The Connection between Sleep Spindles and Seizures in Schizencephaly: A Case Report
- 1. Atatürk University Medical School, Department of Physiology,Turkey
- 2. Atatürk University Medical School, Department of Physiology,Turkey
- 3. Atatürk University Medical School, Department of Physiology,Turkey
- 4. Zonguldak Bülent Ecevit University, Medical School, Department of Endocrinology and Metabolism, Zonguldak, Turkey
Abstract
Objective: The aim of this case report is to define the interaction between sleep and epilepsy (and, sleep spindles and seizures), also the importance of electrophysiological follow up in sleep laboratory and correct diagnostic approach.
Background: Sleep spindles are generated from the thalamus. Sleep spindles and seizures originate from the same type of dynamical system. Schizencephaly is a rare congenital disorder of cell migration with defect in sulcation. It is characterized by gray matter lined clefts.
Results: The case has complaints of short-term paresthesia in right arm at daytime and morning. Brain MRI showed us “Cortical Heterotopia” at left temporoparietal area. In polisomnography (PSG) analysis, there were no sleep spindles, mostly there were delta wave oscillations together with repeated seizures.
Conclusions: The absence of sleep spindles in PSG, presence of epileptic seizures and somatic complaints of the patient together with the appearance of heterotopic area on brain MRI led us to think about the influenced of associative thalamocortical tracts.
Keywords
• Sleep spindle
• Epilepsy
• Correct diagnosis and electrophysiology
Citation
Kayabekir M, Ceylan M, Yalc?n A, Topaloglu O (2021) The Connection between Sleep Spindles and Seizures in Schizencephaly: A Case Report. J Sleep Med Disord 7(2): 1124.
ABBREVIATIONS
AHI:Apnea-Hypopnea Index, EEG: Electroencephalography, MRI: Magnetic Resonance Imaging, NREM: Non Rapid Eye Movement, PSG: Polysomnography, SWD: Spike-Wave Discharges
INTRODUCTION
Sleep spindles are well-known electroencephalography (EEG) phenomena that reflect spontaneous rhythmic activity of thalamocortical neuronal network during Non-REM (NREM) sleep (1–3). In vivo experiments demonstrated a close relationship between sleep spindles and epileptic spike-wave discharges (SWD) (4–5). Sleep spindles are generated from the thalamus. Seizures are a result of abnormal, synchronized attacks of electrical activity in restricted area of connected neurons, called the epileptogenic focus. Several experimental results point to the thalamus having a critical role in the generation of spike and wave discharges. It has been shown that if the thalamus is somehow inactivated these discharges disappear. Actualy, according to this pattern sleep spindle oscillations can turn in to seizures. Sleep spindles and seizures originate from the same type of dynamical system. It has been shown that antagonizing manipulations made on sleep spindles have the same effect on seizures (6-7). SWD are electroencephalographic manifestation of absence epilepsy, and they are triggered by the cortex, opposite to sleep spindles, which are known to originate from the thalamus. In comparison to sleep spindles, SWD are underlain by more intensive excitation and/or synchronization processes in thalamocortical network (8-10). Schizencephaly is a rare congenital disorder of cell migration with defect in sulcation. It is characterized by gray matter lined clefts. Two types have been described in the literature – Closed Lip (Type I), which consists of fused, cleft without hydrocephalous and Open Lip (Type II) consisting of open cleft with hydrocephalous. Diagnosis is made by imaging and magnetic resonance imaging is the most sensitive modality in detecting clefts and the associated anomalies like pachygyria, polymicrogyria, heterotopias, septo-optic dysplasia and absent septum pellucidum (11-15).
Possible etiological factors include teratogenic exposures, viral exposures, genetic mutations, and intrauterine fetal stroke. Risk factors of schizencephaly include young maternal age and the illicit use of alcohol and narcotic substances. The prognosis depends on the size and type of the clefts: Type I has a milder course. It can be asymptomatic or diagnosed only in adult patients and presents with epileptic seizures and mild motor deficits. Type II has a more severe presentation, manifested by epilepsy (often refractory), intellectual disability, varying degrees of paralysis, hemiparesis in unilateral schizencephaly, and quadriparesis in bilateral schizencephaly. The diagnostic method of choice in imaging of schizencephaly is magnetic resonance imaging (MRI). The therapeutic management of both types of schizencephaly is conservative. Surgical treatment is undertaken in some cases with concomitant hydrocephaly or intracranial hypertension (16-17).
Case (History)
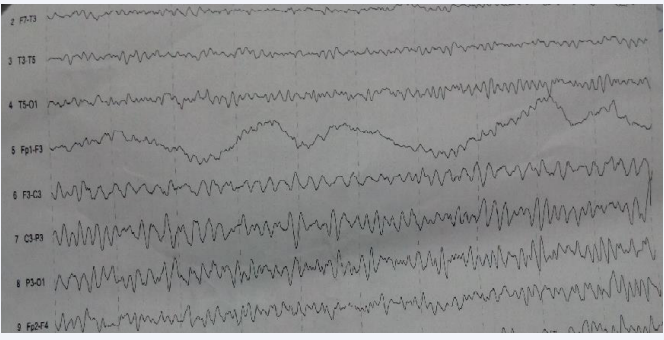
A 23 years old male patient is a student attending university, using left hand dominantly. He had used antidepressants for six months (paroxetine, sertraline) now he is not using the drugs. He has complaints of short-term paresthesia in right arm at daytime and morning. Also there is history of syncope attacks starting with right arm contraction then continuing with loss of consciousness. The patient was diagnosed with Complex Partial Epilepsy (Figure 1).
