic acid (1:5) + 40 % water selected] under the assistance of ultrasonication [52]. The optimized conditions are the type of supra DES (HBD and HBA of supra DES, molar ratio, and water content), solid to liquid ratio, ultrasonication power, frequency and time, centrifugation speed, and time (Table 1, No. 24). Supra DES is selected based on its proper viscosity, high extraction efficiency, large hydrogen bonding network and non covalent bonds. Requisite quantities of white tea cake powder and supra DES are extracted using ultrasonication of necessary power and frequency for required time. Control experiment is performed using ethanol (60%). Extract is centrifuged and supernatant is used for analysis and further processing. Yield of tea polyphenols is found to be 111.36 ± 2.31 mg/g under optimised conditions and higher than the conventional solvent extract. Extraction mechanism is described using SEM study and molecular simulations. ABTS and DPPH radical scavenging activities of supra DES extract are 82 % and 67 % respectively and these are 1.09 and 1.32 times higher than those of ethanol extract. Biofilms are prepared using supra DES extract as a plasticizer for polyvinyl alcohol and chitosan. Two advantages - isolation of tea polyphenols from supra DES extract is not required and dissolution of chitosan in solvent is also avoided. Antioxidant activity of the biofilm containing supra DES extract (tea polyphenols) is determined (ABTS – 38 to 54%; DPPH – 30 to 46%). Maximum extraction efficiency of TP from white tea cakes is attributed to an increase in non- covalent bonds and large hydrogen bond network between HP-β-CD and tea polyphenols through the assistance of lactic acid.
Bioactivity of Catechins
Anticancer activity of silver nanoparticles using NADES green tea extract: Green tea extracts (GTE) are prepared using NADES [glycerol-urea (1:1) + 23% water and betaine:urea (1:2) + 23% water - selected]. NADES GTE used for the preparation of silver nanoparticles (AgNPs). The optimized parameters for the extraction of green teas are the type of NADES (HBD and HBA of NADES, molar ratio, water content), solid to liquid ratio, ultrasound power, ultrasound extraction time and temperature, centrifugation speed, time and temperature, and diluent volume for precipitation (Table1, No. 15). NADES is selected based on its high extraction efficiency, and suitability for the preparation AgNPs with the highest yields [53]. The required quantities of green tea powder and DES are mixed using a vortex mixer. This solution is subjected to extraction using ultrasonication under the optimized conditions of temperature and time. The obtained extract solution is centrifuged using the preferred parameters. The supernatant is collected and diluted using a known quantity of ethanol (50%). The obtained solution is centrifuged under optimal conditions to obtain the DES extract of green tea. The green tea extract is filtered. Equal volumes of the extract and AgNO3 solution are mixed and stirred in the dark for the required time to obtain AgNPs with the NADES extract of green tea. The synthesis efficiency for formation of silver nanoparticles for NADES-based GTE is higher than that of the water-based GTE. NADES-GTE AgNPs formed rapidly and homogeneously with a reduced size [NADES (glycerol-urea)-GTE - 39.12 nm and NADES (betaine-urea)-GTE - 43.11 nm]. The stability of AgNPs is improved owing to the capping ability of NADES on the nanoparticle surface. These are characterized by UV-Vis, FT-IR, XRD, ICP-OES, DLS, and SEM analyses. NADES-GTE AgNPs contained 235 % more catechins than other silver nanoparticles (Water-GTE- AgNPs). NADES- GTE-AgNPs showed potential anticancer activity. The anticancer activity of DES (glycerol-urea)- GTE (60 μg / mL) silver nanoparticles is comparable to doxorubicin (12.5 μg / mL). The other properties of these DES-GTE-AgNPs require further investigation. DES provides two functions (i) higher extraction efficiency for green tea catechins and (ii) easy preparation of stable AgNPs with reduced size and good homogeneity.
CLASSIFICATION OF DES / NADES USED IN SUSTAINABLE OPTIMISED PROCESSING OF TEA
DES / NADES used in the processing of different types of teas are type 3 and type 5 classifications [54 56]. Most of the DES / NADES belong to type 3 (i.e., the combination of a quaternary ammonium salt with a HBD). HBAs are quaternary ammonium salts such as choline chloride, betaine chloride, tetraethyl ammonium chloride and betaine hydrazide hydrochloride (refer supporting information Table S1 for details). HBDs are small organic molecules such as urea, carboxylic acids (e.g., mono- or bicarboxylic acids, or amino acids) or polyols (e.g., diols, triols, glycerol, ethylene glycol or carbohydrates). However, some of the DES / NADES are type 5 (i.e., non-ionic). Both HBA and HBD are small organic molecules (i.e., carboxylic acids, amino acids, phenols, urea, cyclic ketone, sugars). One supra DES (Hydroxypropyl-β-cyclodextrin and L-lactic acid) is reported for the isolation of polyphenols from white tea [52]. Two PDES [polymerizable deep eutectic solvents - citric acid-PPG400 (1:1) + 30% water [50]; PEG200-oxalic acid (2:1) + 20 % water [13] are also used for tea processing.
