Osteoporotic Hip Fracture and Teriparatide: Postoperative Damage and Management
- 1. The Second Clinical Medical School, Zhejiang Chinese Medicial University, China
- 2. The Second Affiliated Hospital of Zhejiang Chinese Medical University, China
- 3. Department of Rehabilitation Medicine, The First Affiliated Hospital, Zhejiang University School of Medicine, China
Abstract
Hip fractures are a significant health concern among the elderly population, characterized by elevated mortality rates, prolonged recovery periods, and substantial postoperative complications. Osteoporosis (OP), a key determinant of increased hip fracture risk and poor recovery outcomes in older adults, leads to deterioration of bone microarchitecture, reduced bone mineral density, and heightened skeletal fragility. This condition substantially impacts patients’ postoperative survival and overall quality of life. Recent advancements in OP management have been notable, with an increasing development of novel pharmacological agents. Teriparatide, which uniquely stimulates bone formation rather than merely suppressing bone resorption, has demonstrated efficacy in mitigating OP-related symptoms and facilitating fracture recovery. Currently, teriparatide is extensively employed in the management of osteoporotic hip fractures. This article presents a narrative review of the etiological factors underlying osteoporotic hip fractures, associated postoperative mortality, and the therapeutic application of teriparatide in fracture management. The objective is to establish a theoretical foundation that may guide the development of enhanced treatment strategies and pharmacological interventions for osteoporotic fractures.
Keywords
• Osteoporosis
• Hip fracture
• Teriparatide
• Chronic Diseases
• Bone-Anaboli
Citation
Dong ZH, Liu C, Man ZW, Wang CJ, Wu ZY, et al. (2026) Osteoporotic Hip Fracture and Teriparatide: Postoperative Damage and Manage ment. JSM Surg Oncol Res 7(1): 1027.
ABBREVIATIONS
OP: Osteoporosis; TPTD: Teriparatide; CKD: Chronic kidney disease; BMD: Bone mineral density; TBS: Trabecular bone score; PEW: Protein-energy wasting; BMI: Body mass index; DVT: Deep vein thrombosis; PE: Pulmonary embolism; AKI: Acute kidney injury; PTH: Parathyroid hormone; PTHrP: Parathyroid hormone- related peptide
INTRODUCTION
Osteoporosis (OP) is a chronic disorder that predominantly affects postmenopausal women and the elderly, and its prevalence has steadily risen over the past decade [1]. OP contributes to fracture risk by compromising bone strength, reducing bone mass, and deteriorating the bone microarchitecture, ultimately leading to heightened skeletal fragility. Consequently, fractures may occur even from low-impact trauma and can affect multiple anatomical sites, with the hip being particularly vulnerable. Within the hip region, the femoral neck,femoral head, and intertrochanteric areas are especially prone to fracture [2,3]. Hip fractures are a frequent injury among the elderly, especially among women, and are associated with substantial negative impacts on overall health [4]. Empirical evidence demonstrates a positive correlation between hip fractures and increased all-cause mortality, with this association being slightly stronger in men than in women. Notably, mortality rates are elevated during the first year following a hip fracture and remain elevated over an extended period [5]. Emerging evidence further indicates that the relationship between OP and hip fractures is more substantial than previously recognized. Given the global demographic shift toward an aging population, osteoporotic hip fractures are poised to increasingly threaten both longevity and quality of life. Accordingly, as the significance of OP prevention in mitigating fracture risk becomes more apparent, there is an urgent need to raise awareness regarding the role of OP in fracture etiology, alongside addressing challenges related to treatment awareness and resource allocation for elderly patients afflicted with this condition [6].
Managing hip fractures predominantly relies on surgical intervention, particularly internal fixation, which has been demonstrated to be effective. Nonetheless, certain risks persist, with complications such as fracture, nonunion, and avascular necrosis of the femoral head occurring with considerable frequency. Even when internal fixation is successfully performed, trauma or compression that compromises the blood supply can precipitate these adverse outcomes [7]. Furthermore, preventing and managing comorbidities and postoperative complications—such as chronic kidney disease (CKD), chronic obstructive pulmonary disease, dementia, heart failure, and diabetes—remain challenging, as these conditions often manifest during the early postoperative period or may develop years following surgery. The sequelae and complications arising from hip fracture surgery can result in chronic pain, functional disability, depression, and elevated mortality rates. Consequently, these adverse effects constitute major health concerns that substantially impact both the life expectancy and quality of life in elderly patients following hip fracture [8].
Pharmacological interventions for fracturemanagement often demonstrate limited targeting specificity or suboptimal efficacy, particularly in the context of osteoporotic fractures such as those of the hip [9]. This limitation is largely attributable to the fact that most patients presenting with osteoporotic fractures are in advanced stages of disease, characterized by markedly reduced bone mineral density and extensive structural deterioration. Consequently, therapeutic agents primarily designed to inhibit osteoclast activity frequently exhibit limited effectiveness under these conditions [10]. Teriparatide (TPTD), an anabolic bone agent, exerts its effects by stimulating osteogenesis rather than suppressing osteoclastic activity. Clinical evidence has demonstrated that TPTD facilitates the repair of bone microarchitecture, mitigates bone loss, fundamentally prevents fracture occurrence, and enhances fracture healing. These characteristics render TPTD particularly suitable for patients with osteoporotic fractures who have a prolonged disease course or are prone to recurrent fractures, conditions under which the bone is severely compromised and antiresorptive therapies demonstrate diminished efficacy. Notably, during the treatment of osteoporotic hip fractures, TPTD has been revealed to exert a significant beneficial effect across multiple clinical parameters [11].
METHODS
This review aims to summarize the literature on the susceptibility to osteoporotic hip fractures, their associated risks and postoperative mortality, and the therapeutic or ameliorative effects of TPTD. A comprehensive literature search was conducted using the electronic databases— PubMed and Sci-Hub, employing keywords such as teriparatide, OP, hip fracture, comorbidities, complications, antiresorptive, anabolic, and fracture recovery. A total of 559 articles were initially identified. Following title screening, 182 articles were selected, and 133 were retained after abstract review. Ultimately, 98 articles were incorporated in the final comprehensive review, with all pertinent references incorporated.
ASSOCIATION BETWEEN OP AND HIP FRACTURE
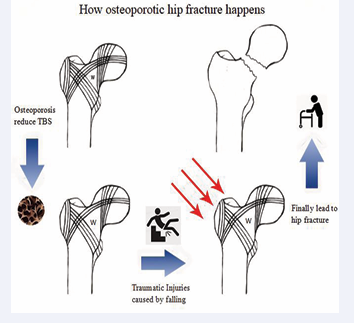
OP is a pathological condition marked by decreased bone mineral density (BMD), degradation of bone microarchitecture, and a consequent loss of bone mass, collectively undermining skeletal integrity [12]. Although hip fractures are often primarily associated with accidental falls, OP represents a critical underlying risk factor. The rapid advancement of OP induces extensive impairment of bone quality and mechanical strength, thereby elevating the risk of falls among affected individuals and directly contributing to an increased incidence of hip fractures and associated mortality rates (Figure 1).
