Pitfalls in Estimating Renal Injury by AKIN, RIFLE, and KDIGO, Overcome by a New Method for Measuring Creatinine Clearance in Acute Kidney Injury
- 1. Department of Nephrology, St. Mary’s Health Center, USA
Abstract
Background: Acute Kidney Injury (AKI) is a common and serious condition encountered in hospitalized patients. The severity of kidney injury is defined by the RIFLE, AKIN, and KDIGO criteria which attempt to establish the degree of renal impairment. The KDIGO guidelines state that the creatinine clearance should be measured whenever possible in AKI and that the serum creatinine concentration and creatinine clearance remain the best clinical indicators of renal function. Neither the RIFLE, AKIN, nor KDIGO criteria estimate actual creatinine clearance. Furthermore there are no accepted methods for accurately estimating creatinine clearance in AKI.
The present study describes a new, unique, and simple method for estimating K in AKI using urine creatinine excretion over an established time interval (E), an estimate of creatinine production over the same time interval (P), and the estimated static glomerular filtration rate (sGFR), at time zero, utilizing the CKD-EPI formula. Using these variables estimated creatinine clearance (Ke) = E/P * sGFR.
Patient examples are provided to highlight the use of this method and its advantage over AKIN, RIFLE, or KDIGO which have the above mentioned shortcomings while often incorrectly classifying the extent of renal injury in the patient with AKI.
Conclusions: The present study provides the practitioner with a new tool to estimate real time K in AKI with enough precision to predict the severity of the renal injury, including progression, stabilization, or improvement in azotemia. It is the author’s belief that this simple method improves on RIFLE, AKIN, and KDIGO for estimating the degree of renal impairment in AKI and allows a more accurate estimate of K in AKI.
Keywords
• Acute kidney injury
• Glomerular filtration rate
• Creatinine clearance
• Estimated K (Ke)
• Actual K
• Static K
• Serum creatinine concentration
• Urine creatinine concentration
• Creatinine excretion
• Creatinine production
• Urine volume
Citation
Mellas J (2016) Pitfalls in Estimating Renal Injury by AKIN, RIFLE, and KDIGO, Overcome by a New Method for Measuring Creatinine Clearance in Acute Kidney Injury. JSM Renal Med 1(1): 1002.
ABBREVIATIONS
AKI: Acute Kidney Injury; GFR: Glomerular Filtration Rate; K: Creatinine Clearance; Ka: Actual K; Ke: Estimated K; Ks: Static K; C: Serum Creatinine Concentration; U: Urine Creatinine Concentration; E: Creatinine Excretion; P: Creatinine Production; Vd: Creatinine Distribution Volume
INTRODUCTION
Acute Kidney Injury (AKI) is a common and serious medical condition encountered in hospitalized patients. When present it contributes significantly to morbidity, mortality, and overall health care costs [1,2].
The international nephrology community has agreed on a classification system for estimating the degree of renal impairment in AKI. These criteria are defined by; the Risk, Injury, Failure, Loss, End stage renal disease (RIFLE), Acute Kidney Injury Network (AKIN) and the Kidney Disease Improving Global Outcomes (KDIGO) criteria. These methods attempt to estimate the degree of kidney injury using a measure of absolute or relative rise in serum creatinine concentration or determining the duration of oliguria [1-12]. Shortcomings to these criteria include the fact that neither method estimates actual glomerular filtration rate (GFR) or creatinine clearance (K) and hence infer the extent of renal injury. Furthermore, by relying on serum creatinine concentration (C) and changes in C (dC/dt), there are inherent errors related to different creatinine kinetics which vary between patients, based on muscle mass, making direct correlations between patients unreliable. Additionally, there are situations where significant falls in GFR are not detected due to a delay in the rise in C. There are also dilution effects of parenteral solutions on C and dC/dt. Finally, the estimate of renal injury is made retrospectively and does not allow one to predict the course of the renal impairment prospectively [13,14]. It is for these reasons that AKIN, RIFLE, KDIGO are not reliable metrics for accurately measuring GFR, K, or the extent of renal injury in AKI.
