Behavior of the Respiratory Gases along Ergospirometry in Transgender Women after Long Term Hormone Therapy: A Cross Sectional Study
- 1. Department of Developmental Endocrinology Unit, University of São Paulo Hospital das Clínicas, Brazil
- 2. Department of Medicine Course, Centro Universitário São Camilo, Brazil
- 3. Department of Cardiac Rehabilitation and Exercise Physiology Unit, University of São Paulo Heart Institute, Brazil
Abstract
For transgender women (TW) undergoing estrogen therapy (ET), physiological responses while exerting physical effort is unknown at many points.
Objective: To evaluate the behavior of respiratory gases along with ergospirometry in TW undergoing long-term GAHT
Methods: A cross-sectional study was carried out with 15 TW (34.2±5.2 yo), 14 cisgender men (CM) and 13 cisgender women (CW). The TW received ET for 14.4 ±3.5 years. Ergospirometric test was performed.
Results: Expiratory volume (VE) was similar among groups in the rest and AT points, and an approximation was observed between the pattern of TW and CW at the peak (P>0.05), both lower than CM (TW vs CM p0.05). The end-tidal CO2 partial pressure (PetCO2) of TW was similar to that of CW during the test (p>0.05), and both were lower than that of CM at all points (p0.05 at peak.
Discussion: The physiological curves of respiratory parameters of TW were mostly similar to the CW group. The VE pattern may be a consequence of ET. The VE/VO2 and VE/CO2 show that TW are adapted to this new condition.
Conclusion: The behavior of TW gases during exertion showed a similar physiological pattern to CW.
Keywords
• Transgender
• Exercise test
• Women
CITATION
Alvares LAM, Santos MR, Souza FR, Santos LM, Mendonca BB, et al. (2024) Behavior of the Respiratory Gases along Ergospirometry in Transgender Women after Long-Term Hormone Therapy: A Cross-Sectional Study. JSM Sexual Med 8(1): 1131.
INTRODUCTION
There are physiological differences in respiratory function between males and females. Sex hormones are involved in the regulation of lung maturation [1], and are observed in both the upper and lower air pathways.
Anatomical differences have been described in the upper airways [2-4], with almost all structures being larger in cisgender men (CM) than in cisgender women (CW). The tracheal cross- sectional area is 29% larger in CM, and in general, the luminal areas of the airways are 14-31% larger in CM [5].
CW have smaller airways relative to lung size than men [6], smaller rib cages [7,8] and smaller dimensions of the chest wall [9].
Since birth, females have smaller lungs than males, with fewer respiratory bronchioles [10], and such differences persist throughout life. As a consequence of the higher area, CM has a larger total number of alveoli and a larger alveolar surface area [11].
These structural differences are associated with functional differences during spontaneous breathing and exercise.
Since such aspects and differences in the structure and function of the respiratory system become critically important during exercise, sex differences in respiratory gas patterns during effort have also been described in recent decades.
In recent years, there has been an increase in the number of transgender women (TW) in sports. Thus, we aim to provide a detailed and comprehensive description of the physiological determinants of respiratory patterns in TW. Because of the complex dynamics of the respiratory system during effort and the known differences between sexes, in-depth analyzes of respiratory gases are necessary.
We hypothesized that TW, who underwent male puberty and are undergoing long-term GAHT, have different respiratory adaptations than CW and CM.
METHODS
The study design and methods have been published previously [15]. The study protocol, available with the full text of this article at https://bjsm.bmj.com, was approved by the Ethical Committee of Clinics Hospital of the Medical School of the University of São Paulo (CH-MSUSP). Written informed consent was obtained from all patients (CAAE 17138719.0.0000.0068).
The evaluators were not blinded, with the exception of those who evaluated the results of the cardiopulmonary exercise tests.