Figure 1 Figure describes how osteoporotic hip fracture Happens.
Impact on bone mass
OP is primarily characterized by reductions in BMD and trabecular bone score (TBS), which together impair the mechanical strength of bone tissue and heighten fracture susceptibility [13]. The hip joint, as a principal weight- bearing structure in the human body, is particularly prone to fractures associated with OP-induced bone loss. Specifically, diminished bone mass within the proximal
femur significantly weakens its load-bearing capacity, rendering it susceptible to fractures even under normal physiological stress [14,15]. During menopause, females experience a decline in osteogenic activity alongside increased osteoclastic resorption, leading to marked deterioration of TBS and an inevitable loss of bone mass. Hormonal fluctuations combined with augmented bone resorption synergistically accelerate bone loss during this period [16, 17]. Among the elderly population, inadequate intake of vitamin D and calcium is prevalent, disrupting calcium homeostasis, stimulating parathyroid hormone secretion, and ultimately resulting in secondary hyperparathyroidism. This condition exacerbates bone loss and elevates the risk of hip fractures [17,18]. Moreover, pharmacological side effects may intensify bone loss; notably, abrupt discontinuation of most anti-resorptive OP therapies without appropriate transitional measures may lead to varying degrees of BMD reduction [19]. Empirical evidence also demonstrates that individuals sustaining hip fractures experience a femoral neck BMD loss exceeding fivefold within the first year post-fracture compared to non-fractured counterparts. Such accelerated bone loss substantially heightens the risk of subsequent fractures, particularly among elderly patients who already exhibit diminished bone density and an increased susceptibility to falls [20].
Impact on bone structure
Skeletal stability is determined by both the macrostructural and microstructural characteristics of bone. From a macrostructural perspective, the femoral neck—a key component of the hip—is relatively slender and therefore susceptible to injury. Its lateral side contains principal tensile trabeculae, while the medial side is composed of principal compressive trabeculae; together with secondary compressive trabeculae, these elements form a sparsely trabeculated triangular region known as Ward’s triangle. OP further compromised the integrity of this structure, rendering it highly vulnerable to fractures under external loading conditions [21,22]. The femoral neck-shaft angle is anatomically adapted to endure substantial vertical loads and to efficiently distribute body weight during normal ambulation. Nonetheless, femoral neck fractures typically arise from external forces applied perpendicular to the femoral axis, lateral impacts to the greater trochanter, or torsional stresses on the leg when the hip joint is abducted. Under these circumstances, the femoral neck is subjected to lateral or twisting forces, generating considerable shear and tensile stresses that predispose this region to injury. Furthermore, an excessively large neck-shaft angle increases compressive forces on the lateral aspect of the hip joint, thereby
amplifying the deleterious effects of external forces and increasing fracture risk [23,24]. At the microstructural level, OP impairs bone architecture by reducing trabecular thickness, disrupting trabecular continuity, and decreasing trabecular number, ultimately causing cortical bone thinning and increased porosity. These structural changes collectively enhance bone fragility and diminish intrinsic bone strength, thereby heightening susceptibility to fractures [25]. Moreover, OP is a chronic condition with a prolonged disease course, during which many patients experience extended periods of immobility in advanced stages. This reduction in physical activity diminishes mechanical loading on bone, impairs bone metabolism and formation, and further contributes to skeletal fragility [26].
Impact on accidental falls
Although falls are not the sole determinant of hip fractures, they constitute the primary precipitating factor for most such injuries. The elderly population is particularly susceptible to falls, with both the incidence of falls and the risk of subsequent hip fractures escalating with advancing age. Simultaneously, the effectiveness of preventive interventions aimed at reducing accidental falls diminishes in this demographic [27,28]. Skeletal muscle weakness is a prevalent clinical issue among older adults, often resulting from prolonged protein-energy wasting (PEW). Clinically, PEW is characterized by reductions in body mass index (BMI) and serum protein concentrations and is frequently observed in patients with chronic kidney disease, especially those undergoing dialysis. The depletion of skeletal muscle mass contributes to lower limb weakness, impaired gait stability, and compromised balance, thereby elevating the risk of falls. Additionally, the diminished muscular protection following a fall heightens the likelihood of sustaining fractures [46]. Additional factors, such as delayed reaction times, visual impairment, and dizziness, further augment both the risk of falls and the inability to grasp objects that might mitigate injury during a fall. The magnitude of impact force exerted on the hip during a fall is a critical determinant of fracture risk. In particular, lateral falls impose the greatest force on the lateral aspect of the hip, transmitting stress from the greater trochanter to the femoral neck, thereby substantially increasing the probability of fracture at this site. Notably, direct impact on the hip during a fall elevates fracture risk by nearly fivefold [29,30]. Moreover, delays in the management of falls significantly affect both the severity and prognosis of hip fractures. In the context of an aging population, the proportion of elderly individuals living alone has risen markedly, heightening the likelihood of delayed treatment and subsequent deterioration of fracture outcomes [31,32].
FACTORS FOR HIGH MORTALITY AFTER HIP FRACTURE SURGERY
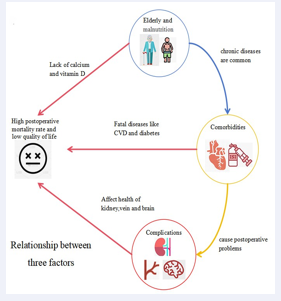
Hip fractures are associated with increased mortality and disability, largely because they compromise the body’s primary weight-bearing joint, impair normal mobility, and hinder the restoration of daily functional abilities. Despite surgical intervention, postoperative mortality and disability rates remain markedly high [33], largely due to the fact that most individuals sustaining osteoporotic hip fractures are elderly and often present with multiple comorbidities, which collectively heighten the risk of postoperative complications. The interplay of these factors significantly impairs patients’ recovery following surgery (Figure 2).
Figure 2 Factors for high mortality after hip fracture surgery
Advanced age and malnutrition
Advanced age has been identified as a key determinant influencing postoperative mortality among patients with hip fractures [34]. Individuals over 80 years of age typically experience a natural decline in the functional capacity of multiple organ systems, accompanied by reduced muscle strength. Empirical evidence suggests that both advanced age and markedly weakened hip flexor muscles are significant contributors to the risk of secondary fractures [35]. Sarcopenia, a condition closely associated with aging, constitutes an additional factor that adversely affects recovery following hip fracture surgery. Research demonstrates that over 30% of individuals aged 70–90 years are affected by sarcopenia, with prevalence in males approximately twice that observed in females. This condition compromises muscle strength and BMI thereby negatively influencing both quality of life and postoperative survival [36]. Malnutrition, characterized by protein deficiency, results in reductions in bone and muscle mass, thereby compromising bone strength and muscular function [37]. Studies indicate that malnutrition is prevalent among the elderly and significantly contributes to elevated postoperative mortality following hip fractures [38]. Moreover, malnutrition impairs tissue repair and immune function, while deficiencies in vitamin D and calcium exacerbate bone loss by disrupting osteogenesis [18,39]. Surgical intervention itself constitutes a form of trauma, typically eliciting an inflammatory response and substantial blood loss. Consequently, elderly patients with concomitant anemia are at increased risk of frailty due to the synergistic effects of anemia and associated cognitive impairments [40].
