Cellular Component Bands of Bungner
- 1. Department of Surgery, Pontifical Xaverian University, Colombia
- 2. Pontifical Xaverian University, School of Medicine, Colombia
- 3. Research Incubator on Wound Healing and Tissue Regeneration – Pontifical Xaverian University, School of Medicine, Colombia
Abstract
Peripheral nerve injuries are common clinical conditions; thus, understanding their pathophysiology and advances in the field of nerve regeneration are important for the optimal treatment of patients. Regenerative events after injury have become increasingly important. A unique phenotypic expression, derived from already present cells, largely affects this process as a key phenomenon for recovering injured nerve function. Here, the available literature is reviewed to better understand this regenerative event and determine the series of cellular and molecular processes occurring at the axonal level. This review is the product of an investigative exercise by a research group in Research Incubator.
Keywords
Schwann cell , Peripheral nerve regeneration ,Walleriana degeneration, Bands of Büngner, Büngner cell
Citation
Nieto L, Torres S, Rios E, Torres I, Camargo S (2021) Cellular Component Bands of Bungner. JSM Clin Case Rep 9(2): 1187.
ABBREVIATIONS
WD: Wallerian Degeneration; SC: Schwann Cell
INTRODUCTION
Peripheral nerve injuries are common and have many causes, including traumatic, congenital, and metabolic causes. Their detection, appropriate diagnosis and treatment are critical for patients’ recovery [1]. Therefore, understanding their pathophysiology, especially the regenerative events that occur after the moment of injury, is critically important for seeking new treatment options, as this type of injury is catastrophic for the patient both personally and professionally [2]. Much remains to be investigated in this field, especially at the cellular and molecular levels, to understand the complex series of processes that occur during axonal regeneration and functional recovery. This knowledge also benefits improving and accelerating these processes for better and faster recovery. This report reviews the discoveries made to date regarding a key cellular phenomenon during the process of peripheral nerve injury and regeneration [3].
In response to peripheral nerve injury, inflammation begins in the nerve cell body, axons and neuromuscular junctions [4]. This is an exceptional regenerative process, evidenced by chromatolysis at the nuclear level, which indicates a metabolic increase, edema of the axonal stump to prepare for growth and Wallerian degeneration (WD) at the end distal to the lesion. This leads to axonal and myelin disintegration [5], as they prepare to receive the axons that begin growing from the proximal end [6].
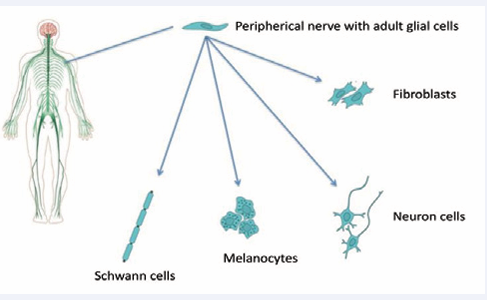
Wallerian Degeneration is usually a transient, acute process in reaction to a lesion, which occurs to regenerate the peripheral nerves. The participation of Schwann cells (SC) (glial cells from the peripheral nervous system) is critical for WD (Figure 1).
Figure 1 Peripheral glial cells are the sources of many neuroglial and mesenchymal cells.
These cells have impressive regenerative properties with a high degree of phenotypic plasticity, triggering a large-scale transformation of the myelin and non-myelin cells from uninjured nerves to repair the Schwann cells of the injured nerves, which is a unique characteristic when promoting repair [7]. This functional recovery contrasts that of the glial cells of the central nervous system, which respond in a way that inhibits repair [8].
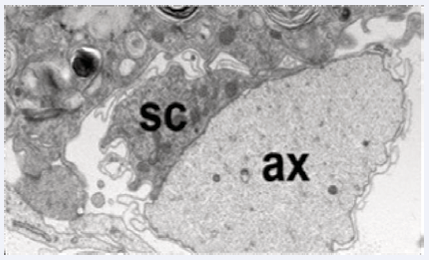
SC genesis, development and differentiation are promoted and depend on molecular signals from the axons (Figure 2).
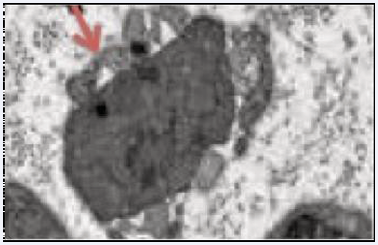
Figure 2 Electron micrograph of a healthy murine nerve showing an intact axon (ax) and its Schwann cell (SC).