Definitions
Transgender Group: In brief, this cross-sectional study, evaluated 15 TW (average age of 34.2 years old, SD ±5.2; average time of GAHT 14.4 years, SD ±3.5) recruited at the Gender Dysphoria Unit of the Division of Endocrinology of Hospital das Clínicas from the Faculty of Medicine of the University of São Paulo. They compared to cisgender control group composed of 27 healthy subjects, of which 13 were CW (35.6±4.1 years of age) and 14 were CM (36.7±3.8 years of age) [Supplementary Table 1,2] matched by age, body mass index (BMI, weight/ [height] 2; (kg/m2) and physical activity level to the TW group according to the International Physical Activity Questionnaire (IPAQ) [12-14] [Supplementary Tables 4 and 5].
All the subjects underwent hormonal tests at the Laboratório de Hormônios e Genética Molecular LIM42 of Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo, body composition assessment by an In Body 720 device (Biospace, Korea) with an 8-point reading through a tactile electrode and ergospirometric test that will be described below.
Cardiopulmonary Assessment: In this article, we focus on these specific parameters: VE (pulmonary ventilation), VCO (CO production), VE/VO2 (ventilatory equivalents for oxygen), VE/VCO2 (ventilatory equivalents for carbon dioxide), VO2/FFM, VO2/ weight, PetO2 (End-Tidal Partial Pressure of Oxygen) and PetCO2 (End-Tidal CO2 partial pressure).
Ergospirometry test: All individuals performed cardiopulmonary exercise testing (Sensor Medics - Vmax Analyser Assembly, Encore 29S) on a treadmill (Ergoline - Via Sprint 150 P) with incremental effort until exhaustion (ramp protocol). During cardiopulmonary exercise testing, cardiovascular performance was continuously evaluated through electrocardiography (Micromed – Cardio PC 13) with twelve leads simultaneously. Any observed electrocardiographic alteration that might compromise the experimental protocol excluded the individual from the study. The maximum functional capacity was determined by peak oxygen consumption (VO2 peak) evaluated at the maximum exercise intensity. The criteria used to consider the ergospirometry test as maximum were a respiratory exchange ratio (RER) > 1.10 and/or maximum heart rate (HRmax) > 95% of the age-predicted HRmax. The anaerobic threshold (AT) was determined at the time the individual presented the lowest ventilatory equivalent value for O2 (VEO2 ) and partial oxygen pressure at the end of expiration (PO2 ) (PetO2 ), before these parameters started to progressively increase, and a nonlinear increase in the RER value. The respiratory compensation point (RCP) was determined at the time the individual presented the lowest value of ventilatory equivalent for CO2 (VECO2 ), before this parameter started to progressively increase, and the maximum value of partial pressure for CO2 at the end of expiration ( PCO2 ) (PetCO2 ), before initiating a progressive decrease in this response.
Ergospirometry tests were carried out by the Cardiovascular Rehabilitation and Exercise Physiology Unit of Instituto do Coração, Hospital das Clinicas of the Faculty of Medicine of the University of São Paulo.
Statistical Analysis
Due to the lack of previous data to guide the sample size estimation, a convenience sample was used. Statistical analysis was performed using GraphPad Prisma 8.4.2. The D’Agostino and Pearson test was used to evaluate whether the variables were parametric or nonparametric. For parametric variables, comparison among the three groups was done through ANOVA, and for comparative sub analyses of each two groups, the Tukey test was used. The Kruskal–Wallis test was used for nonparametric variables and Dunn’s multiple comparison tests was used for comparative sub analyses of two groups. A p value<0.05 was considered indicative of statistical relevance in all tests There was not missing data.
RESULTS
Laboratory Analysis
The Supplementary Table 2, adapted from Alvares [15], shows the haemoglobin levels (g/dL) of the TW, CW and CM were 14.0 ± 0.6, 13.8 ± 0.6 and 15.3 ± 1.1, respectively.
