Science of Urolithiasis: A Multidimensional Integrative Review
- 1. Animal Physiology and Toxicology laboratory, Department of Zoology, Shivaji University, India
Abstract
The trends of urolithiasis have been immerged as noteworthy public health associated social and economic issue, specifically attributed towards the dietary and lifestyle habits. Strange aspects in the history of urolithiasis enlights modern and current changes in its epidemiology. The incidence and rate of urolithiasis exhibited as per the geographical variations which were associate with individual parameters including age, sex, dietary habit and genetic variations. It was found that, more than 15 % of population living in the developing area were victimized for nephrotic alteration in which different types of urinary calculi were prominently documented in early Ayurveda (Sanskrit literature ) during the period of between 3000 BC. to 2000 BC. Since then, problem of stone formation and related pathetic condition in human who has received challenge against medical sector with its multifunction etiology and high rate of recurrence. Crystallization found and reported because of imbalance between promoters and inhibitors. With respect to challenges in the field, there are number of inhibitors of crystal formation including molecules, peptides, proteins and biological derivatives came in to the focus. In general urolithiasis includes supersaturation, nucleation, aggregation and retention of urinary crystals within the nephrotic cells. Sequential steps were modulated by irregular metabolic and biochemical in the process crystallization. Typically, calcium cascade leads to activate undesired biochemical changes for crystallization, which cause cellular damage, along with non-recoverable alternations reported in historical literature. Acute and severe pains, necrotic inflammation and related biological alteration were imposed for pathophysiological and medical emergencies. In a modern era, comprehensive biological and metabolic assessment is crucial step towards diagnosis. Medical and therapeutic impact customize with respect to patients for effective management and limits further undesired damage and recurrence to save the life. To enhance therapeutic approaches, it is essential to gain in-depth knowledge about known facts, new drugs, upcoming techniques and different avenues to overcome the saturation, crystallization and aggregation in nephrotic cell to stop further kidney damage. As a future perspective, understanding of urolithiasis, pathophysiology, metabolism, alterations and drug delivery ending the story up to recovery in the kidney seems to be more challenged but demanding attention to understand smooth and socioeconomic potential of clinical work. To understand, investigate and come up with new trends in biological and clinical sciences to overcome the problem of urolithiasis in association with action of proactive drug, its impact and nature of biomechanics in the nephron, present review has been carried out.
Keywords
• Urolithiasis; Calcification; Biomechanism; Animal Models; Diagnostic Tools; AI.
Citation
Sutar VS, Kamble NA (2025) Science of Urolithiasis: A Multidimensional Integrative Review. J Urol Res 12(2): 1167.
ABBREVIATIONS
KDS: Kidney Stone Disease; GBD: Global Burden of Disease; SDI: Sociodemographic Index; RAAS: Renin Angiotensin Aldosterone System; PCT: Proximal Convoluted Tubule; DCT: Distal Convoluted Tubule; GSS: Guy’s Stone Score; GAGs: Glycosaminoglycan’s; CaOx: Calcium Oxalate; COM: Calcium Oxalate Monohydrate; COD: Calcium Oxalate Dehydrate; SLC26: Solute-Linked Carrier26; MDCK: Madin Darby Canine kidney; RSK: Relative Supersaturation Risk; RTA: Renal Tubular Acidosis; KUB: Kidney, Ureter and Bladder; CT: Computed Tomography; MRU: Magnetic Resonance Urography; IVP: Intravenous Pyelogram; SWL: Shock Wave Lithotripsy; PCNL: Percutaneous Nephrolithotomy; NSAIDs: Nonsteroidal Anti-Inflammatory Drugs; NaOx: Sodium Oxalate; CKD: Chronic Kidney Disease; KOX: Potassium Oxalate; EG: Ethylene Glycol; HLP: Hydroxy-L-Proline.
BACKGROUND
Renal calculi derived from Latin r?n?s, meaning “kidneys” and calculus meaning “pebble” are which solid or crystal aggregation of minerals from the urine [1]. Under clinical physiology term urolithiasis or nephrolithiasis or renal calculus is nothing but a formation of kidney stone biologically anywhere in urinary system [2,3]. After urinary tract infections and prostate disorders, urolithiasis found to be third most common disorder in excretory system [4]. It is the most common, serious urinary track disorder worldwide where younger population is affecting with the high rate [5]. The variation about prevalence rate and occurrence of kidney stone found depending on race, geographical and climatic prominently concentric in hot climatic condition [6,7].
The continent wise data showed that, rate of formation of kidney stone in Asia was 1–5%, in Europe 5–9%, 7–15% in North America, 4 % in China. 20 % in Saudi Arabia and 12 % in India [8,9]. From Asian and African countries “stone belt” have been recognised including Egypt, the Middle East, and parts of Asia. Prevalence of stone disease in these areas reported remarkably at high risk ranging from 10% to 15%. Current data provide information about stone belt dispersed into Sub-Saharan Africa which has been potentially driven by rapid economic growth, urbanization with dietary alteration converting to westernized diet [10]. Recurrence rate of Kidney Stone Disease (KDS) ranges from 6.1% to 66.9% worldwide. Within 10 years timespan, 50 % of affected population undergoes second episode of renal colic [11]. 7.7 % to 12 % world population reported suffered due renal stone disease, where males were more affected than female [12,13]. It was conceded as lifelong disease because of higher chances of recurrence which may leads into chronic renal failure, end-stage kidney disease, type II diabetes mellitus, hypertension, cardiovascular associate pathologies, obesity, urinary acidification and dyslipidaemia [14,15].
Back to the history, kidney stone and its remedies were described in old Sanskrit literature in India like Vedas, Puranas and Samhita. The great Charak Samhita written by Charak had beautifully explained the medical background like anatomy, physiology and pathology of kidney and kidney stone. Acharya Sushrut also had explained characteristics of renal calculi, etiology and symptoms with the scientific base and explanation in his book Sushrut Samhita [16]. The history of kidney stones dates back to ancient civilizations. with early references to the condition appearing in ancient texts and evolving through medical and scientific advancements.
Examinations of Egyptian mummies have reported with number of evidence of kidney and bladder stones. In 1901, English archaeologist E. Smith discovered a 5,000-year old bladder stone at El Amrah, Egypt, also, he reported four cases of such stones among thousands of mummies, suggesting that stone disease was rare in ancient Egypt. The Papyrus Ebers (1500 BC), one of the oldest medical texts from ancient Egypt, is one of the earliest records of kidney stones, describing symptoms related to urinary discomfort and mentioning herbal remedies for treating the condition [17]. The Corpus Hippocraticum, as a collection of around 60 early ancient Greek medical works strongly associated with the physician Hippocrates (400 BC) were found scientifically important contributions to world of urology, including advances in urine analysis, a theory of stone formation, and knowledge of stone removal. It also emphasized ethical care and treating both the body and mind, ideas that are still important today [18].
The Roman physician Claudius Galen (130 AD) showed great interest in diagnosing and treating kidney stones. He emphasized examining symptoms like urine retention and identifying whether the blockage was caused by stones, thick humors, or other issues in the kidneys or ureters. In his work On the Natural Faculties, Galen noted that stones often caused sharp pain between the kidneys and bladder while traveling through the ureters, with pain and urine retention stopping once the stone passed [19].
During the medieval period, Persian scholars like Avicenna also known as Ibn Sina (980–1037 AD) and Abu Bakr Mohammad Ibn Zakariya Razi, known as Rhazes (865 to 925 AD) made significant contributions to the understanding of kidney stones. Avicenna’s Canon of Medicine described using grasping forceps and mechanical litholopaxy to manage bladder stones. He noted that kidney stones responsible for intensive pain than bladder stones. He emphasized the importance of anatomy knowledge during cystolithotomy to minimize the risk of sterilizing the patient [20], while Rhazes emphasized the role of environmental factors and diet in stone formation. He contributed his work on kidney and bladder stones, its diagnosis and therapeutic views which were found similar to modern medical knowledge. He was also skilled and known telling the difference between kidney stones, bladder stones, and other conditions like colitis [21].
The Renaissance and early modern periods saw the development of surgical techniques to remove kidney stones, most notably lithotomy, which involved the surgical extraction of stones. The French surgeon Ambroise Pare (16th century) pioneered surgical approaches to treat kidney stones [22,23]
Considering present scenario and clinical aspects, urolithiasis is one of the Global Burdens of Disease (GBD) with high rate of recurrence percentage. So to overcome this disease, cost of novel technologies, medical management and healthcare was also very high [24]. Economically, the cost of Kidney Stone Disease (KSD) comparative similar and high to the prostate cancer and bladder cancer combined in the United Kingdome [25]. The developmental level of the country was indicated by Sociodemographic Index (SDI). Which can be calculated and analysed by gross domestic product per capita, mean education in age group 15 and onwards and total fertility rate under 25 years old. Generally SDI scored ranges was from 0 to 1. Below the 0.7 indicate burden of urolithiasis. Opposite to this when the SDI value is more than 0.7 indicates complete medical and health system [26]. Thus, the impact of KDS affects overall socioeconomic standard which alternately decline the productivity and nations economy.
METHODOLOGY OF LITERATURE SEARCHIN
We conducted a comprehensive search of electronic databases, including PubMed, Science Direct, Scopus, Google Scholar, and Research Gate, to gather information on various aspects of urolithiasis. The search covered the period from 2000 to 2024. Key terms used in the search included ‘kidney stone,’ ‘urolithiasis,’ ‘nephrolithiasis,’ ‘kidney stone inhibitors,’ ‘kidney stone promoters,’ ‘types of kidney stones,’ ‘recent theories on kidney stone formation,’ ‘formation of kidney stones,’ ‘risk factors,’ ‘diagnosis of renal stones,’ ‘animal models.’ And ‘AI in urolithiasis.’ for kidney stone research.
Human Excretory System and Location of Kidney Stone:
Among human developmental organ differentiation, excretory system proved one of the major organ systems which performs multiple function. One of primary function of excretory system was to remove nitrogenous waste i.e. excretion. As well as these it contributes for homeostasis, maintenance of blood pressure and pH, water and electrolyte balance [27]. It also regulates hormonal coordination like RAAS (Renin – Angiotensin Aldosterone System [28], erythropoiesis by erythropoietin hormone [29], also controlled by the kidneys.
