Cutaneous Plasmacytomas Secondary to Nonsecretory Multiple Myeloma
- 1. Department of Dermatology, Hospital de Braga, Portugal
- 2. Department of Oncology, Hospital de Braga, Portugal
- 3. Department of Pathology Anatomy, Hospital de Braga, Portugal
Abstract
Introduction: Multiple myelomais characterized by neoplastic prolife ration of plasma cells in bone marrow. Cutaneous involvement occurs in only5 to 10% of patients and includes specific rare lesions such as multiple cutaneous plasmacytomas in advanced stages of disease.
Case report: Male patient, 44years-old, diagnosed with nonsecretory multiple myeloma (Durie-Salmon stage III, International Staging System Stage I) for about18 months. No chromosomal or cytological characteristics of poor prognosis were found. Complete response was achieved after conventional chemotherapy, followed by intensive chemotherapy and autologous transplant of stem cells. Ten months after treatment, some erythemato-violaceous nodules and tumors with 2 to 7cmin diameter appeared, with elastic consistency and smooth surface with well-defined limits, slightly painful, localized in lower back, right leg and right forearm. The pathological examination of a nodule was consistent with metastatic cutaneous plasmacytoma, with intense and diffuse expression of CD138and cyclinD1 and frequent expressionforCD79a. Patient was treated with interferon-alpha and subsequently radiotherapy. At the third week of radiotherapy the patient suffered sudden death at his home (autopsy was inconclusive).
Discussion: We present this case because it has occurred in a young adult, with the rare association to the nonsecretory multiple myeloma and for the presentation of exuberant cutaneous metastatic lesions after systemic chemotherapy and autologous transplant of stem cells. These lesions are a sign of poor prognosis and, in this case, the first sign of disease progression, exclusively cutaneous, without bone marrow involvement.
Keywords
Cutaneous plasmacytomas, Multiple myeloma
Citation
Araújo C, Marques H, Fernandes JC, Pardal A, Brito C (2014) Cutaneous Plasmacytomas Secondary to Nonsecretory Multiple Myeloma. J Dermatolog Clin Res 2(3): 1022.
INTRODUCTION
Multiple Myeloma (MM) is a malignant hematologic disorder characterized by monoclonal proliferation of plasma cells. This entity affects men more often than women and tends to occur in older adults (mean age of 68 years) [1].
Cutaneous involvement associated with multiple myeloma occurs in a percentage that varies from 5 to 10% of cases and the lesions are classified in nonspecific and specific [2,3].
The nonspecific cutaneous manifestations of multiple myeloma are shown in table 1.
Table 1: Nonspecific cutaneous manifestations of multiple myeloma.
| Amyloidosis |
| Purpura, |
| Alopecia |
| Ichthyosiform dermatitis |
| Raynaud's phenomenon, |
| Cold urticarial |
| Pyodermagangrenosum |
| Leukocytoclastic vasculitis |
| Anhydrosis |
| Sclerodermiform lesions |
| Subcornealpustulardermatosis |
| Scleromyxedema |
| S.Sweet |
| POEMS (polyneuropathy, organomegaly, M-protein and skin lesions) |
| Angioedema with C1 inhibitor deficiency |
| Plane xanthomas |
| Necrobioticxanthogranuloma |
| Follicular hyperkeratosis |
The specific cutaneous lesions related to multiple myeloma are the extramedullary cutaneous plasmacytomas, which was first reported by Hedinger in 1911 [4]. They can occur by lymphatic or vascular spread or, more frequently, by direct extension to the skin, from underlying bone lesions. In some cases, cutaneous extra medullary plasmacytom as has been described as the presenting sign of multiple myeloma [5].
Any area of the skin can be involved, but it has been reported most frequently on the trunk and abdomen followed by face, scalp, neck and extremities [6-12]. Unusual localizations have been described, including eyelid, tongue, and scrotum and perianal region [13]. Multiple lesions are the rule, although solitary lesions have also been recorded.