Hormonal Date
On the day of sporting ability analysis, the mean TT (ng/dL) levels of the TW, CW and CM were 92.5 (range 12-637), 20.1 (12-41) and 524.3±169.0, respectively
One of the participants had a high level of testosterone at the time of the study. Although she was not blocked on the test day (total testosterone =637 ng/dL), her value was 79 ng/ dL six months before the study [Supplementary Figure 1,2]. In addition, this point did not interfere with her VO2 results [Supplementary Figure 3].
Level of Physical Activity
The level of physical activity was similar among all groups (TW vs. CW, p>0.05; TW vs. CM, p>0.05; CW vs. CM, p=>0.05), with most participants being active or very active in all groups [Supplementary Table 5].
Gases Analysis
All volunteers reached RER > 1.10.
Table 1 shows the main statistical analysis of this study (Figures 1-4).
Table 1: Behavior of the respiratory gases along ergospirometry in transgender women compared to cisgender woman and cisgender man groups.
|
|
Transgender women (n=15) |
Cisgender women (n=13) |
Cisgender men (n=14) |
Comparison |
P |
|
VO2/FFM (ml/kg/min) |
|
|
|
|
|
|
Rest |
5.5 (range 4.5-8.0) |
6.0 (range 4.3-7.8) |
2.9 (range 2.3-5.1) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
AT |
29.5 (range 23.7-59.9) |
31.2 (range 25.0-40.2) |
29.4 (SD±6.5) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
RCP |
43.5 (SD±5.3) |
44.1 (SD±15.0) |
45.8 (SD±6.8) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
Peak |
47.3 (SD±5.1) |
53.3 (SD±8.3) |
52.4 (SD±5.8) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
VO2/Wgt (ml/kg/min) |
|
|
|
|
|
|
Rest |
2.9 (range 2.3-5.1) |
2.4 (SD±0.3) |
3.3 (range 2.2-9.5) |
TW x CW |
** |
|
TW x CM |
NS |
||||
|
CW x CM |
**** |
||||
|
AT |
21.5 (SD±3.9) |
22.1 (SD±3.0) |
21.6 (range 17.1-41.6) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
RCP |
30.9 (SD±5.0) |
33.7 (range 25.7-60.3) |
36.2 (SD±6.7) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
Peak |
33.5 (SD±4.7) |
35.7 (SD±4.7) |
42.0 (range 32.3-49.2) |
TW x CW |
NS |
|
TW x CM |
** |
||||
|
CW x CM |
** |
||||
|
VCO2(L/min) |
|
|
|
|
|
|
Rest |
0.25 (SD±0.05) |
2.05 (SD±0.02) |
0.28 (range 0.20– 0.82) |
TW x CW |
* |
|
TW x CM |
NS |
||||
|
CW x CM |
*** |
||||
|
AT |
1.51 (range 0.97-2.7) |
1.10 (SD±0.22) |
1.6 (SD±0.39) |
TW x CW |
* |
|
TW x CM |
NS |
||||
|
CW x CM |
*** |
||||
|
RCP |
2.61 (SD±0.44) |
2.06 (SD±0.35) |
3.2 (SD±0.66) |
TW x CW |
** |
|
TW x CM |
** |
||||
|
CW x CM |
**** |
||||
|
Peak |
2.92 (SD±0.51) |
2.57 (SD±0.57) |
3.95 (SD±0.62) |
TW x CW |
NS |
|
TW x CM |
**** |
||||
|
CW x CM |
**** |
||||
|
VE (BTPS) (L/min) |
|
|
|
|
|
|
Rest |
12.3 (SD±2.4) |
10.0 (SD±1.5) |
11.8 (SD±3.0) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
AT |
47.7 (SD±12.1) |
36.6 (SD±7.6) |
43.5 (SD±13.4) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