Comorbidities
Patients with OP frequently exhibit poor overall health and often present with multiple comorbidities. In patients with hip fractures, the presence of comorbidities often limits the ability to provide targeted treatment during surgery, making these conditions a major determinant of postoperativesurvivalandqualityoflife[41].Cardiovascular disease is a common condition among the elderly and has a bidirectional relationship with hip fractures, with cardiac dysfunction accounting for a substantial proportion of deaths following hip fracture surgery [42]. Diabetes is a common comorbidity in patients with OP and contributes to bone calcium loss and deterioration of bone structure. Associated complications, such as vision impairment and neuropathy, further increase the risk of falls in the elderly [43,44]. Renal dysfunction also significantly elevates the risk of hip fractures in patients following a fall, with the magnitude of risk correlating with the severity of renal impairment. This increased risk is linked to the effects of renal dysfunction on drug metabolism and excretion, as well as increased bone fragility [45,46]. Dialysis, commonly employed in kidney disease management, can induce vitamin D deficiency and disrupt calcium and phosphorus metabolism, thereby promoting OP [47]. Diabetic nephropathy, a form of chronic kidney disease resulting from diabetes, exerts a greater negative impact on patients. Metabolic disturbances from proteinuria and bone calcium loss due to hypercalciuria increase the risk of hip fractures beyond that observed with diabetes or kidney disease alone [48].
Complications
Postoperative complications, including vascular diseases, infections, and dementia, can arise from a variety of factors. Their incidence is strongly associated with the presence of comorbidities and can further impair patients’ motor function and ability to perform activities of daily living. These complications often prolong recovery and may ultimately contribute to poor prognoses, disease progression, or increased mortality [49].
Deep vein thrombosis (DVT) and pulmonary embolism (PE): DVT is closely linked to the development of PE. Prolonged postoperative immobilization reduces venous blood flow in the lower extremities, promoting platelet aggregation and DVT formation. Furthermore, mobility impairments associated with frailty can exacerbate thrombus development [50,51]. Detached venous thrombi can embolize through the circulatory system to the pulmonary artery, potentially resulting in PE, a life-threatening condition. Individuals with a prior history of PE are particularly susceptible to this complication [52].
Infections: Anesthesia administered during surgical procedures affecting the nervous system induces reflex inhibition, impairing the clearance of foreign materials from the airway and thereby elevating the risk of pulmonary infections, irrespective of whether general or spinal anesthesia is employed [53]. This risk is further intensified by prolonged bed rest, which compromises pulmonary secretion clearance and may result in aspiration pneumonia or respiratory failure, with smokers being particularly vulnerable [54]. Additionally, pressure ulcers represent a prevalent health concern in bedridden elderly populations. Factors such as malnutrition and limited mobility increase skin susceptibility to damage, facilitating the development of pressure ulcers that may progress into chronic wounds. Infections originating from these wounds can further escalate to sepsis or septicemia [55].
Neuropsychiatric manifestations: Older adults demonstrate heightened cognitive vulnerability, rendering them susceptible to delirium and dementia in the context of surgical procedures, anesthesia, and pain. These neuropsychiatric conditions impair the ability of elderly patients to accurately evaluate their own health status, resulting in delays in seeking appropriate medical care and the emergence of related risk factors. Such factors are associated with adverse outcomes, including diminished postoperative functional recovery, increased incidence of infections and complications, and higher mortality rates [56,57].
Acute kidney injury (AKI): Elderly patients often present with clinical conditions such as hypoalbuminemia, hyperkalemia, and an elevated propensity for bleeding,which collectively elevate the risk of AKI following hip fracture surgery. Individuals with CKD, a prior history of CKD, or multiple comorbidities are particularly susceptible to developing AKI [58]. The occurrence of AKI in this population is associated with prolonged hospitalization and heightened mortality, with a notably significant impact on early postoperative mortality [59,60].
ROLE OF TERIPARATIDE IN OP MANAGEMENT
OP is primarily characterized by an imbalance between bone resorption and bone formation. Therapeutic approaches targeting these two processes have led to the development of anti-resorptive and anabolic agents [61]. Anti-resorptive drugs, including bisphosphonates and denosumab, are currently widely used in clinical practice and have been demonstrated to reduce bone turnover markers, decrease fracture incidence, and increase BMD [62]. However, the adverse effects of these therapies cannot be overlooked. Bisphosphonates have been associated with an elevated risk of atypical femoral fractures, osteonecrosis of the jaw, ocular nerve-related complications, and unexpected inflammatory reactions. Denosumab may cause a rapid rebound in bone turnover upon discontinuation, resulting in significant bone loss [63,64]. Moreover, patients with osteoporotic hip fractures often present at an advanced disease stage, characterized by severe deterioration of bone microstructure and markedly diminished bone strength, which substantially limits the therapeutic efficacy of anti-resorptive drugs [65].
Although the conventional management of OP has primarily focused on pharmacological agents that inhibit osteoclastic activity, stimulating osteoblastic activity appears to exert a more pronounced impact. In individuals with OP, males are generally less affected than females, likely due to their higher baseline osteoblastic activity. The augmentation of bone formation confers greater advantages in counteracting bone loss compared to merely decreasing bone resorption [66]. TPTD is one of the few available bone-anabolic agents, and its ability to promote skeletal reconstruction is associated with a significant reduction in the risk of subsequent fractures and other OP- related complications [65]. A substantial body of research has demonstrated that the benefits of TPTD extend beyond fracture repair to include enhancement of bone strength, prevention of secondary fractures, and a consequent decrease in postoperative mortality, thereby improving both patient survival and quality of life (Table 1).
