Residual Collapsibility of The Upper Airways After Pharyngeal Surgery For OSA
- 1. Pneumology Unit, San Jacopo Hospital, Italy
- 2. Head and Neck Department, ENT Unit Director, Santa Maria delle Croci Hospital, RavennaUmberto I Hospital Lugo, AUSL of Romagna, Italy
- 3. Sleep Respiratory Unit, Ospedali Privati Forlì, Forlì, Italy
Abstract
A man with severe OSA (AHI: 56.7; Epworth Sleeping Scale: 11/24) underwent expansion sphincter pharyngoplasty (ESP) proved effective in controlling apnea and hypopnea and daytime sleepiness (AHI: 5.5; Epworth Sleeping Scale: 0/24) at six months after surgery. Otherwise, the nasal cannula inspiratory flow contour signal for almost of the 7 hours of recording time showed Inspiratory Flow Limitation (IFL), an indicator of a residual pharyngeal collapsibility when exposed to negative inspiratory pressure. The IFL seems to be associated with significant physiological changes, such as an increase in end-tidal CO2, esophageal pressure or inspiratory time. To date, clinical studies are warranted to elucidate the clinical role of these physiological changes and guide the best management of these patients. Our case report has a significant educational and clinical value. In fact, the instrumental and clinical resolution of the OSA may hide a residual effect, the collapsibility of upper airway, that has been associated with potential detrimental effect on the health of the subject with OSA. We suggest that in case of residual and persistent IFL it should be necessary to provide a structured management to prevent the occurrence of comorbidities and for an early diagnosis of a possible recurrence of OSA.
Keywords
• Obstructive sleep apnoea; Expansion sphincter pharyngoplasty; Upper airway; Inspiratory flow limitation
Citation
Sanna A, De Vito A, Bosi M (2021) Residual Collapsibility of The Upper Airways After Pharyngeal Surgery For OSA. J Sleep Med Disord 7(1): 1117.
INTRODUCTION
Obstructive sleep apnea (OSA) is a chronic disease widespread in the world with an extraordinarily high prevalence in both sexes [1]. If left untreated, it is a risk factor for stroke, cardiovascular, metabolic and neurocognitive disease, cancer [2,3] and motor vehicle and work-related accidents [4]. OSA is characterized by the collapse of the upper airway (UA) with reduced or cessation of airflow during sleep [5]. Continuous Positive Airway Pressure (CPAP), the treatment of choice for OSA regardless of its severity, prevents pharyngeal collapse with resolution of apnea and hypopnea [6]. In the last few years different OSA phenotypes have been defined, and treatments other than CPAP (UA surgery, oral appliance, positional therapy) have been proposed [7]. One of the latest pharyngoplasty modification is represented by the expansion sphincter pharyngoplasty (ESP), a surgical procedure which stabilize the lateral pharyngeal walls (a frequent site of collapse), by reducing their collapsibility when exposed to negative inspiratory pressure during sleep [8].
CASE PRESENTATION
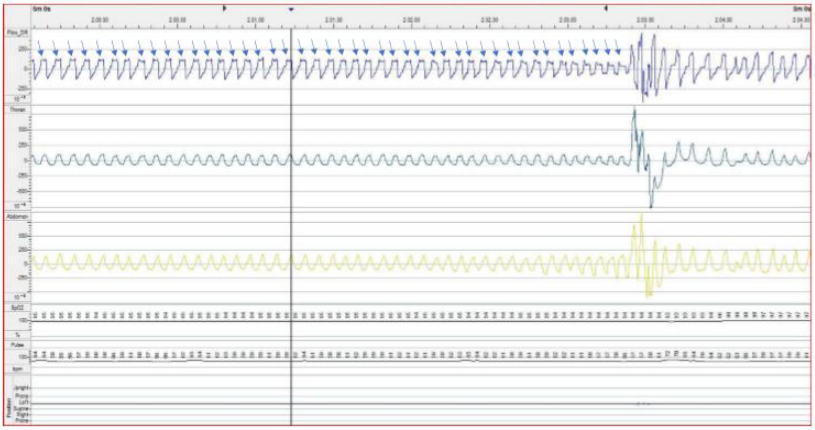
A 56-year-old overweight (body mass index: 29.0) man with severe OSA(AHI: 56.7; Epworth Sleeping Scale: 11/24) refused CPAP treatment. Informed consent was obtained from the patient. He had no history of endocrine, cardiac, or neurological disorders. Drug-induced sleep endoscopy (DISE) showed the lateral pharyngeal walls as the primary site of collapse, and consequently ESP combined with tonsillectomy and nasal septoplasty has been performed. The Home Sleep Testing (HST) carried out six months after ESP was performed, as well the diagnostic one, with an Embletta device (model 2601-1 PDS X10Xact Trace, USA) and showed an improvement in AHI (from 56.7 to 5.5) associated with a an imorovement in Epworth Sleeping Scale (from 11/24 to 0/24). Otherwise, the analysis of HST traces reported IFL, defined as a flattening of the nasal cannula inspiratory flow contour signal, for almost of the seven hours of recording time (Figure 1).
