The Impact of Combined Spinal Epidural Analgesia (CSEA) on Postpartum Depression: A Mendelian Randomization Study
- 1. Department of Anesthesiology, Chengdu Second People’s Hospital, China
Abstract
Background: Postpartum Depression (PPD) develops within the first few weeks or months following delivery and causes severe emotional and psychological problems. Pain has been closely linked to the occurrence of depression. Observational studies have suggested that effective pain relief during childbirth can reduce the incidence of PPD. However, these studies are fraught with numerous confounding factor. Combined Spinal-Epidural Analgesia (CSEA) is a commonly used pain relief method during childbirth. It is currently unclear whether a causal relationship exists between CSEA and PPD.
Methods: An analysis was conducted using five methods in Mendelian randomization (MR) to study the use of CSEA during childbirth and PPD. The data were obtained from the United Kingdom Biobank Database (UKB) for CSEA and from FinnGen for PPD. The analysis included MR Egger, weighted median, Inverse Variance Weighted (IVW), simple mode, and weighted mode. We then conducted exposure heterogeneity testing using Cochran’s Q statistics and assessed the pleiotropy of exposure Single Nucleotide Polymorphisms (SNPs) using MR-Egger.
Results: IVW Odds Ratio (OR): 0.978, 95% confidence intervals (95%CI): 1.031–0.407, P: 0.408. The results of the weighted median (OR: 1.035, 95% CI: 0.995, 1.118, P: 0.377), simple mode (OR: 0.929, 95% CI: 0.773, 1.116, P: 0.435), and weighted mode (OR: 0.995, 95% CI: 0.930, 1.065, P: 0.888) suggest that there is no significant link between CSEA and PPD.
Conclusion: We have concluded that there is no causal link between CSEA and PPD. This information can assist clinical professionals in gaining a better understanding of this condition.
Keywords
• Combined Spinal-Epidural Analgesia; Postpartum Depression; Mendelian Randomization
Citation
Zhang M, Liu S (2025) The Impact of Combined Spinal-Epidural Analgesia (CSEA) on Postpartum Depression: A Mendelian Randomization Study. JSM Pain Manag 3(1): 1009.
ABBREVIATIONS
CSEA: Combined Spinal-Epidural Analgesia; PPD: Postpartum Depression; UKB: Kingdom Biobank database; SNPs: Single Nucleotide Polymorphisms; OR: Odds Ratio; 95% CI: 95% Confidence Intervals; IVW: Inverse Variance weighted; MR: Mendelian randomization; GWAS: Genome-Wide Association Study; RCTs: Randomized Controlled Trials; IVs: Instrumental Variables; LD: Linkage Disequilibrium; EAF: Effect Allele Frequency
INTRODUCTION
Approximately 140 million babies are born globally each year, making the safety and health of pregnant women during the perinatal period a significant public health concern [1]. Postpartum Depression (PPD) refers to severe depressive symptoms or a typical depressive episode occurring at any time during the first year after childbirth in women [2]. The global incidence rate of PPD is between 15% and 25% [3]. Typical postpartum depression occurs within 6 weeks after childbirth and can resolve spontaneously within 3 to 6 months. However, severe cases can persist for 1 to 2 years, and there is a recurrence rate of 20% to 30% with subsequent pregnancies. Typical postpartum depression occurs within 6 weeks after childbirth and can resolve spontaneously within 3 to 6 months [2]. However, severe cases can persist for 1 to 2 years, and there is a recurrence rate of 20% to 30% with subsequent pregnancies. Its clinical features include low mood, anxiety, fatigue, insomnia, changes in appetite, decreased interest in the baby, and maternal role insecurity, among others [4,5]. Previous research has indicated that PPD can have significant effects on both the mother and the offspring, such as lower cognitive function in the offspring and a higher incidence of insecure attachment [5]. Therefore, understanding the potential factors leading to PPD is crucial.There are many potential factors contributing to the occurrence of PPD: psychological environmental factors (such as economic circumstances, and marital status) and biological factors (such as hormones, inflammation,
genetics, and pain) have been shown to be closely associated with the development of PPD [6-8]. It is well known that pain plays a significant role in the onset and progression of depression [9]. The childbirth process involves intense pain, which can have many adverse effects on both the mother and the fetus, including chronic pain, postpartum stress syndrome, and negative psychological and physiological consequences [10-12]. Some studies have suggested that women who experience significant pain during pregnancy, such as back or pelvic pain, may be at a higher risk of developing PPD [13-15]. Chronic pain during pregnancy can contribute to increased stress and discomfort, which in turn can affect a woman’s mental health [9].