RCP |
83.5 (SD±12.8) |
66.1 (SD±11.1) |
92.1 (SD±19.7) |
TW x CW |
* |
|
TW x CM |
NS |
||||
|
CW x CM |
*** |
||||
|
Peak |
102.3 (SD±16.2) |
87.4 (SD±10.2) |
128.8 (SD±18.7) |
TW x CW |
NS |
|
TW x CM |
** |
||||
|
CW x CM |
**** |
||||
|
VE/VO2 |
|
|
|
|
|
|
Rest |
40.5 (SD±5.0) |
43.6 (SD±8.6) |
35.1 (SD±6.6) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
** |
||||
|
AT |
28.0 (range 24.0-39.0) |
28.4 (SD±3.0) |
24.2 (SD±2.6) |
TW x CW |
NS |
|
TW x CM |
** |
||||
|
CW x CM |
** |
||||
|
RCP |
35.0 (range 30.0-48.0) |
35.4 (SD±3.6) |
30.0 (range 27.0-42.0) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
* |
||||
|
Peak |
39.2 (SD±4.4) |
42.1 (SD±4.3) |
38.9 (SD±4.3) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
VE/VCO2 |
|
|
|
|
|
|
Rest |
49.2 (range 42.0-57.0) |
50.6 (SD±9.4) |
40.5 (SD±6.3) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
AT |
32.0 (SD±3.7) |
32.1 (SD±3.1) |
28.0 (SD±3.1) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
RCP |
32.6 (SD±4.1) |
32.9 (SD±2.6) |
28.7 (SD±4.6) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
Peak |
32.8 (SD±4.4) |
33.9 (SD±3.2) |
30.0 (SD±3.3) |
TW x CW |
NS |
|
TW x CM |
NS |
||||
|
CW x CM |
NS |
||||
|
PetO2 (mmHg) |
|
|
|
|
|
|
Rest |
100.6 (SD±3.7) |
100.5 (SD±3.3) |
969.8 (SD±4.1) |
TW x CW |
NS |
|
TW x CM |
* |
||||
|
CW x CM |
NS |
||||
|
AT |
95.69 (SD± 4.3) |
95.4 (SD±2.8) |
90.9 (SD±4.0) |
TW x CW |
NS |
|
TW x CM |
** |
||||
|
CW x CM |
* |
||||
|
RCP |
102.8 (SD±4.1) |
103.7 (SD±2.7) |
100.3 (SD±3.1) |
TW x CW |
NS |
|
Peak |
106.4 (SD±4.5) |
108.2 (SD±2.5) |
105.6 (SD±3.9) |
TW x CM |
NS |
|
CW x CM |
NS |
||||
|
TW x CW |
NS |
||||
|
|
|
|
|
TW x CM |
NS |
|
|
|
|
|
CW x CM |
NS |
|
PetCO2 (mmHg) |
|
|
|
|
|
|
Rest |
29.7 (SD±2.7) |
30.5 (SD±2.5) |
33.7 (SD±3.7) |
TW x CW |
NS |
|
TW x CM |
** |
||||
|
CW x CM |
* |
||||
|
AT |
35.3 (SD± 3.7) |
34.9 (SD±1.7) |
40.1 (SD±3.2) |
TW x CW |
NS |
|
TW x CM |
*** |
||||
|
CW x CM |
*** |
||||
|
RCP |
34.2 (SD±4.2) |
33.7 (SD±2.6) |
37.9 (SD±3.5) |
TW x CW |
NS |
|
TW x CM |
* |
||||
|
CW x CM |
** |
||||
|
Peak |
|
|
|
TW x CW |
NS |
|
|
32.2 (SD±4.4) |
32.1 (SD±3.1) |
35.5 (SD±4.3 |
TW x CM |
NS |
|
|
|
|
|
CW x CM |
NS |
VO2: oxygen consumption; L/min: litre per minute; ml/kg/min: millilitre per kilogram per minute; AT: anaerobic threshold; RCP: respiratory compeNSation point; FFM: fat-free mass; Wgt: weight; VE: volume of expired air; BTPS: body temperature pressure, saturated; VE/VO2 (ventilatory equivalents for oxygen), VE/VCO2 (ventilatory equivalents for carbon dioxide); VCO2: carbon dioxide production VCO2: carbon dioxide production; PetO2 (End-Tidal Partial Pressure of Oxygen) and PetCO2 (End-tidal CO2 partial pressure); mmHg: millimetres of mercury TW: transgender women; CW: cisgender women; CM: cisgender men; NS: non-significant; *p<0.05; **p<0.01; ***p<0.001****p<0.0001.