A real time, easy to perform, reliable and accurate estimate of GFR or K has been an elusive missing metric in AKI. Its usefulness in clinical medicine is clear in that one would have an accurate measure of renal injury, where the course and severity can be predicted in advance. Furthermore one could anticipate a plateau phase followed by a recovery which may also be anticipated well in advance of a changing C. A method to accurately estimate GFR or K, real time, is a welcome and overdue tool in the study of AKI.
The author recently published a paper which describes a method for estimating K in AKI, relying on principals of creatinine mass balance and K estimation using the urine creatinine excretion (E) as opposed to serum creatinine (C). This method is more accurate than those relying on serum creatinine in that urine creatinine concentration (U) and urine volume (V) are measured directly and accurately, and not estimated, while the term static K (Ks) is introduced and represents the creatinine clearance as determined by CKD-EPI, at any time zero in the course of AKI. Details of the method are described in reference [15]. Figure (1) provides an example of the pattern of creatinine excretion in AKI and the logic behind the method for measuring K in AKI, where Ke = Ks x E/P. In this method E is directly measured using a four, six or eight hour urine sample. P is estimated by the following formulas; P (male) = (27-.173 * age)* weight in kg, and P (female) = (25-.175 * age)* weight in kg over 24 hours. Where weight is lean body weight and P falls by 2% for each hospital day [14].
The following example and cases demonstrate the significance of this method for estimating K in AKI and highlights the inherent potential errors in AKIN, RIFLE, and KDIGO for estimating the degree of renal injury in AKI.
Example
In the case of two patients with a urine output of 500 ml in twenty fours, the same value for C at the time tested, and an identical estimation for P, the patient with the greater value for E will have a less severe renal injury for any level of Ks. In a similar way these same two patients with an E/P value of .5 will have very different values for Ke if in one case the Ks at t0 is 100 ml/ min and in the other 40 ml/min where the value for Ke in these patients will be 50 ml/min and 20 ml/min respectively. One can appreciate that Ke = Ks x E/P where the term E/P determines the future direction of the azotemia and Ks provides a baseline value used to determine Ke. In both cases AKIN, RIFLE, and KDIGO would be inaccurate and misleading as to the extent of renal injury.
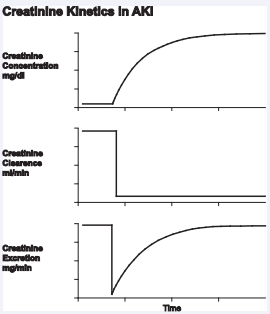
Figure 1 Intuitively, one can appreciate that as you move from left to right along the X-axis, the ratio of creatinine excretion over production (E/P) rises at any time Interval, while the estimated static K (Ks) at the start of that time interval falls. Hence, a rising E/P multiplies by a falling Ks at any time (t) is a constant number and equals the actual K where K=Ks*E/P. In essence, E/P represents a “correction factor for the Ks at any time (t). At the plateau phase, to the far right of the graph, the creatinine no longer rises, E/P=1, thus 1 multiplies by Ke=Ka.
Case 1: A patient with CKD and a baseline C of 4 mg/dl presents with bladder outlet obstruction and a C of 8.7 mg/dl. The bladder is decompressed, the urine output is brisk. The E/P ratio is .7, Ke is 5 ml/min. This suggests lack of recovery with relief of the obstruction, progressive azotemia ensues, and renal replacement therapy is initiated. This case highlights the prognostic value of measuring K real time, something that is not possible with the present classification system.
Case 2: A patient presents with sepsis and shock. On presentation he is resuscitated with crystalloid and given antibiotics. C is 1.5 mg/dl, the urine output is less than 500 ml in the first 24 hours. E/P is .6. Ke is 25 ml/min suggesting the patient will not need renal replacement therapy. C peaks at 2.94 mg/dl and then gradually declines. In this case the presence of oliguria does not correlate with the extent of renal injury. An actual value for Ke is clearly superior to urine volume as a measure of renal injury.
Case 3: A patient presents with acute pancreatitis with a C 3.2 mg/dl. He is resuscitated with crystalloid and colloid. The E/P ratio is .18, and Ke 4 ml/min. Renal replacement therapy is initiated on day 4 with a C level of 5.65. On day 28 the urine output was noted to steadily rise, exceeding 2000 ml per day. It is on day 35 that Ke has risen to 10 ml/min and dialysis is safely terminated with gradual renal recovery noted. In this case the initial low value for Ke shows the extent of renal injury and predicts the need for dialysis. In addition renal recovery is measured far in advance of present methods which rely on following the serial values for C and then deciding on whether dialysis can be stopped. By measuring Ke, recovery is measured days in advance. One can also see that the onset of a diuresis does not necessarily correlate with renal recovery.