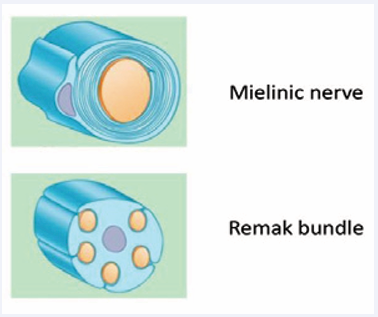
During their life cycle, these cells can change naturally between a differentiated state and an immature or undifferentiated state known as the proliferative phenotype [9]. The differentiated state can be myelinating, which is a one-to-one association between axon and Schwann cell, or non-myelinating, in which multiple small-diameter axons align in the Schwann cell folds, forming Remak bundles [10] (Figure 3).
Figure 3 Transcription factors promote promyelination, in which Schwann cells acquire a one-to-one association with axons (myelinated axons). Other Schwann cells remain unmyelinated by adding multiple axons (Remak bundles).
Both states, and the process of changing between the states, are regulated by intrinsic cellular regulatory factors. Some factors are positive, such as Krox20, Sox10 and Oct6, which promote nerve myelination at the appropriate time during development and reestablish Schwann cell functions after a lesion, while others are negative, such as c-Jun, Sox2, Notch, Krox24 and Pax3, which lead to dedifferentiation and demyelination, thereby blocking the normal induction of myelination by cyclic AMP [11].
SC autophagy, or myelinophagia, during WD involves forming a double membrane for intracellular isolation that envelops a certain cytoplasmic load and transfers it to lysosomes known as autophagosomes for degradation. This autophagy is directed to destroy the myelin contained in the Schwann cells [12]. Thus, the myelin sheath is fragmented into oval segments, and its degeneration continues until it is reduced to intracellular detritus, a process that occurs during the first 5-7 days after injury [13]. The second phase of demyelination occurs by phagocytosis, an extrinsic process that attracts hematogenous macrophages to the injured nerve by Schwann cells, which, along with antibodies and the complement system, degenerate most of the lipids and myelin proteins. Schwann cells also participate in this phase to phagocytize the remaining myelin detritus [14].
Büngner cells
When injury occurs, Schwann cells passively return to a defective or immature cell state by inducing a blockade of factors that maintain their differentiation/myelination [15]. This process is described as double cell dedifferentiation, and along with this process, an alternative cellular repair pathway is activated those changes cellular functions [16]. In other systems, this process is known as transdifferentiation [17], and at the peripheral nervous system level, it results in new cells appearing that specialize in axonal repair (Figure 3), a process that continues to be studied [18].
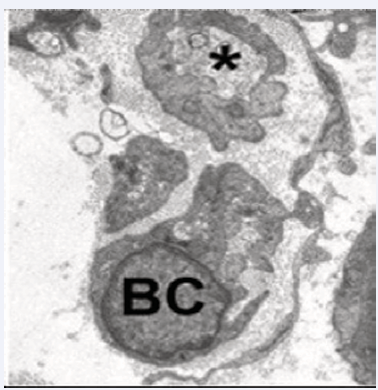
This local axonal process consists of dedifferentiating mature Schwann cells into specialized repair cells called Büngner cells [19] (Figure 4).
Figure 4 Small regenerative axonal buds (asterisk) and adjacent Büngner cells are typical tissue components of an injured peripheral nerve.
This involves an activation state and a new degree of cellular plasticity, representing a new cell type specialized in regenerating peripheral nerves by creating pathways that guide the axon to its target [20] by regeneration tracks called Büngner bands [21] (Figure 5).
Figure 5 Regenerative pathway in the peripheral nerve distal to the lesion, composed of elongated Büngner cells (arrow) or Büngner bands.
These bands carry the nerve fibers to the appropriate destination to restore function. In addition, they favor the survival of the affected peripheral neurons, preserving the axons that would otherwise die as well as opening the bloodbrain barrier to recruit macrophages to the injury site for myelin debridement [22].
Transforming Schwann cells into Büngner cells requires extensive cell reprogramming that depends greatly on demyelination [23,24]. The mTOR pathway is activated in theinjured nerve, which conducts important biosynthetic processes and accelerates catabolism and growth. These functions, along with demyelination, reorganize the morphological and molecular profiles of the myelin and non-myelin cells, converting them into Büngner cells [25].
