Mucinous Tubular and Spindle Cell Renal Cell Carcinoma: About an Observation and Review of the Literature
- 1. Department of Urology B, Mohamed V University, Morocco
Abstract
Mucinous tubular and spindle cell carcinoma is a rare tumor described in the 2004 WHO classification as a new entity with a relatively indolent behavior. We report a case of 60 year old man with a large left upper pole tumor. The patient was treated by left radical nephrectomy. The histologic findings confirmed that it was a tubulo-mucinous carcinoma and spindle cell grade I Furhman contours with non-infiltrating hilar invasion. Because of the favorable prognosis with this type of tumor, mucinous tubular and spindle cell carcinoma must be differentiated from papillary renal cell carcinoma, especially the variant with sarcomatoid dedifferentiation. Our case report of this rare entity ain to draw the attention of pathologists and clinicians to the importance of its diagnosis.
Keywords
Renal carcinoma, Mucinous stroma, Low grade
Cite this article
Himmi Y, Moutah B, Slaoui A, Tarik K, Khalid E, et al. (2022) Mucinous Tubular and Spindle Cell Renal Cell Carcinoma: About an Observation and Review of the Literature. J Urol Res 9(1): 1132.
INTRODUCTION
Mucinous tubular and spindle cell renal cell carcinoma is a rare kidney cancer, which was first described in 1998. It has already been designated in the category of unclassifiable Renal Cell Carcinoma (RCC) [1], in the classification of the World Health Organization (WHO), kidney tumors. In 2004, it was incorporated as a new entity apart from renal cell carcinomas [2]. Although CTM has been reported to have a relatively good prognosis, follow-up data is limited and the clinical behavior of this tumor remains to be established, and there is a need to collect more clinicopathologic characteristics of CTM for better understanding. Thus, we present the case of a tubulo-mucinous and fusiform carcinoma of low malignancy grade in a 60-year-old man with a review of the literature.
OBSERVATION
M BM, 60 years old, chronic smoker at the rate of 30 packs/ year. He had been complaining for 2 months of low-intensity left low back pain associated with a single episode of haematuria, with no other associated urinary or digestive disorder. On clinical examination, the patient was afebrile. His conjunctivae were normally stained, and his abdomen supple. The lymph node areas were free.
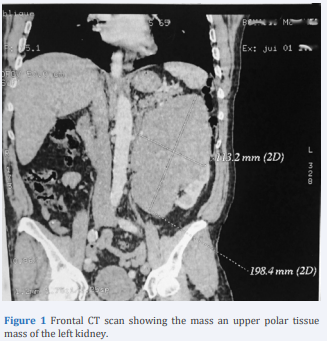
Computed tomography revealed a well-defined superior polar left renal mass, 20 cm in long axis, hypodense before injection, slightly enhanced after injection of the contrast product (Figure 1).
Figure 1 Frontal CT scan showing the mass an upper polar tissue mass of the left kidney.
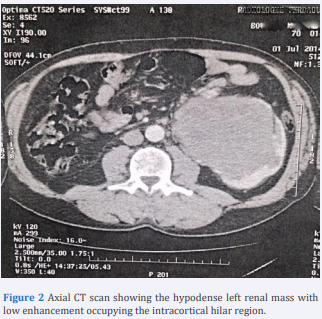
This mass pushes the calicielles cavities and the renal pedicle forwards (Figure 2). With the presence of a 10 mm latero left aortic lymphadenopathy. Biologically, the patient had a hemoglobin level at 13 g/dl, leukocytes at 6400/mm3 , normal renal function with a creatinine level at 8.5 mg. The patient was operated on subcostally. After detachment of the left colic angle, he underwent radical left nephrectomy. The postoperative course was simple.
Figure 2 Axial CT scan showing the hypodense left renal mass with low enhancement occupying the intracortical hilar region.
On macroscopic examination, the kidney is the seat of a 20 cm mass, solid, beige-yellow in color and includes areas of hemorrhagic changes. This tumor was poorly borderline not encapsulated and coming into contact with the renal capsule.
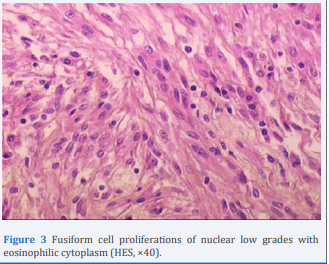
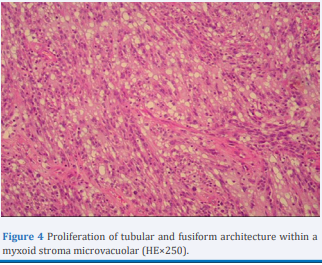
Microscopic examination revealed proliferation of monotonous cubic eosinophilic epithelial cells. The cells are arranged in tubular structures and in parallel cell cords. These tubular structures are continuous with spindle-shaped sectors which are also of low nuclear grades. The microvacuolar stroma contains a mucinous substance with an inflammatory histiocyte infiltrate (Figure 3,4).
Figure 3 Fusiform cell proliferations of nuclear low grades with eosinophilic cytoplasm (HES, ×40).
Figure 4 Proliferation of tubular and fusiform architecture within a myxoid stroma microvacuolar (HE×250).
No vascular emboli were seen in the peri-tumor, nor capsular invasion or sarcomatoid contingent.