Combined spinal-epidural analgesia involves inserting a spinal needle through the dura mater into the subarachnoid space to rapidly and effectively administer local anesthetics through a single injection, providing pain relief [16]. Additionally, an epidural catheter is placed in the epidural space to maintain analgesic effects throughout the labor process [16,17]. Local anesthetics work by blocking sodium ion channels in nerve cell membranes to inhibit the transmission of sensory impulses, thereby producing analgesic effects [18]. Commonly used local anesthetics include bupivacaine, lidocaine, and ropivacaine [18,19].
CSEA offers unique advantages in labor analgesia, including the ability to maintain consciousness during the procedure, and avoiding the risks associated with general anesthesia, such as significant impacts on respiratory and circulatory functions [18]. CSEA rarely leads to a drop in blood pressure. Compared to epidural anesthesia, CSEA can reduce the dosage of drugs used and their impact on motor function, but it carries a higher probability of side effects such as itching, nausea, vomiting, drowsiness, and headaches [20].
Previous research has shown that CSEA can reduce the duration of labor [20]. CSEA is the most commonly used method of anesthesia for maternal labor pain relief, but its association with the occurrence of PPD is currently not clear. Although many researchers have studied the relationship between labor analgesia and postpartum depression, the results of these studies are contradictory. Some observational studies and multicenter, prospective, longitudinal studies suggest that neuraxial labor analgesia can reduce the incidence of PPD; however, other studies have failed to demonstrate this association [21-24].
Mendelian Randomization (MR) is a method that uses measured genetic variations to determine the causal relationship between an exposure factor and an outcome [25]. Therefore, it can effectively avoid the influence of potential confounding factors when investigating the association between combined spinal-epidural analgesia and postpartum depression. We utilized Genome-Wide Association Study (GWAS) summary statistics for both spinal-epidural analgesia and postpartum depression at the genetic level to analyze their causal relationship, making the results more reliable compared to observational studies and Randomized Controlled Trials (RCTs).
METHODS
Overall Study Design
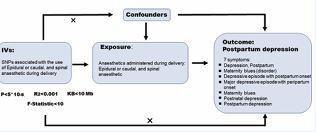
MR is a data analysis technique used for assessing causal inference in epidemiology. It evaluates the causal relationship between the exposure of interest and the outcome of concern by employing genetic variations as Instrumental Variables (IVs). Genetic loci are determined at the moment of conception and remain unaffected by postnatal factors. This determinism is the primary reason MR utilizes genetic loci as analytical tools. Selecting appropriate instrumental variables requires satisfying three conditions: First, instrumental variables must be independent of other confounding factors influencing the outcome variable. Second, instrumental variables should have a close association with the exposure factor. Third, there should be no correlation between instrumental variables and the outcome variable. To identify instrumental variables meeting these criteria, we established criteria based on the values of P, R2, and F-statistics. Since fewer SNPs were retained after applying the condition of P < 5*10-8, we modified the P-value requirement to P < 5*10-6. Subsequently, SNP independence settings were implemented. We performed Clump on all SNPs with the conditions set at Linkage Disequilibrium (LD) R2 < 0.001 and KB < 10 Mb. Finally, SNP statistics strength settings were applied, requiring F-statistics > 10 for all SNPs.
We utilized data from the UK Biobank, which is currently one of the most renowned and open biobanks worldwide, specifically data related to “Anesthetics administered during delivery” (Data-Field 41219). This dataset collected information from 16,657 women who underwent childbirth between June 2013 and March 2023. Among them, we used the GWAS summary statistics data of 320 women who received combined spinal-epidural analgesia as our exposure factor SNPs (Phenotype Code: 41219_6). Additionally, we obtained postpartum depression GWAS data (EFO_0007453) from the FinnGen database, which includes data from 226,707 participants, comprising 11,711 cases and 214,996 controls.
