Usefulness of Metformin in Cancer Treatment: Teaching New Tricks to an Old Dog
- 1. Department of Hematology, General Hospital of Mexico, Mexico
- 2. Experimental Medicine Unit, National Autonomous University of Mexico, Mexico
- 3. Molecular Biology Laboratory, General Hospital of Mexico, Mexico
- 4. Department of Genetics, General Hospital of Mexico, Mexico
Abstract
Metformin was synthesized and marketed since the 50’s and is now one of the most used drugs for the management of noninsulin-dependent diabetes, this thanks to good metabolic control and high safety profile presented, despite the fact that do not know all their mechanisms of action. AMPK activation was identified as the target under which the effects of metformin were explained.
However, from several epidemiological studies that indicated the use of metformin as a protective factor against malignancies or as a factor for better response to chemotherapy regimens is that renewed interest in investigating other mechanisms of action.
This review hopes to provide a current perspective on the mechanisms of action of metformin known so far, highlighting its clinical significance inhibition of AMPK/mTOR/ S6K1 pathway, and reduce the expression of multi-drug resistance (MDR1) genes.
Keywords
• Metformin
• MDR Genes
• TOR Serine-Threonine Kinases
• AMP-Activated Protein Kinases
• Receptor
• IGF Type 1
• Cancer
Citation
Ramos-Peñafiel CO, Santoyo-Sánchez A, Olarte-Carrillo I, Queipo-García GE, Martínez-Tovar A (2016) Usefulness of Metformin in Cancer Treatment: Teaching New Tricks to an Old Dog. J Endocrinol Diabetes Obes 4(1): 1079.
ABBREVIATIONS
ABC: Adenosine Tri phosphate Binding Cassette; AMP: Adenosine Mono Phosphate; AMPK: AMP dependent kinase; ATP: Adenosine Tri Phosphate; CREB: cAMP Response Element-Binding; GABA: Gamma-Amino Butyric Acid; HCC: Hepato Cellular Carcinoma; HER-2: Human Epidermal growth factor Receptor 2; ICC: Intrahepatic Cholangio Carcinoma; IGF: Insulin-like Growth Factor; IGF-1R: Insulin-like Growth Factor 1 receptor; IL6: Interleukin 6; LKB1: Liver Kinase B1; MDR1: Multi-Drug Resistance genes; miRNAs: Micro RNA; mTOR: Mammalian Target of Rapamycin; NF-kB: Nuclear Factor kB; PI3K: Phosphoinositide 3-kinase; Shh: Sonic Hedgehog; STAT3: Signal Transducer and Activator of Transcription.
INTRODUCTION
Cancer is a public health problem placed as one of the top five causes of death worldwide. In 2012, according to Global Registry of Cancer (GLOBOCAN), a total of 14.1 million cases and 8.2 million deaths were associated directly with this disease, being the lung and breast cancer the leading causes of death in both men and women, respectively [1]. One of the main limitations on developing countries are the few available records that can estimate the distribution, mortality, severity or the local situation of each country, in order to perform strategies of effective prevention in the population [2]. The National Cancer Institute estimated that the incidence decreased by 1.5% per year from 2001 to 2010 mainly in the male gender but remained in female gender. It was also estimated that both age and comorbidities are constant factors that influence the risk of death [3]. This same record suggests that the Hispanic population that inhabits in the United States has a lower risk of suffer lung or breast cancer; but in some types, such as cervical cancer, the Hispanic population has an incidence 60% higher than the population non-Hispanic [4]. In our country the Histopathological Registry of Malignant Neoplasms of the 2002 estimated a total of 58.599 deaths associated with cancer, being 12.7% of all deaths; of all cases registered between 1993-2002 the uterine cervix cancer was the leading cause (22% of cases), followed by breast cancer (11%), prostate (6%) and lymphomas (5%). Leukemias only accounted for 2% of all cancers in our country (16.512 cases) [5].
Despite the fact that in the last decade it has been realized a significant progress in the understanding and treatment of a great diversity of tumors, treatment by through personalized medicine based on molecular targets, immunotherapy and inhibitors of cellular organelles such as the proteasome, the cure still seems distant [6]. Other researchers have proposed new mechanisms of action to a diverse drugs, among them metformin is one of the major.