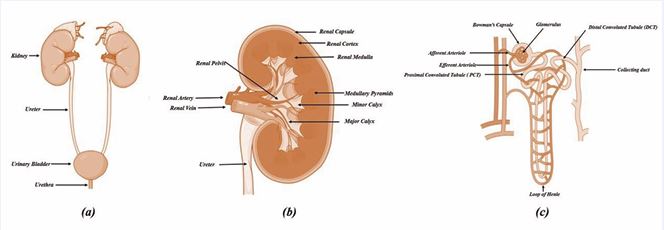
Urinary system situated in upper abdominal cavity on ether site of vertebral column. Figure 1a. showed human excretory system consist pairs of kidneys (Renes) which was in concern with a primary excretory organ that eliminate the nitrogenous waste, pairs of ureters that convey the urine from kidney to the urinary bladder, urinary bladder (Vesica urinaria), where the urine get stored temporarily and urethra who discharge the urine from bladder to outside the body [30]. Kidneys are the prime excretory organ which performs more than 30 different physiological actions [31]. Gross anatomy showed kidney has is dark red in colour, measures about 17 cm in length and 7 cm in width. Histologically it can be differentiated into two regions, outer renal cortex and inner renal medulla.
Major structural architecture found inside the kidneys are renal pyramids, minor calyx, major calyx, renal and renal pelvis as showed in Figure 1b. Renal pyramids are packed with functional unit of the kidney i.e. Nephrons. Nephrons are nothing but filtration unit found combinely in renal cortex and renal medulla. Nephrons are divides into two parts 1) Malpighian Body consist a combination of Bowman’s capsule and network of capillaries i.e. glomerulus. 2) Renal tubules are differentiated structurally and functionally into Proximal Convoluted Tubule (PCT), loop of Henle, Distal Convoluted Tubule (DCT) and collecting duct in Figure 1c [32,33].
Figure 1 a. Human excretory system, b. Internal structure of kidney, c. Ultrastructure of nephron [33].
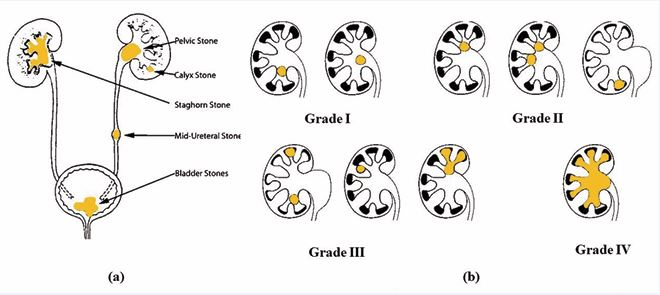
Kidney stones are polycrystalline in nature. 97% of renal stones are found in the kidneys and ureters whereas the remaining 3% found in urinary bladder and urethra [34]. Nephrolithiasis referred when kidney stone found kidneys. Cystolithiasis and ureterolithiasis are the condition where stone found in urinary bladder and urethra respectively [35]. In kidney different structural modifications are seen as non-staghorn and staghorn stones. Non-staghorn stones located in calyx or pelvic [36]. Staghorn stones are large branching stones which occupies renal pelvis and renal calyces [37].
Near about 70% of staghorn stone comprises mixtures of magnesium ammonium phosphate also known as struvite and calcium carbonate apatite [38]. Depending upon location, stone in the ureter were classified as proximal, middle or distal stone Figure 2a [36]. Also, can classified as simple stone which found located in independently in renal pelvises or calyx or complex stone as partial or complete staghorn stone [39].
Figure 2 a. Location of kidney stone in urinary system [36], b) Kidney stone gradation system [41]
Thomas et al. developed the Guy’s Stone Score (GSS)in 2011. GSS is based on complexity, anatomy and position of the stone [40]. As showed in Figure 2b [41]. Kidney stone grade system (GSS) include following gradation as [42],
Grade I - A solitary stone in the mid/lower pole with simple anatomy or a solitary stone in the pelvis with simple anatomy
Grade II - A solitary stone in the upper pole with simple anatomy or multiple stones in a patient with simple anatomy or any solitary stone in a patient with abnormal anatomy
Grade III - Multiple stones in a patient with abnormal anatomy or, stones in a calyceal diverticulum or partial staghorn calculus.
Grade IV - Staghorn calculus or any stone in a patient with spina bifida or spinal injury.
Composition, Types and Shapes of Kidney Stone:
Under the anatomical, morphological and histological investigation inflammatory or pathetic condition reflects stage of urinary crystallization. Nutrition dependent, metabolic products and biproducts also gives information about stone composition. Biochemical and mineral based biproductive entities compositions induce superficial or in-depth inflammation to nephrotic cells. Variety of the kidney stones enlights pathophysiological conditions and can damage different excretory cell, tissue and system [43-45]. On the other hand, the balanced factor like urine sodium, citrate, oxalate, uric acid and calcium and specific gravity associates with stone formation [46]. There are number of biological and metabolic conditions in association with age, weight, height body composition, hormonal fluctuations and even gender differentiation reflects over crystallization. Present review has been made to understand factors influencing to its composite content and type of differentiation.
Composition: In 19th century the chemical characterization of urinary calculi was firstly demonstrated by Austrian chemist J. F. Heller [47]. Review concluded that more than 100 chemical compounds have been identified from urinary stone [48,49]. Documented that, urinary stone comprises two subunits i.e. and Non crystals and Crystals subunits. Non crystals subunits are also called as organic matrix which include Glycosaminoglycan’s (GAGs), lipid, carbohydrates and protein. Non crystals and Crystals subunits combines together to initiate process of kidney stone formation. The contribution of organic matrix in the stone formation process are protein (64 %), nonamino suger (9.6%), hexosamine as glycosamine (5%), water (10%), phospholipid (8.6 %) and inorganic ash (10.4%) [50]. On the other hand, crystal subunits include various forms major chemical compositions includes Calcium, Magnesium, Ammonium phosphate, Uric acid and Cysteine. in which almost 70-75 % of urinary stone are calcium containing stone [51].
Types and Structure of Stone: Based upon presence of major crystals composition, stones can be categories into two groups as I) Calcium, II) Non Calcium stones [52].
Calcium Stone: The high concentration of calcium carbonate in water and low nutrients diet is the major responcible factors for the calcium dependent crystallization and relevent stone formation [53]. As per study, the Calcium Oxalate (CaOx) stone found most common type of urolithiasis which has biochemically combination of calcium and oxalate ions. At high level of calcium responsible for stone, the condition reported as Hypercalciuria, whereas presence of high oxalate in the stone called as Hyperoxaluria [54]. The key contributor i.e. oxalate get excreted in the urine and concederd as major risk factor in formation of CaOx stone. The plasma oxalate hastwo different sources, one as exogenous, mainly dietary sources and other as endogenous which were formed due to various metabolic activities.
The members of anion exchanging family i.e. solute linked carrier26 (SLC26) present in the gut absorbed the dietary oxalate and immediately excreted by kidney [55]. The bio-metabolism in liver (60 to 80 %) red blood cells and ascorbic acid were major endogenous sources for the oxalate [56,57]. Documented Structurally calcium oxalate found in three different crystallographic forms i.e., calcium oxalate monohydrate (COM or whewellite, CaC2O4.H2O ) has dumbbell shaped and calcium oxalate dehydrate ( COM or weddellite, CaC2O4.2H2O ) has envelop shape [58] and calcium oxalate trihydrates (CaC2O4.3H2O). In calcium oxalate crystals, calcium oxalate monohydrate crystals (COM) found as oxalate dependent whereas Calcium Oxalate Dehydrate crystals (COD) was calcium dependent [59].
Clinically it has been observed that after the calcium oxalate, the second most common type of stone are made up of with various combinations like, calcium phosphate, calcium carbonate brushite, gypsum and dolomite. Among which, Calcium phosphate contribute dominant share with 5 % of total combination. Like Calcium oxalate stones, Calcium phosphate stones were not common. It may be caused by various internal factors like hyperparathyroidism, renal tubular acidosis, and urinary tract infections [60]. Calcium Phosphate found in urinary stones as calcium orthophosphates (amorphous carbonated CaP and carbapatites) and brushite (dicalcium phosphate dihydrate). Calcium orthophosphate are mostly dependent on urinary pH dependent (when urine pH was found above 6.5) where for brushite high concentrations of calcium and phosphate were required [61,62].
Non-Calcium Stone:
a) Struvite Stone: Struvite (MgNH4.PO4·6H2O) as a crystalline substance composed of magnesium ammonium phosphate. It comprises 10 to 15% of kidney stone. It was known as “triple phosphate” stones because of identification of three ions i.e. calcium, magnesium, ammonium [63]. The enzyme urease produced by the bacteria species proteus splits urea into ammonia and carbon dioxide and results alkaline urine and favors the struvite formation hence it was also referred as infection stones [64,65]. Reported that, struvite stones infection found more commonly in females than the male with ratio 3:1. In the acute infection (acute pyelonephritis), individuals may suffers from fevers, chills, irritative voiding symptoms, cloudy urine with flank pain. The chronic infection the symptomatic condition might be non-specific like malaise and generalized weakness but if infection and obstruction were prolonged it may lead into Xanthogranulomatous Pyelonephritis (XGP), a rare, chronic kidney infection that causes destruction of renal parenchyma resulting failure of partial or complete kidney [66].
b) Cysteine Stone: Two molecules cysteine combines by disulfide bond and forms Cysteine as amino acid. Cystinuria as inherited condition where the trans epithelial transporters in renal proximal tubules get defected by the genes SLC3A1 and SLC7A9, and leads into higher excretion of cysteine from kidney. Scientifically known as basic reason for the supersaturation of urine by cysteine and leads into formation of cysteine crystals [67,68]. Among all urolithiasis cases, the rate of Cystinuria is 1–2%. But in paediatric patients it is more dominant with 6–8 %. Recent study highlighted revised classification of Cysteine stone as Type I, Non type I and Mixed type of Cystinuria. Type I Cystinuria (Obligate heterozygotic relatives of patients with normal aminoaciduria, Non-Type I Cystinuria (obligate heterozygotic relatives of individuals with high or moderate hyperexcretion of cystine) and Mixed type cystinuria (obligate heterozygotic relatives of individuals with high or moderate hyperexcretion of dibasic amino acids; lysine, arginine and ornithine) [69]. Type I patients have SLC3A1 mutations, Non Type I patients have SLC7A9 mutations, and Mixed type patients have mutations in both SLC3A1 and SLC7A9 [70]. In preventive measure, increased fluid intake greater than 3 liters per day and chemical reaction point of view, limiting the sodium and protein diet and alkaline urine pH can reduce the risk of Cystinuria [71].