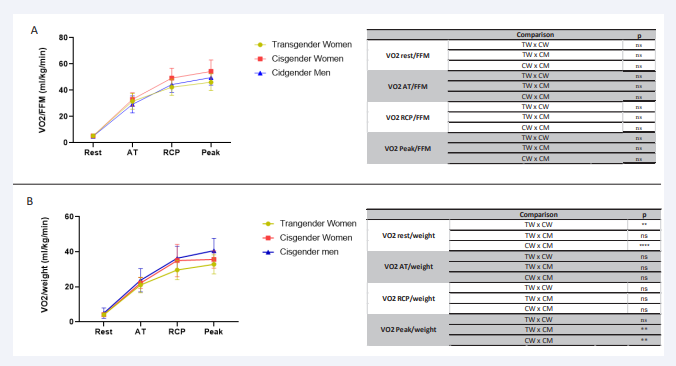
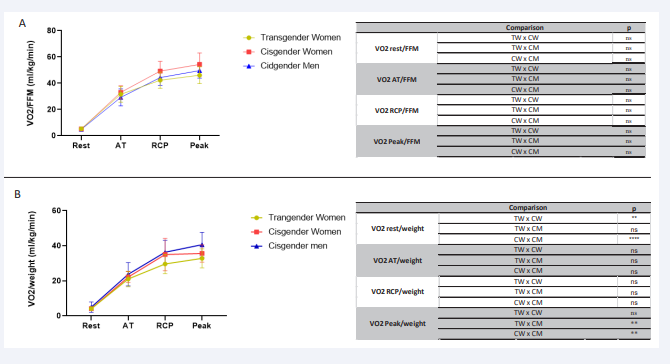
Figure 1: Comparative analysis of VO2/FFM and VO2/weight VO2/FFM from rest to peak during effort in the 3 groups of transgender women, cisgender women and cisgender men during incremental effort from rest to exhaustion. VO2/ FFM (A), VO2/ weight (B). VO2: oxygen consumption; FFM: fat-free mass; Wgt: weight; ml/kg/min: millilitre per kilogram per minute; AT: anaerobic threshold; RCP: respiratory compensation point; ns: non-significant; **p<0.01; ****p<0.0001.
Figure 2: Comparative analysis of pulmonary ventilation and carbon dioxide production from rest to peak during effort in the 3 groups of transgender women, cisgender women and cisgender men during incremental effort from rest to exhaustion. VE (BTPS) (A), VCO2 (B). VE: pulmonary ventilation; BTPS: body temperature, pressure, water vapor saturated; VCO2: carbon dioxide production; L/min: litre per minute; AT: anaerobic threshold; RCP: respiratory compensation point; ns: non-significant; *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
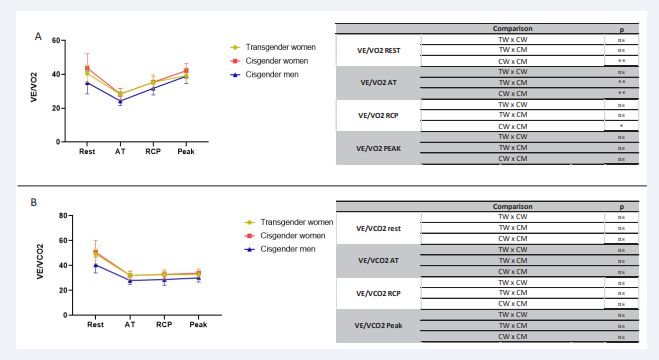
Figure 3: Comparative analysis VE/VO2 and VE/VCO2 from rest to peak during effort in the 3 groups of transgender women, cisgender women and cisgender men during incremental effort from rest to exhaustion. VE/VO2 (A), VE/VCO2 (B). VE/VO2 (ventilatory equivalents for oxygen), VE/VCO2 (ventilatory equivalents for carbon dioxide); L/min: litre per minute; AT: anaerobic threshold; RCP: respiratory compensation point; ns: non-significant
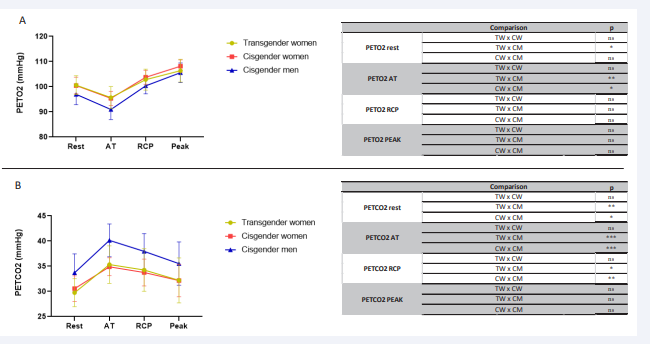
Figure 4: Comparative analysis PetO2 (End-Tidal Partial Pressure of Oxygen) and PetCO2 (End-tidal CO2 partial pressure) from rest to peak during effort in the 3 groups of transgender women, cisgender women and cisgender men during incremental effort from rest to exhaustion. PetO2 (A), PetCO2 (B). PetO2 (End-Tidal Partial Pressure of Oxygen) and PetCO2 (End-tidal CO2 partial pressure); mmHg: millimetres of mercury. AT: anaerobic threshold; RCP: respiratory compensation point; ns: non-significant; *p<0.05; **p<0.01; ***p<0.001.
Body Composition Analysis