Table 1: Researches of teriparatide treatment
|
Authors |
Objective |
Method |
Conclusion |
|
Timothy Graham et al. [71] |
To examine the impact of teriparatide in adult patients with hypoparathyroidism |
Administer and assess up |
Teriparatide treatment demonstrates its efficacy and safety |
|
Cansu Gül Koca and Meryem Kösehasano?ullar [72] |
To evaluate the effects of a single dose of locally administered teriparatide on healing critical- sized defects in rat mandibles |
Animal-controlled experimentation |
Administered TPTD has a positive effect on the integration of allografts |
|
Kim T. Brixen et al. [68] |
To explain the anabolic effects of teriparatide on bone and analyze a new paradigm of anabolic therapy |
A randomized study comprising 1637 women with post-menopausal OP |
Teriparatide constitutes a breakthrough in the treatment of severe OP; which changes several paradigms of bone physiology |
|
Sibi Sanjay et al. [75] |
To evaluate the efficacy of TPTD in hastening fracture healing in elderly patients |
A prospective case–control study on elderly patients with fractures and re-operative bone mineral profile |
TPTD significantly reduces the duration of healing, brings better pain control, and increases the two-minute walk test distances |
|
Laura Guyer et al. [80] |
To evaluate whether teriparatide leads to sustained increases in BMD and TBS, and whether BMD correlates with fracture risk reduction |
The multicenter cohort study assessed the effect of teriparatide administration for 18–24 months, followed by antiresorptive therapy |
Teriparatide led to sustained lower incidences of vertebral, hip, and other fractures after switching to antiresorptive agents. BMD and TBS levels were significantly higher |
|
Ko Chiba et al. [82] |
To investigate the effects of daily teriparatide and weekly high-dose teriparatide on areal BMD, BTM, volumetric BMD, microarchitecture, and estimated strength in patients with postmenopausal OP |
were randomized to receive D-PTH, W-PTH, or BPs for 18 months. Dual-energy X-ray absorptiometry, BTMs, and high-resolution peripheral quantitative CT parameters were evaluated at baseline and after 6 and 18 months of treatment |
D-PTH and W-PTH comparably increased Ct.Th. D-PTH decreased Ct.vTMD and increased Ct.Th and total bone strength. W-PTH increased Ct.vTMD, Ct.Th, and total bone strength to the same extent as D-PTH |
|
Benjamin Z Leder et al. [79] |
To determine whether 24 months of combined denosumab and teriparatide will increase hip and spine BMD more than either individual agent |
The Denosumab and Teriparatide Administration (DATA) randomized controlled trial in which postmenopausal osteoporotic women received teriparatide, denosumab, or both medications for 24 months |
Concomitant teriparatide and denosumab therapy increases BMD more than therapy with either medication alone and exceeds the BMD gains reported with any other therapeutic approach |
|
Junxiong Zhu et al. [98] |
To evaluate whether weekly administration of teriparatide accelerates fracture repair in humans |
Single-center, double-blind, randomized controlled trial |
Researchers predicted a difference in fracture healing time between groups of approximately 1.5 weeks |
Annotation: op: osteoporosis, tptd: teriparatide, bmd: bone mineral density, ct: computed tomography, btm: bone turnover marker, d-pth: daily teriparatide, w-pth: weekly teriparatide, ct.th: cortical thickness, ct.vtmd: cortical volumetric tissue mineral density.
Mechanism of drug action
TPTD is a recombinant form of the N-terminal fragment (1–34) of endogenous human parathyroid hormone (PTH) [67]. Its principal cellular targets are osteoblasts within the skeletal system. TPTD binds to the type 1 parathyroid hormone receptors on the osteoblast surface, thereby stimulating osteoblastic activity. When administered intermittently, PTH signaling directly promotes osteoblast proliferation and differentiation while simultaneously inhibiting apoptosis of these cells [68]. Teriparatide exhibits a biphasic effect on osteogenesis, whereby continuous high-level exposure inhibits bone formation, whereas intermittent low-level exposure stimulates it, necessitating precise regulation of dosing intervals [69]. Empirical evidence suggests that intermittent administration of PTH (1–34) reduces fracture risk, increases BMD, and elevates serum calcium concentrations [70,71]. Furthermore, TPTD exerts a broad anabolic effect by augmenting new bone area, increasing osteoblast numbers, and enhancing overall bone mass, thereby significantly improving bone histological morphology [72]. In addition to directly accelerating new bone formation and promoting appositional growth on pre-existing bone surfaces, teriparatide-mediated activation of PTH receptors triggers intracellular signaling cascades within osteoblasts. This activation stimulates the synthesis and secretion of multiple potent osteogenic growth factors, which create additional sites for bone formation and modulate the local bone microenvironment, thereby promoting trabecular development and cortical bone augmentation [73,74].
Fracture healing and bone repair
Effective fracture management depends on accelerating callus formation and facilitating timely fracture healing. TPTD has been demonstrated to stimulate osteoblastic activity, thereby promoting the formation, growth, and mineralization of the callus, as well as enhancing anabolic metabolism during the bone remodeling phase. In comparison with bisphosphonates, TPTD treatment significantly shortens fracture healing time and exhibits notable efficacy in cases of delayed fracture healing and nonunion [75,76].
The primary therapeutic advantage of TPTD lies in its ability to increase BMD in the hip and lumbar spine, stimulate trabecular bone formation, repair microdamage, and enhance overall bone microarchitecture. Compared with antiresorptive therapies, TPTD produces a more pronounced increase in bone mass and bone turnover markers, thereby substantially lowering fracture risk [77]. However, TPTD administration may induce a degree of bone resorption; therefore, subsequent treatment with appropriate antiresorptive agents such as denosumab or the use of combination regimens incorporating both TPTD and antiresorptive drugs is recommended [78,79]. This combined therapeutic approach has consistently demonstrated efficacy in reducing fracture risk across multiple skeletal sites, including the hip, while concurrently improving patients’ BMD, TBS, overall bone strength, and fatigue resistance [80].
Furthermore, bone formed under the osteogenic influence of TPTD demonstrates higher collagen content, superior microstructural quality, enhanced toughness, and increased resistance to low-energy trauma. These improvements substantially mitigate the prevalent bone fragility observed in patients with various forms of OP and markedly reduce fracture risk [81]. The therapeutic efficacy of TPTD is contingent upon long-term intermittent administration, with optimal outcomes observed following a 24-month treatment course [73]. Evidence suggests that daily administration maximizes trabecular bone formation, whereas weekly dosing effectively maintains cortical BMD. Consequently, the choice of administration regimen needs to be tailored to the patient’s specific degree of bone mass loss and structural damage [82].
Impact on comorbidities and complications
CKD is a common complication in patients with OP, adversely impacting bone density and metabolism, increasing the risk of AKI, and elevating fracture susceptibility. Therapeutic administration of TPTD has been demonstrated to effectively mitigate these conditions and decrease fracture incidence in individuals with CKD [83]. Notably, TPTD treatment significantly shortens the duration of fracture healing. Accelerated and more efficient bone repair facilitates reduced pain levels and enables patients to commence weight-bearing and rehabilitative exercises earlier and with greater safety [84]. Experimental animal models have further revealed that TPTD exerts beneficial effects on skeletal muscle mass and strength, diminishes bone marrow adiposity, and potentiates the positive impact of physical exercise on muscle strength and postoperative functional recovery [85]. Enhanced physical mobility consequently lowers the risk of falls and subsequent fractures. Moreover, expedited restoration of functional activities diminishes complications associated with prolonged immobilization, including aspiration pneumonia, pressure ulcers, and thrombosis. Early achievement of activities of daily living (ADL) contributes to the preservation of cardiopulmonary function and psychological well-being, thereby reducing the prevalence of neuropsychiatric conditions such as delirium, dementia, and depression [86-88]. In managing osteoporotic hip fractures, teriparatide demonstrates a significant reduction in all-cause mortality and a concurrent improvement in patient-reported quality of life (Figure 3).