Morphologically, cutaneous plasmacytomas usually consist of erythematous nodules that may ulcerate or dome-shaped and smooth-surfaced plaques ranging from 1 to 5cm in diameter. Histopathological examination of the cutaneous plasmacytomas in MM revealed two patterns: nodular or diffuse interstitial infiltration [6]. The typical pattern represented by a dense monomorphic dermal plasmacytic infiltrate which is usually separated from the overlying epidermis.
Cutaneous plasmacytomas in patients with multiple myeloma is a rare event, occurring mostly in advanced disease or relapse. Usually indicates aggressive behavior and short survival, coursing to death within 12 months after diagnosis [14].
This patient was diagnosed with an unusual nonsecretory multiple myeloma (Durie-Salmon stage III, International Staging System I), who ten months after complete response with conventional chemotherapy, intensive chemotherapy and autologous transplant of stem cells, presented cutaneous plasmocytomas.
CASE PRESENTATION
A 44years old man, caucasian, presented with lumbago with eight month of evolution. In July 2010 was diagnosed as having nonsecretory multiple myeloma initially presented with osteolytic lesions and vertebral plasmacytoma. There was no relevant personal or family history of malignancy or chronic pharmacologic therapy.
Initial radiological exam was performed, which showed a pathologic fracture over the L3 vertebral body and multiple mild osteolytic lesions over the ribs, C2, C3, D2, L1 e L4. At that time, he was submitted to corrective surgery. Complaints of progressive malaise, loss of appetite, weight loss (more than 10Kg in 6 months) and generalized bone pain subsequently developed.
Initial analytic characteristics are shown in table 2.
Table 2: Initialanalytical characteristics
| Hemoglobin | 15g/dl (13.5-17.0) |
| Leucocytes | 7.7x103 /uL (4-10.5) |
| Platelets | 332x103 /uL (166-308) |
| PTH | <2.5 pg/ml (14-72) |
| Ca2+ | 11.3 mg/dl (8.4-10.2) |
| Lactic dehydrogenase | 411 U/L (240-488) |
| B2-microglobulin | 1760 ng/mL (800-2200) |
| C reactive protein | 10.30 mg/L (<5) |
| Alkaline phosphatase | 137 U/L (40-130) |
| Total protein | 7.6 g/dL (6.4-8.7) |
| Albumin | 4.9 g/dL (3.5-5.2) |
| A/G ratio | 1.6 |
| Immunoglobulin G (IgG) | 685 mg/dl (700-1600) |
| IgM | 24 mg/dl (40-230) |
| IgA | 147 mg/dl (70-400) |
In immunelectrophoresis, no monoclonal bands were observed. Bence-Jones protein was undetectable. There was no renal insufficiency.
Examination of bone marrow from the iliac spine revealed a cellularity proportional to patient age without relevant morphologic alterations and < 10% plasma cells (CD 138+, MUM+ and kappa+).
Histopathologic exam showed bone tissue partly substituted by large aggregates of neo plastic cells with scarce and poorly defined cytoplasm and irregular, hipercromatic and multi lobular nuclei. Immunohistochemical study revealed neo plastic cells with intense and diffuse expression of CD 138 (Figure 1)

Figure 1 Neoplastic cells with intense and diffuse expression of CD 138 in bone marrow biopsy.
and table 1 cycline D1 and also frequent expression of CD 79a. No expression to CD45, CD3, CD 20 or mieloperoxidase.
There weren’t found chromosomal or cytological characteristics of poor prognosis by Fluorescence In Situ Hybridization (FISH).
After clinical staging, it was concluded to be nonsecretory multiple myeloma (Durie-Salmon stage III, International Staging System I) and chemotherapy was initiated with cyclophosphamide, bortezomib and dexamethasone between 08/2010 and 11/2010 followed by intensive chemotherapy and autologous transplant of stem cells in 01/2011 with complete response. A reassessment of bone marrow showed plasma cells < 10% and serial evaluation of protein electrophoresis was negative.
Ten month safter treatment, some erythemato-violaceous nodules and tumors with 2 to 7cmin diameter appeared, with elastic consistency and smooth surface with well-defined limits, slightly painful, localized in lower back, right leg andright forearm (Figure 2).
