Bilateral Duplicated Renal Collecting Systems and Associated Pathology: An Unusual Case Report
- 1. Department of Integrative Physiology and Anatomy, University of North Texas HSCTCOM, USA
- 2. Department of Urologic Surgery, University of North Texas HSC-TCOM, USA
Abstract
A 31-year-old female with a 19 year history of chronic urinary tract infections (UTI), intermittent hematuria, and associated flank pain was referred to our service for evaluation of anatomy of renal system and possible surgical intervention. At the time of the referral, the UTI’s had become more frequent and the flank pain had increased in severity.The following diagnostic studies were employed to elucidate the etiology of the patient’s condition:abdominal and pelvic computed tomography (CT) scans, intravenous pyelogram (IVP), voiding cystourethrogram (VCUG), kidney ureter and bladder x-rays (KUB), flexible cystoscopy, bilateral renal ultrasounds (RUS), cystourethroscopy with sounding, bilateral retrograde pyelograms with fluoroscopy, and ureteroscopy. Our studies revealed an uncommon, bilateral duplication of the urinary collecting systems. Upper and lower pole collecting systems, each with their own ureter, were discovered for both kidneys. Chronic changes of the upper pole collecting system of the left kidney including hydroureter, caliectasis, and renal calculi were also observed. Furthermore, a large ureterocele, measuring 4 cm in length x 2 cm maximumtransverse diameter, was observed in the distal portion of the left upper pole collecting system ureter. This ureter inserted into the proximal band of the urinary sphincter. These findings are consistent with an obstructive process that resulted in chronic damage to the left upper pole collecting system, and presents a difficult clinical scenario to manage. The duplicated
collecting system on the right showed normal function despite its anatomical variation. Based on this finding, we believe that the location where the duplicated ureter inserts into the bladder is more predictive of chronic renal insult and associated pathology versus the presence of the variant alone causing problems.
Keywords
Collecting system; Ureter • Renal; Variant; Pathology; Bilateral; Duplication
Citation
Fraser PR, Bredeweg AC, Stroud R, Rosales AA (2014) Bilateral Duplicated Renal Collecting Systems and Associated Pathology: An Unusual Case Report. JSM Clin Case Rep 2(6): 1062.
INTRODUCTION
Anatomic variation are generallypart of the differential diagnosis when a healthcare professional attempts to successfully diagnosis and treat chronic urologic problems,such as chronic UTI, or chronic pyelonephritis. These conditions can be caused by anatomic variations that create backward pressure on the system. Concrete figures detailing prevalence of ureter duplicationare elusive; however, some studies suggest that unilateral and bilateral duplication occur in approximately 1.5% and 0.3% of the population, respectively. All forms of renal duplication have a 2:1 predominance for females to males. Ureteroceles are commonly linked with renal duplication, but only in unilateral duplications and only on the non-duplicated side. Renal duplications are found to have a significantly increased incidence in chronic pyelonephritis and UTIs [1].
Another group of investigators looked into a set of complicated renal duplications and found that most cases were unilateral. Furthermore, among those unilateral duplications, a predominance of upper pole duplications were found in a 3:1 ratio, with lower pole duplications comprising the smaller group. They also demonstrated that the most common comorbid condition present with a duplicated urinary collecting system was hydronephrosis [2]. Hydronephrosis is the condition resulting from the constant reflux of urine back into the kidney. Blunting of the renal calyces and swelling of the renal pelvises can provide evidence of reflux on IVP or retrograde pyelogram.
One particular case study revealed a total obstruction of both duplicated lower pole moieties in a case of bilateral duplication of the urinary collecting system. The patient was 22-year-old primagravida that experienced sepsis and flank mass two days after a normal vaginal delivery. Angiography and retrograde pyelography were used to observe significant bilateral occlusion of the lower pole moieties and subsequent hydronephrosis in those areas [3]. The group described a spherical mass that contained a curved segment of thickened calcification that blocked flow from lower aspect of the left kidney, and a significantly large inflammatory mass that blocked the lower portion of the right inferior renal fossa.
The treatment of vesicoureteral reflux and/or hydronephrosis in patients with some form of obstruction as a result of complications secondary to partial duplication of urinary collecting system has been controversial. The standard operative procedure for such conditions has traditionally been heminephrectomy in which case the dysfunctional pole of the kidney is removed along with its associated ureters. The duplication of the ureter and the associated renal pelvis is the most common upper urinary tract variation in childhood. When heminephrectomy has been undertaken, in most cases the defunctionalized segment is often left in situ. In such cases, long term follow up has shown that leaving behind the problematic segment causes little to no problems. One group showed that later resection of the defunctionalized segment was unnecessary [4].