c) Uric Acid Stone: Uric acid stones were found smooth, round, yellow- orange in nature. It radiographically transparent until get combined with other molecules like calcium crystals or struvite. Excess of uric acid accumulation called as hyperuricosuria. High purine diet, low urine volume and low urinary pH increases the risk of uric acid stone formation [72]. Uric acid contributes 10–20% of all urolithiasis cases. Near about 300–400 mg/dL uric acid was produced endogenously. Exogenous sources include high purine diet such as meat, animal organs, fish metabolically converts into uric acid ( purine → free nucleic acids → inosinic acid → hypoxanthine → xanthine (by xanthine oxidase) → uric acid) [73,74]. Documented that, those who were facing problem related with obesity and diabetes have high risk of formation of uric acid stone. Acidic urine is common in people with problems in ammonia production or higher acid excretion . Insulin helps to increase ammonia production and activates a process to balance ions. But when there was insulin resistance, ammonia production and transport decrease, causing low urine pH. In people with metabolic syndrome, being overweight and having insulin resistance are key factors in stone formation. Patients with uric acid stones and found their average BMI was 30.0 kg/m² [75].
d) Medicated Stone: A medicated stone also known as drug-induced kidney stone, forms when certain medications or their byproducts crystallize in the urinary tract. Near about 2 % of cases of urinary tract calculi are of Drug-induced kidney stones. These drugs were categories into (1) drugs with that have high renal excretion or poor solubility in urine and (2) drugs that alters the metabolic changes who promote urinary stone formation [76]. Common drugs associated with stone formation include atazanavir and indinavir, antiretroviral medications known to form poorly soluble crystals in the urinary system [77]. Topiramate, used for epilepsy and migraines which elevates urine pH and promote calcium phosphate stone formation [78-80]. Sulfa drugs, such as sulfamethoxazole, can crystallize in acidic urine, especially in dehydrated patients [81,82]. Triamterene, a diuretic, recomonded another common drug can obstruct the urinary tract [83]. Excessive use of calcium-containing antacids or supplements can increase calcium excretion, leading to calcium-based stones [84]. Similarly, high-dose vitamin C and D supplements can raise urinary oxalate levels, increasing the risk of calcium oxalate stones [85,86].
e) Rare Type of Kidney Stones: Rare and uncommon kidney stones can have a variety of compositions, ranging from metabolic byproducts to toxic compounds. These stones often require advanced diagnostic techniques, including specialized imaging or chemical analysis, to identify and differentiate them from more common types of stones. Some of the rare and uncommon stones are as follows
Shape and Structure of Stones: Morphometric and biochemical identification of urine crystals in the laboratory can be done with the help of published photographs and respective solubility test [102]. Urinary crystals found in different shape and size which are dependent on differences in the supersaturation of lithogenic salts, pH value, and content of inhibitors and promoters in the individuals [103].
THEORIES FOR KIDNEY STONE FORMATION
Kidney stone formation is a multifactorial process influenced by various biological, chemical, and environmental factors. Over the years, multiple theories have been proposed to explain the mechanisms behind nephrolithiasis. These theories reflect advancements in molecular biology, imaging, and biochemistry, providing a deeper understanding of stone pathogenesis. The key theories are:
Free Particle Theory: Theory was proposed by Vermeulen and Lyon (1968) and documented that, formation stone take place at the narrow portion of the nephron i.e. tubular lumen of the distal nephron, where crystal forming component spontaneously precepted by means of supersaturated urine and begins to aggregate and initiate progressive growth in the morphological feature when urine passes though the kidney. When it reached into the collecting duct due to the enlargement in size it get obstructed. In special cases like cystinuria and CaOx stones the individuals shows high urinary levels of cystine and oxalate and lead into intratubular nucleation [106,107].
Fix Particle Theory: This theory was proposed by Finlayson and Ried (1978). Biochemically, calcium oxalate and calcium phosphate crystals were commonly found in the urine of effected person similar to that of normal healthy person [108]. According to this theory, at the site of the renal tissue injury (due to crystal, virus or bacteria), supersaturated urine crystals were attached to the renal epithelium which lead to formation of aggressive stone [109].
Randall’s Plaque Hypothesis: The theory was given by Alexander Randall (1937) suggested that, accumulation of the calcium phosphate on the tip of the renal papillae gave an ideal foundation for the formation of the calcium oxalate stone because calcium phosphate frequently found at the core of calcium oxalate stone [110,111]. More specifically low urinary pH, low urinary volume and increased calcium level act as promoter which attracts the organic matrix rich in proteins and lipids and membrane bound vesicle, collagen fibres and some components from extracellular matrix to form a aggressive stone structure [112].
Blocked Lymphatic Theory: Concept and theory was proposed by Reginald J. Car (1954). Theory emphasized the importance of the renal lymphatic system in preventing the accumulation of precipitating salts. Under normal conditions, the lymphatic system helps maintain the balance of substances in the kidney. However, when the lymphatic system becomes dysfunctional, salts accumulate in the renal interstitium, facilitating stone formation. This theory, although less explored in recent times, has gained interest in studies investigating renal interstitial fluid dynamics and their relationship with nephrolithiasis [113,114].
Vascular Hypothesis: Its new vascular hypothesis of stone formation. The vascular hypothesis proposes that changes in renal blood flow, especially fluctuations from laminar to turbulent flow, can cause injury to the renal vasculature. This vascular injury may lead to the formation of atherosclerotic-like conditions, promoting calcification and stone formation. Recent studies suggest that the vasa recta, through which blood flows in the kidney and damaged by these fluctuations, contributing to the pathogenesis of nephrolithiasis [115].
Oxidative Stress Theory: Oxidative stress has recently been implicated as a key factor in kidney stone formation. Reactive Oxygen Species (ROS) produced in the kidney can lead to renal epithelial cell damage, apoptosis, and the initiation of crystal formation. The imbalance between ROS and antioxidant defences can accelerate rate the deposition of calcium oxalate crystals. Above theory emphasizes the role of oxidative damage in kidney stone pathogenesis, highlighting the potential therapeutic benefit of antioxidants in preventing stone formation [116,117].
Immune Reactive Theory: Emerging evidence suggests that the immune system plays a significant role in kidney stone formation. Specifically, macrophage activation and differentiation in response to crystal deposition may contribute to the inflammatory environment in the kidney, promoting stone growth. Inflammatory cytokines and the recruitment of immune cells to the site of stone formation may enhance crystal aggregation. This theory points to the complex interplay between the immune system and nephrolithiasis, providing a potential target for immunomodulatory therapies [118-120].
Gut Microbiota Theory: Recent studies have highlighted the role of gut microbiota in kidney stone formation. Certain bacteria, such as Oxalobacter formigenes, known to metabolize oxalate, potentially reducing the risk of calcium oxalate stone formation. Alterations in the composition of gut microbiota, or dysbiosis, were found linked with high risk of kidney crystallization. The gut–kidney axis, through which microbial metabolites influence renal function, represents a novel area of research in nephrolithiasis [121-123].
Matrix Theory: Matrix theory propose aggregation of organic compound within the renal interstitium, such as glycoproteins and mucoproteins, provided substrate for crystal nucleation. These macromolecules facilitated aggregation of mineral crystals, promoting stone formation. Alterations in the composition or concentration of these organic matrices may influence the propensity to develop kidney stones. This theory emphasized the role of extracellular matrices in the pathogenesis of nephrolithiasis [124,125].
Inhibitor Deficiency Theory: This theory underscores the importance of natural inhibitors found in urine, such as citrate, magnesium, and certain glycoproteins, in preventing stone formation. Generally inhibitors play a crucial role in preventing the aggregation of crystals by binding to them and elevate the mechanism of excretion. A deficiency or dysfunction of these inhibitors, such as in conditions like hypocitraturia, significantly increases the risk of stone formation. Clinical evidence has showed that restoring normal levels of urinary inhibitors proved to be a key strategy in managing and regulation of high risk for nephrolithiasis [126-129].
BIOMECHANISM OF CRYSTALLIZATION
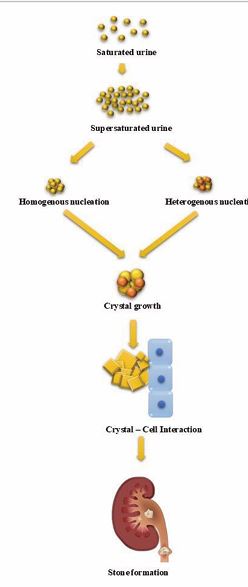
Kidney stone formation, or biomineralization found to be a complex biochemical process which has been critically governed by two primary factors: promoters and inhibitors. Metabolically promoters are components that facilitate crystallization, including low urine volume, acidic urine (low pH), and elevated levels of calcium, sodium, oxalate, and urate. Opposite to this, inhibitors were factors that counteract the biochemical processes involved in stone formation. Common inhibitors include inorganic substances like citrate and magnesium, as well as organic components such as urinary prothrombin fragment, glycosaminoglycans, and osteopontin. The process of kidney stone formation progresses through a series of well-documented steps: saturation, where solutes get dissolved in urine; supersaturation, when their concentration exceeds solubility limits; nucleation, the formation of initial crystals; crystal growth or aggregation, where crystals grow or cluster together; crystal retention, where crystals adhere to renal epithelium or remain trapped in tubules; and finally, stone formation, where the aggregated mass developed into a clinically significant crystal formation and bioaccumulation [130,131].
a) Saturation and Supersaturation: Supersaturation is a key concept in kidney stone formation, defined as the ratio of the concentration of dissolved salts in urine to their solubility. It serves as a measure of urine’s potential to precipitate and aggregate solid crystals. Supersaturation not only reflects the mineral composition of kidney stones but also provides valuable insight into the risk of stone formation. Higher levels of supersaturation indicate a greater likelihood of crystal nucleation, growth, and aggregation, making it a critical factor in understanding and predicting urolithiasis [132,133]. Most of study reveled that, the initial driving force for the formation of stone found duo to saturation of urine itself. When the salts added into the solvent it starts to dissolve. At the typical point no more salt get dissolve into the solvent. This particular point called as saturated point of solvent to the salt. If more amount of salt is added in the solvent, there was a initiation of crystallization called as thermodynamical solubility product (Ksp) [134]. Calcium oxalate monohydrate, calcium phosphate such as Octo calcium phosphate, uric acid and struvite contribute dominant share for supersaturation [135].
b) Crystals Nucleation: The biochemical process of urinary stone formation begins with the aggregation of supersaturated excretory particles, such as free atoms, ions, and salt molecules, which cluster together and form a crystal nucleus. Nucleation, the process of crystal formation, can be divided into two types. Primary nucleation occurs when crystals of the precipitating phase form spontaneously, without the involvement of pre-existing particles. Primary nucleation further classified as homogeneous nucleation, where nucleation occurs spontaneously when supersaturation reaches an adequate level. On the other hand, secondary nucleation occurs when nucleation initiates on an existing surface, such as epithelial cell membranes, cell debris, other crystals, red blood cells, or the urine stream. By literature, type of nucleation is known as heterogeneous nucleation [136,137]. Documented that, Disfunctioning of mitochondria alters the mechanism of energy production, regulation of oxidative stress and intracellular calcium homeostasis and a key factor forth formation of crystal nucleus.
c) Crystal growth and Aggregation - Urinary stones form through crystal growth, where crystals in urine aggregate to form hard masses. Crystallization begins when a crystal nucleus forms in the nephrocytes and exposure to urine promotes further growth through encrustation. Crystal growth can occur through two mechanisms. The free particle mechanism involves crystals growing and aggregating within the urine of renal tubules. As the crystals enlarge, they become too large to be excreted and remain trapped within the kidney. However, they act as inhibitors when it comes to crystal aggregation and growth, preventing the crystals from becoming larger. Similarly, Tamm-Horsfall glycoprotein, depending on its stage of aggregation, may either promote or inhibit crystal formation. Considering dual role of glycoprotein, it highlights the complex and dynamic nature of stone formation, where the same substance can influence the process in different ways depending on the phase of crystal growth.