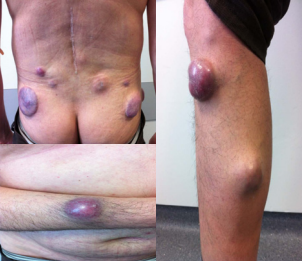
Figure 2 Metastatic cutaneous plasmacytoma localized in lower back, right leg and right forearm.
The pathological examination of a nodule was consistent with metastatic cutaneous plasmacytoma, with intense and diffuse expression of neo plastic cells for CD138 and cyclinD1 and frequent expression for CD79a. Some lesions underwent spontaneous remission and some appeared in other locations.
No medullary plasmacytosis or serum immunoelectrophoretic changes were observed; urine immunofixation was also negative. Analytic characteristics at time of tumor recurrence are shown in table 3.
Table 3: Analytical characteristics at time of tumor recurrence
| Hemoglobin | 13.8g/dl (13.5-17.0) |
| Leucocytes | 4.7x103 /uL (4-10.5) |
| Platelets | 252x103 /uL (166-308) |
| Ca2+ | 8.6 mg/dl (8.4-10.2) |
| Lactic dehydrogenase | 204 U/L (240-488) |
| B2-microglobulin | 2143ng/mL (800-2200) |
| Total protein | 6.7 g/dL (6.4-8.7) |
| Albumin | 3.9 g/dL (3.5-5.2) |
| A/G ratio | 1.6 |
| Immunoglobulin G (IgG) | 525 mg/dl (700-1600) |
| IgM | 16.9 mg/dl (40-230) |
| IgA | 147 mg/dl (70-400) |
A further skeletal survey revealed enlargement of some osteolytic lesions in D7, L5, sacrum and iliac crests.
Patient was submitted to interferon-alpha and subsequently radiotherapy. At the third week of radiotherapy the patient suffered sudden death at his home (autopsy was inconclusive for the cause of death).
DISCUSSION
There are 4 types of plasma cell neoplasia: classic multiple myeloma (MM), extra medullary plasmacytoma without MM, solitary plasmacytoma of bone, and plasma cell leukemia. Cutaneous involvement may be seen in all 4 types of plasma cell neoplasia. Extra Medullary Plasmacytomas (EMP) is seen in approximately 7 percent of patients with multiple myeloma at the time of diagnosis and additional 6 percent of patients will develop extra medullary plasmacytomas later in the disease course [15-16].
Cutaneous involvement in multiple myeloma has been recognized in association with all classes of myeloma proteins: IgG, IgM, IgA and IgD; heavy chains; and kappa and lambda light chains [17]. Evidence suggest that subtype IgG is probably the most common producing cutaneous dissemination as is the most frequent immunoglobulin among myeloma. However, some authors have mentioned a greater predominance of IgA (35%) in cases with cutaneous infiltration [3,18]. Those with IgD as well as light chain subtypes and nonsecretors seem to have the most aggressive behavior [6].
Secondary cutaneous plasmocytoma usually represents dissemination of late stage multiple myeloma [6-10,18].
Non secretory myeloma is a rare variant of multiple myeloma (1-5%). It is characterized by the absence of M-protein in the serum and urine, bone marrow plasmacytosis and end organdamage. Of these, the majority (approximately 85 percent) will have M-protein that can be detected in the cytoplasm of the neoplastic plasma cells by immunochemistry, but have impaired secretion of this protein. The other 15 percent do not have immunoglobulin detectable in the plasma cells (ie, nonproducer myeloma). Hypogamamaglobulinemia has frequently been described and may be a sign of defective immunoglobulin production. There is minimal to no risk of myeloma kidney, but they are at risk for other complications of multiple myeloma [19].
We noted the case because has occurred in a young adult, with the rare association to the nonsecretory multiple myelomaand for the presentation of exuberant cutaneous metastatic lesions after multiple therapeutic regimens. This patient presented initially with vertebral plasmacytoma causing vertebral fracture and osteolytic lesions, symptoms of general malaise and bone pain.
Immunohistochemical staining in extramedullaryplasmacytoma shows the typical monoclonality of the neoplastic cells, characterized by a “myeloma–like” immunophenotype [20-23].