SIGNIFICANT EFFECTS ON THE USES OF DES / NADES AS ALTERNATE GREEN SOLVENTS FOR SUSTAINABLE PROCESSING OF BIOACTIVE CONSTITUENTS FROM TEA
Important observations during extraction of bioactive phytochemicals
Effects on substrate: The liberation of intracellular substances is controlled by plant cells. The plant cells contain cellulose, hemicellulose and pectin. DES / NADES extraction increased the yields of bioactive compounds in shorter times. Distraction of raw material matrix structure, due to pre-treatment / dissolution of cellulose structure / particle size reduction along with the breakage and / or formation of porous structure during use the of DES /NADES, is disclosed by scanning electron microscopy [9,12,17,20,21,33,52]. DESs / NADESs penetrate deep into the matrix, and the mass transfer increases and releases the components. Low pH can promote hydrolysis and increase the penetration ability of DESs / NADESs. Changes in the microstructures of the DES / NADES- treated samples are observed, indicating that the DES / NADES broke the cells and cell walls, and dissolved the weak bonds in the samples during extraction, as indicated by SEM. However, DES/ NADES with a neutral pH showed higher extraction yields of catechins owing to their solubility in DES / NADES [17].
Considerations during solvent screening, selection, water addition: Polarity, viscosity, solubility, surface tension, and extractability are important properties to consider in the selection of DES / NADES as an extraction solvent, which can be adjusted based on the consideration of the molecular structure of the target compounds, with the selection of suitable HBA and HBD [18,19]. The addition of water to a certain level reduces the viscosity, improves the diffusion of bioactive molecules, increases the polarity and maintains the supramolecular complexes between HBD and HBA, which is favorable for high extraction efficiency. Excessive addition of water destroys the hydrogen bonding interactions between HBD and HBA and weakens the eutectic structure of the DES / NADES. This phenomenon reduces the extraction yields. Hence, an appropriate quantity (generally upto a maximum of 50%) of water can be added to DES / NADES to increase the yields [9,13,29,33 35,39,44,47,48,50,52,53]. Phenolic compounds, both free and bound, are extracted using selected DES with assistance of vortex. The target compounds are separated from the DES extracts using an XAD-12 macroporous resin [42]. Both free and bound phenolic compounds from the DES extracts exhibited good antioxidant capacities. However, bound phenolic compounds display different antioxidant properties and significantly support their antioxidant capacity [43]. Molecular dynamics simulations (MDS) and density functional theory (DFT) revealed the interactions between the bioactive molecules and DES / NADES, which helped the movement of bioactive molecules from the aqueous phase to the DES / NADES phase [36,50]. The COSMO-SAC methodology is an effective screening tool for determining the extractability of functional components and solvent selection for bioactive component extraction [41]. Interactions of DESs / NADESs with target molecules from green tea are reported according to MDS and DFT and obtained a theoretical basis [36]. Computational studies using the COSMO-RS-DARE approach to determine the solubility of bioactive compounds in NADES are reported [40]. The solubility computation performed using the COSMO-RS-DARE approach for caffeine solubility values in NADES agreed with the experimental data.
Effects during microwave assistance: In DES MAE, temperature is a critical factor. An increase in temperature decrease the viscosity of the DES / NADES, and its penetration into the sample matrix improves. The mass transfer of target molecules into the DES /NADES increases because of the weakening of the intermolecular interactions in the sample. However, an excess increase in temperature degrades the biomolecules. Furthermore it requires a lower amount of solvent and shorter extraction time [30,50]. Bioactive phytochemicals (i.e., epigallocatechin gallate, epicatechin gallate, theanine, caffeine) are isolated from tea residues using citric acid PPG 400 (DES-MAE) with microwave power assistance efficiently and simultaneously. Efficient extraction is explained using Fick’s second law. These are separated by liquid-liquid extraction, and aqueous two phase system. Separation of individual compounds is described using molecular dynamics simulations. Individual compounds are purified using resins. Maximum quantity of theanine is isolated with the used DES owing to electrostatic interaction and hydrogen bonding between citric acid and theanine [50].