Case 4: An elderly patient presents with signs of hypovolemia. He is resuscitated with isotonic crystalloid. On day 1 C is 1.45 mg/dl. The patient is oliguric and on day 2 C has risen to 3.81 mg/dl. The E/P ratio is 2.7; Ke is 43 ml/min predicting rapid renal recovery. On days three and four C falls to 2.55 and 1.63 mg/dl respectively consistent with rapid improvement in K as reflected by the favorable E/P ratio and Ke. Here one sees that renal recovery is imminent. AKIN, RIFLE, and KDIGO would significantly over estimate the extent of renal injury.
Case 5: A middle aged man with alcoholic hepatitis presents with hepatic decompensation. On day one C is .68 mg/dl. By day 4 C has risen to 3.35 mg/dl, E/P is less than .1, and Ke is essentially zero. On day five RRT is initiated due to progressive azotemia and symptomatic fluid excess. On day 25 C is 3.57 mg/dl, E/P ratio is .67, and Ke is 10 ml/min despite persistent oliguria. This suggests that C will continue to rise in the short term and renal function has begun to recover. As predicted C rises and peaks at 5.1 mg/ dl on day 27. On day 30 the patient remains oliguric yet E/P is now 1.3 and Ke is 30. This is followed by a gradually falling C as predicted by these last measurements. Once again this example shows the prognostic effect of measuring Ke and the often misleading meaning of urine volume in predicting the extent of renal damage.
Case 6: A young patient with CKD due to chronic glomerulonephritis and a baseline C of 2 mg/dl presents with superimposed pre-eclampsia with C 2.45 md/dl on day 1. She undergoes emergency C section. On day 3 C has risen to 2.81 mg/ dl, E/P is .86, and Ke is 20 ml/min. Using the steady state formula for K = P/C; solving for C gives a value of 4.2 mg/dl which reflects the peak C value when E/P = 1 and K can be calculated directly. In this case the patient’s actual peak C was 4.1 mg/dl followed by a gradually falling C. Here we see a case where peak C can be predicted with reasonable accuracy. This reasoning clearly adds a significant level of knowledge and sophistication to the assessment of the patient with AKI.
Case 7: An elderly patient presents with a massive pulmonary embolism with hypotension. He is treated with pressors, crystalloid and anticoagulants. On day 1 C is .81 mg/dl. On day 3 C has risen to 2.31, the patient is oliguric, the E/P ratio is .3, Ke is 10 ml/min and C continues to rise as predicted. The patient is given high dose loop diuretic and responds with a brisk diuresis. Repeat testing reveals an E/P ratio of .25, Ke of 6 ml/min. Despite the presence of a diuresis these results suggest progressive renal injury as reflected by a continued rising C. In this example one sees that the onset of a diuresis does not necessarily equate to renal function recovery.
Case 8: An elderly patient with CKD and a baseline C of 2 mg/ dl is hospitalized with pneumonia, sepsis, and respiratory failure. C on day 1 is 4.61 mg/dl and 4.63 mg/dl on day 2. The patient is mechanically ventilated, while receiving antibiotics, and pressors. The E/P ratio is 1.1 and Ke 16 ml/min. This predicts a gradual improvement if renal function reflected by a gradually falling C. Renal replacement therapy is not needed. Measuring Ke adds a level of assurance that the renal injury is limited and barring further renal insults the patient may be able to avoid dialysis.