In our case neoplastic plasma cells of MM involving the skin showed a fairly homogeneous immunophenotype characterized by strong immunorreactivity for CD 138 andcycline D1 and frequent expression of CD 79a. It remains to be demonstrated whether CD138 immunoexpression is related to prognosis [24].
Cutaneous metastatic spread was evident despite bone marrow remission. Treatment with an autograft may have contributed to the cutaneous relapse. A few cases of cutaneous metastatic plasmacytomas have developed after chemotherapy for myeloma and autologous stem cells [25]. After myeloablative therapy, re-infusion to reconstitute the marrow could seed myeloma cells into the extramedullary tissues and altered interaction between cytokines, adhesion molecules and cellular immunity [26]. Early relapse observed in this case pose three hypothesis: skin tissue may work as a sanctuary to malignant plasma cells; immune deficiency state induced by autologous transplantation may promote tumor proliferation and autograft collected cells may contain myeloma cells which could have a different organ tropism and seeded in the skin. A stem cell harvest purged of myeloma cells, by monoclonal antibodies, might theoretically avoid this possible complication of treatment [27].
Although novel effective agents are available for myeloma treatment, the management of cutaneous plasmacytomas is disappointing, probably by biological differences between intramedullary and extramedullary disease. It depends on various factors including the number of lesions, their location, stage of underlying multiple myeloma and whether there is coexistent bone marrow involvement [26]. Single nodules can be resected but radiotherapy is effective for large or deep lesions [6].Other treatments include intralesional steroid and α-TNF [28,29]. In the presence of extensive involvement, chemotherapy in conjunction with radiotherapy remains the treatment of choice [6]. Adjuvant α-interferon has been tried as primary treatment but also in relapsed myeloma after an autologous stem cell transplant. The mechanisms underlying antitumor activity of α-IFN may be mediated by antiproliferative and immunomodulatory effects but biologic heterogeneity in myeloma may have important clinical implications for response to IFN. The initial results reported superior progression-free survival and overall survival in patients receiving IFN-α, however, with longer follow-up, the benefit has not been sustained [30-32]. The integration of IFN with other therapy following maximal cytoreduction has not been adequately tested.
Extramedullary cutaneous plasmacytomaoccurring late in the diseasecourse at multiples sites suggests haematogeneous spread and carries a poor prognosis. Our case illustrates the clinical aggressiveness of a non secretory multiple myeloma relapsing and the short survival once skin metastases develop, in this case,the first signof disease progression, exclusively cutaneous, without bone marrow involvement.
CONCLUSION
In conclusion, cutaneous involvement by multiple myeloma occurs infrequently and usually heralds a poor prognosis regardless of the various forms of therapy.
The application of immunohistology to the spectrum of plasma cell disorders and an expanding spectrum of cytogenetic abnormalities enhanced by fluorescent in situ hybridization technology reshape the way we define plasma cell disorders as their clinical relevance becomes clearer.
The diagnosis of cutaneous extramedullary metastatic plasmacytoma is important, particularly in bone marrow– negative patients, since it indicates persistence or progression of the disease and for which new therapeutic options are needed.
REFERENCES
2. BULEFARB SM. Cutaneous manifestations of multiple myeloma. AMA Arch Derm. 1955; 72: 506-522.
4. Hedinger E. ZurFrage des Plasmacytomas. Frankfurt Z Path. 1911; 7: 343-350.
9. Tüting T, Bork K. Primary plasmacytoma of the skin. J Am Acad Dermatol. 1996; 34: 386-390.
17.Piette WW. Myeloma, paraproteinemias, and the skin. Med Clin North Am. 1986; 70: 155-176.
21.Petruch UR, Horny HP, Kaiserling E. Frequent expression of haemopoietic and non-haemopoietic antigens by neoplastic plasma cells: an immunohistochemical studyusing formalin-fixed, paraffinembedded tissue. Histopathology. 1992; 20: 35–40.









































































































