The FFM of the TW (55.1±5.5 kg) was intermediate between the CW (40.6±4.1kg) and CM (63.7±5.4 kg) (TWvsCW p<0.0001; TWvsCM p<0.001; and the CMvsCW p p<0.0001).
DISCUSSION
This is the first study to demonstrate the respiratory physiology from de rest to the peak of respiratory parameters in TW undergoing ergoespirometric test and complement the previous published data on VO2peak of this group [15].
VE is the volume of air moved in and out of the lungs (respiratory rate x tidal) [17]. At rest, the TW group was similar to the other groups, and during effort, the CW and TW patterns were similar. This means that the possibility of expanding flow resembled the female pattern.
The volume variations in male lungs are larger than those in female lungs [18,19]. Men have significantly larger mean values for all pulmonary variables, both volumes and flows [1]. The differences between women and men impact the development of flow, the regulation of lung volume, pressure swings and the consequent work of breathing.
Estrogen can be associated with the reduction of these respiratory values. A translational study demonstrated that tidal volume was significantly increased in male wild-type mice compared with female wild-type mice; however, this pattern was reversed in alpha-ERKO mice (estrogen receptor knockout) [8]. These data suggest that functional disruption of ER-alpha leads to changes in a variety of respiratory parameters and that this nuclear receptor may be a critical regulator of breathing and respiratory rhythmogenesis in mice [2].
Females’ smaller airway diameter and lung volume result in lower peak expiratory flow and vital capacity. Therefore, women have a smaller maximal flow-volume loop. Their capacity to generate increased ventilation during exercise is smaller than men’s.
The regulation of lung volume during exercise is an important factor, as it reflects the strategy by which tidal volume is achieved and contributes to the work of breathing.
The smaller female vital capacity implies a lower maximal tidal volume to be achieved during exercise, compensated for by a higher respiratory rate to adequately ventilate the lungs. The volume of CO2 produced by TW from rest to exhaustion demonstrated a peculiar pattern, being different from the value in the other groups in most analyses.
