Perineal Ultrasound in Postprostatectomy Incontinence: An Overview of Technique and Practice
- 1. Department of Urology, Hospital Hochsteiermark, Austria
- 2. Private Institute for Manual Therapy, Austria
- 3. Faculty of Applied Sciences Joanneum, Department of Physiotherapy, Austria
Abstract
Introduction: Continence after prostatectomy depends on a combination of preoperative patients’ characteristics, surgical techniques and intraoperative details. The integrity of pelvic floor muscles and striated urethral sphincter significantly influence the recovery of continence after surgery. An accurate assessment of possible risk factors aids in identifying patients with a higher likelihood of postoperative incontinence and those who would benefit from a personalized postoperative rehabilitation. This overview aims to describe the use of perineal ultrasound in the evaluation of anatomy and function of the male pelvic floor, in particular after prostatectomy.
Methods: Literature search through the MEDLINE, Ovid, and Embase databases
Main outcome measures: Literature review and clinical research studies on the use of perineal ultrasound in the diagnostic and pelvic floor training programs for postprostatectomy incontinence.
Results: The bladder neck and urethra, the bulb of penis and the anorectal junction can be easily identified during perineal ultrasound. They represent important reference landmarks, and their displacement correspond to activation of the pelvic floor muscle, in particular the striated urethral sphincter, bulbospongiosus and puborectalis muscles. Real time dynamic imaging provides an individualized assessment of the coordinated function of pelvic floor muscles.
Clinical implications: Activation of the striated urethral sphincter, bulbospongiosus muscle and puborectalis muscle can be reliably observed by perineal ultrasound. Perineal ultrasound gives feedback during pelvic floor muscle training leading to early detection of failure in specific muscle activation and helps to develop individually tailored rehabilitation programs in postprostatectomy incontinence.
Strengths and limitations: Research about motor control of the male pelvic floor based on ultrasound techniques provide a new understanding in pathophysiology and rehabilitation of postprostatectomy incontinence. To date there are only few studies using perineal ultrasound as a diagnostic and functional feedback tool in the management of postprostatectomy incontinence.
Conclusion: Perineal ultrasound is an easy and reproducible diagnostic tool for the assessment of the anatomy of the male pelvis and pelvic floor muscles. Real time dynamic imaging provides a precise assessment of the coordinated function of the striated urethral sphincter, bulbospongiosus and puborectalis muscles. It gives feedback of pelvic floor muscle training leading to early detection of failure in specific muscle activation and helps to develop individually tailored rehabilitation programs in postprostatectomy incontinence.
Keywords
Male pelvic floor, Pelvic floor muscle training, Perineal ultrasound, Prostatectomy, Urinary incontinence
ABBREVIATIONS
PPI: Post-Prostatectomy Incontinence; RP: Radical Prostatectomy; PFM: Pelvic Floor Muscle; BN: Bladder Neck; FUL: Functional Urethral Lenght; PFMT: Pelvic Floor Muscle Training; SUS: Striated Urethral Sphincter; BS: Bulbospongiosus Muscle; PR: Puborectalis Muscle; ARJ: Anorectal Junction.
Citation
Dalpiaz O, Sammer A, Zeder R, Spreitzhofer EM, Gödl-Purrer B (2022) Perineal Ultrasound in Postprostatectomy Incontinence: An Overview of Technique and Practice. JSM Sexual Med 6(5): 1098.
INTRODUCTION
Post-prostatectomy incontinence (PPI) represents a challenging matter for both patients and urologists, and is a significant socio-economic and psychological burden. Depending on the definition of incontinence and the length of follow-up, up to 40% of men experience incontinence after radical prostatectomy (RP), from which the majority recovers full continence after 6-12 months postoperatively [1]. Continence after RP is a complex issue, relying on a combination of preoperative patients’ characteristics, surgical approach and intraoperative details. The integrity of the striated urethral sphincter (SUS) and pelvic floor muscles (PFM) significantly influences the recovery of continence after surgery [2,3]. An accurate preoperative assessment of possible risk factors associated with PPI supports identifying patients with a higher probability of PPI and those who would benefit from preventive strategies in the preoperative setting and personalized rehabilitation postoperatively.