Metformin and Cancer
Metformin is a widely antidiabetic drug used to treat the type 2 Diabetes mellitus. Initially, Galega officinalis (French Lila) was used by herbalists to treat polyuria and halitosis, but it was not until the late fifties, where three drugs were synthesized from their active ingredients: phenformin, metformin and buformin (Figure 1).
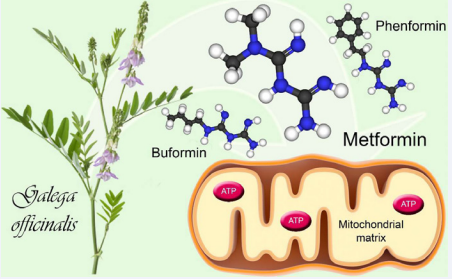
Figure 1 Biguanides extracted from the French lilac.
Chemically Metformin is a synthetic biguanide and since 1995 is the only available biguanide in the United Kingdom and the United States [7]. Despite so extensive use, their mechanisms of action are under study yet, but it is known that mitochondrial activity is their main molecular target [8]. This drug has a positive charge that accumulates in the mitochondrial matrix and inhibits the Complex I of the electron transport chain avoiding the oxidation of NADH with the consequent decrease of ATP levels. AMPK (Adenosine 5’-monophosphate-activated protein kinase) is activated due this change in ATP levels, carrying to the cell of an anabolic state to a catabolic state, restoring their energy balance [9,10].
The interest of their potential use as anti-tumor drug was originated in several epidemiological cohort studies of patients with type 2 diabetes mellitus, in which the use of metformin alone or in combination with sulfonylureas decreased the risk of developing cancer, compared to those exposed to insulin [11]. These findings have been replicated in many epidemiological reports around the world almost always with consistent results. In this point, the benefits of the use of metformin on the cancer can place on two sceneries: the first, patients who have an increased risk of developing cancer (e.g. Diabetes) and the second, those patients who already have the disease and we want to potentiate the treatment or prevent relapse.
Studies in cancer patients
Diabetes is one major risk factors to develop malignancies. Baur and cols conducted one of the largest studies about diabetes and cancer risk. With collected data from 3188 primary care physicians in Germany they found that the prevalence of cancer was higher in the group of patients with type 2 diabetes (66 / 1,242) compared with people without diabetes (185 / 6,025) (5.1% versus 3%, p <0.001). Odds ratio to develop cancer were lower in diabetic patients who were treated with metformin as monotherapy (OR, 1.04; 95% CI, 0.46 - 2.39) than to those with others oral treatments (OR, 2.26; 95% CI, 1.24 - 4.13).
In the prospective cohort of this population, the risk ratio for dying from cancer was lower in the metformin treatment group (RR, 1.34; 95% CI, 0.42- 4.25) compared to those with any other treatment that did not include Metformin (RR 3.51; 95% CI, 2.09- 5.88) [12]. Meanwhile, Zhang ZJ and cols reported a lower risk of colorectal neoplasia in patients with type 2 diabetes mellitus who were treated with metformin according to findings reported in five trials (RR, 0.63: 95% CI, 0.5- 0.79) [13]. Other examples are the study conducted by Chaiteerakij Rat the Mayo Clinic (Rochester, MN) where they are evaluated 612 cases of patients with ICC and 594 controls. In univariate analysis, factors such as cirrhosis (OR, 21.8; 95% CI, 5.3- 90.5, P <0.001), infection by hepatitis C virus (OR, 6.4; 95% CI: 1.4 - 28.5, P <0.001),diabetes (OR, 3.5; 95% CI, 2.2-4.9; P <0.001) and smoking (OR 1.5; 95% CI, 1.2-1.8; P = 0.02) were the ones that showed greater influence on the development of the ICC. Regarding the use of metformin, those diabetic patients using metformin counted with a lower risk of developing ICC compared with those who did not use (OR, 0.2; 95% CI, 0.1-0.4 p <0.001) [14] . Zhang and colleagues conducted a meta-analysis of the metformin effects over the risk of HCC in diabetic patients (4 case-control studies and 3 cohort studies).They conclude that metformin has a protective effect versus another anti diabetic therapies (RR 0.24, 95% CI, 0.13- 0.46, p <0.001) reducing by up to 76% the risk for HCC. Interestingly, when considering only cohort studies the association was not consistent [15]. About the metformin benefits against breast cancer, Col NF et al based on seven studies (4 cohorts and 3 case-controls), all studies evaluated metformin versus any other treatment (sulfonylureas, insulin and