Figure 3 Impact on comorbidities and complications
Additionally, in cases where osteoblast activity is suppressed due to high-dose vancomycin— commonly employed to prevent surgical site infections post-fracture—combination therapy with TPTD has demonstrated significant ameliorative effects [89].
Adverse effects
TPTD is generally well tolerated and associated with relatively mild adverse effects. The most frequently reported primary side effects include injection site pain, mild headache, dizziness, and nausea [70,74]. A notable concern following the discontinuation of TPTD therapy is the loss of BMD. Although prolonged administration of TPTD tends to yield more favorable outcomes, patients with OP—who typically exhibit reduced metabolic capacity—may experience excessive drug accumulation. Such accumulation may contribute to hypercalcemia and hypercalciuria, and potentially promote drug dependence, which could negatively impact bone density following treatment cessation. These effects appear to be more pronounced among female patients. Consequently, the implementation of antiresorptive therapy subsequent to TPTD treatment is considered essential [90,91]. Furthermore, research has suggested that the concurrent use of TPTD and red yeast rice supplements may result in elevated transaminase levels. It is hypothesized that TPTD may increase the risk of adverse hepatic reactions to monacolin K; similar combined use is contraindicated
[92]. Additionally, due to the osteoclastic activity induced by TPTD at high concentrations, a slight reduction in bone mass may be observed during the initial phase of treatment; however, this phenomenon does not need to be classified as an adverse effect.
Prospect of osteoanabolic agents
Currently, teriparatide is widely recognized as the most commonly employed anabolic agent in OP management, whereas the therapeutic potential of other anabolic therapies remains under active investigation. Romosozumab—a novel monoclonal antibody—has demonstrated efficacy in promoting bone growth, increasing BMD, and facilitating fracture healing. However, its clinical application is limited by reports of severe adverse effects, including cardiovascular dysfunction [93,94]. Abaloparatide, a parathyroid hormone-related peptide (PTHrP) analog structurally similar to teriparatide, exhibits enhanced metabolic activity that produces potent bone-forming effects, leading to greater reductions in fracture risk and more pronounced improvement in BMD [95,96]. Although developing and producing anabolic agents remain incomplete, and notable side effects persist, their ability to stimulate new bone formation is of central therapeutic importance. Moreover, the relatively rapid onset of action of these agents can be optimized through combination therapy with antiresorptive drugs, offering the best therapeutic effect. Therefore, anabolic agents continue to represent a promising option for managing and preventing osteoporotic fractures [65,97,98].
CONCLUSION
This review examines the impacts of OP on mortality following hip fractures and surgical interventions. Although hip fractures are predominantly attributed to falls, the contribution of OP-related bone degradation and structural impairment is a critical factor that warrants consideration in the pathogenesis of this condition. Such pathological changes not only elevate the risk of falls among the elderly but also increase the likelihood of fractures subsequent to a fall. The postoperative mortality rates associated with hip fractures are notably high, primarily due to the advanced age of most patients, often accompanied by organ dysfunction, severe malnutrition, and multiple comorbidities. Such comorbidities, including cardiovascular and cerebrovascular impairments, can exacerbate complications, resulting in infections, thrombosis, mental disorders, renal failure, and other severe outcomes.
Furthermore, this review elucidates the mechanisms of action and clinical efficacy of TPTD in osteoporotic fracture management. Anabolic therapy with TPTD has the potential to fundamentally ameliorate the effects of OP by enhancing bone strength and promoting fracture healing, thereby facilitating earlier participation in rehabilitation and daily activities. This therapeutic approach may consequently reduce the incidence of comorbidities and complications, prevent subsequent fractures, decrease postoperative mortality, and improve the overall quality of life in patients with hip fractures. Although there have been reports of suboptimal efficacy and adverse effects associated with TPTD, underscoring the necessity for further research to comprehensively evaluate its therapeutic benefits and safety profile, its favorable efficacy and sustained clinical utilization of TPTD suggest a promising future for bone- anabolic pharmaceuticals.
AVAILABILITY OF DATA AND MATERIALS
The datasets generated and/or analysed during the current study are available in the Medline repository, [https://pubmed.ncbi.nlm.nih.gov/].
CONFLICT OF INTEREST
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
AUTHOR CONTRIBUTIONS
Z-HD: Data Curation, Methodology, Investigation, Visualization, Writing - Original Draft, Writing - Review & Editing. CL: Data Curation, Investigation, Writing – review & editing. Z-WM: Data Curation, Investigation, Writing – review & editing. C-JW: Methodology, Data Curation, Writing – review & editing. Z-YW: Methodology, Formal Analysis, Writing – review & editing. Y-YL: Methodology, Investigation, Writing – review & editing. Y-FY: Methodology, Project Administration, Formal Analysis, Writing – review & editing. B-BT: Methodology, Project Administration, Writing – review & editing. KL: Methodology, Project Administration, Writing – review & editing. SW: Resources, Funding Acquisition, Visualization, Writing - Review & Editing. Z-EW: Resources, Visualization, Writing - Review & Editing. JW: Validation, Writing – review & editing. F-QQ: Validation, Writing – review & editing. X-LS: Conceptualization, Funding Acquisition, Supervision, Writing - Review & Editing. L-LZ: Conceptualization, Funding Acquisition, Supervision, Writing - Review & Editing.
FUNDING
This study was supported by Key R&D Program ofZhejiang (2026C02A1083), Clinical Research Program of Zhejiang Provincial Administration of Traditional Chinese Medicine (2026ZL0459).
ACKNOWLEDGMENTS
The authors of this manuscript sincerely appreciate Professor Xiao-Lin Shi and Professor Wei-Feng Hu of the Second Affiliated Hospital of Zhejiang Chinese Medical University
REFERENCES
- Harris K, Zagar CA, Lawrence KV. Osteoporosis: Common Questions and Answers. Am Fam Physician. 2023; 107: 238-246.
- Srivastava M, Deal C. Osteoporosis in elderly: prevention and treatment. Clin Geriatr Med. 2002; 18: 529-555.
- Johnston CB, Dagar M. Osteoporosis in Older Adults. Med Clin North Am. 2020; 104: 873-884.
- Black DM, Rosen CJ. Clinical Practice. Postmenopausal Osteoporosis. N Engl J Med. 2016; 374: 254-262.