This Schwann cell plasticity and Büngner cell appearance are regulated by a complex of signaling pathways and transcription factors that are activated at the distal end of the lesion as a response to injury [26].
Signaling pathways
Three signaling pathways, known as MAPK, have been described. First, the JNK pathway involves a serine-threonine kinase that phosphorylates the c-Jun factor, improving its activity and expression. Second, the Ras-MAPK pathway negatively regulates cell differentiation and myelination in Schwann cells [27,28]. Third, the p38 kinase pathway, with promyelinating action, is required to maintain the balance between promyelination and dedifferentiation [29,30] The primary transcription factors involved are c-Jun, Sox2 and the protein kinase Erk1/2, which induces generation of these cells and leads to a high expression of inflammatory cytokines, monocyte chemotactic protein 1 (MCP-1), macrophage inhibitor factor 1 and interleukin 1 (IL-1) [31,32]
c-Jun Factor
The transcription factor c-Jun is an antagonist of the promyelinating transcription factor, Krox-20 (also known as Egr2), and of promyelinic signals, such as elevation of cyclic AMP. c-Jun is upregulated during injury, leading to WD onset, demyelination or myelinophagia, and regulation of important neurotrophins, such as glial-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor, artemin and Sonic-hedgehog (Shh), and cell surface proteins, such as P75NTR, L1, NCAM and N-cadherin, molecules that may mediate Schwann cell and axonal interactions [33-35]
By promoting massive SC dedifferentiation into their multipotent precursors, phenotypic expression of Büngner repair cells and their neurotrophic and environmental support for effective axonal growth and neuronal repair at the proximal end of the lesion can also control the axonal regeneration rate [36,37]. In addition, c-Jun affects the genomic expression of Schwann cells, a function that is restricted during nerve injury and other pathological conditions, such as demyelinating diseases [38].
Sox2 Factor
The transcription factor Sox2 acts synergistically with the c-Jun factor to inhibit myelination and is critical for forming a “nerve bridge” through the lesional defect that will guide the proximal axon growth to the distal end [39]. This is mediated between the pre-formed Büngner cells and nerve fibroblasts, activating the N-cadherin to relocate and the injured nerve roots to collectively migrate [40,41]
DISCUSSION
Understanding the cellular and molecular mechanisms of SC interaction with axons components is essential by controlling axonal growth and regeneration and provides how to understand into the nerve repair and regeneration after different types of injuries [42]. Phenotypes changes during the process of nerve regeneration, are fundamental by optimize the different repair phases. One of those phenotypic modifications of the original SC, is necessary by formed the bands of Büngner, structure necessary by the axonal growth. For this process is relevant the expression of various molecular intracellular signals involved in SC motility and migration. The factor ERK 1/2 is important in mediating migration of SCs, and factor MAPK is a way of facilitating SC motility. It helpful in the expression of specific cytokine o pathways involved in these characteristics in the formation of bands de Bungner and promoting nerve regeneration [43]. Consequently, recued expression of these factors, mainly c-Jun and Sox-2 factors, reduce growth support and Büngner phenotype expression [44,45]. Therefore, the bands of Büngner they are formed by the phenotype change of SC, the Bungner cell, and for this to be fulfilled, it is necessary the expression of different types of factors, mediated by signals from distal stump of injured nerve and the process of wallerian degeneration.
CONCLUSION
SC are the key factors in regenerating peripheral nerves by returning to an undifferentiated state and giving rise to Büngner cells, which specialize in organizing and guiding axonal growth from one nerve stump to another as well as regulate transcription factors and molecular signals [42,45].
The characteristics exhibited by the c-Jun factor allow us to conclude that it acts as a “global regulator” of the repair program that leads to regenerating injured nerves. Understanding how c-Jun and other factors control this cellular repair, how these factors interact with other signals, and how their manipulation could increase the repair process and improve nerve repair has important clinical significance both for peripheral nerve lesions of various etiologies and for diseases that involve these tissues.
REFERENCES
- Kidd GJ, Ohno N, Trapp BD. Biology of Schwann cells. Handb Clin Neurol. 2013; 115: 55-79.
- Carr MJ, Johnston AP. Schwann cells as drivers of tissue repair and regeneration. Curr Opin Neurobiol. 2017; 47: 52-57.