sulfonylurea combination) in patients with breast cancer. Metformin shows a protective factor (OR 0.83, 95% CI, 0.71 to 0.97) in all the studies, and higher in those patients with >3 years of intake the drug (n = 4 studies, OR 0.75, 95% CI; 0.62- 0.91) [16]. In this context Liu and co-workers described the positive effect of the addition of Metformin for decreasing the proliferation of breast cancer cell lines with negative receptor triple [17]. Recently, Koh and collaborators described onto the same cell line the effects of a compound derived from metformin (metformin GABA, metformin-pregabalin, gabapentin-metformin) with similar anti-tumor effects but 100 times higher [18]. Due this effects many another researchers have explored their effect on survival mainly in breast cancer HER-2 positive. Kim and collaborators in a cohort study showed major survival on the treatment arm with metformin (202 diabetic patients) versus control arm (6,967 non-diabetic subjects) and with another type of treatment (184 diabetic patients), also this last group showed a greater tendency to metastasis (RR 5.37, 95% CI 1:88 to 15.28) [19]. However, Oppong et al in Canada studied 313 patients with breast cancer and diabetes mellitus, after 87 months of follow-up did not identify differences in survival, relapse-free survival among the use of different types anti diabetic drugs [20]. Nowadays it is too easy find oncological clinical literature about use of metformin against cancer. The most solid evidence of the benefits we have so far are in breast cancer, colorectal cancer, ovarian and endometrial cancer, although it is need develop most powerful tests to validate this therapeutic effect [21].
Antitumor effect of metformin
Despite the wide variety of epidemiological studies, the understanding of antitumor effects has been derived from many assays in cell lines. The classic therapeutic target of metformin hydrochloride is indirect inhibition of mitochondrial action by blocking the Complex I of the respiratory chain, avoiding oxidation of NADPH with the consequent reduction in the production of ATP and AMP accumulation. A high AMP/ ATP ratio promotes the activity of LBK1 (Liver Kinase B1, also called Serine/Threonine Kinase 11 - STK11) responsible for phosphorylation and activating AMPK (AMP-activated protein kinase), a metabolic sensor that inhibits cellular processes that require large amount of energy present in many tissues such as liver, muscle, adipocytes, pancreas and in many neoplastic cells [22,23].
Activated AMPK phosphorylates TSC2 (Tuberous Sclerosis Complex 2) which stimulates the activity of the GTPaseRheb (Ras homolog enriched in brain) with subsequent inactivation of mTOR (mammalian target of rapamycin), essential for cell proliferation due that regulate expression of cyclin D1, HIF-1alpha / 2alfa (Hypoxia-inducible factors), and so-called transcription factors Yamanaka. OCT4, KLF4, SOX2, c-Myc [24–26]. Also has been reported that regulates transcription of microRNAs (miRNAs) which influence with various transcription factors as FOXO or CREB [10,24,25]. This effect on the miRNAs has been described by Feng et al in the cell line HCT 116 (colon cancer) where the addition of metformin and fluorouracil (5-FU) increased levels of a tumor suppressor gene called Spry2 through suppression of micro RNA-21 (miR-21) [27].
According to Vazquez-Martin, the effects of metformin are preserved even in those cells that have started with oncogenic changes [28].This was corroborated in 2010 by Memmott et al, using mice exposed to 4-methylnitrosamino-1- (3-pyridyl)- 1-butanone, a carcinogen tobacco, and were subsequently treated with metformin. At the liver, Metformin activated to AMPK and mTOR was inhibited, but in lung Metformin inhibited phosphorylation of insulin growth factor receptor (IGF-1/1R), Akt and ERK (extracellular signaling factor) pathways [29]. IGF-1/1R inhibition has also been beneficial in other cell lines. Xie et al reported that the addition of metformin on endometrial cancer cell lines increased expression of progesterone receptor which is negatively regulated by the IGF-1 [30] . These findings have been tested in clinical trials where metformin improves the effectiveness of targeted chemotherapy against hormone receptors, perhaps it is operating under the same mechanism described above [31].