This morphological and histological analysis concluded that Furhman Grade I was tubulomucinous and fusiform carcinoma of the kidney.
DISCUSSION
Mucinous tubular and spindle cell carcinoma is a rare malignant epithelial tumor of the kidney, recognized since 2004 as a new entity of renal cell carcinoma. CTM has a female predominance and a good prognosis [3]. More than 80 cases have been listed in the literature [2]. The rare morphology of CTM has produced much confusion in the past.
The tubular architecture of the CTM showing focal papillae is strong evidence that may be in favor of papillary renal cell carcinoma. In cases with a predominant fusiform configuration, this appearance can lead to confusion with leiomyomas or even sarcomas [4].
CTM presents clinically with a clinical symptomatology that combines haematuria, lumbar pain and a palpable mass [5]. Radiological explorations found calcifications in 34% of cases, a figure six times higher than in other kidney tumours. There is also a greater frequency of invasion of the retroperitoneal space and greater tumor hypovascularization. But in general, there are no specific imaging criteria for the diagnosis of CTM. These features may resemble other variants of renal cell carcinoma, such as chromophobe cell carcinoma or papillary carcinoma of nothing, which have a less favorable prognosis. However, renal CTM should be suspected.
Faced with a large, well-circumscribed mass, the enhancement is weak after injection of the contrast product, with a weak or intermediate signal on T2-weighting, especially if in association with nephrolithiasis [1]. The size of the CTM is variable. It ranges from less than 1.0 cm in diameter to more than 18.0 cm, with most tumors measuring between 2.0-4.0 cm in the longest axis [6]. Macroscopically, the CTM are essentially of medullary location, well limited, firm, and often homogeneous, with a whitish-grey or more rarely brownish appearance. Hemorrhagic or necrotic changes are rare [7].
Histologically, the architecture is compact at low magnification with tubular sectors in continuity with fusiform sectors seeming to come from the compression of the tubes [8-10]. The tubes are stretched, interconnected, and sometimes arranged in parallel cellular cords, close to the structure of the loop of Henlé. They are composed of cuboidal cells with eosinophilic or clarified cytoplasm, centered by an oval or elongated nucleus without a prominent nucleolus. Mitoses are rare. The fusiform sectors can in places evoke a proliferation of smooth muscle cells. Between the tubes, a myxoid stroma is visible, positive for alcian blue. A few foci of inflammatory cells, notably containing foamy macrophages, may be present.
These tumors have a complex phenotype, expressing a wide variety of epithelial markers (EMA, AE1/AE3, CK7, CK19), and distal nephron markers (EMA, CK 19, E-cadherin).
The immunohistochemical profiles of these tumors reported in the literature are not clearly defined, they are inconsistent or even contradictory due to the heterogeneity of these tumors and the insufficient number of cases studied [8,11,12]. Indeed, the expression of the RCC Ma is inconstant; a recent study showed that 92% of the cases studied expressed this marker 4. This figure was 45% and only 7% in other series. Other markers are also of variable expression such as EMA, CD 15 and PS 100.
It is obvious that tumors with a similar morphological appearance may have different immunohistochemical expression without this necessarily implying that they are different. Several classifications of tumors in different organs have been mainly based on their morphology, the immunophenotype not always being exactly the same for tumors classified in the same type. Some authors suggest classifying tumors with the morphological characteristics of Mucinous tubular and spindle cell carcinoma according to the expression of different markers [13]. However, the expression of these antigens does not seem to be sufficient to separate tumors with the same clinical presentations, microscopic and evolutionary characteristics.
In the literature, cytogenetic data indicate various chromosomal losses and associated gains, but no loss of 3p or trisomy 7 and/or trisomy 17. Using comparative genomic hybridization and FISH (fluorescence in hybridization situ), we find characteristic combinations generally involving the loss of chromosomes 1, 4, 6, 8, 13 and 14 and the gains of chromosomes 7, 11, 16 and 17 [14].
The prognosis, according to data from the literature and in agreement with the histological appearance of low grade, seems favorable; however, cases with local recurrence, distant and regional metastases associated with the presence of metastatic lymphadenopathy have been reported in the literature [11,15- 17]. Recently, 5 cases of CTM with sarcomatoid differentiation and aggressive behavior have been reported in the literature, among these five cases three had distant metastases with fatal outcome for the patients [16,18]. It can be suggested that the changes observed in CTMs with sarcomatoid differentiation are related to the biobehavior of the kidney CTM. But given the low number of published cases and the duration of follow-up of these cases, the true biological potential and the morphological criteria that define it remain unknown [19]. Surgical resection is therefore always recommended, associated with careful followup of the patient.
CONCLUSION
MTSCC is a new entity in the pathological classification proposed by the WHO in recent years. It is essential to recognize CTM because of its favorable prognosis. Further cytogenetic and immunohistochemical studies as well as greater clinical experience are needed for a better characterization of these tumors.
REFERENCES
4. MacLennan GT, Cheng L (2008) Neoplasms of the kidney. In:Bostwick DG, Cheng L (eds) Urologic surgical pathology, 2nd edition . Mosby– Elsevier, Philadelphia, pp 104–6
14.Srigley JR, Eble JN, Grignon DJ, Hartwick RWJ: Unusual renal cell carcinoma (RCC) with prominent spindle cell change possibly related to the loop of Henle. Mod Pathol. 1999; 12: 107.