Poorly Cohesive Carcinoma within Hyperplastic Polyp of the Stomach
- 1. Institute of Geriatric Surgery, Università Campus Bio-Medico, Italy
- 2. Institute of Pathology, Università Campus Bio-Medico di Roma, Italy
Abstract
A 75-year-old woman with a signet-ring cell carcinoma in a gastric hyperplastic polyp is reported. Endoscopic biopsies of the gastric lesion revealed glandular foveolar hyperplasia and ulceration, with focal severe cytological atypias in sparse glands. Total gastrectomy showed the presence of a polyp histologically characterized by hyperplastic glands and the presence of signet-ring cell carcinoma within the stroma. Cancer was limited to the polyp lesion. Extensive sampling of other regions of the stomach was negative for neoplasia. From a review of the literature, only 4 cases of signet ring carcinoma within a gastric hyperplastic polyp were reported. Our case points the focus on the relevance of extensive endoscopic sampling of hyperplastic polyps in order to rule out underlying neoplasia.
Keywords
• Gastric polyps
• Gastric carcinoma
Citation
Capolupo GT, Taffon C, Mascianà G, Carannante F, Crescenzi A, et al. (2017) Poorly Cohesive Carcinoma within Hyperplastic Polyp of the Stomach. JSM Med Case Rep 2(1): 1004.
INTRODUCTION
Gastric polyps account for 2% to 3% of gastric examinations, usually as incidental findings [1]. Hyperplastic polyps typically arise in stomach in a background of chronic gastritis and are the second most common gastric polyps [2]. Large polyps can have superficial ulceration or hemorrhage. Hyperplastic polyps usually develop within the body or antrum. It is believed that they represent an exuberant regenerative response of the gastric foveolar cells [3]. Hyperplastic polyps rarely progress to gastric carcinomas; malignant transformation however, although rare is well documented [4].
CASE PRESENTATION
A 75-year-old woman with a recent cardiac ischemia was admitted in our hospital for a cardiologic check-up; clinical and laboratory examination detected a severe normocytic normochromic anemia (Hb 5.6 g/dl). The patient reported recent hematochezia. A colonoscopy and an upper endoscopy were performed, the latter showing a bleeding protruding lesion in the gastric fundus, measuring about cm 2,5 in diameter. The lesion appeared soft, partly ulcerated with fibrin deposition consistent with bleeding (Figure 1)
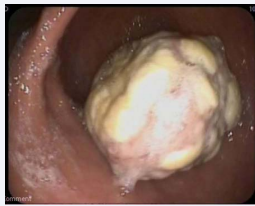
Figure 1: Endoscopic image of gastric polyp.
Biopsies of the lesion were submitted to the department of Pathology for histologic evaluation. Colonoscopy didn’t show relevant alterations.
Histological examination of the gastric biopsies showed hyperplastic polyp mucosae with heavy inflammatory infiltrate. Foveolar hyperplastic pits showed isolated foci of high grade epithelial dysplasia characterized by globet cells with depolarized and hyperchromatic nuclei within the basal membrane, there wasn’t evidence of chronic gastritis or H.pylori infection.
Although there was no evidence of infiltrative cancer, considering the severe anemia, the hemodynamic instability, the endoscopic appearance and size of the lesion, the patient was submitted, prior informed consent, to total gastrectomy.
Before the gastrectomy procedure, the patient was submitted to CT scan resulted negative for other pathologies or distant metastasis.
After surgical procedure the patient was discharged on the 10th post-operative day in good general clinical condition.
Definitive histology was performed on the surgical specimen. The stomach showed a 2,5 cm in diameter reddish protruding lesion in the fundus, at 1 cm from the proximal resection margin. The lesion was totally sampled according to the Japanese rules for gastric cancer [4] embedded in paraffin. 3 µm thick sections were cut for standard histology and immunohistochemical studies.
Histologically, the polypoid lesion was characterized by a proliferation of the surface foveolar cells lining exaggerated, elongated, and distorted pits that extend from the surface deep into the lamina propria with mucosal expansion. The glands developed extensive infoldings, branching in bizarre ways producing either a serrated appearance or dilated cysts, occasionally quite prominent. Smooth muscle fibers arborized in the lamina propria. Mucosa appeared edematous, infiltrated by single atypical discohesive epithelial elements, and characterized irregular and pleomorphic nuclei, eccentrically placed, with evident nucleolus, and ample eosinophilic cytoplasm and sporadically with classical signet-ring morphology. Plasma cells, lymphocytes and eosinophils were also present (Figure 4)
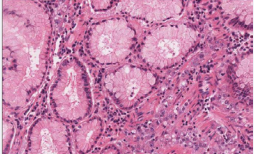
Figure 4: Hematoxylin-eosine staining.
