Drug Survival Rates of Systemic Therapies in the Management of Adult Atopic Dermatitis
- 1. Health, Royal Melbourne Hospital, Melbourne, Australia
- 2. Department of Dermatology, Royal Melbourne Hospital, Australia
- 3. Department of Epidemiology and Preventive Medicine, Monash University, Australia
- 0. Both authors contribute equally
Abstract
Atopic dermatitis, Atopic eczema, Immunosuppressive, Drug survival, Mycophenolate mofetil
KEYWORDS
• Atopic dermatitis
• Atopic eczema
• Immunosuppressive
• Drug survival
• Mycophenolate mofetil
CITATION
Lolatgis H, Anderson L, Martyres R, Liew D, Gillard J, et al. (2017) Drug Survival Rates of Systemic Therapies in the Management of Adult Atopic Dermatitis. J Dermatolog Clin Res 5(4): 1107
ABBREVIATIONS
AD: Atopic Dermatitis; FLG: Filaggrin Gene; SCORAD: Scoring Atopic Dermatitis; DLQI: Dermatology Life Quality Index.
INTRODUCTION
Atopic dermatitis (AD) is a chronic inflammatory skin disease characterized by pruritic, scaly, and relapsing lesions. The disease affects 3-7% of adults and 15-20% of children [1]. It often has a genetic predisposition. Mild cases respond to a combination of therapies consisting of topical glucocorticoids, antibiotics, topical calcineurin inhibitors, and antihistamines.
In moderate to severe cases of atopic dermatitis, systemic immunosuppression using steroid sparing agents is often required to achieve adequate disease control. The time to drug discontinuation, or ‘drug survival,’ with these immunosuppressive agents is often limited by an array of side effects and/or ineffectiveness.
The pathogenesis of atopic dermatitis is multifactorial. However, it is currently accepted that a defective skin epidermal barrier results in increased trans-epidermal water loss and increased colonization and penetration by microorganisms and allergens, evoking inflammatory responses [2]. A strong association is recognized with loss-of-function mutations in the filaggrin gene (FLG) which leads to impairment in skin barrier formation, thereby facilitating trans-epidermal penetration of allergens and allergic sensitization [3]. T helper type 2 (Th2) cells, Th1, Th17 and Th22 cells are also involved in the pathogenesis [4,5].
For disease that is resistant to, or minimally improved by emollients, topical glucocorticoids and calcineurin inhibitors, systemic treatment options are considered. Oral corticosteroids are used as first line for acute flares and initial stabilization. Longterm maintenance for chronic or recurrent disease requires oral steroid sparing agents. Available agents include cyclosporine, azathioprine, methotrexate, and mycophenolate mofetil.
In Australia, the decision of which drug to use as first line therapy is individual-based, influenced predominantly by the patient’s comorbidities and the various side effect profiles of each drug. The Australian Pharmaceutical Benefits Scheme also has an influence on choice. For example, cyclosporine remains the only steroid-sparing agent listed and subsidized for atopic dermatitis. As such, it has traditionally been used first-line in conjunction with, or following oral corticosteroid treatment. Furthermore, the historical high cost associated with mycophenolate mofetil has deterred its use in the past. Recent changes to the availability and patency of mycophenolate have made it a more economical agent to use.
When prescribing these treatments, dermatologists must consider the likelihood that the treatment they are prescribing will continue to be a suitable option in the long term.
Drug survival’ is defined as the period in which a given drug continues to be an adequate treatment for a specific patient. ‘Drug survival’ is determined by whether the patient adheres to the drug and may be limited by discontinuation secondary to safety issues or lack of effectiveness. The decision to discontinue therapy is decided on a case-by-case basis.
The rationale behind undertaking this study is centered around the lack of effective systemic agents available and the pattern of recurrent failure for these agents to achieve longer term success. The aims of the study are to identify which agents are surviving the longest and why, and formulating a suggested algorithm for treatment using this data.
MATERIALS AND METHODS
We performed a retrospective cohort analysis of 44 patients in our clinic aged over 18 years of age. To be included in the study, patients were deemed to have moderate or severe atopic eczema by a dermatologist, and were receiving systemic immunosuppressive maintenance therapy (cyclosporine, azathioprine, methotrexate, or mycophenolate mofetil) through the Department of Dermatology outpatient clinic at The Royal Melbourne Hospital.