Figure 3 Biomechanism of Stone formation [142-144].
d) Crystal – Cell Interaction - [140] reported that, when fully grown crystals attach to the surface of the renal epithelium, this process known as retention or crystal cell interaction. In vitro studies have demonstrated this phenomenon, particularly through crystal-cell adhesion experiments using Madin Darby Canine Kidney (MDCK) strain I cell line cultures. Experimentally, reports have concluded that healthy and intact epithelial cells were nonadherent, meaning that crystals do not easily attach to the surface of normal renal epithelial cells. However, if there was any injury or wounding to the epithelial surface, the surrounding hyaluronan-rich pericellular matrix plays a crucial role in promoting crystal-cell adhesion. Under cellular interaction matrix facilitates the binding of crystals to the damaged cells, contributing to the retention of the crystals and potentially aiding in the formation of kidney stones.
Cell injury and apoptosis – Available literature provides information that, crystals in the renal tubules can directly damage the epithelial cells, triggering a cascade of intracellular events. Crystals can caused mechanical damage to the cell membrane, leading to increased oxidative stress, inflammation, and mitochondrial dysfunction. Cellular damage biologically leads to initiation or activation of apoptotic pathways, causing the affected cells to undergo programmed cell death. In particular, extrinsic and intrinsic apoptotic pathways may be activated in response to crystal-induced injury. Biochemically extrinsic pathway get triggered by crystal adhesion to cell surface receptors, while the intrinsic pathway is activated by cellular stress, such as oxidative damage or mitochondrial dysfunction [141]. Exposure to high level of oxalate or calcium oxalate crystals induces epithelial cellular injury lead to apoptosis where prominent and cell death found leading to inflammations of nephrocytes and serve kidney damage [49].
KIDNEY STONE AND DIET
It is clear that, Formation of kidney stone is associate with age, gender, genetics, lifestyle, environmental factors, geographical location and dietary habitat [145]. Among which nutritional exposure component were reported most dominant factors involved in the increased frequency of risk of kidney stone among the population [146,147], reported that, the diet with animal source protein may cause metabolic acidosis and further progress into nephropathy. It reduces glomerular filtration rate.
Table 1: Rare Type of kidney stones
|
Sr. No |
Name of the stone |
Causes |
Effects/ Symptoms |
References |
|
1 |
Xanthine Stones |
Deficiency of enzyme Xanthine oxidase |
Hypouricemia and hypouricosuria |
[87] |
|
2 |
Ammonium Urate Stones |
Irritable Bowel Syndrome (IBS), laxative abuse, Obesity, Urinary diversion, Pregnancy, Chemotherapy, and Recurrent Urinary tract infections |
CKD, UTIs, IBS, Gout, and even Urothelial carcinoma. |
[88] |
|
3 |
Matrix Stones |
Previous surgery for stone disease and/ or recurrent UTIs, especially due to Proteus mirabilis or Escherichia-coli |
Similar to calcium nephrolithiasis, flank pain and UTI |
[89,90] |
|
4 |
Silica Stones |
Magnesium Trisilicate, Antacid medication contains Silica, Silica rich diet, Urine pH, and concentration of silicate |
Acidic urine. Secondary UTIs may occur as a result of mucosal irritation |
[91,92] |
|
5 |
Calcium Carbonate Stones |
Metabolic disorder, Hypomagnesuria, Hypercalciuria, Hyperuricemia, Hyperuricosuria, and Hyperoxaluria. |
Urinary Tract Infection (UTI) Pyuria, Hematuria, Proteinuria. Abdominal pain radiating to the scrotum or labia, Fever, Nausea, and Vomiting |
[93] |
|
6 |
Lipid Stones |
Low High Density Lipoprotein (HDL) and high Triglycerides level |
lower urinary pH, Associate with urinary uric acid |
[94,95] |
|
7 |
Silicon Dioxide (Quartz) Stones |
Excessive intake of Magnesium trisilicate in food or Medications |
Urine alkalinization, Promotes crystallization of urine components such as calcium phosphate by inhibition of carbonate dehydratase. |
[96,97] |
|
8 |
Opaline silica |
Kaolin consumption, |
Silica nephropathy |
[98,99] |
|
9 |
2,8-Dihydroxyadenine (2,8-DHA) |
Genetic deficiency of enzyme Adenine Phosphoribosyl Transferase (APRT) |
Chronic Kidney Disease (CKD) that can progress to End-Stage Renal Disease (ESRD). |
[100,101] |
Table 2: Schematic presentation of various shapes of kidney stones [104,105]
|
|
||
|
Fig. No. |
Type of Crystal |
Shape |
|
1 |
Calcium Oxalate Monohydrate ( COM) |
Oval, Biconvex, and Dumbbells to Elongated rods |
|
2 |
Calcium Oxalate Dihydrate ( COD) |
Bipyramidal or Envelope-shaped |
|
3 |
Calcium Phosphate (CaP) |
Prisms, Rosettes, Needle-like or Splinter-like, or Sticks or Rods |
|
4 |
Struvite |
Coffin lid shape |
|
5 |
Uric acid |
Rho boards, Barrels, Rosettes, 4-sided plates, Needles, Rounds, or Parallelograms. |
|
6 |
Cystine |
Hexagonal plates. |
On the other hand the regular intake of neutral diet rich with plants can increases stone inhibitors like urine pH, volume, phytate, citrate, potassium and magnesium. In the chemical analysis of kidney stone from various regions diverse element were identified like calcium (Ca), magnesium (Mg), manganese (Mn), copper (Cu), iron (Fe), zinc (Zn), strontium (Sr), sodium (Na),potassium (K), phosphorus (P), sulphur (S) and chlorine (Cl) in Indian kidney stone patients. phosphorus (P), sulphur (S), silicon (Si), zinc (Zn) and titanium (Ti) were common elements of stones founds in Turkish patient . (Ca) and (P) were dominant component found in Iranian patients, Along with Calcium (Ca) some metals like Na (1.56%), K (0.9%), Mg (3.08%), Fe (1.17%), Al (0.49%), Zn (0.7%), Cu (0.19%),Mn (0.029%), P (10.35%), S (1.88%), Sr (0.306%), Mo (0.2%), Cr (0.146%), Co (0.05%) and Ni (0.014%) were identified in from the patients from Jorden.
Table 3: Genes associated with kidney stone disease.
|
Sr. No |
Type of Disorder |
Gene responsible |
Type inheritance |
|
1 |
Familial hypercalciuria |
ADCY10, VDR |
Autosomal dominant |
|
2 |
Autosomal dominant hypocalcemia |
CASR, GNA11 |
Autosomal dominant |
|
3 |
Bartter syndrome |
NKCC2 (SLC12A1), ROMK (KCNJ1), CLCNKB, BSND, CASR, CLCN5 |
Autosomal dominant and Autosomal recessive or X linked recessive |
|
4 |
Dent disease |
SLC34A1, SLC34A3, SLC9A3R1 |
X linked recessive |
|
5 |
Familial hypomagnesaemia with hypercalciuria and nephrocalcinosis |
CLDN16, CLDN19 |
Autosomal recessive |
|
6 |
Cystinuria |
SLC3A1, SLC7A9 |
Autosomal dominant or Autosomal recessive |
|
7 |
Hyperuricosuria |
HRPT1, PRPS1 |
X linked recessive |
|
8 |
Xanthinuria |
XDH, MOCOS, MOCS1, MOCS2, GPHN |
Autosomal recessive |
|
9 |
Failed urinary acidification |
SLC4A1, ATP6VB1, ATP6VA4, CA2 |
Autosomal dominant or Autosomal recessive |
|
10 |
Hyperoxaluria |
AGXT, GRHPR, HOGA1, SLC26A1 |
Autosomal recessive |
Lin [148], reported in his study enlighted various dietary factors responsible to enhancement and prevention of process of formation of kidney stone. The intake of red meat have high risk of stone formation than the poultry and fish consumption. The daily and high intake of fruits, fibres and vegetables reduces the risk of stone formation. But in case of spinach due to its rich oxalate content The risk of nephrolithiasis observed. In liquid intake, consumption of 2 litters of water per day may reduces the risk of stone formation almost half.
Legay C [149], reported that inadequate amount of fluid intake always higher the risk of stone formation. Some of the beverages act as enhancer while some are reducers in the stone forming mechanism. Non alcoholic beverages like tea, coffee and alcoholic beverages like beer and wine are act as reducers to the stone forming and decreases the risk. Overall he concluded that urine dilution and concentration of beverages decides where leading to decreasing or increasing the risk. The most common oxalate rich food includes vegetables, legumes, fruits, cereals and pseudocereals, nuts, herbs and spices. Specifically The spinach have a rich source of oxalate in it. it was observed that cooked spinach has (364 mg/100 g) amount of oxalate found than the raw spinach (1959 mg mg/100g ) which pooves the food processing also concederd as a factor responsible for kidney stone formation [150].
