Tuberculin Skin Test Response in Patients Newly Diagnosed with HIV in Cotonou, Benin: A Cross-Sectional Study
- 1. Department of Medicine, National Hospital for Tuberculosis and Pulmonary Diseases, Benin
- 2. Department of Medicine, University of Nigeria Enugu Campus, Nigeria
- 3. Department of Pulmonary and Critical Care Medicine, University of Washington, USA
Abstract
Objective: To determine the prevalence of positive tuberculin skin test among patients newly diagnosed with HIV and to evaluate how it varies across CD4+ T cell ranges.
Methods: A cross sectional study was conducted in 2012-2013 on persons who tested positive for HIV after voluntary counselling and testing in four HIV clinics in Cotonou. Data on Tuberculin skin test (TST) in duration, CD4+T cell count, sex, age and presence of Bacille Calmette Guerin (BCG) post-vaccination scar were collected.
Results: A total of 644 persons living with HIV (PLWH) were included out of which 301 persons (46.7%) had CD4+ T cell below 200 cells/μl. Overall, participants were more often female (69.7%) and had a mean (SD) age of 35.9 (9.8) years. The TST-positivity rate was 34.0 % (30.5- 37.8) when a 5mm TST cut-off was used. In any range of CD4T+ cell count, the odds (unadjusted and adjusted) of having a positive TST were relatively similar across TST cut-offs. Consistently, the odds of having a positive TST decreased with lower levels of CD4+ T cell count.
Conclusion: Only 1 in 3 patients newly diagnosed with HIV in Benin had a positive tuberculin skin test at the time of diagnosis. This needs to be taken into account for the implementation of Isoniazid Preventive Therapy among PLWH in Benin.
Keywords
• Tuberculin skin test
• HIV
• Isoniazid preventive therapy
• Benin
Citation
Wachinou AP, Affolabi D, Iroezindu MO, Agodokpessi G, Ade S, et al. (2016) Tuberculin Skin Test Response in Patients Newly Diagnosed with HIV in Cotonou, Benin: A Cross-Sectional Study. JSM Trop Med Res 1(1): 1001.
INTRODUCTION
In developing countries, Tuberculosis (TB) is the leading cause of deaths among persons living with HIV (PLWH) [1,2]. In 2012, a total of 320,000 deaths due to HIV-associated TB was reported worldwide. The African region bears the heaviest burden of the TB/HIV co-infection considering that almost 75% of PLWH worldwide reside in Africa [2].
Isoniazid preventive therapy (IPT) is one the key components of the TB/HIV treatment strategies recommended by the World Health Organization (WHO) to reduce the burden of TB in PLWH [2,3]. A systematic review of prophylaxis treatment has demonstrated that IPT reduces the risk of active TB by 62% in PLWH with positive Tuberculin Skin Test (TST) while there was no evidence of positive effect for individuals with negative TST [4].Similar findings have been reported from previous studies [5,6]. Based on those findings, the WHO in 1998 recommended IPT for PLHIV with positive TST [7]. However, TST is often not widely performed in resource-constrained settings [3]. For example, in Benin, TST is performed only in two cities (Cotonou and Parakou).
In order to simplify the IPT recommendation and circumvent the challenge of TST, WHO in 2011 revised its guidelines, stating that TST is no longer a requirement for IPT initiation in PLWH [8]. While this new recommendation should foster the implementation of IPT among PLWH, it will also result in IPT being initiated for many individuals who cannot benefit from it (those with negative TST). Furthermore, many of these latter individuals may actually experience negative side effects from the Isoniazid and/or the possibility of development of INH resistance [3]. In Benin, the proportion of PLWH who are TST positive and who could then benefit from IPT if implemented systematically is unknown.
We report on a cross-sectional study to determine the prevalence of tuberculin skin test positivity in patients newly diagnosed with HIV and to evaluate how it varies across CD4+ T cell ranges.