Effects during ultrasonication assistance: The hydrodynamic force is enhanced with an increase in ultrasonic power to a certain stage in DES-UAE. It disrupts cell walls and increases the yields. An excessive increase in ultrasound power raises the bubbles owing to cavitation. This reduces the transmission of ultrasound in the DES / NADES media and reduces the yields. An appropriate ultrasonication time facilitates the complete dissolution and release of the target compounds [17,33,39,44,49,52]. However, excessive ultrasonication may degrade bioactive compounds. Several classes of compounds are isolated using chlorine chloride-based DESs via ultrasonication (DES-UAE). DES extracts are purified using D101 Macroporous resin and analyzed by HPLC and LC-MS. The types of compounds include polyphenols, alkaloids, proteins, amino acids, monosaccharides, catechins, theaflavins, thearubigins, and theabrownins. Choline chloride-ethylene glycol (NADES) is found to be the most suitable for the extraction of most bioactive compounds from Dianhong Congou black tea [49]. The yields of Anji white tea polysaccharides (TPs) are high using the DES-UAE method and the total carbohydrate content in TPs is also high. The molecular weights of TPs obtained using DES-UAE extraction are lower than those obtained using hot water extraction. The cavitation effects due to ultrasound treatment broke the glycosidic chain of high MW polysaccharides, and the MWs of TPs from DES-UAE extraction are reduced. Low-MW TPs exhibited higher antioxidant activities. DES-extracted TPs exhibited higher hypoglycaemic and inhibitory activities against α-amylase and α-glucosidase than hot water-extracted polysaccharides [47].
Observations during use of hollow fiber membrane for separation: A hollow fiber membrane (HF-LPME) filled with DES is used to extract bioactive compounds efficiently. Hydrogen bonding between the functional groups of NADES and the target analyte, as well as π-type hydrogen bond between the polar groups of NADES and the conjugated aromatic rings in caffeic acid are important factors for efficient extraction. The high viscosity of NADES decreases with the addition of methanol (30%) and allows the free movement of analyte molecules [46].
Explanations during use of magnetically imprinted polymers for separation: Magnetic Imprinted Polymers have been reported to adsorb catechins from green tea solutions. The adsorption efficiency is high. These could be used for upto five cycles [32]. Magnetic dispersive micro-solid phase extraction based on magnetic agarose nanoparticles and deep eutectic solvents has been used for the isolation of bioactive compounds. Adsorption is due to hydrogen bonding between the polar groups on the MANPs and phenolic hydroxyl groups of target molecules. Furthermore, π-type hydrogen bonds have been reported between the hydroxyl functional groups of MANPs and the conjugated aromatic rings of bioactive compounds [45]. Deep eutectic solvent (DES) based-molecularly imprinted resins cellulose nanofiber aerogels (MIRs@CNF AG) could adsorb bioactive compounds. MIRs@CNF AG displayed aqueous stability and compressibility. Hence, it is useful for the separation of bioactive compounds in aqueous environment. Hydrogen bonding, electrostatic attraction, and π-π stacking forces are involved in the adsorption mechanism, as per density functional theory. These can be used up to five cycles without decrease in the quantity and, the solvent is saved [35]. DES-Fe3 O4 /MIPs are used as adsorbents in magnetic solid-phase extraction for the separation of theobromine, theophylline, catechin hydrates and caffeic acid from green tea extracts. Recovery values are high [37,38]. DES based Fe3 O4 /molecularly imprinted polymers are used for the separation of alkaloids from green tea extracts in excellent yields [37]. Caffeine is extracted using UAE-DES. Fick’s second law of diffusion kinetics model for active compounds extraction is confirmed using this method. Macroporous resins (XAD-200) are used for the recovery of target compounds from the DES extract. The recovery yields are good [39].
Use of membranes for separation of oxidised highly polymeric polyphenols: Thearubigins and theabrownins fractions containing abundant quantities of highly polymeric tea pigments are isolated from black tea NADES extract using membranes. These fractions exhibited effective antioxidant activity owing to the presence of higher quantities of highly oxidised polymeric polyphenols. These could be useful in food, pharmaceutical, medicinal and cosmetic industries [9].
Important observations during the utilisation of tea waste for lignin: Antimicrobial and antioxidant lignins are isolated from tea residue waste using NADES pretreatment followed by solvent extraction. Utilisation of tea waste can avoid environmental pollution [12]. Lignin is separated from tea stem using PDES pretreatment and extraction. Molecular weight and nanoparticle size of lignin are reduced owing to intervention of PDES as per density functional theory and useful for the preparation of fluorescent lignin. Remaining cellulose is easily convertible to levulinic acid. Agricultural waste is utilised [13].
Noteworthy points during separation of tea enzymes
Tea polyphenol oxidase (PPO) is isolated and purified from the tender leaves and buds of tea plants using selected DES via a three-phase partitioning (TPP) system. The selection of DES is important. The extraction / standing time and pH of the solution are critical for the TPP formation. The purified PPO is in the middle phase. The recovery of PPO and purification folds are good with the use of DES. The upper-phase DES can be recovered and re- used [10]. Tea peroxidase is isolated and purified from the tender leaves and buds of tea plants using selected HDES via a triple phase partitioning (TPP) system. The selection of HDES is important. The extraction / standing time and pH of the solution are critical for the TPP formation. The purified tea peroxidase is in the middle phase. The recovery of tea peroxidase and purification folds are superior with the use of HDES, when compared to the tea peroxidase produced using t-butanol TPP system. The upper-phase DES can be recovered and re-used, indicated its applicability on industrial scale [11].