Perineal ultrasound (PUS) represents a long-established imaging in the evaluation of urinary incontinence and pelvic floor dysfunction in women [4]. PUS has the advantage of dynamic imaging, with visualization of the pelvic anatomy both at rest and during focused instructions. About ten years ago the utilization of PUS began in the evaluation of the pelvic anatomy and PFM function in men [3,5,6]. Also activity of PFM and SUS has been assessed reliably with PUS, as previously reported [7,8]. Recently, functional changes after prostatectomy have been investigated by PUS [5,7,9]. However, the use of PUS in the diagnosis and rehabilitation of PPI has to be confirmed.
This overview is targeted on description of the technique of PUS, its application and image interpretation, and summarizes the currently available literature discussing the potential diagnostic and therapeutic role of PUS in PPI.
METHODS
A literature search was performed on MEDLINE, Ovid and Embase databases for English articles published since 2000. The search terms used were various combinations of the following: “(trans)perineal ultrasound,” “male pelvic floor” “prostatectomy” “urinary incontinence” “pelvic floor muscle training” “male sling”. A total of 74 articles were reviewed by two authors, 18 relevant articles on PUS and male pelvic floor were identified and considered for discussion.
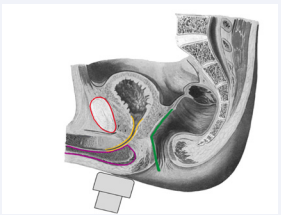
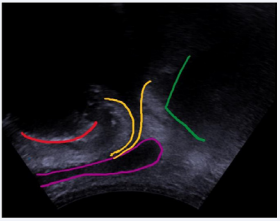
Figure 1a: Schematic representation of transducer placement on the perineum and imaged structures in the midsagittal plane in male after prostatectomy: pubic symphysis (red), bladder neck and proximal urethra (yellow), corpus spongiosum and bulb of penis (pink), ano-rectal junction (green).
Perineal ultrasound imaging – static view
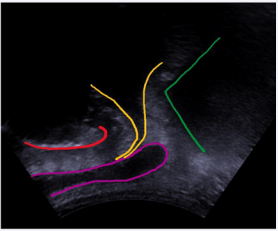
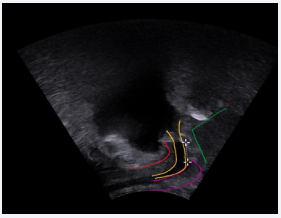
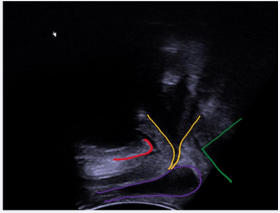
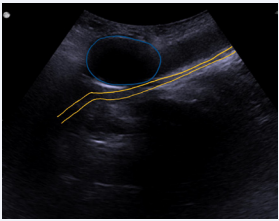
Two-dimensional images of the pelvic floor muscles are acquired using a low frequency (3,5-6 MHz) curved array transducer placed on the perineum in the midsagittal plane (Figure 1a). The technique has been widely described and validated [10]. Patients are routinely investigated in a supine position with their legs bent and slightly rotated outwards. PUS can also be easily performed in a standing position, as previously reported [9,10]. The standard midsagittal view includes from ventral to dorsal the symphysis pubis (SP), the bladder neck (BN), the urethra and bulbus, and the anorectal junction (ARJ) (Figure 1a,1b). The SP represents an easy static reference point for orientation and identification of the above-mentioned anatomical structures and it serves as reference landmarks during dynamic investigation. The BN and the urethra can be simply identified with the partially filled bladder. A standardized bladder volume is not necessary. The functional urethral length (FUL) as a prognostic factor for PPI can be easily measured before and after RP [11,12]. The functional (membranous) urethral length is calculated as the distance between the bladder neck (vesico-urethral junction) and the urethra at the penile bulb (Figure 2). The configuration of the BN can be featured during PUS. A bladder neck funneling is an usual finding in patients after RP, as reported in (Figure 3). The prognostic role of funneling in PPI is still not clear.