MATERIALS AND METHODS
Study Setting and Population
This study was nested with in a larger cross-sectional study which was conducted from June 2012 to April 2013 at Cotonou, the economic capital of Benin Republic. Participants were persons who voluntarily presented at four HIV clinics for HIV counselling and testing. We consecutively enrolled HIV-infected individuals who were aged 18 years or more who did not report any history of tuberculosis. HIV antibody test was done using Determine (Abbott, Japan) and Genie III (Biorad, France) was used as confirmatory test.
Ethical consideration
The study was approved by the National Ethics committee of Benin. All participants provided written informed consent.
Data collection
CD4+ T cell count: We assessed CD4+ T-lymphocyte count (CD4+T cell count) using a Cyflow apparatus (Partec, Germany) and subsequently categorized CD4+ T cell count into three groups: <200 cells/µl, 200-499 cells/µl and ≥500 cells/µl according to the Centers for Disease Control and Prevention (CDC) classification system [9].
TST in duration: To assess tuberculin skin test positivity, trained health workers initially injected patients with 0.1 ml of Tuberculin PPD RT 23 (Statens Serum Institute, Copenhagen, Denmark) intradermally on the ventral aspect of the left forearm. After 3-5 days, we assessed TST in duration with the ballpointpen method using a plastic ruler graduated in millimetres. We established four TST positive cut-offs of 0mm, 5mm, 10 mm and 15 mm.
Additional variables: We collected demographic data such as sex and age (years) by self-report. We assessed Bacille Calmette-Guérin (BCG) vaccination status by checking the presence or absence of BCG post-vaccination scar.
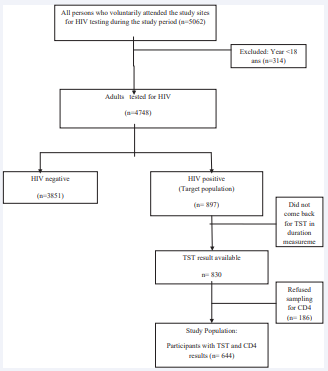
Recruitment process: All the participants who fulfilled inclusion criteria were recruited. Among the 897 patients who tested positive for HIV during the period of our study, 830 had TST measurement (Figure 1).
Figure 1: Study population flow diagram
From these, we excluded 186 for whom a CD4+ T cell count was not available, leaving a final study sample of 644 patients.
Statistical analysis: We analysed the data with STATA 12 (Stata Corp LP, College Station, Texas).
We used Chi- square for comparison of TST-positivity prevalence across CD4+ T cell count ranges. Logistic regression was used to quantify the magnitude of the relationship between TST cut-offs and CD4 level adjusting for potential confounders. Level of significance was set at 5%.
RESULTS
Characteristics of study participants
Of the 644 patients newly diagnosed with HIV who were included in the analysis, 301 (46.7%) had low CD4+T cell count (<200 cells/µl) and 69.7 were female. Mean (SD) age was 35.9 (9.9) years, with those having low CD4+ T cell counts being somewhat older with 37.3(9.8) years (p=0.0007). Participants aged between 25 and 45 years represented almost 75% (474) of the participants at any of the CD4+ T cell count ranges. Other characteristics including sex did not differ across CD4+ T cell ranges. BCG scar was present less often among patients with low CD4+ T cell count. Table (1)
Table 1: Baseline characteristics of study participants.