Another study analyzed the effectiveness of nonsurgical management of vesicoureteral reflux and/or hydronephrosis restricted to segments of kidney associated with duplication of the urinary collecting system. In that study, the group included unilateral and bilateral duplications, all of which were experiencing some form of reflux and dysfunction. Reflux was graded on a scale of I-V, and all groups were included in the analysis. The patients were divided in three groups: no treatment, antibiotics, and surgical intervention. Outcomes were shown to be statistically equal across the three groups, with the antibiotic group having less UTIs. These findings caused the investigators to conclude that conservative management of vesicoureteral reflux is preferred over surgical intervention [5].
Despite the obvious lean away from surgical intervention in the case of duplicated urinary collecting systems, a group of urological surgeons showed that a new technique known as the modified psoas hitch with Lich Gregoironlay is an effective technique for ureter reconstruction. The group conducted a retrospective analysis of 20 patients that had undergone modified psoas hitch with Lich-Gregoironlay during reconstruction of the ureter. The group showed resolution of symptoms and no long-term sequelae such as chronic flank pain, recurrent pyelonephritis, hydronephrosis, or compromised renal function [6]. The technique was limited to ureteral reconstruction; however, the technique could be utilized in some cases of renal reconstruction in which a segment of kidney is defunctionalized while attempting to utilize the segment of ureter for the purpose of lengthening an existing ureter.
Our case study provides a variety of anatomic variations and associated pathology which utilize to illustrate the delicate and often controversial aspects of managing a patient with complicated duplication of urinary collecting systems with concomitant chronic pyelonephritis and persistent flank pain. Furthermore, the unusual nature of the variant in our case presents a unique clinical dilemma, and requires an thorough understanding of the condition, the associated complications, and the available treatment modalities to successfully manage the patient.
CASE REPORT
In the routine evaluation of a 31-year-old Caucasian American female for chronic UTI and chronic flank pain an anatomic variant was discovered on imaging studies. Multiple diagnostic studies were utilized in the study of this patient. Studies included: abdominal and pelvic computed tomography (CT) scans, intravenous pyelogram (IVP), voiding cystourethrogram (VCUG), kidney ureter and bladder x-rays (KUB), flexible cystoscopy, bilateral renal ultrasounds (RUS), cystourethroscopy with sounding, bilateral retrograde pyelograms with fluoroscopy, and ureteroscopy. The aforementioned studies revealed an uncommon, bilateral duplication of the urinary collecting systems. Upper and lower pole collecting systems, each with their own ureter, were discovered for both kidneys. Chronic changes of the upper pole collecting system of the left kidney including hydroureter, caliectasis, and renal calculi were also observed. Furthermore, a large ureterocele, measuring 4 cm in length x 2 cm maximumtransverse diameter, was observed in the distal portion of the left upper pole collecting system ureter. This ureter inserted into the proximal band of the urinary sphincter. Both left ureters were observed to be dilated.
DISCUSSION
Chronic UTI, chronic pyelonephritis, ureteroceles, hydroureter, caliectasis, chronic flank pain, and renal calculi are important conditions that each alone pose a threat to the kidneys. When they occur in concert the threat is magnified, and the management complicated. Such a case has potentially devastating effects to the well-being of the patient. A treatment plan must be carefully constructed to include consideration for the best result with the least amount of invasive therapy as possible. The diagnosis of the anatomical variant in this case, required a great deal of invasive procedures, each with their own set of risks. Fortunately, the information gleaned was essential to rescuing the kidneys from the viscous cycle that had already begun to damage them.