Factors Affecting / Risk Factors of Kidney Stone
Daily Fluid Intake: Under balanced food intake, hydration and dietary habits play a pivotal role in reducing the risk of kidney stone formation, with sufficient water intake being the cornerstone of prevention. Studies have reported and highlight that consuming more than 2500 ml of water daily, coupled with achieving a urine output of at least 2000ml/day, which can significantly reduces the prevalence of kidney stones [151,152]. This underscores the importance of adequate hydration in maintaining urinary tract health. Interestingly, at the amount of water consumed found crucial, physiochemical parameter, such as its hardness, appears to have minimal impact on kidney stone risk, emphasizing that it is the volume of fluid intake that matters most [153].
Alongside hydration, dietary choices are equally influential in modifying the risk of kidney stones. Carbohydrate enriched beverages, including both non-cola and cola sodas, have been shown to increase the risk of kidney stone formation by 23–33% when consumed daily, compared to less frequent intake [154]. This suggests that avoiding or minimizing these beverages proved to be critical preventive measure to restrict the crystallization and deposition in kidney.
Rodal K [155], reviewed that, Caffeine showed promise in protecting kidney health by reducing kidney stone risk and preventing fibrosis. Studies reveal that caffeinated coffee and tea lower stone risk by 10 % and 8 % per 240 ml serving, respectively. Caffeine also inhibits calcium oxalate crystal growth and adhesion to kidney cells by reducing annexin A1 levels. Additionally, it protects against renal fibrosis by reducing oxidative stress and restoring normal cellular function during hypoxia. These findings highlight caffeine’s potential as a preventive agent for kidney stones and fibrosis. Phytochemically various fruit juices like orange, apple, and grapefruit significantly decreases Relative Supersaturation Risk (RSK) for development of calcium oxalate stone [156].
Supersaturated Urine: Saturation and supersaturation crystallization occurs when stone-forming salts like calcium oxalate, calcium phosphate, uric acid, or cystine aggregate and precipitate in the urine at high concentrations, leading to significant crystal development. Crystallization get influenced by several physiochemical factors, including urine volume, pH, density, viscosity, fluidity and the presence of inhibitors or promoters of crystallization. Hypovolumia, often due to dehydration, seasonal accumulation of concentrated solutes and turbid volume, all these were reported as risk factors of supersaturation, which can provoke calcification and stone formation. Urine pH plays a significant role, with acidic urine promoting uric acid and cystine stones, while alkaline urine favours calcium phosphate stones. Calcium oxalate stones commonly form in neutral to slightly acidic conditions, depending on the availability of calcium and oxalate ions [157,158].
Hypercalciuria: In normal condition concentration of calcium in extracellular fluid found ranges between 8.5 to 10.5 mg/dl (2.1 -2.5 mM) in which 50 % of serum calcium found associate with plasma protein, small amount of calcium contributes to forms citate and remaining are in the free ion form. [159]. Scientifically hypercalciuria as a pathological condition where excess amount of calcium reported the urine hypercalciuria, condition which has been characterized by excessive calcium excretion in the urine 0.1 mmol Ca/kg/d (4 mg/kg/24 h), plays a central role in the inflammation of nephrocyte and crystallization, especially calcium-based stones such as calcium oxalate and calcium phosphate [160]. These types of kidney stones were the most common, comprising the majority of stone cases. The primary mechanism by which hypercalciuria contributes to stone formation may be due to increased in urinary calcium concentrations, which raises the supersaturation of calcium salts. Supersaturation reported as critical factor in stone formation, as it promotes the crystallization of calcium salts and their aggregation into larger stones [161].
Genetic factors play a significant role in hypercalciuric nephrolithiasis such as monogenic condition, particularly in childhood. Several monogenic biological conditions, such as Bartter syndrome, Dent’s disease, Autosomal Dominant Hypocalcemic Hypercalciuria (ADHH), hypophosphatemic hypercalciuric nephrolithiasis, and familial hypomagnesemia with hypercalciuria, been linked to mutations in key renal transporters, channels, and receptors. For example, Bartter syndrome involves mutations in the NKCC2 co-transporter, ROMK channel, or CaSR, while Dent’s disease results from mutations in CLC-5. Clinically ADHH is caused by activating mutations in CaSR, and familial hypomagnesemia found linked to paracellin-1 mutations. Likewise some genetic factors along with acquired condition disrupt calcium and ion handling in the kidneys, increasing the risk of hypercalciuria and kidney stones. Several insights were focused and provided valuable information pertaining to mechanisms regulating calcium reabsorption and the predisposition to nephrolithiasis [162].
Gameel A [163], reported that, Hypercalciuria, or excessive calcium in urine, found key risk factor for kidney stones, with prevalence in children ranging from 46% to 69%. It often runs in families, suggesting an autosomal dominant inheritance pattern. The urine calcium to creatinine ratio generally used to diagnose hypercalciuria, with higher ratios indicating the condition and helping guide treatment to prevent kidney stones. Dietary factors also play a significant role; high sodium or calcium intake can increase urinary calcium excretion [164]. For instance, excessive sodium intake can lead to increased calcium loss in urine because sodium and calcium were both filtered by the kidneys and compete for reabsorption [165]. Moreover, an excess of vitamin D can lead to elevated calcium absorption in the intestines, resulting in higher circulating calcium levels, which, in turn, can contribute to hypercalciuria [166].
Hyperthyroidism: Hyperthyroidism, Parathyroid Hormone (PTH) abnormalities, and kidney stone formation are intricately linked through their effects on calcium metabolism and renal function. Clinically hyperthyroidism reported as pathological condition characteries by increased level of thyroid hormones in the body. Excessive thyroid hormone production in hyperthyroidism leads to an increased metabolic rate, weight loss, palpitation, tremor and bone metabolism also. Number of case studies revealed that patients with hyperthyroidism were associate with hypercalcemia with mild to moderate level. Chen and Kohri [167,168] documented that, not only hypercalciuria but also promoters present in urine from people with hyperparathyroidism promote the formation of calcium oxalate crystals and kidney stones. Elevated thyroid hormones enhance bone resorption, leading to a higher release of calcium into the bloodstream. Biologically, hypercalcemia can stimulate the parathyroid glands to release more PTH in an attempt to normalize calcium levels. PTH increases calcium release from bones and promotes calcium reabsorption in the kidneys, which, in combination with the hypercalcemia reported in hyperthyroidism and was found factor responsible for formation of calcium based kidney stones or crystallization [169-171]. Increased calcium load may lead to nephrocalcinosis, clinical condition where calcium deposits accumulate in the kidneys, further impairing renal function and increasing the risk of both kidney stones and chronic kidney disease [172].
Renal Tubular Acidosis: To maintain homeostasis of body along with lungs, kidney serve as vital organ for maintenance of acid base balance by reabsorbing filtered bicarbonate ions and removing hydrogen ions. Renal Tubular Acidosis (RTA) reported pathological condition where renal tubules were unable to reabsorb and filter the bicarbonate and hydrogen ions respectively result in more acidic urine [173]. In renal pathophysiology, RTA can be classified in 4 types as of Types 1: RTA due to defect in distal tubule, Type 2 RTA due to defect in proximal tubule. Type 3 RTA found very rare and type 4 RTA was characterized by abnormal excretion of acid and potassium involved in the collecting duct, which leads to pathological condition of hyperkalemic acidosis [174,175], documented that, Kidney stone occurs only in type 1 RTA where hydrogen ion gradients were defective in distal tubule. In distal RTA (dRTA), the urine remains alkaline due to the kidney’s failure to excrete hydrogen ions effectively. Biochemically alkaline environment, combined with low urinary citrate levels as a natural inhibitor of stone formation and increased risk for urolithiasis. Scientist have reported that, dRTA was associated with hypercalciuria, further promoting stone formation. Magni [176-178] documented that, In contrast, Type IV RTA is less commonly linked to kidney stones. A study found that patients with type IV RTA had lower urinary pH and calcium excretion compared to controls, suggesting that impaired kidney function in type IV RTA reduces the excretion of stone-forming substances, lowering stone formation risk. Overall, while RTA can increase the risk of kidney stones, the specific risk varies depending on the type of RTA and the associated metabolic disturbances.
Hyperoxaluria: Hyperoxaluria as metabolic condition characterized by elevated oxalate levels in the urine 40 to 45 mg/day (>0.45- 0.50 mmol/day) significantly increased the risk of crystallization and aggregation [179]. The primary role of oxalate in calcification relates with tendency to form insoluble calcium oxalate crystals, which can deposit and bioaccumulate in kidney and urinary tract, leading to urolithiasis. Over the time, these depositions and precipitations can cause nephrocalcinosis which contribute to kidney inflammation and damage leading chronic kidney disease in severe cases [180].In the literatures, hyperoxaluria is classified into two main types: primary and secondary. Primary hyperoxaluria as an inherited metabolic disorder formed due to genetic mutations, leading to excessive oxalate production. Further subdivided into three types: Type 1, which was found caused by mutations in the AGXT gene; Type 2, associated with mutations in the GRHPR gene; and Type 3, linked to mutations in the HOGA1 gene. Secondary hyperoxaluria, on the other hand, was due to increased oxalate absorption from the gastrointestinal tract, often related to gastrointestinal disorders like Crohn’s disease, celiac disease, or bariatric surgery [181,182].
Cystinuria: As per available literature cystinuria reported as rare genetic disorder caused by mutation in genes responsible for transporters of proximal tubule dibasic amino acid. Under clinical condition, biochemically facilitates reabsorption of cysteine, ornithine, lysine, and arginine from tubular fluid [183,184], documented that, the name “cystine” comes from its original description as “bladder calculi” Out of four cysteine was reported as least soluble hence precipitate out and forms renal stone [185].
Deficiency of Stone Inhibitors: In calcium base stone, citrates were reported as is well inhibitor [186,187], reported in his study that, when potassium citrate was administrated to the patients who has uric acid lithiasis, prevention of calcium oxalate nephrolithiasis may be done by reducing urinary saturation and spontaneous precipitation of calcium oxalate. Zukerman [188], documented that, The low urinary citrate excretion less than 320 mg (1.67 mmol) per day for adults as hypocitraturia clinically as known risk factor for the development of kidney stones. Under pathological condition, urinary citrate binds with urinary calcium leading to decreased level of free ionic calcium. Also urinary citrate interact with calcium oxalate crystals and inhibit crystal aggregation crystal growth [189].