In pancreas cancer cell lines, Rozengurt E. et al posit that metformin disrupts crosstalk between insulin/IGF-1 receptor and G protein–coupled receptors associated to IGF-1 [32]. Dirat and collaborators point to the Rac1 GTPase like a molecular target for metformin in prostate cancer. The addition of the drug in a cell essay caused the inhibition of Rac 1 GTPase by blocking cell signaling pathways as P-REX1, cAMP, CXCL12 / CXCR4 [33]. Recently, Nakamura et al added Metformin to blocking the expression of proteins involved in the signaling Sonic hedgehog (Shh) pathway in BxPC3 human pancreatic cancer cells [34]. Similar results have been obtained in breast cancer cells [35].
Furthermore, Lin and collaborators in cell lines of lung cancer also showed that the addition of Metformin inhibits the phosphorylation of pathway mediated by IL6/STAT3avoiding transcription of pro-survival factors and increasing the cisplatin-mediated toxicity, this last effect was mediated by several independent mechanisms of the mTOR pathway [36].
All these mechanisms of action are illustrated in Figure 2.
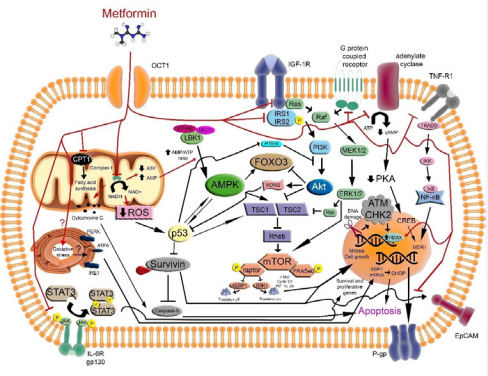
Figure 2 Signaling pathways as therapeutic associated target in cancer treatment with metformin
Other possible targets for Metformin: Multi-drug resistance (MDR1) genes The family of multidrug resistance genes
it is a superfamily of 49 ABC (adenosine triphosphate (ATP) – Binding Cassette) human genes which are classified in 8 subfamilies ranging from ABC-A to ABC-G and ANSA (arsenite and antimonite transporter) based on the homology degree of their sequences [37]. These genes are specialized in energy-dependent cellular transport and participate in a wide range of events such as the removal of harmful substances, secretion of toxins, mobilization of ions and peptides and cell signaling [38]. However, some antitumor drugs (Vinca alkaloids like vincristine and vinblastine; anthracyclines as daunorubicin and doxorubicin; Epipodophyllotoxins like etoposide and teniposide; tyrosine kinase inhibitors as imatinib) also are substrates for this pumps, particularly for the gp-P170, an efflux systems via an energy-dependent mechanism present in different types of cancer [38,39]. In 1994, Glazer and collaborators established a positive association between the expression of MDR1 and high concentrations of cAMP [40]. Subsequently this group use a selective AMPK inhibitor to verify the phenotypical change with a reduced expression of drug resistance genes [41]. Scala et al used 8-Cl-cAMP (an analog of cAMP) for achieve a lower metabolic activity in cancer cell lines and to induce effectively the down regulation of MDR1 [42]. Although tumor cells resistant to chemotherapy are a major problem for oncologist, there are only few clinical studies about this. Parissenti and collaborators evaluated a pharmacological inhibitor of cAMP in the MCF-7breast cancer cell line which is resistant to doxorubicin [43]. Despite being a common cellular metabolic pathway, only in the MCF-7 cell line has been associated the effects of Metformin over expression of MDR1 and glycoproteins-P by at least two mechanisms: The first pathway begins with activation of the AMPK that decrease the adenylate cyclase activity which in turn reduces the activity of PKA (protein kinase A) thereby preventing last phosphorylation of CREB needed to carry out transcriptional functions [44,45]. In a second way, Metformin reduce levels of NF-kB through inhibition of TNF-alpha receptor signaling cascade [45].
Metformin and its association with proteins that inhibit apoptosis
Survivin is one of the proteins involved in the inhibitors mechanisms of apoptosis that are active in both normal and pathological states. Its main role occurs during cell mitosis, specifically in the G2 / M stage by inhibiting the caspase 9 activity [46]. During tumor development, the expression ratio of survivin is inverse, various mutations on survivin has also been associated with tumor growth [47,48]. A large number of drugs that was mentioned perform synergy with metformin, but one in particular shows an antagonistic effect. Lesan y collaborators in the cell line MKN-45 showed that the addition of metformin in conjunction with cisplatin, increased the levels of survivin and mTOR. This antagonistic effect is justified by the interference of cisplatin mediated to Metformin not being a good option for the treatment of gastric cancer [49]. In another model of gastric cancer, Han and collaborators evaluated the effect in three gastric cancer cell lines (MKN-28 SGC-7901, BGC-823) with addition of Metformin, suggesting that the effect of this on apoptosis is mediated by inhibition of survivin mediated by effect on via mTOR [50] (Figure 2).