Patients had to have at least a 16 month diagnosis of moderate or severe atopic eczema based on the diagnostic criteria discussed by Hanfin and Rajka [6].
The enrollment period was between January 2012 and April 2015. Patients in the cohort who had been on immunosuppressive therapy prior to this date as documented in medical records also had these treatment series included. This included data for some patients for up to ten years.
Patients who received their oral immunosuppressive agents for reasons other than atopic dermatitis were excluded. Patients continued on topical treatments and systemic prednisolone as deemed necessary and appropriate by the treating dermatologist. Those who used prednisolone simultaneously were included. Those who used a combination of two steroid sparing immunosuppressive agents at any one time were excluded.
Patients were reviewed every 3-6 months for clinical assessment of disease and to record side effects and toxicity related to their treatment. Scoring Atopic Dermatitis (SCORAD), Dermatology Life Quality Index (DLQI) data at each review were inconsistently recorded.
The primary outcome measured was systemic treatment discontinuation for any cause. This was assessed for and determined by the clinician. These were recorded as four categories (1=toxicity, 2=ineffectiveness, 3=non-compliance, 4=remission).
Survey and chart review data were recorded for each clinic visit. Demographic data, number of treatments tried, length of time on each treatment, and the reason for treatment cessation were analyzed using simple descriptive statistics.
RESULTS
The final sample size consisted of forty-four patients. One hundred and two patients were excluded from the study because they did not meet the inclusion criteria outlined above. Patient characteristics, percent of patients exposed to each drug, and average length of time exposed to each drug are shown in tables 1 and 2,
|
Table 1: Patient Characteristics. |
|
|
|
Number (%) of patients (n = 44) |
|
Male sex |
31 (70.5) |
|
Female sex |
13 (29.5) |
|
Mean age |
43 years |
|
Personal history of atopy |
39 (44.3) |
|
Atopic dermatitis from childhood |
22 (50) |
|
Table 2: Overview of individual therapies: Number (%) of patients (n=44) and average length of time exposed to each drug. |
||||
|
|
Treatment Type |
|||
|
|
Cyclosporine |
Azathioprine |
Methotrexate |
Mycophenolate Mofetil |
|
Number (%) of patients exposed to drug (n=44) |
30 (68) |
20 (45) |
26 (59) |
18 (41) |
|
Mean length of time exposed to drug (weeks) |
119 |
109 |
109 |
58 |
respectively. As expected, cyclosporine was the most commonly trialed drug amongst the cohort, and probably due to more recent access, mycophenolate was trialed the least. On average, each patient was exposed to 2.6 of the 4 systemic immunosuppressant drugs during their clinical course, which suggests poor long term adequacy overall. Reasons for individual treatment cessation are highlighted in figure 1.

Figure 1 Reasons for treatment cessation recorded under four categories: toxicity, ineffectiveness, non-compliance and remission (a) Cyclosporine (n=24) (b) Azathioprine (n=18) (c) Methotrexate (n=16) (d) Mycophenolate Mofetil (n=7).
Interestingly, of the 18 patients who were treated with mycophenolate mofetil, only 4 stopped treatment due to adverse events or side effects. Two patients achieved remission with mycophenolate (11%). 11 of the 18 patients remained on the drug at the time of last data collection (61%), indicating a likelihood of treatment success. The trend towards survival of mycophenolate is further demonstrated in the Kaplan-Meier curve(Figure 2).
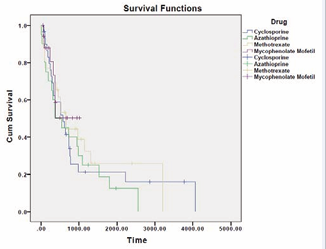
Figure 2 Kaplan-Meier Curve demonstrating the trend towards survival mycophenolate, compared with methotrexate, cyclosporine, and azathioprine.
The results for mycophenolate mofetil were skewed because a large proportion of our patient population who had tried mycophenolate mofetil (61%) remained on the drug at the time of data collection, suggesting success of therapy. This group could not be included in the calculation measuring length of time to cessation. The sample size for mycophenolate mofetil discontinuation was subsequently very small (5 patients), showing an average survival time of 52 weeks for the cases that failed treatment.