- Monk KR, Feltri ML, Taveggia C. New insights on Schwann cell development. Glia. 2015; 63: 1376-93.
- Jessen KR, Mirsky R. The repair Schwann cell and its function in regenerating nerves. J Physiol. 2016; 594: 3521-31.
- Grinsell D, Keating CP. Peripheral nerve reconstruction after injury: A review of clinical and experimental therapies. Biomed Res Int. 2014; 2014: 698256.
- Gomez-Sanchez JA, Pilch KS, van der Lans M, Fazal SV, Benito C, Wagstaff LJ, et al. After Nerve Injury, Lineage Tracing Shows That Myelin and Remak Schwann Cells Elongate Extensively and Branch to Form Repair Schwann Cells, Which Shorten Radically on Remyelination. J Neurosci. 2017; 37: 9086-9099.
- Evans EB, Brady SW, Tripathi A, Hoffman-Kim D. Schwann cell durotaxis can be guided by physiologically relevant stiffness gradients. Biomater Res. 2018; 22: 14.
- Xiao Y, Faucherre A, Pola-Morell L, Heddleston JM, Liu TL, Chew TL, et al. High-resolution live imaging reveals axon-glia interactions during peripheral nerve injury and repair in zebrafish. Dis Model Mech. 2015; 8: 553-64.
- Namgung U. The role of Schwann cell-axon interaction in peripheral nerve regeneration. Cells Tissues Organs. 2014; 200: 6-12.
- Miyamoto Y, Torii T, Kawahara K, Inoue M, Morimoto T, Yamamoto M, et al. Data on the effect of in vivo knockdown using artificial ErbB3 miRNA on Remak bundle structure. Data Brief. 2017; 12: 313-319.
- Feltri ML, Poitelon Y, Previtali SC. How Schwann Cells Sort Axons: New Concepts. Neuroscientist. 2016; 22: 252-65.
- Chen P, Piao X, Bonaldo P. Role of macrophages in Wallerian degeneration and axonal regeneration after peripheral nerve injury. Acta Neuropathol. 2015; 130: 605-18.
- Jessen KR, Mirsky R. The Success and Failure of the Schwann Cell Response to Nerve Injury. Front Cell Neurosci. 2019; 13: 33.
- Gomez-Sanchez JA, Carty L, Iruarrizaga-Lejarreta M, Marta Palomo- Irigoyen, Marta Varela-Rey, Megan Griffith, et al. Schwann cell autophagy, myelinophagy, initiates myelin clearance from injured nerves. J Cell Biol. 2015; 210: 153–168.
- Jessen KR, Mirsky R. The repair Schwann cell and its function in regenerating nerves. J Physiol. 2016; 594: 3521-31.
- López-Leal R, Díaz-Viraqué F, Catalán RJ, Saquel C, Enright A, Iraola G, et al. Schwann cell reprogramming into repair cells increases miRNA-21 expression in exosomes promoting axonal growth. J Cell Sci. 2020; 133: jcs239004.
- Jessen K, Mirsky R. The Schwann cell lineage: Cellular transitions during development and after injury. In: Kettenmann H, Ransom B, eds. Neuroglia. Oxford: Oxford University Press; 2012: 159–171.
- Bolívar S, Navarro X, Udina E. Schwann Cell Role in Selectivity of Nerve Regeneration. Cells. 2020; 9: 2131.
- Nocera G, Jacob C. Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol Life Sci. 2020; 77: 3977- 3989.
- Kaucká M, Adameyko I. Non-canonical functions of the peripheral nerve. Exp Cell Res. 2014; 321: 17–24.
- Torii T, Miyamoto Y, Yamauchi J. Cellular Signal-Regulated Schwann Cell Myelination and Remyelination. Adv Exp Med Biol. 2019; 1190: 3-22.
- Monje PV. The properties of human Schwann cells: Lessons from in vitro culture and transplantation studies. Glia. 2020; 68: 797-810.
- Weiss T, Taschner-Mandl S, Bileck A, Slany A, Kromp F, Rifatbegovic F, et al. Proteomics and transcriptomics of peripheral nerve tissue and cells unravel new aspects of the human Schwann cell repair phenotype. Glia. 2016; 64: 2133-2153.
- Salzer JL. Axonal regulation of Schwann cell ensheathment and myelination. J Peripher Nerv Syst. 2012; 17: 14-9.