Statistical analysis
We conducted a merge of the selected SNPs related to women who received combined spinal-epidural analgesia during childbirth and SNPs associated with postpartum depression (Supplementary Material 1). Within the postpartum depression SNPs, we filtered out those related to combined spinal-epidural analgesia. Subsequently, a harmonization process was carried out to align the allele directions of exposure SNPs and outcome SNPs. Based on EAF (effect allele frequency) size, we excluded palindromic SNPs that could not determine direction and incompatible SNPs. In the end, we identified a total of 76 relevant SNPs (Supplementary Material 2). Following this, we proceeded with MR estimation. MR estimation involved five methods: MR Egger, weighted median, inverse variance weighted, simple mode, and weighted mode. IVW (Inverse Variance weighted) was the primary analysis method. We then conducted exposure heterogeneity testing using Cochran’s Q statistics and assessed the pleiotropy of exposure SNPs using MR-Egger. If no heterogeneity was observed, we employed a fixed-effects model; if heterogeneity was present, a random-effects model was utilized. The effects of combined spinal-epidural analgesia on postpartum depression are presented as Odds Ratios (ORs) with corresponding 95% confidence intervals (95% CIs). For the entire MR analysis, we used R 4.3.1 software, along with the integrated TwoSampleMR package.
RESULTS
Postpartum depression encompasses seven types of symptoms: 1. depression, postpartum 2. maternity blues (disorder) 3. depressive episode with postpartum onset 4. major depressive episode with peripartum onset 5. maternity blues 6. postnatal depression 7. Postpartum depression. Through MR analysis, it was found that there is no causal relationship between the use of combined spinal-epidural analgesia for pain relief during childbirth and postpartum depression (Table 1, Figure 1)..
Figure 1 Overview Diagram of Mendelian Randomization Analyses. Genetic data on combined spinal–epidural analgesia and postpartum depression
Table 1: Summary of Results for Five Methods in MR Analysis.
|
Method |
Used of SNPs |
OR |
95%CI |
Pval |
|
MR Egger |
76 |
0.986 |
(0.921,1056) |
0.693 |
|
Weighted Median |
76 |
1.035 |
(0.959,1.118) |
0.377 |
|
Inverse Variance weighted |
76 |
0.978 |
(1.031,0.407) |
0.408 |
|
Simple Mode |
76 |
0.929 |
(0.773,1.116) |
0.435 |
|
Weighted Mode |
76 |
0.995 |
(0.930,1.065) |
0.888 |
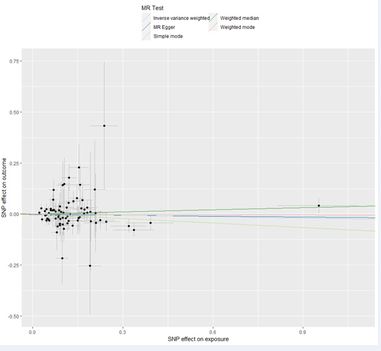
The IVW method is the primary approach for predicting the impact of combined spinal-epidural analgesia on postpartum depression. The results showed a P-value of 0.408 for IVW, with an OR value of 0.978 and a 95% confidence interval (95%CI) of 1.031 to 0.407. Therefore, this suggests that CSE does not influence the occurrence or development of postpartum depression. Similarly, the weighted median (OR: 1.035, 95% CI: 0.995, 1.118, P: 0.377), simple mode (OR: 0.929, 95% CI: 0.773, 1.116, P: 0.435), and weighted mode (OR: 0.995, 95% CI: 0.930, 1.065, P: 0.888) all indicated that there was no statistically significant association between CSEA and PPD (Table 1, Figure.2).
Figure 2 Scatter plots of MR analyses.Genetic data on combined spinal– epidural analgesia and postpartum depression.
Heterogeneity Analysis
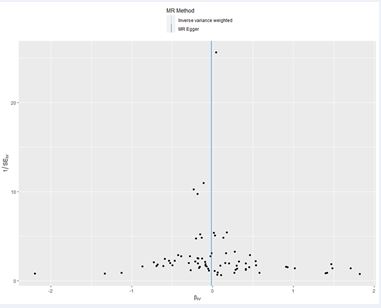
We conducted an examination of horizontal pleiotropy related to SNPs associated with CSEA using the Cochran Q statistics method. The statistical analysis resulted in a p-value of 0.454, indicating the absence of heterogeneity (Figure 3).