Figure 1b: PUS standard midsagittal view: pubic symphysis (red), bladder neck and proximal urethra (yellow), corpus spongiosum and bulb of penis (pink), ano-rectal junction (green).
Figure 2: Bladder neck (yellow) representation during PUS
Figure 3: Measurement of the lenght of functional urethra after radical prostatectomy
Dynamic perineal ultrasound
Based on current knowledge, the striated urethral sphincter (SUS), the puborectalis (PR) and the bulbospongiosium (BS) muscle have the potential to support urethral closure [3,9]. The functional urethra, the anorectal junction and the bulb of penis represent important reference landmarks (Figure 4a). The displacement of these landmarks correspond to the activation of the SUS, PR, and BP, as previously described by Stafford and Hodges in different papers [3,10,13,14]. In particular, the activity of the striated urethral sphincter (SUS) can be easily observed during voluntary contraction. During activation of SUS the urethra draws dorsally. The puborectalis muscle (PR) draws the urethra ventrally against the SP and elevates the bladder and the ARJ moves forwards and upwards. The bulbospongiosus muscle (BS) compresses the bulb of penis to constrict the urethra distal to the SUS from dorsal to ventral, in the opposite direction to SUS. In this way, the urethral bulb appears thinner and longer achieving an optimal urethral closure, as demonstrated in (Figure 4b).
Figure 4a: Schematic representation of bladder neck and urethra (yellow), bulbus (pink) and anorectal junction (green) at rest. The displacement of these landmarks correspond to the activation of the striated external sphincter (yellow), bulbospongiosus (pink) and puborectalis (green) muscles.
Figure 4b: Colored arrows indicate direction of landmark displacement during voluntary contraction: activity of striated external sphincter (yellow), bulbospongiosus (pink) and puborectalis (green) muscles.
Perineal ultrasound in the postoperative rehabilitation
Several papers and meta-analysis showed that PFM training (PFMT) either alone or in addition to biofeedback and/or pelvic floor muscle electric stimulation is effective for treating PPI, significantly reducing the time to continence recovery [15,16]. Current evidence of perioperative PFMT focusses on training of the ventral complex of the male pelvic floor, in particular SUS and BS. Not only integrity of PFM and SUS but also their correct activation significantly contributes to continence status after RP. As above described, PUS allows an easy and reproducible identification of landmarks and a visualization of activity of SUS and BS, as previously described. Thus, it represents an effective method for teaching correct activity and specific exercise of PFM, to concentrate patients’ awareness during training in a real time visual feedback.
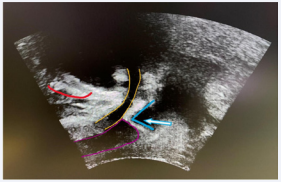
Figure 5a: Synthetic slings appear highly hyperechogenic on PUS. PUS show the sling (arrows) in relation to urethra (yellow), supporting the BS muscle and repositioning of the functional urethra (yellow).
PUS following surgery for postprostatectomy incontinence
Male slings or compression devices such as artificial urinary sphincter or balloons have been implanted to treat PPI with satisfactory outcomes. Slings restore continence in males either by urethral compression and/or by repositioning the bulb of urethra [17]. The wide-weave material of slings is highly echogenic and often accompanied by an acoustic shadow produced by the tape. It is therefore easily identifiable, as previously described [18]. In the midsagittal plane, the suburethral portion of the sling and the identification of the correct position of the sling/balloon in relation to the urethra and /or the less or more effective urethral compression can be assessed, as reported in the (Figure 5a,5b). In the coronal view, the full length of the sling can be visualized. Real-time dynamic imaging provides important information regarding the position and function of the sling during voluntary contraction.
Figure 5b: PUS in the midsagittal plane showing the balloon (blue) of an adjustable male sling slightly compressing the urethra (yellow).