Table 1: Patient with CKD due to hypertension with baseline C 4 mg/dl. He presents with bladder outlet obstruction. The bladder is decompressed with a catheter. Ke suggests no improvement in renal function and RRT is initiated
| Hospital Day | 1 | 2 | 3 | 4 | |
| S Creatinine | 4 | 8.72 | 8.75 | 9.18 | 9.52 |
| U Volume | 700(8 hrs) | ||||
| Ks | 7 | ||||
| E/P | 0.74 | ||||
| Ke | 5 | ||||
| Notes | |||||
| 2010 | RRT |
Table 2: Patient presented with septic shock and is resuscitated with crystalloid, pressors and antibiotics. Oliguria is noted with a fractional sodium excretion of 3%. Ke suggests the insult is not severe enough to warrant RRT and the onset of recovery is noted after several days. Ke correlates with Ks at the peak level for C.
| Hospital Day | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| S Creatinine | 1.23 | 1.5 | 2.27 | 2.84 | 2.94 | 2.71 | 2.52 | 2.35 | 2.23 | 1.71 |
| U Volume | 85(4hrs) | |||||||||
| Ks | 43 | 22 | ||||||||
| E/P | 0.58 | |||||||||
| Ke | 25 | |||||||||
| Notes | Peak C |
Table 3: Elderly male with severe hypovolemia, oliguria, and rising C he is resuscitated with IV crystalloid. Ke suggests rapid renal recovery.
| Hospital Day | 1 | 2 | 3 | 4 |
| S Creatinine | 1.45 | 3.81 | 2.55 | 1.63 |
| U Volume | 350(4 hrs) | |||
| Ks | 16 | |||
| E/P | 2.7 | |||
| Ke | 43 | |||
| Notes | IV fluids |
Table 4: 48 yr male with alcoholic hepatitis and oliguric ischemic ATN, ARDS, sepsis, on CVVH and then intermitent HD for several weeks.Patient later extubated, off pressors, with urine output 200 to 400 ml in 24 hours. Note that patient is oliguric on days 25 and 30 with a reversal of the E/P ratio suggesting recovery on day 30.
| Hospital Day | 1 | 4 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 | 31 | 32 |
| S Creatinine | 0.68 | 3.35 | 2.76 | 2.28 | 3.57 | 4.27 | 5.1 | 3.5 | 3.54 | 3.74 | 3.36 | 2.83 |
| U Volume | < 50 | 100 | 150 | |||||||||
| Ks | 20 | (8hrs) | (8hrs) | |||||||||
| E/P | <. 1 | 15 | 20 | |||||||||
| Ke | 0 | 0.67 | 1.3 | |||||||||
| Notes | Start RRT | HD | Transient Hypotension | 10 | Last HD | 30 |
Table 5: 24 yr pregnant female with CKD due to chronic GN with baseline creatinine 2. On day 1 she has emargency C section due to fetal disttress. On day 3 her Ke is estimated as 18-20 ml/min. Using the formula K=P/C, solving for C her peak serum creatinine is estimated as 4.2 to 4.6. Recovery begins with creatinine peaking ay 4.1.
| Hospital Day | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| S Creatinine | 2.45 | 2.55 | 2.81 | 3 | 3.14 | 3.64 | 3.71 | 4.11 | 4.06 | 3.84 |
| U Volume | 1200 | |||||||||
| Ks | (8 hrs) | |||||||||
| E/P | 24 | |||||||||
| Ke | 0.86 | |||||||||
| Notes | 18-20 | Peak Creatinine | Home |
Table 6: Elderly man with massive pulmonary embolism and pressor dependent hypotension. Initially oliguric, converted to non oliguric state with loop diuretic. One sees no correlation between Ke and urine volume
| Hospital Day | 1 | 2 | 3 | 4 | 5 | 6 |
| S Creatinine | 0.81 | 1.69 | 2.31 | 3.12 | 3.8 | 4.05 |
| U Volume | 110 | 450 (8 hrs) | ||||
| Ks | (8 hrs) | 24 | ||||
| E/P | 33 | 0.25 | ||||
| Ke | 10 | 6 | ||||
| Notes | Oliguria Lasix 200 mg x 1 | Non oliguric | ||||
| Hospital Day | 1 | 2 | 3 | 4 5 |
||
| S Creatinine | 4.61 | 4.63 | 4.33 | 3.99 | 3.68 | 3.63 |
| U Volume | 300(8 hours) | |||||
| Ks | 14 | |||||
| E/P | 1.1 | |||||
| Ke | 15- 16 | |||||
| Notes | Off Pressors | |||||
| Elderly male with pneumonia, Sepsis, and respiratory failure with histoy of CKD and baseline creatinine 2 mg/dl. | ||||||
DISCUSSION
AKI is a common, serious, and often devastating disorder resulting in significant morbidity, increased risk of mortality, and excessive healthcare costs as a consequence of complications which may occur as a result [1,2]. It remains one of the most challenging disorders encountered in medicine often requiring complex knowledge of pathophysiology across several organ systems and medical subspecialties. In the analysis of a patient with AKI, the physician is faced with determining etiology, pathophysiology, extent of renal injury, and reversibility whenever possible [16,7,8]. To this end, the AKIN, RIFLE, and KDIGO criteria have been proposed as the best estimates of renal injury [1-12]. These criteria do not establish pathophysiology or reversibility. Rather, the extent of renal injury is estimated by an absolute or relative rise in C, the duration of oliguria, or the need for RRT without signs of renal recovery. Neither GFR nor K is actually measured. Shortcomings to these criteria include potential errors with C and dC/dt due to dilution effects as well as differences in muscle mass and creatinine kinetics from patient to patient. Furthermore these estimates of renal injury are retrospective and have little impact on forecasting the actual extent of renal injury or the course of the azotemia during an episode of AKI [13,14].