Figure 3 Funnel plot for examining heterogeneity of SNPs associated with CSEA.
Horizontal Pleiotropy Analysis:
We examined horizontal pleiotropy related to SNPs associated with CSEA using the MR-Egger method. The calculated p value was 0.691. Therefore, in the study aiming to predict the causal relationship between SNPs related to CSEA during childbirth and PPD, there was no influence from confounding factors (Figure 2).
DISCUSSION
In this study, we conducted a Mendelian randomization analysis to examine the causal relationship between the use of effective epidural analgesia during childbirth at the genetic level and the occurrence and development of postpartum depression. The conclusion drawn from our analysis is that there is no statistically significant correlation between them. This finding differs from the results of many previous observational studies. By studying the impact of CSEA on PPD at the genetic level, we eliminated interference from various other confounding factors, providing a more direct indication of the causal relationship between CSEA and PPD.
Previous studies have indicated that there are various potential causes for the occurrence of PPD. These include rapid hormonal changes following childbirth, genetic factors,and social factors such as economic factors and marital/family relationships [26-29].
Pain has been confirmed as an independent risk factor for the onset of depression. The pain experienced during childbirth is intense, and previous studies have analyzed the correlation between effective pain relief methods during childbirth and the occurrence of postpartum depression. The conclusions drawn by various research teams have also been inconsistent.
Jianlan Mo’s team conducted a meta-analysis of 19 observational studies involving 8,758 parturients. The results showed that perinatal pain can increase the risk of PPD [OR = 1.43, 95% CI (1.23, 1.67), P < 0.05], but the use of epidural analgesia during childbirth can effectively reduce the occurrence of PPD [OR = 0.42, 95% CI (0.33, 0.55), P< 0.05] [21]. The results of a prospective cohort study showed that the incidence of PPD among parturients who received epidural analgesia during childbirth was significantly lower than that among those who underwent natural childbirth [30]. However, there was also an RCT involving 130 parturients that found no statistically significant difference in the probability of developing PPD between those who received epidural analgesia and those who did not six weeks after childbirth (27.7% vs. 16.9%; Fisher’s exact P = 0.103) [31]. Jian Wang’s team also demonstrated that neuraxial labor analgesia does not reduce the risk of PPD occurrence (OR = 0.84, 95% CI: 0.58 1.23) [32]. Riazanova’s team explained the connection between effective labor analgesia and PPD occurrence at the physiological level. They assessed the levels of stress hormone cortisol in the perinatal serum. The results showed that in parturients who received epidural anesthesia during childbirth, both the early postpartum pain intensity and cortisol levels were reduced. However, the impact on the probability of developing PPD six weeks postpartum was minimal [33].
Due to ethical concerns, there have been few randomized controlled experiments exploring the relationship between labor analgesia and postpartum depression to date. Observational research methods have been the primary approach in studying the connection between pain and PPD, but their reliability is questionable due to the presence of confounding variables, high heterogeneity, and limitations such as misclassification errors in the analysis process.This study overcomes the limitations of previous research through MR analysis. Starting from a genetic level, it provides a better explanation of the causal relationship between labor analgesia and PPD. The research results indicate that effective labor analgesia does not impact the occurrence and development of PPD. The occurrence of PPD may be associated with other factors. Certainly, this study has its own limitations. First, the GWAS data used in this study from parturients who received combined spinal-epidural analgesia cover a wide range, and many details about the anesthesia, such as the type and amount of local anesthetics used during CSEA, are not reflected in the GWAS. Second, because the data in this study were collected from populations in European and American countries, there may be biases in the results when applied to parturients of different ethnicities and in other countries. For example, previous studies have reported that Japanese parturients who received analgesia during vaginal delivery had a higher probability of developing PPD six months postpartum than those who did not receive analgesia during delivery (OR: 1.233, 95% CI: 1.079–1.409) [34]. The authors explained that this might be due to a maternal myth in Japanese culture, which believes that pain relief during childbirth helps to form strong maternal instincts. This belief leads to fewer Japanese parturients using analgesia during childbirth, and as a result, those who do use it may experience social and familial psychological pressures later on, making them more susceptible to PPD [34].