DISCUSSION
Integrity and function of SUS and PFM contribute significantly to continence status after prostatectomy. Roll et al firstly reported sonographic findings of the muscles of the male pelvic floor and described reliability for perineal sonographic image acquisition and analysis technique for evaluation of superficial structures of the male pelvic floor [6]. Until now, several studies confirmed PUS as an easy and reproducible method for the evaluation of PFM function [3,5,7,10]. In 2012 Stafford et al described the dynamics of pelvic floor contractions through PUS in ten healthy continent men. They validated displacement of standardized landmark, such BN and ARJ during PUS. Anatomical considerations suggested that these are caused by contraction of the pelvic floor muscles, in particular SUS and BS, improving the understanding of the male urinary continence mechanism [3]. In a following paper, the same authors presented a new method for quantifying the displacement of pelvic structures during PFM contraction, confirming the high repeatability of parameters during PUS. An important point of discussion was the reproducibility of magnetic resonance imaging measured displacements with PUS in one proband [10]. PPI occurs usually during events which increase intra-abdominal pressure, for example by coughing. Assessment of PFM dynamics in men during voluntary and evoked coughs is important to understand incontinence. Coughing in healthy men activates a complex pattern of urethral movements that can be attributed to shortening and lengthening of different PFM, as described. Urethral dynamics differed between voluntary and evoked cough, suggesting that voluntary coughing may not be an adequate test for assessment of PPI [19].
All discussed papers focused on the importance of the ventral muscle complex of the pelvic floor for maintaining continence. In particular, they confirmed the role of SUS, BS and PR muscles in urethral constriction, the relation between displacement observed on ultrasound, the activation of PFM and also the validity of their interpretation in healthy men [13]. The role of preoperative puborectal muscle function assessed by transperineal ultrasound in UI was also discussed by Neumann and O´Callaghan. They concluded that the PR muscle does not play a role in the recovery of continence after RP which may help to explain the negative findings of many studies of PFM training for PPI [20]. A few years later, Stafford et al focused their research on continence mechanism in men with PPI. Dynamic features of PFM contraction seems to be associated with continence status after prostetectomy. Continent men provided evidence of greater displacement of SUS, BS and PR muscles during evocated cough and sustained maximal voluntary contraction. Greater SUS shortening was associated with continence and was able to compensate, to some degree, for poor function of the PR and BS muscles. Otherwise, when a deficit in shortening SUS is present, continence might still be achieved by enhanced BS and PR muscle activation, except for patients with very poor function of SUS [9,21]. These observations would significantly impact muscle training and rehabilitation.
Recently, the relationship between pre- and postprostatectomy measurements of PFM function and the development of early PPI has been investigated in sixty men referred for preoperative PFM training before RP [14]. They focused their attention on SUS and confirmed that shortening of SUS is a more important determinant of continence than other muscles, as reported in previous study [9,14]. In the same study, compression of bulb by BS did not differ between continent and incontinent men pre- and post-prostatectomy. BS shortening during coughing during preoperative examination was also negatively associated with PPI. Furthermore, RP changed some features of ARJ, but not motion of SUS or BS, as reported in other studies [8]. Postoperatively, the authors found a reduced displacement of ARJ, arising from structural changes and intraoperative reconstructive techniques [14].
Several studies confirmed a correlation between postoperative FUL and incontinence severity [12,22]. The FUL can be easily measured by PUS before and after prostatectomy as shown in the figure. A recent paper discussed whether the membranous (functional) urethral length is predictive of postprostatectomy incontinence (PPI) requiring surgery such as artificial urinary sphincter or male sling. The authors concluded that patients who had surgery for PPI had a significantly shorter FUL and sphincter volume than continent controls. In particular, men with an functional urethral lenght >17 mm are unlikely to require surgery for PPI whereas an FUL <12 mm significantly increases the risk for needing surgery for PPI. However, the authors utlized T2-weighted magnetic resonance imaging scans for imaging [23]. FUL can be easily assessed by PUS with a high repeatability [9]. Stafford et al in 2022 investigated the relationship beetween preand post prostatectomy measurements of PFM function and their prognostic role in incontinence. Severity of incontinence was negatively correlated with urethral length [14]. However, the absence of correlation between SUS and urethral lenght would suggest an independent impact on continence. The FUL cut-off between continent and incontinent patient has to be defined in further studies.