Neutrophils were especially prominent in surface ulcerated areas; vascular proliferations resembling granulation tissue developed superficially near areas of inflammation. Within the foveolar hyperplastic lining there were areas of dysplastic changes as seen in the bioptic samples, with low and high grade intraepithelial neoplasia with features of signet ring cell (Figure 2-4)
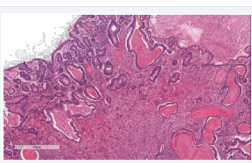
Figure 2: Hematoxylin-eosine staining.
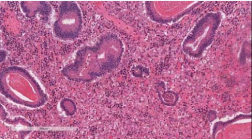
Figure 3: Hematoxylin-eosine staining
Immunohistochemistry was performed by the streptavidinbiotin method. The antibodies used were mouse monoclonal antibodies against Pan-cytokeratins (clone MNF116) and CD68 (clone PG-M1) all from Dakocytomation, Denmark. Sections were treated with LSAB2 (Dakocytomation) and reaction product was revealed by. 3-3-Diaminobenzidine (DAB). Sections were counterstained with haematoxylin.
Discohesive atypical epithelial elements resulted positive for Pan-CK and negative for CD68 at the immunohistochemical analysis. (Figure 5,6)
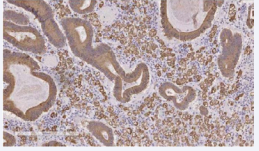
Figure 5: Immunohistochemistry study.
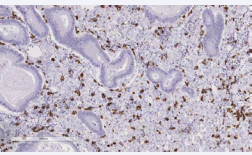
Figure 6: Immunohistochemistry study.
Diagnosis of gastric carcinoma, poorly cohesive type, arising in hyperplastic polyp was made.
Extensive sampling of other regions of the stomach showed atrophic, follicular, active and micro-erosive chronic gastritis of severe grade, associated to extensive type I and II intestinal metaplasia.
DISCUSSION
Hyperplastic polyps are a quite common form of gastric polyps [5]. They develop around gastric remnants, ulcers, or gastroenterostomy stomas. It is believed that they represent an exuberant regenerative response of the gastric foveolar cells. Rare polyps are large and simulate carcinoma. Most hyperplastic polyps arise on a background of chronic gastritis. Atypia is either absent or minimal and if present is of regenerative in nature. Neutrophils are especially prominent in ulcerated areas. Vascular proliferations resembling granulation tissue develop superficially near areas of inflammation. These reparative changes may resemble low-grade dysplasia or adenomas.
Hyperplastic polyps rarely progress to gastric carcinomas with an incidence of no more than 2% [6]. It is generally acknowledged that hyperplastic polyps do not transform in carcinoma, although they may occasionally be associated with gastric cancer [7-9]. The appearing of adenomatous and dysplastic foci, followed by the cancerous lesion is considered to be related to the size and macroscopic type of the hyperplastic polyp [7,8,10-16].
Anyways, an association of poorly differentiated adenocarcinoma or signet ring carcinoma with hyperplastic polyp is reported, with particular regard to familiar form.
From a review of the literature, only 4 cases of signet ring carcinoma arising within a gastric hyperplastic polyp were reported [12,13,15,16], one of them describing a case of multifocal signet-ring carcinoma in an inverted hyperplastic polyp [12].
Our case report a poorly cohesive gastric carcinoma with combined signet ring cells and pleomorphic eosinophilic cells, raised in a gastric hyperplastic polyp and associated with foci of foveolar high grade dysplasia in the surface epithelium. The latter was the only worrying feature observed at the time of endoscopic sampling of the lesion. Accuracy in endoscopic sampling and histological examination are critical points to reveal pre-neoplastic changes and allow do not miss underling invasive carcinoma.
This case emphasizes the importance for endoscopists to obtain extensive bioptic sampling from gastric polyps, especially if associated with chronic gastritis, in order to allow an adequate histological examination. Reactive hyperplasia need to be accurately screened to rule out foveolar dysplasia. The microscopic evidence of foveolar dysplasia (type2 gastritis associated dysplasia) strongly suggests a close observation of the patient being the finding a precursor lesion of invasive neoplasia.
ACKNOWLEDGEMENTS
Dr. G. Capolupo - Dr. G. Masciana – Dr. F.Carannante: Patient care and managment Dr. C.Taffon: Image contribution.
Prof. M. Caricato Dr. A. Crescenzi: revision and final approval of the manuscript This work received no specific funding.








































































































