- Benito C, Davis CM, Gomez-Sanchez JA, Turmaine M, Meijer D, Poli V, et al. STAT3 Controls the Long-Term Survival and Phenotype of Repair Schwann Cells during Nerve Regeneration. J Neurosci. 2017; 37: 4255- 4269.
- Jessen KR, Mirsky R, Lloyd AC. Schwann Cells: Development and Role in Nerve Repair. Cold Spring Harb Perspect Biol. 2015; 7: a020487.
- Qian T, Wang X, Wang Y, Wang P, Liu Q, Liu J, et al. Novel miR-sc4 regulates the proliferation and migration of Schwann cells by targeting Cdk5r1. Mol Cell Biochem. 2018; 447: 209-215.
- Monje PV, Sant D, Wang G. Phenotypic and Functional Characteristics of Human Schwann Cells as Revealed by Cell-Based Assays and RNA- SEQ. Mol Neurobiol. 2018; 55: 6637-6660.
- Salzer JL. Switching myelination on and off. J Cell Biol. 2008; 181: 575–577.
- Li J, Zhang Z, Wang J, Du S, Yao D, Cao R, Cui S. Protein Kinase Cα Promotes Proliferation and Migration of Schwann Cells by Activating ERK Signaling Pathway. Neuroscience. 2020; 433: 94-107.
- Martini R, Klein D, Groh J. Similarities between inherited demyelinating neuropathies and Wallerian degeneration: An old repair program may cause myelin and axon perturbation under nonlesion conditions. Am J Pathol. 2013; 183: 655–660.
- Yu H, Zhu L, Li C, Sha D, Pan H, Wang N, Ma S. ERK1/2 and AKT are vital factors in regulation of the migration of rat Schwann cells. J Vet Med Sci. 2015; 77: 427-32.
- Hantke J, Carty L, Wagstaff LJ, Mark Turmaine, Daniel K Wilton, Susanne Quintes, et al. c-Jun activation in Schwann cells protects against loss of sensory axons in inherited neuropathy. Brain. 2014; 137: 2922–2937.
- Figlia G. c-Jun in Schwann Cells: Stay Away from Extremes. J Neurosci. 2018; 38: 3388-3390.
- Blom CL, Mårtensson LB, Dahlin LB. Nerve injury-induced c-Jun activation in Schwann cells is JNK independent. Biomed Res Int. 2014; 2014: 392971.
- Allodi I, Udina E, Navarro X. Specificity of peripheral nerve regeneration: interactions at the axon level. Prog Neurobiol. 2012; 98: 16-37.
- Huang L, Quan X, Liu Z, Ma T, Wu Y, Ge J, et al. c-Jun gene-modified Schwann cells: upregulating multiple neurotrophic factors and promoting neurite outgrowth. Tissue Eng Part A. 2015; 21: 1409-21.
- Arthur-Farraj PJ, Latouche M, Wilton DK, Quintes S, Chabrol E, Banerjee A, et al. c-Jun reprograms Schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron. 2012; 75: 633-47.
- Roberts SL, Dun XP, Doddrell RDS, Mindos T, Drake LK, Onaitis MW, et al. Sox2 expression in Schwann cells inhibits myelination in vivo and induces influx of macrophages to the nerve. Development. 2017; 144: 3114-3125.
- Kim HA, Mindos T, Parkinson DB. Plastic fantastic: Schwann cells and repair of the peripheral nervous system. Stem Cells Transl Med. 2013; 2: 553–557
- Arter J, Wegner M. Transcription factors Sox10 and Sox2 functionally interact with positive transcription elongation factor b in Schwann cells. J Neurochem. 2015; 132: 384-93.
- Jessen KR, Arthur-Farraj P. Repair Schwann cell update: Adaptive reprogramming, EMT, and stemness in regenerating nerves. Glia. 2019; 67: 421-437.
- Roberts SA, Lloyd AC. Aspects of cell growth control illustrated by the Schwann cell. Curr Opin Cell Biol. 2012; 24: 852-7.
- Torres-Mejía E, Trümbach D, Kleeberger C, Dornseifer U, Orschmann T, Bäcker T, et al. Sox2 controls Schwann cell self-organization through fibronectin fibrillogenesis. Sci Rep. 2020; 10: 1984.
- ?tef?nescu O, Enescu D, Lasc?r I. Schwann cell cultures: Recent advances and novel approaches to the reconstruction of peripheral nerve defects. Rom J Morphol Embryol. 2012; 53: 467–471.