- Katsoulis M, Benetou V, Karapetyan T, Feskanich D, Grodstein F, Pettersson-Kymmer U, et al. Excess mortality after hip fracture in elderly persons from Europe and the USA: the CHANCES project. J Intern Med. 2017; 281: 300-310.
- de Jong-Holthuijsen S, Ravensbergen WM, Achterberg WP, Appelman-Dijkstra NM, Gussekloo J, Poortvliet RKE. Initiation of anti-osteoporosis medication following hip fracture in older adults: a systematic review and thematic synthesis of qualitative studies from patient and healthcare professional perspectives. Age Ageing. 2025; 54: afaf237.
- Daskalakis II, Bastian JD, Tosounidis TH. Time Is Bone: Missed Opportunities for Secondary Prevention After a Hip Fracture. J Clin Med. 2025; 14: 5816.
- Guzon-Illescas O, Perez Fernandez E, Crespí Villarias N, Quirós Donate FJ, Peña M, Alonso-Blas C, et al. Mortality after osteoporotic hip fracture: incidence, trends, and associated factors. J Orthop Surg Res. 2019; 14: 203.
- Crilly RG, Kloseck M, Chesworth B, Mequanint S, Sadowski E, Gilliland J. Comparison of hip fracture and osteoporosis medication prescription rates across Canadian provinces. Osteoporos Int. 2014; 25: 205-210.
- Bodenner D, Redman C, Riggs A. Teriparatide in the management of osteoporosis. Clin Interv Aging. 2007; 2: 499-507.
- Borges JL, Freitas A, Bilezikian JP. Accelerated fracture healing with teriparatide. Arq Bras Endocrinol Metabol. 2013; 57: 153-156.
- Consensus development conference: diagnosis, prophylaxis, and treatment of osteoporosis. Am J Med. 1993; 94: 646-650.
- Glaser DL, Kaplan FS. Osteoporosis. Definition and clinical presentation. Spine (Phila Pa 1976). 1997; 22: 12S-16S.
- Bliuc D, Nguyen ND, Alarkawi D, Nguyen TV, Eisman JA, Center JR. Accelerated bone loss and increased post-fracture mortality in elderly women and men. Osteoporos Int. 2015; 26: 1331-1339.
- Guerado E, Cruz E, Cano JR, Crespo PV, Alaminos M, Sánchez-Quevedo Mdel C, et al. Bone mineral density aspects in the femoral neck of hip fracture patients. Injury. 2016; 47: S21-S24.
- Karlamangla AS, Burnett-Bowie SM, Crandall CJ. Bone Health during the Menopause Transition and Beyond. Obstet Gynecol Clin North Am. 2018; 45: 695-708.
- Chavassieux P, Meunier PJ. Mécanismes de la perte osseuse dans les ostéoporoses [Mechanisms of bone loss in osteoporosis]. Rev Prat. 1995; 45: 1083-1088.
- Meunier PJ, Chapuy MC, Arlot ME, Delmas PD, Duboeuf F. Can we stop bone loss and prevent hip fractures in the elderly? Osteoporos Int. 1994; 1: 71-76.
- Cosman F, Langdahl B, Leder BZ. Treatment Sequence for Osteoporosis. Endocr Pract. 2024; 30: 490-496.
- Dirschl DR, Henderson RC, Oakley WC. Accelerated bone mineral loss following a hip fracture: a prospective longitudinal study. Bone. 1997; 21: 79-82.
- von Meyer GH. The classic: The architecture of the trabecular bone (tenth contribution on the mechanics of the human skeletal framework). Clin Orthop Relat Res. 2011; 469: 3079-3084.
- Skedros JG, Brand RA. Biographical sketch: Georg Hermann von Meyer (1815-1892). Clin Orthop Relat Res. 2011; 469: 3072-3076.
- Augat P, Bliven E, Hackl S. Biomechanics of Femoral Neck Fractures and Implications for Fixation. J Orthop Trauma. 2019; 33: S27-S32.
- Brownbill RA, Ilich JZ. Hip geometry and its role in fracture: what do we know so far? Curr Osteoporos Rep. 2003; 1: 25-31.
- Hollensteiner M, Sandriesser S, Bliven E, von Rüden C, Augat P. Biomechanics of Osteoporotic Fracture Fixation. Curr Osteoporos Rep. 2019; 17: 363-374.
- Greer RB 3rd. Wolff’s Law. Orthop Rev. 1993; 22: 1087-1088.
- Lorentzon M, Johansson H, Harvey NC, Liu E, Schini M, Vandenput L, et al. Diminishing returns of fall reduction for hip fracture prevention in older age. Osteoporos Int. 2025; 36: 1749-1751.
- Nilsson M, Eriksson J, Larsson B, Odén A, Johansson H, Lorentzon M. Fall Risk Assessment Predicts Fall-Related Injury, Hip Fracture, and Head Injury in Older Adults. J Am Geriatr Soc. 2016; 64: 2242-2250
- Nasiri Sarvi M, Luo Y. Sideways fall-induced impact force and its effect on hip fracture risk: a review. Osteoporos Int. 2017; 28: 2759-2780.
- Yang Y, Komisar V, Shishov N, Lo B, Korall AM, Feldman F, et al. The Effect of Fall Biomechanics on Risk for Hip Fracture in Older Adults: A Cohort Study of Video-Captured Falls in Long-Term Care. J Bone Miner Res. 2020; 35: 1914-1922.
- Birge SJ. Osteoporosis and hip fracture. Clin Geriatr Med. 1993; 9: 69- 86.
- Jarman MP, Sokas C, Dalton MK, Castillo-Angeles M, Uribe-Leitz T, Heng M, et al. The impact of delayed management of fall-related hip fracture management on health outcomes for African American older adults. J Trauma Acute Care Surg. 2021; 90: 942-950.
- chroeder JD, Turner SP, Buck E. Hip Fractures: Diagnosis and Management. Am Fam Physician. 2022; 106: 675-683.
- Aslan A, Atay T, Aydo?an NH. Risk factors for mortality and survival rates in elderly patients undergoing hemiarthroplasty for hip fracture. Acta Orthop Traumatol Turc. 2020; 54: 138-143.
- Yang H, Yuan Y, Ge R, Shi L, Si F, Chen Y. Risk factors and predictors of contralateral hip fracture after surgical treatment in elderly patients. Front Aging. 2025; 6: 1633184.
- Cakmak S, Turkmen I, Kulali F, Saglam N, Unnu SC, Basat SU. Examination of osteosarcopenia in patients over 65 years old with hip fracture. North Clin Istanb. 2025; 12: 327-336.
- Rizzoli R, Bonjour JP. Unterernährung und Osteoporose [Malnutrition and osteoporosis]. Z Gerontol Geriatr. 1999; 32: 31-37.