Ureteral duplication is the consequence of abnormal development of the ureteric bud (UB). For this discussion, we will focus on the processes behind complete ureteral duplication as opposed to partial duplication. The ureteric bud, induced by the metanephric mesenchyme, arises from the distal portion of the mesonephric duct around the 5th week of gestation. The UB further branches and elongates developing into the ureters, renal pelvis, calyces, and collecting ducts. Embryological basis of complete ureteral duplication may be explained by the development of two separate ureteral buds from a single nephric duct, either by increased induction stimulus or inadequate suppression of induction. Studies have shown that normal renal development depends on reciprocal interactions between the UB and metanephric mesenchyme. The metanephic mesenchyme stimulates outgrowth of the UB primarily through production of glial-derived neurotrophic factor (GDNF) [7]. GDNF, acts on its receptor c-RET, inducing proliferation and branching of UB cells. Many genes play a role in this inductive process and abnormal increases in inductive stimulus is a possible explanation for formation of multiple ureteric buds. Another aspect associated with inductive signaling relates to proper positioning of the UB and regulation of inductive stimuli. Several factors have been implicated in this process and will be discussed independently. Two negative regulators of UB formation include bone morphogenic protein 4 (BMP-4) and Sprouty1. BMP-4 is expressed in the mesenchyme surrounding the nephric duct acts locally to inhibit ectopic budding by antagonizing GDNF action [8]. Embryos lacking BMP-4 have duplicated ureters, and organ cultures show blockade of UB formation in GDNF-soaked beads [9]. Sprouty1 is a negative regulator of a downstream signaling cascade activated by GDNF. Studies show that embryos lacking Sprouty1 have supernumerary UB formation [9]. FoxC1 is produced by the metanephric mesenchyme adjacent to the nephric duct and acts to restrict the activity of GDNF. Loss of FoxC1 is associated with ectopic ureteric buds and formation of duplicated ureters [9]. Fibroblast growth factors also appear to play an important part in ureter devlopment. The loss of fibroblast growth factor receptor 2 (Fgfr2) in animal models often leads to multiple ureteric buds and anomalies including duplicated ureters [10]. Normally Fgfr2 is expressed in stromal cells adjacent to the nephric duct and main ureteric bud trunks and is important in ensuring that there is a single, properly positioned ureteric bud [10]. In one particular study involving mice with Fgfr2 deletions, 67% of the embyros had two ureteric buds from one nephric duct [10]. Deletion of Fgfr2 did not change expression of GDNF, BMP-4, or other major regulators of ureteric bud formation suggesting an alternative critical role in this process [10]. Many different genes are involved with proper ureter development and any alteration in induction or regulation of induction for primary formation of the ureteric bud could be an explanation for complete ureteral duplication (Figure 1,2,3).
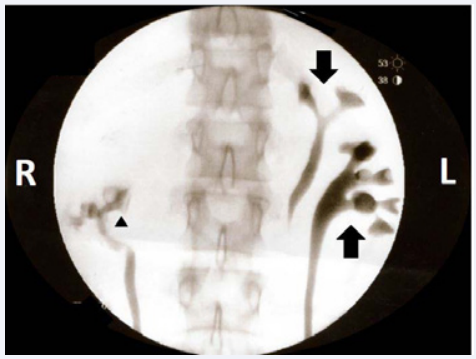
Figure 1 Fluoroscopic image of retrograde cystoscopy showing duplicated urinary collecting system of the left kidney. The upper pole moiety (downward arrow) and the lower pole moiety (upward arrow) can be observed on the side of the image labeled L. The lower pole moiety of the right collecting system is visualized as well (triangle).
Figure 2 Fluoroscopic imaging of retrograde cystoscopy shows a duplicated urinary collecting system of the right kidney. The upper pole moiety (horizontal arrow) and the lower pole moiety (upward arrow) are clearly visualized along with their own ureters.
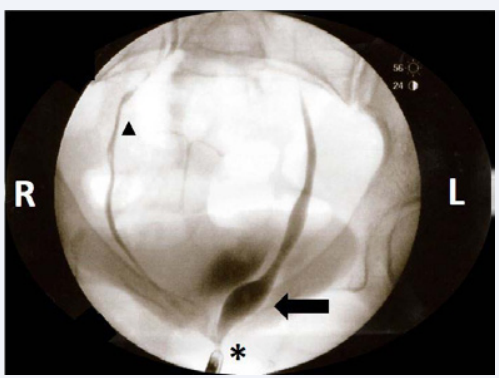
Figure 3 Fluoroscopic imaging of a retrograde cystoscopy details the ureteral orifice of a supernumerary ureter inserting into the proximal portion of the external sphincter muscle of the female urethra (asterisk). The tip of the cystoscope can be seen just inferior and to the left of the asterisk. A significant ureterocele can be observed to be filled with dye (arrow) just proximal to the insertion into the external sphincter muscle of the female urethra. One of the supernumerary ureters for the right collecting system can be observed ascending from the urinary bladder to the right kidney (triangle). The other right ureter was not visualized in this image as the dye from that injection had dissipated.
Our patient had learned to cope with most of her symptoms and only sought treatment when the pain was unbearable or if she developed systemic symptoms, such as the fevers associated wit her chronic pyelonephritis. This behavior is typical of a patient dealing with a life-long illness, but it presents the possibility that while she sought treatment for significant illness, she was not seeking treatment for minor and moderate symptoms. Mild and moderate renal diseases are significant with respect to the health of the kidneys as well as the problems associated with damage to the system, and should be investigated whenever possible. The kidneys have an amazing amount of reserve capacity that allows them toperform their function in the face of constant chemical and mechanical insults. The downside to the fantastic renal reserve is that kidney disease is often not detected until significant damage has already occurred. We were compelled to obtain the full picture of what was troubling our patient as she had almost two decades of chronic insult to her urinary tract. Complete metabolic profiling revealed good renal function, which was encouraging to the patient and our team.