Key observations through the use of DES extracts for bioactivities and applications.
DES-based GTE are used for the preparation of AgNPs with good stability, homogeneity, and reduced size. Silver nanoparticles of DES-based green tea extracts with bioactive compounds have shown potential anticancer activity [53]. Green tea NADES extracts (enriched with antioxidant catechins) provided chitosan films with good properties, and the surface of the biofilms became rougher with an increase in green tea polyphenol content. These films may be useful in the food industry [51]. Supra DES (HP-β-CD - lactic acid) extract containing tea polyphenols is isolated from white tea cakes under the assistance of ultrasonication (UAE-DES). Antioxidant supra DES extract is used as a plasticizer directly for the preparation of biofilms using polyvinyl alcohol and chitosan. Tea polyphenols are not isolated from supra DES extract and chitosan is not dissolved in solvent [52].
Most of the compounds used to produce DES / NADES are approved under various safety regulations in different countries. NADESs had a significant impact on the extraction of target compounds with higher yield, premium quality, and quantity from tea. Further, use of DES / NADES reduces extraction time and solvent quantity, when used along with other suitable techniques or independently. The application of DES and NADES in the food and allied industries is a promising area of sustainability and for clean production of products with bioactive phytochemicals from tea.
DES/NADESS-MAJOR ADVANTAGES
Main advantages of DES / NADES are easy and low-cost production procedures, maintainable production (which does not produce waste), flexibility of DESs (it is superior, as these are used in different extraction techniques), extraction capabilities, and higher recovery of bioactive compounds [57]. DESs are designer solvents and several amalgamations / mergers are possible. It is possible to regulate physico-chemical characteristics for precise purposes. The comprehensive polar range, versatility, high degree of solubilization strength for different target compounds, low harmfulness, biodegradable / recyclability, non-volatility, and non-flammability are advantages of DES/NADES. DES / NADES as extraction solvents provide higher yields of target compounds, when compared to conventional solvents (Refer table S2 supporting information) [9,17,18,30,31,33,34,39,41
,42,47,49,50,53]. Further bioactive phytochemicals are stable in DES / NADES extracts [17,58]. Solubility of the bioactive phytochemicals is increased in the presence of DES / NADES [40]. Bioactivities of NADES extracts are higher, when compared to conventional solvent extracts [47,51-53]. The economics of DES/NADES are comparable to those of conventional solvents [19,28,59,60]. The disadvantages are the high viscosity of DES/NADESs in selected cases could be deterring; however, this problem is resolved appropriately by the addition of sufficient water. However, the addition of excess water may reduce the yields of bioactive compounds as the interactions between the sample and DESs decrease due to excess water. DESs possess supramolecular characteristics, with a water content of less than 50%. Ultimately, water addition can reduce the cost of production, as it reduces the consumption of DES [18,30,31]. Target compounds are recovered from DES extracts using different resins [30,39,42,49]. Tea residue waste and agricultural waste tea stem is used for the production of bioactive lignins. In selected cases, DES can be re-used, and the solvent is saved [10-12,35]. Currently, the application of DES/NADESs on an industrial scale is continuously improving, as the extracts are being used directly (many of the ingredients of the DESs are in the GRAS category) without expensive downstream purification steps. Scale-up studies on the use of DES have been initiated and parameters are being optimized. Economic considerations have also been discussed [10,11,17].
LIMITATIONS
Many combinations of HBAs and HBDs are possible. Hence, characterization of DESs as well as generalization of their properties is not an easy task. High Viscosity makes handling difficult in select cases (isolation and purification of bioactive molecules). DES/NADESs have almost zero vapor pressure, and the recovery of the target compound can be difficult in selected cases. DES / NADES can extract minerals, trace elements along with phytochemicals, when these are present in the plants. Presence of toxic trace elements is a problem in the DES / NADES extracts of bioactive phytochemicals, which are useful for food, pharmaceutical, medicinal and cosmetic industries. However, this limitation can be solved by monitoring the extraction of these elements with judicious selection of DES / NADES components [61,62]. DES display low toxicity levels, when compared to conventional organic solvents. NADES exhibit lowest toxicity, as these are formed from primary metabolites. Hence NADES are more suitable alternatives for applications in food, pharmaceutical, medicinal and cosmetic industries. It is vital to select the components of DES / NADES and their concentrations to lessen the dangers. Biodegradability is one of the important aspect of DES / NADES. Easily biodegradable DES / NADES are safe for the above industrial applications.