- Malafarina V, Reginster JY, Cabrerizo S, Bruyère O, Kanis JA, Martinez JA, et al. Nutritional Status and Nutritional Treatment Are Related to Outcomes and Mortality in Older Adults with Hip Fracture. Nutrients. 2018; 10: 555
- Cianferotti L, Bifolco G, Caffarelli C, Mazziotti G, Migliaccio S, Napoli N, et al. Calcium in Elderly Patients before and after a Hip Fracture and Their Impact on the Musculoskeletal System: A Narrative Review. Nutrients. 2024; 16: 1773.
- Sheehan KJ, Williamson L, Alexander J, Filliter C, Sobolev B, Guy P, et al. Prognostic factors of functional outcome after hip fracture surgery: a systematic review. Age Ageing. 2018; 47: 661-670.
- Schrøder CK, Hjelholt TJ, Møller H, Madsen M, Pedersen AB, Kristensen PK. Comorbidity and Quality of In-Hospital Care for Hip Fracture Patients. J Am Med Dir Assoc. 2022; 23: 671-677.
- Wu J, Zhang Y, Wang J, Zhang Q, Jiang J, Jiang Q, et al. Bidirectional association between cardiovascular disease and hip fracture: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2025; 25: 366.
- Chau DL, Edelman SV, Chandran M. Osteoporosis and diabetes. Curr Diab Rep. 2003; 3: 37-42.
- Dominguez LJ, Muratore M, Quarta E, Zagone G, BarbagalloM. Osteoporosi e diabete mellito [Osteoporosis and diabetes]. Reumatismo. 2004; 56: 235-241.
- Tang CH, Chou CY. Hip fracture in patients with non-dialysis chronic kidney disease stage 5. Sci Rep. 2021; 11: 20591.
- Covino M, Vitiello R, De Matteis G, Bonadia N, Piccioni A, Carbone Let al. Hip Fracture Risk in Elderly With Non-End-Stage Chronic Kidney Disease: A Fall Related Analysis. Am J Med Sci. 2022; 363: 48-54
- Lee S, Kang Y, Hong YA, Shin SJ, Kwon SH, Chung S, et al. Associated factors of osteoporosis and the impact of osteoporosis on all-cause mortality in incident hemodialysis older patients. Kidney Res Clin Pract. 2026; 45: 110-119.
- Lee SE, Yoo J, Kim KA, Han K, Choi HS. Hip Fracture Risk According to Diabetic Kidney Disease Phenotype in a Korean Population. Endocrinol Metab (Seoul). 2022; 37: 148-158.
- Zidrou C, Vasiliadis AV, Rizou S, Beletsiotis A. Second hip fracture in older adults: incidence and risk factors. Eur J Orthop Surg Traumatol. 2023; 33: 1599-1606.
- Navarrete S, Solar C, Tapia R, Pereira J, Fuentes E, Palomo I. Pathophysiology of deep vein thrombosis. Clin Exp Med. 2023; 23: 645-654.
- Zhang H, Wu F, Sun J, Liu Y, Zhou Z, et al. The impact of frailty evaluation on the risk of venous thromboembolism in patients with hip fracture following surgery: a meta-analysis. Aging Clin Exp Res. 2023; 35: 2413-2423.
- Lin YC, Lee SH, Chen IJ, Chang CH, Chang CJ, Wang YC, et al. Symptomatic pulmonary embolism following hip fracture: A nationwide study. Thromb Res. 2018; 172: 120-127.
- Fukuda T, Imai S, Shimoda S, Maruo K, Nakadera M, Horiguchi H. Aspiration pneumonia and anesthesia techniques in hip fracture surgery in elderly patients: A retrospective cohort study using administrative data. J Orthop Surg (Hong Kong). 2022; 30: 10225536221078622.
- Bon J, Nouraie SM, Smith KJ, Dransfield MT, McDonald ML, et al. Lung-Specific Risk Factors Associated With Incident Hip Fracture in Current and Former Smokers. J Bone Miner Res. 2020; 35: 1952- 1961.
- Jaul E. Assessment and management of pressure ulcers in the elderly: current strategies. Drugs Aging. 2010; 27: 311-325.
- Harris MJ, Brovman EY, Urman RD. Clinical predictors of postoperative delirium, functional status, and mortality in geriatric patients undergoing non-elective surgery for hip fracture. J Clin Anesth. 2019; 58: 61-71.
- Hou M, Zhang Y, Chen AC, Liu T, Yang H, Zhu X, et al. The effects of dementia on the prognosis and mortality of hip fracture surgery: a systematic review and meta-analysis. Aging Clin Exp Res. 2021; 33: 3161-3172.
- McKeag P, Spence A, Hanratty B. ACUTE KIDNEY INJURY FOLLOWING SURGERY FOR HIP FRACTURE. Acta Ortop Bras. 2020; 28: 128-130.
- Agar A, Gulabi D, Sahin A, Gunes O, Hancerli CO, K?l?c B, et al. Acute kidney injury after hip fracture surgery in patients over 80 years of age. Arch Orthop Trauma Surg. 2022; 142: 2245-2252.
- Rantalaiho I, Gunn J, Kukkonen J, Kaipia A. Acute kidney injury following hip fracture. Injury. 2019; 50: 2268-2271.
- Aibar-Almazán A, Voltes-Martínez A, Castellote-Caballero Y, Afanador-Restrepo DF, Carcelén-Fraile MDC, López-Ruiz E. Current Status of the Diagnosis and Management of Osteoporosis. Int J Mol Sci. 2022; 23: 9465.
- Reid IR, Billington EO. Drug therapy for osteoporosis in older adults. Lancet. 2022; 399: 1080-1092.
- Vargas-Franco JW, Castaneda B, Rédiní F, Gómez DF, Heymann D, Lézot F. Paradoxical side effects of bisphosphonates on the skeleton: What do we know and what can we do? J Cell Physiol. 2018; 233: 5696-5715.
- Ferrari S, Langdahl B. Mechanisms underlying the long-term and withdrawal effects of denosumab therapy on bone. Nat Rev Rheumatol. 2023; 19: 307-317.
- Ebina K, Etani Y, Noguchi T, Nakata K, Okada S. Clinical effects of teriparatide, abaloparatide, and romosozumab in postmenopausal osteoporosis. J Bone Miner Metab. 2025; 43: 3-9.
- Seeman E. During aging, men lose less bone than women because they gain more periosteal bone, not because they resorb less endosteal bone. Calcif Tissue Int. 2001; 69: 205-208.
- Eriksen EF, Keaveny TM, Gallagher ER, Krege JH. Literature review: The effects of teriparatide therapy at the hip in patients with osteoporosis. Bone. 2014; 67: 246-256.
- Brixen KT, Christensen PM, Ejersted C, Langdahl BL. Teriparatide (biosynthetic human parathyroid hormone 1-34): a new paradigm in the treatment of osteoporosis. Basic Clin Pharmacol Toxicol. 2004; 94: 260-270.