Most studies present PUS in prone position. The influence of body position on dynamics of the pelvic floor during perineal ultrasound has been exanimated. Feedback for PFM is possible in different body position (standing or sitting) but the position needs to be standardized for comparative assessment [24].
Ultrasound and/or magnetic resonance imaging can also be used after PPI surgery. Male slings provide a minimally invasive procedure with satisfying outcomes [17]. The management of failure or complications after sling placement is challenging. Imaging has an important complementary role in evaluating position and function of the sling. The use of ultrsasound in assessing sling failure and complications is currently in its early stages and is still debated even in female [25,26]. Likewise, visualizing the sling in continent patients would support followup and experience in interpretation of difficult cases and causes of failure. A recent review discusses the current literature with the aim to identify the benefits and limitations of ultrasound and MR and to evaluate the role of each imaging modality for patients with sling complications. The role of ultrasound in prediciting female incontinence, as well as assessing pain, voiding dysfunction and tape complication has been discussed in detail. Ultrasound provides a readily available, inexpensive, and easy to perform imaging modality available in most hospitals. Magnetic resonance imaging has the benefit that the images are acquired according to standard protocols and can more easily be reviewed by other clinicians [27].
PFM training alone or in combination with biofeedback and / or electrostimulation for men undergoing RP has been confirmed as first line treatment for PPI, significantly accelerating the recovery of continence [14]. PUS is increasingly used by physiotherapists for treatment programs and biofeedback of PFM activation [14]. For the use of PUS during rehabilitation, Stafford et al investigated the relationship between sonographic observations of displacements of specific landmarks and their relationship to EMG amplitudes for PFM [9,13,14]. Rehabilitation seems to be more effective when it´s focused on training of the ventral compartment of the PFM, targeting to recruit the SUS and BS muscles. Hodges et al published a protocol for a randomized controlled trial comparing urethral training with the use of PUS to conventional training and no training. The authors hypothesized that selective training and individualized functional training of the pelvic floor incorporated in functional tasks would reach superior results than the usual management [28]. The content of PFMT programs determines its efficacy. In a systematic review, Hall et al found a substantial heterogeneity in content and quality of reporting of the components of exercise regimes. The substantial variation in treatment regimens used in clinical trials might contribute to the fact, that to date there is no sound evidence for the efficacy of PFM training in PPI. In particular, only few PFMT regimes focus on training of the ventral compartment in particular SUS and BS, and fewer use training with PUS as biofeedback control [29]. As above discussed, Stafford et al investigated differences in the function of PFM in continent and incontinent patients after RP. Men who were continent achieved greater shortening of the SUS, PR, and BC muscles than the incontinent group, particularly during voluntary contractions and coughing. This also could be a useful clinical target for individualized treatment programs, focusing in ventral muscle compartment [21].
There is increasingly evidence that effective PFM training programs should focus on SUS and BS rehabilitation. PR muscle training seems to be of minor importance.
PUS should be used to interpret activation of PFM during different instructions and can be recommended as a biofeedback measurement to teach and control PFM training. The realtime visual feedback would also help patients in learning and visualizing activation of SUS and BS. In this way, patients could optimize the pattern of activation of the PFM and improve the adherence to exercise. Further research is required to confirm the diagnostic and therapeutic utility of PUS in PPI. The possibility of having a reliable and accessible imaging tool, such as ultrasound, for visualizing pelvic floor structures and muscles, could help us understand the physiopathology of PPI and plan the most appropriate therapeutic strategy.
CONCLUSION
Perineal ultrasound is an easy and reproducible method for the assessment of the male pelvic anatomy and pelvic floor. Through real time dynamic imaging PUS provides a precise assessment of the coordinated function of SUS, BC and PR. PUS gives feedback on PFM training leading to early detection of failure in specific muscle recruitment and helps to develop individually tailored rehabilitation programs in PPI. PUS should be routinely implicated as a diagnostic and rehabilitative tool in postprostatectomy incontinence.