Development of strategies to enhance the biodegradability of DES / NADES is an important task for researchers.
More toxicity / biodegradability studies on DES / NADESs are required to avoid environmental pollution [59,63]. In vitro toxicity studies have been reported; however, in vivo toxicity studies are required for several DESs for further applications in food / health / cosmetic products. The starting materials of the DES / NADES can indicate the biodegradability of the final DES / NADES to some extent. However, this approach is not yet comprehensive. Computational predictive methods must be developed for these purposes.
CONCLUSIONS
Tea is one of the most widely used beverages, next to water, and is produced systematically in the plantation industry worldwide. Recently, several studies have reported on the use of DES / NADES in tea and tea waste processing. Most studies have focused on the extraction of bioactive phytochemicals. Selected analytical methods for estimating bioactive molecules using DES / NADES have also been reported. The solubilization and stabilization of bioactive molecules have also been described. Conductor- like screening models (i.e., COSMO-RS, COSMO-RS-DARE, COSMO-SAC) are used for these studies for screening and selection of DES / NADES. Several types of DES / NADES are used in the tea processing (e.g., alcohol based, acid based, urea based, sugar based, vinyl pyrrolidone based, amino acid based, ternary, supra DES and polymerizable DES). Extractions are explained using Fick’s second law. Solute-solvent interactions are described using molecular dynamics simulations and density functional theory. Most of these studies have indicated that the use of DES / NADES is efficient in the respective method for tea processing. The extraction times and solvent usage are reduced. The time required for the estimation of the target compounds is also decreased. The solubility of bioactive molecules in the aqueous DES/NADES extract is improved. The stability of phenolic bioactive compounds increased owing to DES / NADES intervention. Selected modern techniques (magnetically imprinted polymers, nanofiber aerogels, magnetic agarose nanoparticles, molecularly imprinted resins with cellulose nanofiber aerogels, magnetic solid phase extraction, and hollow fiber-liquid phase microextraction) have been reported for the exclusive adsorption of key bioactive components from extracts. Selected extracts with target molecules are used directly as drugs because DES / NADES are safer than organic solvents. Furthermore, volatile organic solvents can be replaced in several examples, as the derived products (in particular, extracts from NADES) can be directly used in the food, pharmaceutical, and cosmetic products. Most of the constituents of NADES are safe, as reported by different regulations. NADES are generally nonvolatile and harmless. The addition of water makes it possible to select NADES because most of the NADES are fully water- soluble and miscible. Several types of bioactive phytochemicals [e.g., catechins, xanthine alkaloids, flavonoids, theaflavins, thearubigins, theabrownins, lignin, celluloses, sugars, aminoacids and polysaccharides) are separated from varieties of teas and tea waste and their solubility is also improved. Tea NADES extracts are directly used in the preparation of chitosan films, which could be useful in food industry. Tea enzymes (i.e., polyphenol oxidase, peroxidase) are isolated with good purity from tea two leaves and bud using DES / NADES. Anticancer activity potential of silver nano particles of NADES green tea extract is comparable to synthetic compounds. Bioactive lignin is isolated from tea residue waste (which can reduce environmental pollution), and agricultural waste (i.e., tea stem). NADES are safe at high doses, when compared to routine organic solvents. Several constituents of NADES received GRAS certification (e.g., fructose and, glucose). Because of the high extraction capacity and low toxicity of their components, NADES are suitable for the extraction of bioactive compounds, flavors and fragrances. Derived extracts could be safely used as additives in foods, drug carriers in pharmaceuticals, and in cosmetics. Biofilms with antioxidant activity are reported using NADES extracts, which are useful for food industry (i.e., edible food packaging). The use of DES/NADES is expected to increase in the future owing to these advantages. Pilot-scale extraction using deep eutectic solvents and evaluation of economic feasibility studies are initiated. Researchers have used DES/NADES as alternative solvents during tea processing for various purposes (e.g., extraction, estimation of target compounds in complex matrices, enhancement of solubility of bioactive compounds in aqueous media), despite minor toxicological problems. The main reason is that they are safer than traditional solvents. The application of DES/NADES is improving in sustainable tea processing.
CREDIT AUTHORSHIP CONTRIBUTION STATEMENT
Lingamallu Jaganmohanrao: Conceptualization, Visualization, Investigation, Methodology, Formal analysis, Validation, Supervision, Writing – original draft, Writing – review, editing
Supplementary Flies
REFERENCES
- Jaganmohanrao L, Ramalakshmi K. Chemical composition and pharmalogical, medical properties of tea. In book: Recent Trends in Soft Beverages. 2011: 101-112.