| CD4 count (cells/µl) | <200 | 200-499 | ≥500 | p | |||
| Number of participants (n=644) | |||||||
| Age (yrs), mean (SD) | 37.3 | (9.8) | 35.5 | (9.7) | 33.9 | (8.3) | 0.0007 |
| Age categories (yrs), n (%) | |||||||
| 18-24 | 19 | (6.3) | 14 | (10.1) | 21 | (10.3) | 0.005 |
| 25-34 | 107 | (35.5) | 59 | (42.2) | 105 | (51.5) | |
| 35-44 | 113 | (37.5) | 42 | (30.2) | 48 | (23.5) | |
| 45-54 | 37 | (12.3) | 18 | (12.9) | 18 | (8.8) | |
| ≥ 55 | 25 | (8.3) | 6 | (4.3) | 12 | (5.9) | |
| Sex, F (%) | 200 | (66.4) | 97 | (69.8) | 152 | (74.5) | 0.15 |
| BCG scar present, n (%) | 200 | (66.4) | 113 | (81.2) | 152 | (74.5) | 0.0035 |
| CD4 count, mean (SD) | 94.4 | (56.2) | 266.3 | (45.2) | 561.9 | (172.7) | 0.0000 |
| Total per CD4 range, n (%) | 301 | (46.7) | 139 | (21.6) | 204 | (31.7) | |
Prevalence of TST positivity across CD4 level ranges
Overall, the prevalence of a positive TST was 38.2%, 34.0 %, 25.3% and 15.4% for cut-offs of 0 mm, 5 mm, 10 mm and 15 mm, respectively (Table 2).
Table 2: Variation of TST-positivity with different cut off rate across CD4 levels.
| CD4 cell count (cells/µl) | <200 | 200-499 | >=500 | Total | p |
| (n=301) | (n=139) | (n=204) | (n=644) | ||
| TST cut-off | |||||
| ≥0 mm | 29.9% | 43.2% | 47.1% | 38.2% | 0.002 |
| ≥5 mm | 25.9% | 38.8% | 42.6% | 34.0% | 0.002 |
| ≥10 mm | 18.9% | 29.5% | 31.9% | 25.3% | 0.002 |
| ≥15 mm | 10.6% | 16.5% | 21.6% | 15.4% | 0.003 |
Regardless of the cut-off used, positivity was consistently greatest for those with CD4 cell counts ≥ 500 cells/µland least for those with cell counts <200 cells/µl.The corresponding odds ratios, adjusted for age category, sex, presence of BCG scar, are shown in Table (3)
Table 3: Multivariable Odds Ratio for TST Positivity by CD4 level determined by Logistic Regression.
| Adjusted OR (95% CI) | |||
| CD4 level | |||
| <200 | [200-500] | ≥500 | |
| TST>0 | 0.43 | 0.82 | 1 |
| 0.29-0.62 | (0.53-1.28) | ||
| TST≥5 | 0.42 | 0.80 | 1 |
| (0.28-0.62) | (0.51-1.26) | ||
| TST≥10 | 0.45 | 0.84 | 1 |
| 0.29-0.69) | (0.52-1.36) | ||
| TST≥15 | 0.39 | 0.69 | 1 |
| (0.23-0.65) | (0.39-1.21) | ||
| *Adjusted for age category, sex, presence of BCG scar | |||
and were very similar across TST cut-offs, particularly for cut-offs of 0, 5, and 10mm.
Compared to those with CD4+ T cell counts > 500 cells/µl, patients with CD4+ T cell counts < 200 cells/µlhad odds ratios for a positive TST ranging from 0.39 to 0.45, and all were highly statistically significant. Odds ratios for patients with CD4+ T cell counts between 200 and 499 cells/µl ranged from 0.69 to 0.82 and were not statistically significant.
DISCUSSION
We found an overall prevalence of positive TST of 34% when a 5mm cut-off was used among HIV-infected patients without history of tuberculosis in Cotonou. The prevalence of positive TST was associated with ranges of CD4+ T cell count such that the prevalence of positive TST decreased with lower CD4+ T cell counts. In addition, the odds of having a positive TST were generally similar at any TST in duration cut-off for all participants in a specific range of CD4+ T cell count.
It has been shown that IPT is more effective in PLWH with positive TST [4,6,10]. As the WHO revised guidelines has stipulated that performance of TST is no longer mandatory before initiating IPT [8], it is important to know: i) what is the prevalence of TST positive in HIV patients ii) how this prevalence varies across CD4 counts given the fact that almost half, especially in the context of resource-constrained settings. If WHO revised guidelines are applied in extenso, overall, our data suggest that only approximately 1 in 3 PLHIV (1 in 4 in PLWH with CD4+ T cell count <200 cells/µl) would benefit from IPT if implemented without TST assessment. Yet, as shown in our study, almost half of patients newly diagnosed with HIV in our setting, belong to that low range of CD4+ T cell count. The question that needs to be addressed here is: “is it ethical and cost-effective to set up an intervention knowing that only one third of the targeted population could derive benefit from it?”