Genetics: Near about 10 % of individuals were affected due to a family history of kidney stone disease and congenital genetic disease associate with kidney stone [190-192], in his study documented various genetic factors associated with nephrolithiasis as,
Associate Diseases
Considering biological, pathological and environmental risk factors for developing kidney stones can be categorized as non dietary, dietary, and urinary risk factors responsible for calcification. Along with genetical anomalism, non dietary factors include family history of stone disease, primary Hyperparathyroidism, obesity, Diabetes mellitus and Gout were major reported associate diseases with kidney stone [193-195]. Urolithiasis is associated with several other diseases, including urinary tract infections (UTIs) [196], Chronic Kidney Disease (CKD) [197], and Inflammatory Bowel Disease (IBD) [198]. It can also be linked to conditions such as sarcoidosis [199], medullary sponge kidney (MSK) [200], and celiac disease [201].
Medicated Induction: It is rare type of where several medication may contribute kidney stone disease such as guaifenesin, sulfa drugs (sulfadiazine), indinavir, atazanavir, silicate (antacids), ceftriaxone, and uricosuric agents [202].
PROGNOSIS AND DIAGNOSTIC TOOLS
Under clinical investigation, individuals suffering from kidney disease often experience intensive pain in the lower back, below the ribs, which may radiate to the groin and thigh regions. Common related symptoms include frequent and painful urination, urine that found pink, red, or brown in colour, cloudy or foul-smelling urine, and visible blood in the urine (hematuria). Additional symptoms can include nausea, vomiting, sweating, fever, and chills, which may suggest an associated infection [203]. The diagnosis of kidney stone can be done with three ways i.e. blood testing, urine testing and imaging. Eyre [204], observed and concluded that, presence of renal calculi should primarily be assessed through serum calcium measurement. Other blood screening tests, such as serum phosphate, serum bicarbonate, and serum potassium, are deemed unnecessary for the analysis of renal calculi, as they may provide false reassurance. Furthermore, a 24 hour urine analysis is regarded as the standard of care for the prevention of nephrolithiasis.
The urine analysis identify and predict the risk risks factors associate with nephrolithiasis [205,206]. Clinically noted that, stone less than 5 mm have high passage rate than that of larger size stone. Imaging tools not only confirm the presence of stones but also play a critical role in initial diagnosis, treatment planning, and post-therapeutic management [207]. Various imaging modalities were used to diagnose and manage kidney stones, each tailored to specific clinical needs. Ultrasound is a non-invasive, radiation-free tool often used as the first-line method for detecting stones in the kidneys and bladder, particularly in pregnant women and children [208]. Abdominal X-rays and plain radiography of the Kidney, Ureter, and Bladder (KUB) were cost-effective and quick options for identifying radiopaque stones, though they were less effective for identifying radiolucent or small stones [209,210]. Computed Tomography (CT), especially non-contrast CT, was considered the gold standard due to its high sensitivity and specificity, offering detailed insights into stone size, location, and complications [211].
Advanced techniques like Dual-energy CT can differentiate stone composition, aiding in tailored treatment plans [212]. Magnetic Resonance Urography (MRU) found to be an alternative for patients requiring radiation-free imaging, providing excellent soft-tissue [213]. In contrast, while Intravenous Pyelogram (IVP) offers functional and anatomical details but has largely been replaced by CT urography [214]. Fluoroscopy and retrograde pyelography are typically used intraoperatively or in complex cases [215,216], whereas isotope renography evaluates kidney function and obstruction [217]. Emerging modalities, such as contrast-enhanced ultrasound and endoscopic ultrasound, enhance visualization in specific scenarios [218,219]. Overall, these imaging techniques were found supportive for accurate diagnosis, effective treatment planning, and long-term management of kidney inflammation and diseases.
TREATMENT AND PREVENTIVE MEASURES
By therapeutic perspective, the choice of treatment for kidney stones were depends on their size and location. Generally, stones smaller than 5 mm can pass spontaneously through the urine with increased fluid intake (approximately 4 to 5 liters per day) [220,221]. Therapeutically it has been studied that, stones larger than 5 mm may require more invasive treatment options, including Shock Wave Lithotripsy (SWL), Ureteroscopy (URS), Percutaneous Nephrolithotomy (PCNL), or in some cases, Open or laparoscopic surgery [222].
1. Shock Wave Lithotripsy (SWL): By diagnostic and treatment point of view SWL reported as non-invasive procedure used to treat nephrotic calcification and stones in the urinary tract. The treatment involves using high energy shock waves, typically generated outside the body (extracorporeal), to break down stones into smaller fragments. These shock waves pass through the body without damaging surrounding tissues but effectively break the stone into pieces small enough to be passed through the urine [223]. SWL is generally used for stones smaller than 2 cm and located in the kidneys or upper urinary tract. SWL proved its potential effectiveness for stones that were hard but not overly dense [224]. During the procedure, the patient was placed in a bath of water or a gel-filled cushion to help transmit the shock waves to the stone [225,226].
Recent modifications, such as the development of wide focal zone shock wave generators and dual-pulse lithotripters, have enhanced operative outcomes [227]. While these advancements have made lithotripsy a successful procedure, several postoperative complications have been reported. Butt AU [228,229], have highlighted issues such as the passage of lithiasis fragments, infections, renal tissue anomalies, and hypertension following the procedure. The main advantage of SWL reported as that it is non-invasive and does not require surgical cuts, with relatively short recovery time, where most patients recover to normal activities within a few days [230]. However, SWL may not be effective for large or dense stones, and some discomfort or tissue damage can occur. In cases where the stone is not fully broken up, additional treatments may be necessary. The success rate of SWL depends on factors such as stone size, type, and location, as well as the patient’s overall health. Generally, smaller stones have a higher success rate [231].
2. Endoscopy: Endoscopy is a broad term for invasive diagnostic and operative procedures used to examine and treat the interior surfaces of organs and body cavities by inserting a tube-like instrument, often equipped with a light and camera. This technique allows doctors to visually inspect areas of the body that are otherwise difficult to reach and perform surgeries with minimal incisions, leading to faster recovery times and less trauma for the patient. Endoscopy is widely applicable in several medical fields, including Arthroscopy, which is used to examine and treat joints; Bronchoscopy, which is used for the examination of the respiratory tract; Colonoscopy, which allows doctors to examine the digestive tract, particularly the colon; Colposcopy, a procedure for inspecting the female reproductive tract, especially the cervix; and Cystoscopy, which is used to examine the urogenital tract, including the bladder and urethra [232].
3. Ureteroscopy (URS): One specific type of endoscopy is Ureteroscopy, where a thin, rigid or flexible fibre-optic instrument is inserted into the ureter to examine and treat urinary stones or other abnormalities. Therepautically, ureteroscopy were commonly used to manage blockages caused by stones in the ureter, the tube that connects the kidney to the bladder. Procedure can be used to visualize the stone directly and, depending on its size and location, can involve removing or breaking the stone into smaller pieces using laser technology. Success of the procedure depends on several factors, such as the type of stone (whether it is hard or soft), its size, and the diameter of the ureter, as a narrower ureter can make access more challenging [233]. Despite its effectiveness, ureteroscopy comes with risks. Scientist have reported some potential complications which include ureteral avulsion (the tearing or detachment of the ureter), injury to the ureteral wall, excessive bleeding, and the possibility of instrument breakage during the procedure. [234].
4. Percutaneous Nephrolithotomy (PCNL):
Percutaneous means “through the skin”, general term used to describe procedures that involve making a small incision in the skin to access internal organs or structures. One such procedure reported in Percutaneous nephrolithotomy, where endoscopic technique used to remove kidney stones that were too large to pass through the urinary tract naturally. It has been observed that during PCNL, a passage or tract created from the skin directly to the kidney, typically using a needle or a small incision. The protocol allowed the surgeon to access the kidney and removed stones that were larger than 20 mm in size, which would not be amenable to less invasive methods like shock wave lithotripsy or ureteroscopy [235].
PCNL particularly found effective for large or complex kidney stones, as it provides direct access to the stone, allowing it to be fragmented or removed entirely [236]. Postoperative complications and side effects of PCNL can vary in severity, but they are important to consider for both the patient and the healthcare provider. Clinical practitioners have reported number of common complications include extravasation, where urine or other fluids leak out from the kidney or urinary tract into surrounding tissues, potentially leading to infection or inflammation. Blood transfusions may be necessary in 21.0% to 32.1% of cases due to significant blood loss during the procedure [237]. Most of the serious risks seems to be observed for sepsis (a life-threatening infection that spreads throughout the body), colonic or pleural injury, which can occur if the procedure inadvertently damages the colon or lung [238,239]. Additionally, patients with diabetes or gross obesity may face increased risks during recovery due to factors like slower wound healing, an increased risk of infection, and complications related to anesthesia [237].
5. Open or Laparoscopic Surgery (LS): Open or laparoscopic surgery involves making an incision on the back wall of the lower abdomen to remove urinary stones. These procedures are rarely recommended and are typically considered only when less invasive treatments, such as Shock Wave Lithotripsy (SWL), Ureteroscopy (URS), or Percutaneous Nephrolithotomy (PCNL), have been unsuccessful [240,241].
6. Retrograde Intrarenal Surgery (RIRS): Retrograde Intrarenal Surgery (RIRS) reported and recommended under highly advanced endoscopic technique where a flexible ureteroscope is used to access stones within the kidney [242]. A laser is then employed to fragment the stones into small pieces for natural passage. This procedure is entirely endoscopic, avoiding any external incisions, making it ideal for patients seeking a minimally invasive option with rapid recovery [243]. Digital ureteroscopes and high-definition imaging systems have significantly improved the accuracy and success of RIRS. Additionally, robotic-assisted RIRS emerging as a promising innovation for precise stone management in complex cases [244,245].7. Robotic and Laparoscopic Surgeries: Robotic and laparoscopic surgeries were reserved for complex or refractory cases, such as large stones associated with anatomical abnormalities or those requiring reconstructive procedures [246]. Robotic platforms, like the Da Vinci system, provide superior dexterity and three dimensional visualization, enabling precise stone removal and tissue preservation. These techniques were found less invasive than traditional open surgeries, offering shorter hospital stays, reduced pain, and faster recovery [247]. Recent advancements in robotic-assisted surgery allow for improved handling of intricate cases, such as simultaneous removal of stones and correction of urinary tract abnormalities [248].
PREVENTIVE MEASURES IN UROLITHIASIS
As per available literature and preventive measures in urolithiasis were multifaceted, involving dietary, lifestyle, and medical strategies to reduce the risk of stone formation or recurrence. Adequate hydration was found to be one of the most critical components, with a recommended daily urine output of 2000 to 2500 ml dilute stone-forming substances. Generally, water or aqua therapy reported as preferred choice, though citrus based fluids like lemonade can provide additional benefits by increasing urinary citrate, a natural inhibitor of stone formation. Timed hydration throughout the day, including before bed suggested to helps prevent nocturnal urine concentration, a common contributor to stone risk [249,250].