- Purkait MK, Haldar D, Debnath B. Advancement in the extraction of bioactive compounds from tea leaves. In Technological Advancements in Product Valorization of Tea Waste. 2023: 37-65.
- Bae J, Kim N, Shin Y, Kim SY, Kim YJ. Activity of catechins and their applications. Biomed Dermatol. 2020; 4: 10.
- Sharma V, Jaganmohanrao L. A thought on the biological activities of black tea. Crit Rev Food Sci Nutr. 2009; 49: 379-404.
- Zhao T, Li C, Wang S, Song X. Green Tea (Camellia sinensis): A Review of Its Phytochemistry, Pharmacology, and Toxicology. Molecules. 2022; 27: 3909.
- Ganeshpurkar A, Saluja A. The pharmacological potential of catechins. Indian J Biochem Biophys. 2020; 57: 505-511.
- Nuryana I, Ratnakomala S, Fahrurrozi, Juanssilfero AB, Andriani A, Putra FJN, et al. Catechin contents, antioxidant and antibacterial activities of different types of indonesian tea (camellia sinensis). Ann Bogor. 2020; 24: 106-113.
- Luo Q, Luo L, Zhao J, Wang Y, Luo H. Biological potential and mechanisms of Tea’s bioactive compounds: An Updated review. J Adv Res. 2024; 65: 345-363.
- Wang D, Liu Y, Zeng B, Xu Y, Cao S, Luo Y, et al. Compositional and biochemical activity evaluation of highly polymerized tea pigments in black tea based on natural deep eutectic solvent extraction, Food Chem: X. 2025; 27: 102413.
- Xu Y, Zeng B, Xiao S, Wang D, Liu Y, Chen S, et al. Purification of polyphenol oxidase from tea (Camellia sinensis) using three-phase partitioning with a green deep eutectic solvent. Food Chem X. 2024; 23: 101720.
- Liu Y, Wang H, Xu Y, Wang D, Zhu X, Wang M, et al. Menthol-based hydrophobic deep eutectic solvents for the purification of tea peroxidase using a three-phase partition method. LWT-Food Sci. Technol. 2025; 218: 117528.
- Ghosh S, Pramanik K. Extraction of lignin from sustainable lignocellulosic food waste resources using a green deep eutectic solvent system and its property characterization. Int J Biol Macromol. 2025; 307: 142094.
- Zhou PR, Zheng XP, Du YP, Chai Y, Zhang YC, Zheng YZ. Effective pretreatment of tea stem via poly-deep eutectic solvent for promoting platform molecule production and obtaining fluorescent lignin. Int J Biol Macromol. 2025; 297: 139922.
- Aijaz M, Keserwani N, Yusuf M, Ansari NH, Ushal R, Kalia R. Chemical, Biological, and Pharmacological Prospects of Caffeic Acid. Biointerface Res Appl Chem. 2023; 13: 324.
- Zhang S, Jin J, Chen J, Ercisli S, Chen L. Purine alkaloids in tea plants: component, biosynthetic mechanism and genetic variation. Beverage Plant Res. 2022; 2: 9.
- Koina IM, Sarigiannis Y, Hapeshi E. Green extraction techniques for the determination of active Ingredients in tea: current State, challenges, and future perspectives. 2023; 10: 121.
- Jeong KM, Ko J, Zhao J, Jin Y, Yoo DE, Han SY, et al. Multi-functioning deep eutectic solvents as extraction and storage media for bioactive natural products that are readily applicable to cosmetic products. J Clean Prod. 2017; 151: 87-95.
- Heng Z, Baokun T, Kyungho R. Extraction of catechin compounds from green tea with a new green solvent. Chem Res Chin Univ. 2014; 30: 37-41.
- Fernandez MDLA, Boiteux J, Espino M, Gomez FJV, Silva MF. Natural deep eutectic solvents-mediated extractions: The way forward for sustainable analytical developments. Anal Chim Acta. 2018; 1038: 1-10.
- Zainal-Abidin MH, Hayyan M, Hayyan A, Jayakumar NS. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Anal Chim Acta. 2017; 979: 1-23.
- Jablonsky M, Skulcova A, Malvis A, Simac J. Extraction of value-added components from food industry based and agroforest biowastes by deep eutectic solvents. J Biotechnol. 2018; 282: 46-66.
- Kalyniukova A, Holusa J, Musiolek D, Kadukova JS, Wasylka JP, Andruch V. Application of deep eutectic solvents for separation and determination of bioactive compounds in medicinal plants. Ind Crop Prod. 2021; 172: 114047.
- Szopa D, Wróbel P, Witek-Krowiak A. Enhancing polyphenol extraction efficiency: A systematic review on the optimization strategies with natural deep eutectic solvents. J Mol Liq. 2024; 404: 124902.