DISCUSSION
Drug survival’ can be defined as the period during which a drug continues to be an effective treatment for a specific patient. While numerous studies have discussed the advantages and disadvantages of systemic atopic dermatitis treatments, fewer have investigated the reasons for discontinuation of treatment. Our study has primarily looked at the length of time each drug has been used for therapy, and the reasons behind discontinuation of use.
Overall, cessation was predominantly a result of ineffectiveness, side effects, or toxicity. In the case of all four treatments, a total of 94 treatment series were administered. Only 6 out of the 94 ceased due to achieving remission. Our study results mirror previous findings: the discontinuation of systemic therapies in the treatment of severe AD is primarily a result of gradual loss of effectiveness, followed by side effects and toxicities [7].
In our study, 61% of patients treated with mycophenolate mofetil have continued treatment to date, compared to 20%, 10%, and 30% for cyclosporine, azathioprine, and methotrexate, respectively. These trends are illustrated in the Kaplan-Meier analysis in figure 2. We note that these results could have been influenced by differing sample sizes and length of time of drug exposure.
However, recent trends in management have seen an increase in the use of mycophenolate mofetil due to fewer side effects when compared with other systemic agents [8-10]. Studies directly comparing mycophenolate with cyclosporine have shown that the clinical efficacy of mycophenolate as maintenance therapy isas effective as cyclosporine, with a longer relapse free period and a more favorable side effect profile. Patients using cyclosporine showed an increase in SCORAD, used more prednisolone, and experienced a lower quality of life than patients in the mycophenolate group [11].
It is clear that more targeted therapies with lower side effect profiles are required in order to better treat patients with this encumbering disease. Recent trials investigating new immune modulatory agents in the treatment of atopic dermatitis have shown great promise. These have included the IL-4 and IL-13 inhibitor Dupilumab [12], and JAK-STAT pathway inhibitor Tofacitinib [13]. Another study investigating an alternative IL13 blocker, Lebrikizumab [14], recently closed, and results are pending.
LIMITATIONS OF THIS STUDY
Our study was limited by the small sample size of 44 patients. Follow up was often different between treatment groups and hence data and scoring measurements were sometimes inconsistent and at different intervals. Dosages were also variable across the cohort. Cyclosporine ranged from 50-200mg BD, methotrexate doses ranged from 5-25mg/week, azathioprine varied between 25-150mg/day, and mycophenolate mofetil ranged between 500-2500mg BD.
The database quality depended on the extensiveness of medical records, and at times this was incomplete. Scoring Atopic Dermatitis (SCORAD) and Dermatology Life Quality Index (DLQI) data at each review were only intermittently recorded; this data would have been valuable in the context of medication changes. As this was a retrospective study, issues of bias were a possibility. Our study was not blinded, therefore creating the possibility for bias.
CONCLUSION
In atopic dermatitis, current steroid-sparing systemic agents have demonstrated limited drug survival as a result of a constellation of side effects, toxicities, and limited efficacy.
We highlight that the use of mycophenolate mofetil is not currently first line, and that this may need to be revised. Use of mycophenolate mofetil has been limited because of cost and lack of experience. In Australia, the cost barrier has now been overcome with the expiry of the drug patent. Change of current practice to consider mycophenolate as a steroid-sparing agent early on should be considered. We recommend a review of the Australian guidelines, particularly in the use of cyclosporine. In practice, we have observed cyclosporine to be lacking in effectiveness and have significant side effects. It therefore may not be as favorable to be used as first line or standard of care.
With data unraveling the pathogenesis of atopic dermatitis accumulating rapidly, targeted trials looking into therapies to address these discoveries are being published. Larger and more robust trials exploring newer and targeted treatments of specific immune checkpoint pathways will hopefully improve the management of this disease in the near future.








































































































