Dietary modifications play a vital role in stone prevention. Sodium intake should be restricted to less than 2,300 mg per day to reduce urinary calcium excretion, which contributes to calcium stone formation [251]. Biochemically maintaining an adequate calcium intake from dietary sources (1,000–1,200 mg daily) found essential, as low calcium intake can paradoxically increase oxalate absorption and stone risk [252]. For calcium oxalate stones, reducing oxalate-rich foods such as spinach, rhubarb, and nuts was advisable, especially when paired with calcium rich foods to minimize oxalate absorption [253]. It has been noted that, individuals prone to uric acid stones, a low-purine diet emphasizing fruits, vegetables, and legumes helps alkalinize the urine and reduce stone formation [254,255].
Lifestyle changes further complement dietary strategies. Maintaining a healthy body weight, BMI, beverages and engaging in regular physical activity reduces metabolic abnormalities associated with stone formation [256,257]. Stress management and adequate sleep were recommended, as both factors influence hormonal balance in association with urinary pH [258].
Medical management was often tailored to the specific type of stone and underlying risk factors. Thiazide diuretics were found effective for individuals with high urinary calcium levels, while potassium citrate or bicarbonate can alkalinize urine to prevent uric acid and cystine stones [259,260]. In rare cases, medications like allopurinol for hyperuricemia or thiol drugs for cystine stones may be required. [261,262]. For struvite stones, managing recurrent urinary tract infections with antibiotics and urease inhibitors were found critical.
Long-term monitoring and individualized care are key to effective prevention. Regular follow-ups with healthcare providers, including metabolic evaluations with 24-hour urine testing, help identify ongoing risk factors and guide preventive strategies. High-risk groups, such as children, post-bariatric surgery patients, or individuals with rare metabolic disorders, require specialized approaches. Similarly, combination of lifestyle adjustments, dietary changes, and medical interventions, the recurrence of nephrolithiasis can often be significantly reduced, improving patient outcomes and quality of life.
EXPERIMENTAL MODELS UNDER UROLITHIATIC INVESTIGATION
Between 1901 to 2020, total 222 scientist were awarded with Nobel prize in the field of Physiology or Medicine. Out of 222, 186 researcher used animal model in their study [263]. In the animal bioscience experimental models especially, mammals were widely recommended and used because they share similar physiological, pathological and behavioural characters with humans [264]. Various scientifically proven vertebral experimental models including Rat ( Rattus norvegicus), Mice (Mus musculus), Guinea pig (Cavia porcellus), Hamster (Cricetus cricetus) from order Rodentia, Domestic fowl (Gallus gallus) from class Aves, Zebrafish ( Danio rerio) from family cyprinidae, frog and toad (Rana tigrina) from order Anura have been commonly accepted and used for the experimental work [265-268], reported that , Rat (Rattus norvegicus), Mice (Mus musculus), Fruit fly (Drosophila melanogaster) , Pig ( Sus scrofa domesticus) and dog ( Canis lupus familiaris) were well studied experimental models for the urolithiasis. Not only experimental animal but in vitro cellular types were also recommended as models for urolithiatic study which provides important and effective data which can be alternative source for laboratory animals [269]. Nucleation, aggregation and growth are crucial steps in the formation of renal crystal can be studied by various invitro crystallization system [270]. Crystallization systems, including, gel-based models, and advanced microfluidic platforms, replicate conditions of urine and renal flow, enabling real-time observation of crystal dynamics [271,272]. The interactive study between stone crystal and cell membrane have been studied frequently by cultured kidney epithelial cell lines in urolithiasis [273]. Cell culture models, such as MDCK, HK-2, and NRK-52E cell lines, are widely used to explore crystal adhesion, oxidative stress, inflammation, and cell injury, providing insights into crystal-cell interactions [274-277]. Artificial urine systems simulate the ionic composition of human urine to study crystallization kinetics and test inhibitors or promoters of stone formation [278].
DRUG INDUCED UROLITHIASIS
Different types and forms of lithogenic components were used to induce renal calculi in experimental models including Sodium oxalate (NaOx), Glycolic acid, Ethylene glycol and Hydroxy-L-proline [267-279] demonstrate 7.5 mg/kg and 10mg/kg body weight cold sodium oxalate in 200 ml distilled water to the human volunteers results in crystalluria. Sodium oxalate rich diet in experimental rat induces Chronic Kidney Disease (CKD) with reduction of glomerular filtration rate, increased serum urea level and proteinuria [280,281] demonstrate, administration of potassium oxalate (KOX) ( 1.5 mol/lit) in the rat leads nephrocalcinosis. Ethylene glycol (EG) (C2H6O2) termed as toxic alcohol due to its adverse effect on the body [282].
Acute toxicity of in the experimental animals is similar to the human results ataxia, convulsions, metabolic acidosis and coma whereas chronic exposure of EG leads into renal physio alterations [283]. Ethylene glycol metabolically converted into glycolate, glyoxylate, oxalate. The final product combines with calcium and forms calcium oxalate [284]. Intentional or accidental exposure (0.1 ml/kg or 95% EG solution) results severe symptomatic conditions, in addition 1-2 ml/kg (~1500 mg/kg) dose was concederd as lethal [285]. Hydroxy-L-proline (HLP), a component amino acid of collagen and precursor of metabolism of HLP potentially occurs in mitochondria of hepatocytes and proximal tubule cells forms glyoxylate [286,287], demonstrate in his study, that administration of HLP into rats and pig results renal and urinary changes and CaOx nephrolithiasis.
ARTIFICIAL INTELLIGENCE (AI) IN UROLITHIASIS
In modern science, Artificial Intelligence (AI) found to be revolutionizing facility for management of excretory related natural and artificial problem pertaining to advancement in of diagnosis, treatment, prevention, and research to upgrade the health status of mankind. One of AI’s key strengths is its ability to rapidly assess huge datasets as a timebound and critical task, that would be nearly impossible for humans to perform efficiently. This capability is particularly valuable in clinical settings where time is crucial and precision is paramount [288,289]. Under diagnostic applications, AI-powered tools were found significantly improving imaging techniques. AI upgrades quality of detection, classification and differentiation of kidney stone types through advanced imaging modalities such as CT scans, ultrasounds, and X-rays. Now a days automated diagnostic systems not only reduce processing time but also uplift the diagnostic accuracy, helping clinicians identify complications associated with stones more efficiently [290-292].
AI plays crucial role in predicting probabilities of stone recurrence with assessing patient condition. By analysing a wide range of data including metabolic profiles, genetics, and lifestyle factors machine learning models offer personalized risk assessments that enable proactive interventions [293,294]. Furthermore, AI-driven simulations were assisting in preoperative planning for minimally invasive treatments like Extracorporeal Shock Wave Lithotripsy (ESWL) and ureteroscopy. These AI models help optimize treatment outcomes, reduce complications, and guide clinical decision-making [295].
In prevention strategies, AI-powered applications and mobile health technologies are providing personalized recommendations for diet and hydration. Wearable devices that track hydration levels and urinary output, coupled with AI, can issue alerts to patients, helping them mitigate the risk of stone formation by maintaining optimal fluid balance [296,297].
Worldwide AI’s contributions extend beyond clinical practice to research, where it accelerates the analysis of kidney stone morphology. This has led to advancements in targeted drug development aimed at preventing and treating stones more effectively [298-300]. In preclinical studies, AI helps to enhance the design and analysis of animal models, improving their accuracy in replicating human kidney stone pathology and providing better insights into treatment mechanisms [301].
Apart from all above, AI-integrated telemedicine platforms are enabling remote monitoring of patients, facilitating continuous care through virtual consultations. AI based platforms are enhancing patients engagement by providing educational tools even personalized care plans as per requirement of patient [302].
Despite challenges like data privacy, biases, and integration into clinical workflows, as per information available, we found that, AI has great potential in clinical platform including urolithiasis. So it can be widely recommended for to upgrade the lifestyle of mankind in future.
CONCLUSION
Present form of study, reviews the literature on urolithiasis, exploring its historical context, causes, and management strategies. Urolithiasis is influenced by race, geographic, climatic, and lifestyle factors, with variations in the chemical composition, size, and density of urinary stones contributing to its severity. Key mechanisms such as crystal formation, nucleation, and cell interactions shed light on the pathology and progression of the condition.
The review emphasizes the need for a multidisciplinary approach to better understand and management of urolithiasis. Future research should focus on refining animal models, exploring novel treatments, and advancing diagnostic tools. Artificial intelligence presents significant opportunities for early detection, personalized treatment, and prevention, offering the potential to transform patient outcomes. Advancements in this field will improve targeted therapies and enhance quality of life for those affected by urolithiasis.
REFERENCES
- Kathale S, Pawde Y, Meher TR. Case report of an unusually large renal calculus. J Evolution Med Dental Sci. 2015; 4: 879.
- Ramarao N, Vijaya T, Nallani Venkata, Manoharan Sathish Kumar, Babu Ankem. Urolithiasis and Its Causes-Short Review. J Phytopharmacol. 2013; 2: 1-6.
- Kassaw AB, Belete M, Assefa EM, Tareke AA. Prevalence and clinical patterns of urolithiasis in sub-saharan Africa: A systematic review and meta-analysis of observational studies. BMC Nephrol. 2024; 25: 334.
- Khalili P, Jamali Z, Sadeghi T, Esmaeili-Nadimi A, Mohamadi M, Moghadam-Ahmadi A, et al. Risk factors of kidney stone disease: A cross-sectional study in the southeast of Iran. BMC Urol. 2021; 21: 141.
- Barnela SR, Soni SS, Saboo SS, Bhansali AS. Medical management of renal stone. Indian J Endocrinol Metab. 2012; 16: 236-239.
- Parmar MS. Kidney stones. BMJ. 2004; 328: 1420-1424.
- Dawson CH, Tomson CR. Kidney stone disease: Pathophysiology, investigation and medical treatment. Clin Med (Lond). 2012; 12: 467- 471.
- Moftakhar L, Jafari F, Ghoddusi Johari M, Rezaeianzadeh R, Hosseini SV, Rezaianzadeh A. Prevalence and risk factors of kidney stone disease in population aged 40-70 years old in Kharameh cohort study: A cross-sectional population-based study in southern Iran. BMC Urol. 2022; 22: 205.