- Lee J, Jung D, Park K. Hydrophobic deep eutectic solvents for the extraction of organic and inorganic analytes from aqueous environments. Trends Anal Chem. 2019; 118: 853-868.
- Wen Q, Chen JX, Tang YL, Wang J, Yang Z. Assessing the toxicity and biodegradability of deep eutectic solvents. Chemosphere. 2015; 132: 63-69.
- Vanda H, Dai Y, Wilson EG, Verpoorte R, Choi YH. Green solvents from ionic liquids and deep eutectic solvents to natural deep eutectic solvents. C R Chim. 2018; 21: 628-638.
- Ferreira IJ, Oliveira F, Jesus AR, Paiva A, Duarte ARC. Current methodologies for the assessment of deep eutectic systems toxicology: Challenges and perspectives. J Mol Liq. 2022; 362: 119675.
- Benvenutti L, Zielinski AAF, Ferreira SRS. Which is the best food emerging solvent: IL, DES or NADES? Trends Food Sci Technol. 2019; 90: 133-146.
- Fuad FM, Nadzir MM, Kamaruddin AH. Hydrophilic natural deep eutectic solvent: A review on physicochemical properties and extractability of bioactive compounds. J Mol Liq. 2021; 339: 116923.
- Li J, Han Z, Zou Y, Yu B. Efficient extraction of major catechins in Camellia sinensis leaves using green choline chloride-based deep eutectic solvents. RSC Adv. 2015; 5: 93937-93944.
- Bajkacz S, Adamek J, Sobska A. Application of deep eutectic solvents and ionic liquids in the extraction of catechins from tea. Mol. 2020; 25: 3216.
- Ma W, Row KH. Solid-phase extraction of catechins from green tea with deep eutectic solvent immobilized magnetic molybdenum disulfide molecularly imprinted polymer. Mol. 2020; 25: 280.
- Luo Q, Zhang JR, Li HB, Wu DT, Geng F, Corke H, et al. Green Extraction of Antioxidant Polyphenols from Green Tea (Camellia sinensis). Antioxidants. 2020; 9: 785.
- Martinovi? M, Krgovi? N, Neši? I, Žugi? A, Tadi? VM. Conventional vs. Green Extraction Using Natural Deep Eutectic Solvents-Differences in the Composition of Soluble Unbound Phenolic Compounds and Antioxidant Activity. Antioxidants (Basel). 2022; 11: 2295.
- Ma Y, Liu X, Xu X, Wu M, Sun J, Niu N, et al. A compressible and under water super elastic hydrophilic molecularly imprinted resin composite cellulose nanofiber aerogel to separate and purify catechins from tea. Chem Eng J. 2023; 472: 145043.
- Kohani M, Raissi H, Zaboli A, Hashemzadeh H. Exploring the potential of deep eutectic solvents for extracting bioactive compounds fromtea: Insights from molecular dynamics simulations. J Mol Liq. 2024; 393: 123589.
- Li G, Wang X, Row KH. Magnetic Solid-phase Extraction with Fe3O4/ Molecularly Imprinted Polymers Modified by Deep Eutectic Solvents and Ionic Liquids for the Rapid Purification of Alkaloid Isomers (Theobromine and Theophylline) from Green Tea. Mol. 2017; 22: 1061.
- Li G, Row KH. Ternary deep eutectic solvent magnetic molecularly imprinted polymers for the dispersive magnetic solid-phase microextraction of green tea. J Sep Sci. 2018; 41: 3424-3431.
- Cai C, Li F, Liu L, Tan Z. Deep eutectic solvents used as the green media for the efficient extraction of caffeine from Chinese dark tea. Sep Purif Technol. 2019; 227: 115723.
- Jelinski T, Cysewski P. Quantification of Caffeine Interactions in Choline Chloride Natural Deep Eutectic Solvents: Solubility Measurements and COSMO-RS-DARE Interpretation. Int J Mol Sci. 2022; 23: 7832.
- Cui Z, Djocki AVE, Yao J, Wu Q, Zhang D, et al. COSMO-SAC-supported evaluation of natural deep eutectic solvents for the extraction of tea polyphenols and process optimization. J Mol Liq. 2021; 328: 115406.
- Wang X, Jia W, Lai G, Wang L, Contreras MDM, Yang D. Extraction for profiling free and bound phenolic compounds in tea seed oil by deep eutectic solvents. J Food Sci. 2020; 85: 1450-1461.
- Wang X, Contreras MDM, Xu D, Jia W, Wang L, Yang D. New insights into free and bound phenolic compounds as antioxidant cluster in tea seed oil: Distribution and contribution. LWT-Food Sci Technol. 2021; 136: 110315.
- Liu Y, Liu HY, Yang X, Zhu F, Wu DT, Li HW, et al. Green extraction, chemical composition, and in vitro antioxidant activity of theabrownins from Kangzhuan dark tea. Curr Res Food Sci. 2022; 5: 1944-1954.