Metformin new tricks
Knowledge of the antitumor effects of metformin is increasingly complex and extensive, focusing on AMPK activation and blocking the mTOR pathway. Furthermore, Singhal and colleagues evaluated the effects of metformin over the growth of Mycobacterium tuberculosis. In their model, Mycobacterium cell growth was stopped due to the AMPK activation, producing reactive oxygen species which facilitated the lysosomal fusion [51]. These results have driven proposals to incorporate metformin into treatment regimens against tuberculosis, especially those multiresistant bacilli [52].
DISCUSSION AND CONCLUSION
At present, Metformin is considered the first line treatment of nondependent-insulin diabetic patients around the world [53]. Its indications have been extended over time in other pathologies as in gestational diabetes, pre-diabetes and polycystic ovary syndrome [54–56]. Although in the clinic has been used for over 50 years, knowledge about its mechanism of action is not yet well elucidated [57]. Since its initial appearance in Diabetology, the official journal of the European Association for the Study of Diabetes in June 2009, four studies raised the risk of developing cancer in diabetic patients and the impact of different therapeutic strategies, including Metformin [58]. Initially considered by its AMPK activation mechanisms [59] until discovery the involvement of signaling pathways such as mTOR, MERK / ERK, STAT3, PI3K / AKT in cellular mechanisms of proliferation, differentiation, and more recently with effects on apoptosis or stopping the expression of various flow pumps mediated by factors such as surviving [47,60]. The therapeutic possibilities for this old biguanide based on its multiple mechanisms of action have spread not only in oncology, also in infectious diseases such as tuberculosis or even to consider it as a drug that can reduce the aging [61]. In conclusion, according to the evidence on cancer cell lines, metformin apparently has many anti-tumor properties that cover the main mechanisms of cancer genesis and progression, (59) including metabolic depletion of tumor cell (Figures 3) [62].
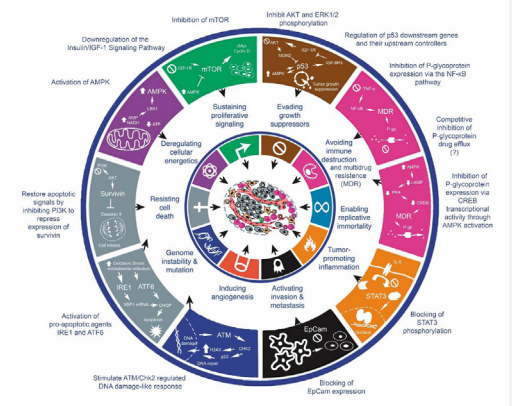
Figure 3 Anti-tumoral effects of metformin (Adapted from: Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell, 2011; 144: 646–74).
Its excellent safety profile coupled to an extensive clinical experience of use will make the oncologist and physicians reconsider its effectiveness in cancer treatment.
ACKNOWLEDGEMENTS
We acknowledge the support National Council for Science and Technology (CONACyT) for the support provided (projects 80085 and 162269) for ongoing research into the General Hospital of Mexico.
REFERENCES
2. Parkin DM. The role of cancer registries in cancer control. Int J Clin Oncol. 2008; 13: 102-111.
25. Serrano M. Metformin and reprogramming into iPSCs. Cell Cycle. 2012; 11: 1058-1059.
55. Bansal N. Prediabetes diagnosis and treatment: A review. World J Diabetes. 2015; 6: 296-303.
61. Anisimov VN. Metformin: do we finally have an anti-aging drug? Cell Cycle. 2013; 12: 3483-3489.
62. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144: 646-674.









































































































