The study conducted an MR analysis to explore the relationship between CSEA and PPD. This research has shed light on the potential causes of PPD, which can aid clinical anesthesiologists, obstetricians, and psychiatrists in better understanding the condition. Additionally, this study opens the door for further investigation into highly correlated factors that contribute to PPD, ultimately helping to prevent it.
Availability of Data and Materials
Data related to Anesthetics administered during delivery (Data-Field 41219) from UK Biobank. Data related to postpartum depression (EFO_0007453) from the FinnGen database.
Author Statement
MY Z created, prepared and wrote the initial draft of the article. SX L contributed in the supervision and mentorship of the lead author. All authors read and approved the final manuscript.
Funding
This work was supported by the Sichuan Medical Association’s Research in the fields of oncology, anesthesia Program (Hengrui) [SC202402].
REFERENCES
- Blencowe H, Cousens DipMathStat S, Mikkel Z Oestergaard, Doris Chou, Ann-Beth Moller, Rajesh Narwal, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012; 379: 2162-2172.
- O’Hara MW, McCabe JE. Postpartum depression: current status and future directions. Annu Rev Clin Psychol. 2013; 9: 379-407.
- Gelaye B, Rondon MB, Araya R, Williams MA. Epidemiology of maternal depression, risk factors, and child outcomes in low-income and middle-income countries. Lancet Psychiatry. 2016; 3: 973-982.
- Beck CT. The effects of postpartum depression on maternal-infantinteraction: a meta-analysis. Nurs Res. 1995; 44: 298-304.
- Murray L. The impact of postnatal depression on infant development.J Child Psychol Psychiatry. 1992; 33: 543-561.
- Yeaton-Massey A, Herrero T. Recognizing maternal mental health disorders: beyond postpartum depression. Curr Opin Obstet Gynecol. 2019; 31: 116-119.
- Pearlstein T, Howard M, Salisbury A, Zlotnick C. Postpartumdepression. Am J Obstet Gynecol. 2009; 200: 357-364.
- Yim IS, Tanner Stapleton LR, Guardino CM, Hahn-Holbrook J, Dunkel Schetter C. Biological and psychosocial predictors of postpartum depression: systematic review and call for integration. Annu Rev Clin Psychol. 2015; 11: 99-137.
- Bair MJ, Robinson RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003; 163: 2433- 2445.
- Sng BL, Sia ATH. Maintenance of epidural labour analgesia: The old, the new and the future. Best Pract Res Clin Anaesthesiol. 2017; 31: 15-22.
- Hawkins JL. Epidural analgesia for labor and delivery. N Engl J Med.2010; 362: 1503-1510.
- Jones L, Othman M, Dowswell T, Alfirevic Z, Gates S, Newburn M, et al. Pain management for women in labour: an overview of systematic reviews. Cochrane Database Syst Rev. 2012; 2012: CD009234.
- Lim G, LaSorda KR, Farrell LM, McCarthy AM, Facco F, Wasan AD. Obstetric pain correlates with postpartum depression symptoms: a pilot prospective observational study. BMC Pregnancy Childbirth. 2020; 20: 240.
- Lim G, Farrell LM, Facco FL, Gold MS, Wasan AD. Labor Analgesia as a Predictor for Reduced Postpartum Depression Scores: A Retrospective Observational Study. Anesth Analg. 2018; 126: 1598-1605.
- Eisenach JC, Pan PH, Smiley R, Lavand’homme P, Landau R, HouleTT. Severity of acute pain after childbirth, but not type of delivery, predicts persistent pain and postpartum depression. Pain. 2008; 140: 87-94.
- Roofthooft E, Rawal N, Van de Velde M. Current status of the combined spinal-epidural technique in obstetrics and surgery. Best Pract Res Clin Anaesthesiol. 2023; 37: 189-198.
- Simmons SW, Taghizadeh N, Dennis AT, Hughes D, Cyna AM. Combined spinal-epidural versus epidural analgesia in labour. Cochrane Database Syst Rev. 2012; 10: CD003401.