- Majidi SM, Hadjmohammadi MR. Development of magnetic dispersive micro-solid phase extraction based on magnetic agarose nanoparticles and deep eutectic solvents for the isolation and pre- concentration of three flavonoids in edible natural samples. Talanta. 2021; 222: 121649.
- Nia NN, Hadjmohammadi MR. Amino acids- based hydrophobic natural deep eutectic solvents as a green acceptor phase in two- phase hollow fibre-liquid microextraction for the determination of caffeic acid in coffee, green tea, and tomato samples. Microchem J. 2021; 164: 106021.
- Xia B, Liu Q, Sun D, Wang Y, Wang W, Liu D. Ultrasound-Assisted Deep Eutectic Solvent Extraction of Polysaccharides from Anji White Tea: Characterization and Comparison with the Conventional Method. Foods. 2023; 12: 588.
- Wang M, Wang J, Zhou Y, Zhang M, Xia Q, Bi W, et al. Ecofriendly mechanochemical extraction of bioactive compounds from plants with deep eutectic solvents. ACS Sustain Chem Eng. 2017; 5: 6297- 6303.
- Zhao Z, Wen M, Jiang V, Su S, Xu YQ, Han Z, et al. Extraction effects ofeight deep eutectic solvents on dianhong black tea: From chemical composition analysis to antioxidant and α-glucosidase inhibitory assessments. Food Biosci. 2024; 61: 104923.
- Yang Y, Guan M, Fang M, Gu Y, Yi L, Ren D. Simultaneous extraction and selective separation of catechins, caffeine and theanine from waste tea residue facilitated by citric acid- based deep eutectic solvent. Sep Purif Technol. 2025; 360: 130918.
- Alves TFP, Teixeira N, Vieira J, Vicente AA, Mateus N, Freitas V, et al. Sustainable chitosan packaging films: Green tea polyphenolic extraction strategies using deep eutectic solvents. J Clean Prod. 2022; 372: 133589.
- Huang D, Chen L, Chen X, Huang X, Yang Y, Liu J, et al. Supramolecular deep eutectic solvents as green media for efficient extraction of tea polyphenols and its application in bio-active film. Food Chem. 2025; 465: 141904.
- Liu Y, Kang S, Li K, Chen J, Bae B, Hwang I, et al. Ecofriendly and enhanced biogenic synthesis of silver nanoparticles using deep eutectic solvent- based green tea extracts. J Clean Prod. 2022; 379: 134655.
- Mannu A, Blangetti M, Baldino S, Prandi C. Promising Technological and Industrial Applications of Deep Eutectic Systems. Mater. 2021; 14: 2494.
- Abranches DO, Coutinho JAP. Type V deep eutectic solvents: Design and applications. Curr Opin Green Sust Chem. 2022; 35: 100612.
- Gao Y, Fan M, Cheng X, Liu X, Yang H, Ma W, et al. Deep eutectic solvent: Synthesis, classification, properties and application in macromolecular substances. Int J Biol Macromol. 2024; 278: 134593.
- Aktas H, Kurek MA. Deep eutectic solvents for the extraction of polyphenols from food plants. Food Chem. 2024; 444: 138629.
- Obluchinskaya ED, Pozharitskaya ON, Zakharova LV, Daurtseva AV, Flisyuk EV, Shikov AN. Efficacy of Natural Deep Eutectic Solvents for Extraction of Hydrophilic and Lipophilic Compounds from Fucus vesiculosus. Molecules. 2021; 26: 4198.
- Dheyab AS, Abu Bakar MF, AlOmar M, Sabran SF, Hanafi AFM, Mohamad A. Deep eutectic solvents (DESs) as green extraction media of beneficial bioactive phytochemicals. 2021; 8: 176.
- Ferreira C, Sarraguça AM. A comprehensive review on deep eutectic solvents and its use to extract bioactive compounds of pharmaceutical interest. Pharm. 2024; 17: 124.
- Shikov AN, Shikova VA, Whaley AO, Burakova MA, Flisyuk EV, Whaley AK, et al. The Ability of Acid-Based Natural Deep Eutectic Solvents to Co-Extract Elements from the Roots of Glycyrrhiza glabra L. and Associated Health Risks. Mol. 2022; 27: 7690.
- Shikov AN, Obluchinskaya ED, Flisyuk EV, Terninko II, Generalova YE, Pozharitskaya ON. The Impact of Natural Deep Eutectic Solvents and Extraction Method on the Co- Extraction of Trace Metals from Fucus vesiculosus. Mar Drugs. 2022; 20: 324.
- Sharma A, Lee BS. Toxicity test profile for deep eutectic solvents: A detailed review and future prospects. Chemosphere. 2024; 350: 141097.