Figure 1: Daytime EEG: Irregular spike, spike wave (or arousal EEG) activities were determined on left frontal, parietal and occipital electrodes. No difference was shown by hypoventilation and fotic stimulation.
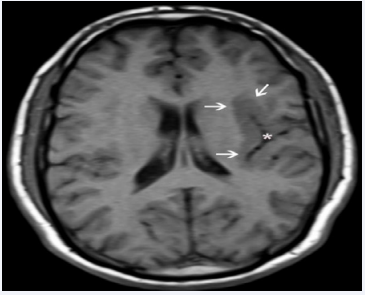
After using of carbamezapin 600 mg 2x1, there were no epileptic seizures in day-time for four months. He has complaints of palpitation sweating, fear, left frontal pain continuing for 45- 60 minutes, on the bed before sleeping. He has teeth clenching at night. Brain MRI showed us “Cortical Heterotopia” at left temporoparietal area (Figure 2).
Figure 2: Axial T1 weighted MR image shows left temporo-parietal heterotopic gray matter (arrows) due to a closed-lip schizencephaly (asterisk).
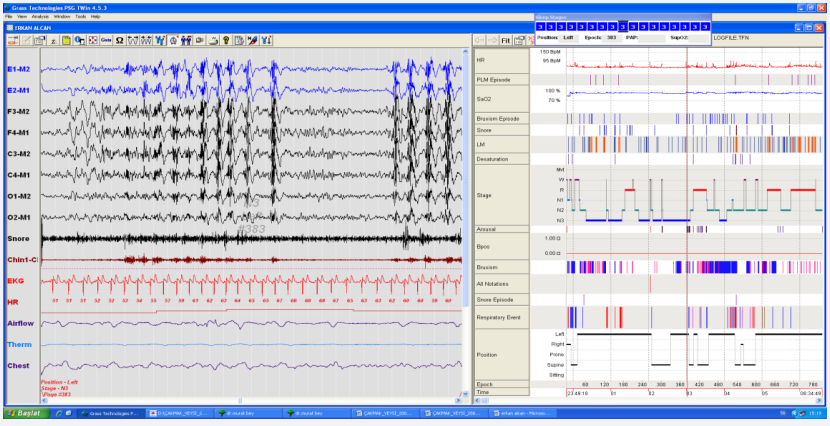
In polisomnography (PSG) analysis, there were no sleep spindles, mostly there were delta wave oscillations together with repeated seizures, bruxism, arousals, central and obstructive sleep apnea until morning (AH?: % 7,6) (Figure 3).
Figure 3: The image of delta wave and epileptic seizure in PSG record continuing an epoch (The Center of Sleep Disorders, Archive of Electrophysiology Laboratory, 2016, Erzurum, Turkey).
DISCUSSION
Sleep spindle oscillations can turn in to seizures. Sleep spindles and seizures originate from the same type of dynamical system. It has been shown that antagonizing manipulations made on sleep spindles have the same effect on seizures (6-7). Schizencephaly is a central nervous system malformation that most often presents as epilepsy. Though a majority of patients have well-controlled seizures, some patients may develop refractory epilepsy with uncontrolled breakthrough seizures and associated risks, including sudden unexpected death in epilepsy. Several experimental results point to the thalamus having a critical role in the generation of spike and wave discharges. It has been shown that if the thalamus is somehow inactivated these discharges disappear. In comparison to sleep spindles, SWD are underlain by more intensive excitation and/or synchronization processes in thalamocortical network (8-10, 17). In our patient, the absence of sleep spindles in PSG, presence of epileptic seizures and somatic complaints of the patient together with the appearance of heterotopic area on brain MRI led us to think about the influenced of assosiative thalamocortical tracts. It was attractive that sleep spindles were not produced. The presence of clinical somatic and emotional symptoms, epileptic seizures determined in PSG though absence of seizures at daytime through the drugs, of the totally absence of sleep spindles point to the existence of subcortical ectopic area. Subcortical heterotopic neurons due to schizencephaly may be shown by MR tractography.
CONCLUSION
As a result; this case is so important in regard to understand the physiopathological basis of the relationship between sleep spindle and epilepsy and to lead us to make new clinical studies.
REFERENCES
1. Steriade M. Neuronal Substrats of Sleep and Epilepsy, CUP, 2003.
2. De Gennaro L. and Ferrara M. Sleep spindles: an overview, Sleep Med Rev. 2003; 7: 423-440.
3. Destexhe A. and Sejnowski TJ. Thalamocortical Assemblies, OUP. 2001.
11. Veerapaneni P, Veerapaneni KD, Yadala S. Schizencephaly. Stat Pearls. 2021.
14. Griffiths PD. Schizencephaly revisited. Neuroradiology. Neuroradiology. 2018; 60: 945-960.