- Sng BL, Kwok SC, Sia AT. Modern neuraxial labour analgesia. CurrOpin Anaesthesiol. 2015; 28: 285-289.
- Macfarlane AJR, Gitman M, Bornstein KJ, El-Boghdadly K, Weinberg G. Updates in our understanding of local anaesthetic systemic toxicity: a narrative review. Anaesthesia. 2021; 76: 27-39.
- Pascual-Ramirez J, Haya J, Pérez-López FR, Gil-Trujillo S, Garrido- Esteban RA, Bernal G. Effect of combined spinal-epidural analgesia versus epidural analgesia on labor and delivery duration. Int J Gynaecol Obstet. 2011; 114: 246-250.
- Mo J, Ning Z, Wang X, Lv F, Feng J, Pan L. Association between perinatal pain and postpartum depression: A systematic review and meta-analysis. J Affect Disord. 2022; 312: 92-99.
- Liu ZH, He ST, Deng CM, Ding T, Xu MJ, Wang L, et al. Neuraxial labour analgesia is associated with a reduced risk of maternal depression at 2 years after childbirth: A multicentre, prospective, longitudinal study. Eur J Anaesthesiol. 2019; 36: 745-754.
- Kountanis JA, Vahabzadeh C, Bauer S, Muzik M, Cassidy R, Aman C, etal. Labor epidural analgesia and the risk of postpartum depression: A meta-analysis of observational studies. J Clin Anesth. 2020; 61: 109658.
- Eckerdal P, Kollia N, Karlsson L, Skoog-Svanberg A, Wikström AK, Högberg U, et al. Epidural Analgesia During Childbirth and Postpartum Depressive Symptoms: A Population-Based Longitudinal Cohort Study. Anesth Analg. 2020; 130: 615-624.
- Sekula P, Del Greco MF, Pattaro C, Köttgen A. Mendelian Randomization as an Approach to Assess Causality Using Observational Data. J Am Soc Nephrol. 2016; 27: 3253-3265.
- Bloch M, Schmidt PJ, Danaceau M, Murphy J, Nieman L, Rubinow DR. Effects of gonadal steroids in women with a history of postpartum depression. Am J Psychiatry. 2000; 157: 924-930.
- Mehta D, Newport DJ, Frishman G, Kraus L, Rex-Haffner M, Ritchie JC, et al. Early predictive biomarkers for postpartum depression point to a role for estrogen receptor signaling. Psychol Med. 2014; 44: 2309- 2322.
- Guintivano J, Arad M, Gould TD, Payne JL, Kaminsky ZA. Antenatal prediction of postpartum depression with blood DNA methylation biomarkers. Mol Psychiatry. 2014; 19: 560-567.
- McClain L, Farrell L, LaSorda K, Pan LA, Peters D, Lim G. Genetic associations of perinatal pain and depression. Mol Pain. 2019; 15: 1744806919882139.
- Tong S, Rao C, Min S, Li H, Quan D, Chen D, et al. Obstetric anesthesia clinic childbirth course combined with labor epidural analgesia is associated with a decreased risk of postpartum depression: a prospective cohort study. BMC Anesthesiol. 2022; 22: 389.
- Kaur A, Mitra S, Singh J, Sarna R, Pandher DK, Saroa R, et al. Pain, stress, analgesia and postpartum depression: Revisiting the controversy with a randomized controlled trial. Saudi J Anaesth. 2020; 14: 473-479.
- Wang J, Zhao G, Song G, Liu J. Association between neuraxial labor analgesia and postpartum depression: A meta-analysis. J Affect Disord. 2022; 311: 95-102.
- Riazanova OV, Alexandrovich YS, Ioscovich AM. The relationship between labor pain management, cortisol level and risk of postpartum depression development: a prospective nonrandomized observational monocentric trial. Rom J Anaesth Intensive Care. 2018; 25: 123-130.
- Suzumori N, Ebara T, Tamada H, Matsuki T, Sato H, Kato S, et al. Relationship between delivery with anesthesia and postpartum depression: The Japan Environment and Children’s Study (JECS). BMC Pregnancy Childbirth. 2021; 21: 522.