DISCUSSION
In OSA, the combination of anatomical and functional sleeprelated factors (ineffective pharyngeal dilator muscle, a low threshold for arousal to airway narrowing, unstable control of breathing due to a high loop gain) results in narrowing of the UA, whose hallmark feature is IFL, the first step in progressively reducing flow to zero, as observed in apnea [5]. Characterization of OSA from an anatomic and sleep-related functional point of view makes it possible to phenotype OSA and treat it in a targeted way [7]. All different therapies acting on the UA (CPAP, oral appliance, UA surgery) are aimed at preserve the UA patency during sleep when exposed to the collapsing negative inspiratory pressure. Our patient, in which DISE showed the collapse of the pharyngeal side walls, underwent ESP that proved effective in controlling apnea and hypopnea and daytime sleepiness as shown by the objective and clinical testing carried out at six months after surgery (AHI: 5.5; Epworth Sleeping Scale: 0/24). However, it did not seem equally effective in stabilize the pharynx, as shown by the persistent IFL (Figure 1). The occurrence of IFL is an indicator of a residual pharyngeal collapsibility when exposed to negative inspiratory pressure. A number of different flow limitation shapes have been recognized in patients with OSA. By combining the shape of flow limitation with negative effort dependence, defined as the reduction from peak to mid inspiratory flow with increasing respiratory effort, identification of the anatomical site where the UA is collapsing during sleep should be possible [10]. We do not know whether the ESP resulted in residual collapse of the pharyngeal side walls or in a change of the dynamic behavior of the UA with other anatomical sites (tongue, epiglottis or soft palate) that have become more compliant, i.e. most easily collapsible. However, the shape of the flow limitation (Figure 1) suggests an UA obstruction resulting from a posterior fall of the tongue [10]. Persistent IFL, already described in OSA treated with UA surgery [11], oral appliance [12] or CPAP [13] may be a possible residual effect, whatever the treatment applied, with possible significant physiological changes, such as an increase in end-tidal CO2 , esophageal pressure or inspiratory time [13]. To date, clinical studies are warranted to elucidate the clinical role of these physiological changes and guide the best management of these patients. The case we present shows an IFL, for almost of the recording time that expresses a sleep-related UA narrowing due to a residual pharyngeal collapsibility even after a soft-tissue UA surgical procedures considered effective in stabilizing the pharyngeal side walls. Although IFL has already been described in OSA subjects under treatment [11-13], our case has a significant educational and clinical value. In fact, the instrumental and clinical resolution of the OSA may hide a residual effect, the collapsibility of UA as shown by the persistent IFL, which seems to be associated with physiological changes [13] with potential long-term detrimental effect on the health of the subject with OSA. IFL should be also considered as an indicator of a possible recurrence of OSA needing an update of the therapeutic strategy. We suggest that: 1) in cases where a residual and persistent IFL occurs a DISE can be carried out to assess whether the dynamic behavior of the UA has changed; and 2) a long-term follow-up with HST should be provided in all cases of OSA treated with UA surgery.
REFERENCES
1. Benjafield AV, Ayas NT, Eastwood PR, Heinzer R, Ip MSM, Morrell MJ, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med. 2019; 7: 687-698.
2. Bonsignore MR, Baiamonte P, Mazzuca E, Castrogiovanni A, Marrone O. Obstructive sleep apnea and comorbidities: a dangerous liaison. Multidiscip Respir Med. 2019; 14: 8.
3. Marin-Oto M, Vicente EE, Marin JM. Long term management of obstructive sleep apnea and its comorbidities. Multidiscip Respir Med. 2019; 14: 21.
4. Sanna A. Obstructive sleep apnoea, motor vehicle accidents, and work performance. Chronic Respir Dis. 2013; 10: 29-33.
5. Osman AM, Carter SG, Carberry JC, Eckert DJ. Obstructive sleep apnea:current perspectives. Nat Sci Sleep. 2018; 10: 21-34.
6. Sullivan CE, Issa FG, Berthon-Jones M, Eves L. Reversal of obstructive sleep apnoea by continuous positive airway pressure applied through the nares. Lancet. 1981; 1: 862-865.
7. Sutherland K, Kairaitis K, Yee BJ, Cistulli PA. From CPAP to tailored therapy for obstructive sleep apnoea. Multidiscip Respir Med. 2018; 13: 44-56.
8. Pang KP, Woodson BT. Expansion sphincter pharyngoplasty: a new technique for the treatment of obstructive sleep apnea. Otolaryngol Head Neck Surg. 2007; 137: 110-114.
9. Kapur VK, Auckley DH, Chowdhuri S, Kuhlmann DC, Mehra R, Ramar K, et al. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2017; 13: 479-504.
10.Genta PR, Sands SA, Butler JP, Loring SH, Katz ES, Demko BG, et al. Airflow Shape Is Associated With the Pharyngeal Structure Causing OSA. Chest. 2017; 152: 537-546.
11.Li Y, Ye J, Han D, Cao X, Ding X, Zhang Y, Xu W, Orr J, et al. PhysiologyBased Modeling May Predict Surgical Treatment Outcome for Obstructive Sleep Apnea. J Clin Sleep Med. 2017; 13: 1029-1037.
12.Godoy LBM, Palombini L, Poyares D, Dal-Fabbro C, Guimarães TM, Klichouvicz PC, et al. Long-Term Oral Appliance Therapy Improves Daytime Function and Mood in Upper Airway Resistance Syndrome Patients. Sleep. 2017; 40.
13.Calero G, Farre R, Ballester E, Hernandez L, Daniel N, Montserrat Canal JM. Physiological consequences of prolonged periods of flow limitation in patients with sleep apnea hypopnea syndrome. Respir Med. 2006; 100: 813-817.