VE/VO2 , the ratio between pulmonary ventilation and O2 consumption [17], presented the same pattern in CW and CM described in the literature, and TW presented the same pattern as the CW group. Since VE/VO2 reflects the ventilatory need for a certain O2 consumption level [17], we can observe that, despite the possible anatomical changes, the ventilatory efficiency is preserved in TW.
In the same way, VE/VCO2 , which represents the ventilatory need to eliminate a certain amount of CO2 produced by active tissues [17], at rest, AT and RCP were similar between the 3 groups. At the peak, they presented the patterns already described, and no difference was seen between groups.
VO2 peak/FFM is the best way to show VO2 functional status because it evaluates the physiological ability of the tissue to uptake oxygen. This ratio of TW group was statistically equal of the other groups at all points, which.
The PETO2 of TW was similar to the CW pattern at rest, and during exercise, both patterns approached the CM pattern. The same was observed in PETCO2 . The perfusion within the pulmonary system of TW was in the female pattern.
In this study, we describe that TW without a history of pulmonary and/or cardiac diseases on prolonged use of hormone therapy have a VE [Supplementary Table 3], with an intermediate value between the values of CM and CW. One explanation for this different VE pattern in TW would be a reduction of the caliber of the airways by estrogen, which is a potential bronchoconstrictor [2].
LIMITATIONS
Our study sample is not a true random sample and was made of volunteers because the casuistry is hard to find, which can be a weak point of our study. However, we believe that the approach of the current study in developing models could be done in other places of the world. Since this study analysed active transgender, the presented findings cannot be extrapolated to athletes.
CONCLUSION
In this small cohort of nonathletic TW, who previously underwent male pubertal development followed by long-term estrogen therapy, the behavior of gases and heart rate during exertion, analyzed through ergospirometry, showed a similar physiological pattern to that of CW in most analyses. Changes in anatomy and functional phenotypes caused by estrogen have been elucidated by in-depth physiological studies
ACKNOWLEDGEMENTS
The authors are grateful to the Gender Dysphoria Unit of the Division of Endocrinology of Hospital de Clínicas da Faculdade de Medicina da Universidade de São Paulo (HCFMUSP), Laboratório de Hormônios LIM42 do HCFMUSP and Instituto do Coração (InCor) do HCFMUSP.
Funding sources
This research was supported by grants from the Coordination for the Improvement of Higher Education Personnel (CAPES/ PNPD; institutional fellowship Grant 88887.362860/2019-00 to LAMA), and by National Council for Scientific and Technological Development (CNPq) Grants 308873/2018-1 (to SD).
Data sharing statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Authors’ contributions
Study conception and design: Alvares LAM, Domenice S; Data acquisition and analysis: Alvares LAM, Santos MR, Souza FR, Santos LM, Alves MJNN; Manuscript drafting: Alvares LAM, Domenice S; Critical revision: Domenice S, Costa EMF, Mendonca BB.
Ethics
This study was approved by the Ethical Committee of Clinics Hospital of the Medical School of the University of São Paulo (CH-MSUSP). Written informed consent was obtained from all patients (CAAE 17138719.0.0000.0068).
Patient involvement
None or not applicable
Questions and Answers
- What are the new findings?
This article shows an in-depth analysis of the gaseous behavior of transgender women during effort.
- How might it impact clinical practice in the near future?
These findings could help in analyses of respiratory parameters of transgender women during rest and effort.
-What is already known about this topic?
There are no data about the physiological behavior of gases of transgender women.
-What this study adds?
This study adds to our knowledge about respiratory physiology in transgender women
REFERENCES
- Seaborn T, Simard M, Provost PR, Piedboeuf B, Tremblay Y. Sex hormone metabolism in lung development and maturation. Trends Endocrinol Metab. 2010; 21: 729-738.
- Card JW, Zeldin DC. Hormonal influences on lung function and response to environmental agents: lessons from animal models of respiratory disease. Proc Am Thorac Soc. 2009; 6: 588-595.