- Vall H, Patel P, Parmar M. Teriparatide. In: StatPearls. StatPearls Publishing; 2025.
- Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001; 344: 1434-1441.
- Graham T, Shoback DM, Abbott L, Lubitz S, Edelson G, Haider A, Ing SW, et al. Real-World Treatment Patterns and Outcomes in Palopegteriparatide Treatment for Patients With Hypoparathyroidism. Endocr Pract. 2025; 31: 1568-1575.
- Koca CG, Kösehasano?ullar? M. Evaluation of single-dose applied teriparatide effect on bone healing with histomorphometric and micro-ct analysis. J Craniomaxillofac Surg. 2021; 49: 98-103
- Lindsay R, Krege JH, Marin F, Jin L, Stepan JJ. Teriparatide for osteoporosis: importance of the full course. Osteoporos Int. 2016; 27: 2395-2410.
- Stroup J, Kane MP, Abu-Baker AM. Teriparatide in the treatment of osteoporosis. Am J Health Syst Pharm. 2008; 65: 532-539.
- Sanjay S, Pradeep P, Thiyagarajan, Raghavendran B, Ahmed R. Effect of Teriparatide in Fracture Healing in Elderly. J Orthop Case Rep. 2025; 15: 387-393.
- Puvvada CS, Soomro FH, Osman HA, Haridi M, Gonzalez NA, Dayo SM, et al. Efficacy and Safety of Teriparatide in Improving Fracture Healing and Callus Formation: A Systematic Review. Cureus. 2023; 15: e37478.
- Chen JF, Yang KH, Zhang ZL, Chang HC, Chen Y, Sowa H, et al. A systematic review on the use of daily subcutaneous administration of teriparatide for treatment of patients with osteoporosis at high risk for fracture in Asia. Osteoporos Int. 2015; 26: 11-28.
- Tsai JN, Uihlein AV, Lee H, Kumbhani R, Siwila-Sackman E, McKay EA, et al. Teriparatide and denosumab, alone or combined, in women with postmenopausal osteoporosis: the DATA study randomised trial. Lancet. 2013; 382: 50-6.
- Leder BZ, Tsai JN, Uihlein AV, Wallace PM, Lee H, Neer RM, et al. Denosumab and teriparatide transitions in postmenopausal osteoporosis (the DATA-Switch study): extension of a randomised controlled trial. Lancet. 2015; 386: 1147-1155.
- Guyer L, Lehmann O, Wenger M, Oser S, Studer U, Steiner C, et al. Long- term impact of teriparatide on bone mineral density, trabecular bone score, and fracture risk relative to total hip T-score: A two-decade, registry-based cohort study. Bone. 2025; 195: 117445.
- Blick SK, Dhillon S, Keam SJ. Spotlight on teriparatide in osteoporosis. BioDrugs. 2009; 23: 197-199.
- Chiba K, Okazaki N, Kurogi A, Watanabe T, Mori A, Suzuki N, et al. Randomized controlled trial of daily teriparatide, weekly high-dose teriparatide, or bisphosphonate in patients with postmenopausal osteoporosis: The TERABIT study. Bone. 2022; 160: 116416
- Sabaghian T, Delkash P, Rahmannia M, Shahidi Bonjar AH, Centis R, D’Ambrosio L, et al. Efficacy and Safety of Anti-Osteoporotic Agents across CKD Stages: A Meta-Analysis of Randomized Clinical Trials. Kidney Blood Press Res. 2024; 49: 581-587.
- Huang TW, Chuang PY, Lin SJ, Lee CY, Huang KC, Shih HN, et al. Teriparatide Improves Fracture Healing and Early Functional Recovery in Treatment of Osteoporotic Intertrochanteric Fractures. Medicine (Baltimore). 2016; 95: e3626.
- Sato C, Miyakoshi N, Kasukawa Y, Nozaka K, Tsuchie H, Nagahata I, et al. Teriparatide and exercise improve bone, skeletal muscle, and fat parameters in ovariectomized and tail-suspended rats. J Bone Miner Metab. 2021; 39: 385-395.
- Che YJ, Qian Z, Chen Q, Chang R, Xie X, Hao YF. Effects of rehabilitation therapy based on exercise prescription on motor function and complications after hip fracture surgery in elderly patients. BMC Musculoskelet Disord. 2023; 24: 817.
- Agarwal N, Feng T, Maclullich A, Duckworth A, Clement N. Early mobilisation after hip fracture surgery is associated with improved patient outcomes: A systematic review and meta-analysis. Musculoskeletal Care. 2024; 22: e1863.
- Kuru T, Olçar HA. Effects of early mobilization and weight bearing on postoperative walking ability and pain in geriatric patients operated due to hip fracture: a retrospective analysis. Turk J Med Sci. 2020; 50: 117-125.
- Tsuji K, Kimura S, Tateda K, Takahashi H. Protective effect of teriparatide against vancomycin-induced cytotoxicity in osteoblasts. J Orthop Sci. 2023; 28: 1384-1391.
- Lin J, Tjokrosaputro PP, Ang BH, Chew CK, Xu C. Prolonged hypercalcemia induced by teriparatide: a case report and literature review. Osteoporos Int. 2025; 36: 1763-1766.
- Murphy PZ, Iranikhah M, Deas CM, Freeman MK. Fracture Risk Following Discontinuation of Teriparatide: A Review of the Literature. Consult Pharm. 2018; 33: 365-375.
- Pappalardo F, Fantini L, Caruso V. Elevation of transaminases associated with teriparatide treatment: a case report. Eur J Hosp Pharm. 2022; 29: 290-293.
- Wu D, Li L, Wen Z, Wang G. Romosozumab in osteoporosis: yesterday, today and tomorrow. J Transl Med. 2023; 21: 668.
- Lim SY. Romosozumab for the treatment of osteoporosis in women: Efficacy, safety, and cardiovascular risk. Womens Health (Lond). 2022; 18: 17455057221125577.
- Gonnelli S, Caffarelli C. Abaloparatide. Clin Cases Miner Bone Metab. 2016; 13: 106-109.
- Tella SH, Kommalapati A, Correa R. Profile of Abaloparatide and It’s Potential in the Treatment of Postmenopausal Osteoporosis. Cureus. 2017; 9: e1300.
- Kostenuik PJ, Binkley N, Anderson PA. Advances in Osteoporosis Therapy: Focus on Osteoanabolic Agents, Secondary Fracture Prevention, and Perioperative Bone Health. Curr Osteoporos Rep. 2023; 21: 386-400.
- Zhu J, Zhang C, Jia J, Yuan W, Zhang M, Leng H, et al. Effect of weekly teriparatide injections on osteoporotic fracture healing: protocol for a double-blind, randomised controlled trial. BMJ Open. 2021; 11: e043137.