A comprehensive cost effectiveness analysis of measures to control TB/HIV done in South Africa suggests that IPT used in a targeted or untargeted way is cost-effective compared to not implementing IPT [11]. This may not automatically apply to other settings with lower prevalence both for HIV infection and tuberculosis. Cost-effectiveness of IPT implementation in HIV patients in our setting is therefore needed to inform decision making process.
Other strategies not necessarily based on IPT may also need to be considered in situations where TST is not available or could not be done in PLWH.
For Howard and et al., the low prevalence of positive TST among PLWH especially in persons with CD4T+ cell count <200 cells/µl is predominantly due to energy (i.e. an all-or-nothing phenomenon) rather than due to a gradual diminution in TST diameter during progressive immunodeficiency [12]. A similar finding was made by Cobelens et al., in PLWH diagnosed with active TB, suggesting that the TST reactivity is similar in PLWH whether in the context of TB infection or active TB [13]. In PLWH with active TB, the all-or-nothing phenomenon has been explained by antigen-specific responsiveness and an association with interleukin-10 (IL-10)–mediated suppression of T cell proliferation [14]. Whether this could be generalized to PLWH with latent TB infection is unknown.
Our data also suggest that increasing the cut-off from 0 mm or 5 mm to 15 mm does not significantly increase the proportion of PLWH who test positive for TST. This finding is consistent with data from a systematic review based on 34 different study populations [12]. In a study on patients with active TB, a similar finding has been reported by Cobelens et al., [13] suggesting that our finding could be generalized in PLWH notwithstanding if they have TB infection or active TB disease.
The strengths of our study include the reasonably large number of persons newly diagnosed with HIV infection recruited. In addition, all the health workers who carried out the tuberculin skin testing were trained and supervised throughout the data collection to ensure good quality of the TST results. Moreover, the study was reported following the STROBE (Strengthening the reporting of Observational Studies in Epidemiology) guidelines.
Nevertheless, our study had some limitations. We did not perform laboratory investigations to rule out active TB diagnosis. However, we believe that in routine context, a symptombased screening could be sufficient to raise suspicion of active tuberculosis in PLWH. As demonstrated by Getahun et al., a clinical screen based on the absence of all the symptoms of current cough, night sweats, fever or weight loss can identify a subset of PLWH who have a very low probability of having TB disease. This screening rule has a sensitivity of 79% and a specificity of 50% and the negative predictive value can reach 97.7% (95%CI: 97.4–98.0) [15].
In conclusion, we found that overall 34 % of persons newly diagnosed with HIV in Benin had positive TST when a 5 mm cut-off was considered, suggesting that only 1 in 3 PLWH could really derive benefit from IPT if implemented without knowing TST status in advance. A cost effectiveness analysis based on data from our setting is therefore needed to inform the decision making process for IPT implementation among PLWH.
AUTHORS CONTRIBUTIONS
APW and AD designed the study, supervised data collection and drafted the manuscript; JT and RA were involved in data collection; WB performed data analysis; DHA helped for data analysis and revised the manuscript; MO, GA, SA, GA, AS critically revised the manuscript for intellectual content. All authors read and approved the final manuscript.
ACKNOWLEDGMENTS
We want to acknowledge Cecilia Patino, William M. Vollmer, Sandra Money, Stephen Gordon, Sonia Buist for their support through the 2014 Global Course organised by the American Thoracic Society.
REFERENCES
2. Global Tuberculosis Report. Geneva, Switzerland. 2013.
12. Kerkhoff AD, Gupta A, Samandari T, Lawn SD. The proportions of people living with HIV in low and middle-income countries who test tuberculin skin test positive using either a greater or equal to 5 mm or a greater or equal to 10 mm cut-off: a systematic review. BMC Infect Dis. 2013; 13: 307.