- Rowley JA, Sanders CS, Zahn BR, Safwan Badr M. Gender differences in upper airway compliance during NREM sleep: role of neck circumference. J Appl Physiol. 2002; 92: 2535-2541.
- Brown IG, Zamel N, Hoffstein V. Pharyngeal cross-sectional area in normal men and women. J Appl Physiol. 1986; 61: 890-895.
- Sheel AW, William Sheel A, Dominelli PB, Molgat-Seon Y. Revisiting dysanapsis: sex-based differences in airways and the mechanics of breathing during exercise. Exp Physiol. 2016; 101: 213-218.
- Mead J. Dysanapsis in normal lungs assessed by the relationshipbetween maximal flow, static recoil, and vital capacity. Am Rev Respir Dis. 1980; 121: 339-342.
- Bellemare JF, Cordeau MP, Leblanc P, Bellemare F. Thoracic Dimensions at Maximum Lung Inflation in Normal Subjects and in Patients with Obstructive and Restrictive Lung Diseases. Chest. 2001; 119: 376-386.
- Bellemare F, Jeanneret A. Sex differences in thoracic adaptation to pulmonary hyperinflation in cystic fibrosis. Eur Respir J. 2007; 29: 98-107.
- Romei M, Mauro AL, D’Angelo MG, Turconi AC, Bresolin N, Pedotti A, et al. Effects of gender and posture on thoraco-abdominal kinematics during quiet breathing in healthy adults. Respir Physiol Neurobiol. 2010; 172: 184-191.
- Becklake MR, Kauffmann F. Gender differences in airway behaviour over the human life span. Thorax. 1999; 54: 1119-1138.
- Thurlbeck WM. Postnatal human lung growth. Thorax. 1982; 37: 564- 571.
- Matsudo SM, Matsudo VR, Araujo T. Nível de atividade física da população do Estado de São Paulo: análise de acordo com o gênero, idade, nível socioeconômico, distribuição geográfica e de conhecimento. Rev Bras Cienc Mov. 2002; 10: 41-50.
- Matsudo S, Araújo T, Matsudo V, Andrade D, Andrade E, Oliveira LC, et al. Questionário Internacional de Atividade Física (IPAQ): estudo de validade e reprodutibilidade no Brasil. Rev Bras Ativ Fis Saúde. 2001; 6: 5-18.
- Matsudo SM, Matdsudo VR, Araújo T, Andrade D, Andrade E, Oliveira L, et al. Nível de atividade física da população do Estado de São Paulo: análise de acordo com o gênero, idade, nível socioeconômico, distribuição geográfica e de conhecimento. Rev Bras Cienc Mov. 2002; 10: 41-50.
- Alvares LAM, Santos MR, Souza FR, Santos LM, Mendonça BB de, Costa EMF, et al. Cardiopulmonary capacity and muscle strength in transgender women on long-term gender-affirming hormone therapy: a cross-sectional study. Br J Sports Med. 2022; 56: 1292- 1298.
- Cerqueira LS, Nogueira FS, Carvalho J, Pompeu FAM. Resposta da cinética de consumo de oxigênio e da eficiência mecânica delta de homens e mulheres em diferentes intensidades de esforço [Internet]. Revista Brasileira de Medicina do Esporte. 2011; 17: 274-278.
- Herdy AH, Ritt LEF, Stein R, Araújo CGS de, Milani M, Meneghelo RS, et al. Cardiopulmonary Exercise Test: Background, Applicability and Interpretation. Arq Bras Cardiol. 2016; 107: 467-481.
- Quanjer PH, Hall GL, Stanojevic S, Cole TJ, Stocks J. Age- and height- based prediction bias in spirometry reference equations. Eur Respir J. 2012; 40: 190-197.
- Quanjer PH, Brazzale DJ, Boros PW, Pretto JJ. Implications of adopting the Global Lungs Initiative 2012 all-age reference equations for spirometry. Eur Respir J. 2013; 42: 1046-1054..