A real time, easy to perform, reliable, and accurate estimate of GFR or K has been an elusive missing metric in AKI. Its usefulness in clinical medicine is clear in that one would have an accurate measure of renal injury, where the course and severity can be predicted in advance. Furthermore one could anticipate a plateau phase followed by a recovery which may also be anticipated well in advance of a changing C. A method to accurately estimate GFR or K, real time, is a welcome and overdue tool in the study of AKI.
The method used in this paper measures E using a four, six, or eight hour timed urine specimen, P is estimated, and Ks equals estimated GFR at time zero by any standard GFR estimating equation such as MDRD or CKD-EPI [17-21]. From this one can measure K in AKI where Ke = Ks x E/P and the extent of renal injury can be predicted with accuracy.
The examples given above highlight the utility of this method in estimating renal function at any time while showing that AKIN, RIFLE, and KDIGO, by being static indicators of renal function in a dynamic and non-steady state, often misrepresents the extent of renal injury. In cases 2, 4, and 5 one sees situations where patients with normal or near normal serum C levels have low values for Ke showing that significant renal injury can be detected at an early stage. There is no other measure of renal function or injury that has been show to accomplish this with accuracy and predictability.
There are other examples where C is elevated but Ke is actually higher than anticipated in patients with a functional and reversible cause for AKI as is seen in example 4. In this case AKIN, RIFLE, and KDIGO would overestimate the degree or renal impairment.
There are also examples where urine output does not predict renal function as in the case of renal function recovery in the patient who remains oliguric with a falling C, likely due to the presence of liver disease where creatinine is excreted in a small urine volume due to enhanced renal absorption of tubular ultrafiltrate as a consequence of the liver pathology, while E is rising due to renal function improvement as seen in example 5. In a different situation, as in example 3, one sees the onset of a brisk diuresis does not predict renal recovery in dialysis dependent AKI and that only a rising E is associated with the onset of recovery.
The accuracy of this method requires an accurate measure of E and estimate for P, while Ks relies on the accuracy of standard GFR estimating equations such as MDRD or CKD-EPI. It is also important to know that the value for Ke estimates K only at the time of the actual measurement. In that many patients with AKI are clinically unstable it is possible the patient may suffer abrupt additional renal insults. In this case repeat measures of Ke would show falling values serving to demonstrate the presence of additional renal insults.
CONCLUSION
The method described in this paper provides an accurate and easy to perform tool for estimating K in the patient with AKI. These examples serve to highlight many of the shortcoming of AKIN, RIFLE, and KDIGO which are overcome by this real time, easy to perform method to accurately measure K in AKI.
The author has no direct or indirect financial arrangement with any product or subject reported in this manuscript. The research was funded entirely by the author. There was no outside funding source.








































































































