- Sofia, Nalini H, Manickavasakam K, Walter Thomas M. PREVALENCE AND RISK FACTORS OF KIDNEY STONE. Global J Res Analysis. 2016; 5:183-187.
- Hailay Abrha, Kidanu Kalayou, Takele Goitom, Zereabruk Kidane, Mebrahtom Guesh, Aberhe Woldu, et al. Urinary Stone and its Associated Factors in Northern Ethiopia. 2024.
- Wang K, Ge J, Han W, Wang D, Zhao Y, Shen Y, et al. Risk factors for kidney stone disease recurrence: A comprehensive meta-analysis. BMC Urol. 2022; 22: 62.
- Zhang L, Zhang X, Pu Y, Zhang Y, Fan J. Global, Regional, and National Burden of Urolithiasis from 1990 to 2019: A systematic analysis for the global burden of disease study 2019. Clin Epidemiol. 2022; 14: 971-983.
- Teklehaimanot SZ, Bahta B, Baye NT. Prevalence of kidney stones among patients presenting to orotta medical surgical national referral hospital. Research Square: 2021.
- Saifullah M, Noor H, Waqas Iqbal M, Khalid N, Sohail M, Akmal M, Mehboob Subhani G. Prevention of renal stone recurrence: Knowledge and practice patterns among medical officers of DHQ hospital, Faisalabad. Esculapio J SIMS. 2023; 16: 9-14.
- Geraghty RM, Cook P, Roderick P, Somani B. Risk of metabolic syndrome in kidney stone formers: A comparative cohort study with a median follow-up of 19 years. J Clin Med. 2021; 10: 978.
- Sohgaura AK, Bigoniya P. A Review on Epidemiology and Etiology of Renal Stone. Am J Drug Discovery Development. 2017; 7: 54-62.
- López M, Hoppe B. History, epidemiology and regional diversities of urolithiasis. Pediatr Nephrol. 2010; 25: 49-59.
- Poulakou-Rebelakou E, Rempelakos A, Tsiamis C, Dimopoulos C. ″I will not cut, even for the stone″: Origins of urology in the Hippocratic Collection. Int Braz J Urol. 2015; 41: 26-29.
- Bloom DA, Milen MT, Heininger JC. Claudius Galen: From a 20th century genitourinary perspective. J Urol. 1999; 161: 12-19.
- Mazengenya P, Bhikha R. The genitourinary and reproductive systems: Interpretation of Avicenna’s (980-1037 AD) treatise in the Canon of Medicine. Morphologie. 2020; 104: 196-201.
- Changizi Ashtiyani S, Cyrus A. Rhazes, a genius physician in diagnosis and treatment of kidney calculi in medical history. Iran J Kidney Dis. 2010; 4: 106-110.
- Shah J, Whitfield HN. Urolithiasis through the ages. BJU Int. 2002; 89:801-810.
- Androutsos G. La lithiase urinaire et son traitement par Ambroise Paré (1509-1590) [The lithiasis of urinary tract and its treatment by Ambroise Paré (1509-1590)]. Prog Urol. 2004; 14: 109-117.
- Qian X, Wan J, Xu J, Liu C, Zhong M, Zhang J, et al. Epidemiological trends of urolithiasis at the Global, Regional, and National Levels: A population-based study. Int J Clin Pract. 2022; 2022: 6807203.
- Gillams K, Juliebø-Jones P, Juliebø SØ, Somani BK. Gender differences in Kidney Stone Disease (KSD): Findings from a Systematic Review. Curr Urol Rep. 2021; 22: 50.
- Li S, Huang X, Liu J, Yue S, Hou X, Hu L, et al. Trends in the Incidence and DALYs of Urolithiasis From 1990 to 2019: Results from the global burden of disease study 2019. Front Public Health. 2022; 10: 825541.
- Bohme K, Foster P, Hick V, Kay I. The kidney and its role in water and electrolyte balance, A. Waller, Functional Physiology. Butterworth- Heinemann Ltd. 1993; 168.
- Remuzzi G, Perico N, Macia M, Ruggenenti P. The role of renin- angiotensin-aldosterone system in the progression of chronic kidney disease. Kidney Int Suppl. 2005; 99: S57-S65.
- Obeagu Emmanuel. Erythropoietin and Kidney Diseases: A Review. 2016; 33: 760-792.
- Williams, Gray H. The Urinogenital System, P Willianms, and R Warwick (36th Edition), Gray’s Anatomy. Churchill Livingstone. 1980; 1387-1388.
- Sateesh J, Guha K, Dutta A. Regenerating re-absorption function of proximal convoluted tubule using microfluidics for kidney-on-chip applications. SN Appl Sci. 2020; 2: 39.
- Waugh A, Grant A. The Urinary System (11th Edition), Gray’s Anatomy. Churchill Livingstone Elsevier. 2010; 330-331.
- Keogh L, Kilroy D, Bhattacharjee S. The struggle to equilibrate outer and inner milieus: Renal evolution revisited. Ann Anat. 2021; 233: 151610.
- Fisang C, Anding R, Müller SC, Latz S, Laube N. Urolithiasis--an interdisciplinary diagnostic, therapeutic and secondary preventive challenge. Dtsch Arztebl Int. 2015; 112: 83-91.
- Dobre M. Evaluation of the chemical composition of renal stones. Analele Universit??ii, Dun?rea de Jos” Gala?i, Fascicula XVII - Medicin?. 2014; 111-119.
- Evan AP. Physiopathology and etiology of stone formation in the kidney and the urinary tract. Pediatr Nephrol. 2010; 25:831-841.
- Diri A, Diri B. Management of staghorn renal stones. Ren Fail. 2018; 40: 357-362.
- Gupta P, Vachharajani T, Ketkar M. LEARNING FROM IMAGES Staghorn Calculi. The Open Urology Nephrol J. 2013; 6: 6-7.
- Tefekli A, Ali Karadag M, Tepeler K, Sari E, Berberoglu Y, Baykal M, et al. Classification of percutaneous nephrolithotomy complications using the modified clavien grading system: Looking for a standard. Eur Urol. 2008; 53: 184-190.
- Moreno-Palacios J, Avilés-Ibarra OJ, García-Peña E, Torres-Anguiano JR, Serrano-Brambilia EA, López-Sámano VA, et al. Rearrangement of the Guy’s stone score improves prediction of stone-free rate after percutaneous nephrolithotomy. Turk J Urol. 2018; 44: 36-41.
- Wu WJ, Okeke Z. Current clinical scoring systems of percutaneous nephrolithotomy outcomes. Nat Rev Urol. 2017; 14: 459-469.
- Thomas K, Smith NC, Hegarty N, Glass JM. The Guy’s stone score-- grading the complexity of percutaneous nephrolithotomy procedures. Urol. 2011; 78: 277-281.
- Gunasekaran S, Natarajan R, Nayagam AS. Multidisciplinary Res. 2021; 2.
- Chhiber N, Sharma M, Kaur T, Singla Sk. Mineralization in health and mechanism of kidney stone formation. Int J Pharmaceutical Sci Invention. 2014; 3: 2319-671825.
- Aggarwal R, Srivastava A, Jain SK, Sud R, Singh RK. Renal Stones: A Clinical Review. EMJ Urol. 2017.
- Finkielstein VA, Goldfarb DS. Strategies for preventing calcium oxalate stones. CMAJ. 2006; 174: 1407-1409.
- Koirala S. Significance of analysing chemical composition of renalstones. J Pathology Nepal. 2014; 4.
- Cloutier J, Villa L, Traxer O, Daudon M. Kidney stone analysis: “Give me your stone, I will tell you who you are!”. World J Urol. 2015; 33: 157-169.
- Alelign T, Petros B. Kidney Stone Disease: An Update on Current Concepts. Adv Urol. 2018; 2018: 3068365.
- Aggarwal KP, Narula S, Kakkar M, Tandon C. Nephrolithiasis: Molecular mechanism of renal stone formation and the critical role played by modulators. BioMed Res Int. 2013; 2013: 292953.
- Kamboj P, Aggarwal M, Puri S, Singla SK. Effect of aqueous extract of Tribulus terrestris on oxalate-induced oxidative stress in rats. Indian J Nephrol. 2011; 21: 154-159.
- Keshavarzi B, Yavar Ashayeri N, Moore F, Irani D, Asadi S, ZarasvandiA, Salari M. Mineralogical composition of urinary stones and their frequency in patients: Relationship to Gender and Age. Minerals. 2016; 6: 131.
- Pachanaa K, Wattanakornsiri A, Nanuam J. Application of small caltrops (Tribulus terrestris) to inhibit calcium oxalate monohydrate crystallization. Scienceasia. 2010; 36: 165-168.
- Khan FN, Haider MF, Singh MK, Sharma P, Kumar T, Neda EN. A comprehensive review on kidney stones, its diagnosis and treatment with allopathic and ayurvedic medicines. Urology Nephrology Open Access J. 2019.
- Ivanovski O, Drüeke TB. A new era in the treatment of calcium oxalate stones? Kidney Int. 2013; 83: 998-1000.
- Chen T, Qian B, Zou J, Luo P, Zou J, Li W, et al. Oxalate as a potent promoter of kidney stone formation. Frontn Med. 2023; 10: 1159616.
- Peerapen P, Thongboonkerd V. Kidney Stone Prevention. Adv Nutr (Bethesda, Md.). 2023; 14: 555-569.
- Singh VK, Jaswal BS, Sharma J, Rai PK. Analysis of stones formed in the human gall bladder and kidney using advanced spectroscopic techniques. Biophy Rev. 2020; 12: 647-668.
- Bilbault H, Haymann J P. Experimental models of renal calcium stones in rodents. World J Nephrol. 2016; 5: 189-194.
- Peikes A. The Formation and Manifestation of Kidney Stones. The Science J Lander College of Arts and Sciences. 2020; 14: 79-85.
- Lee AJ, Yoo EH, Bae YC, Jung SB, Jeon CH. Differential identification of urine crystals with morphologic characteristics and solubility test. J Clin laboratory analysis. 2022; 36: e24707.
- Daudon Michel, Jungers Paul, Bazin Dominique. Stone Morphology: Implication for Pathogenesis. AIP Conference Proceedings. 2008; 1049: 199-215.
- Flannigan R, Choy WH, Chew B, Lange D. Renal struvite stones-- pathogenesis, microbiology, and management strategies. Nature reviews. Urol. 2014; 11: 333-341.
- Gupta S, Shamsher SK. Kidney stones: Mechanism of formation, pathogenesis and possible treatments. J Biomol Biochem. 2018; 2: 1-5.
- Katsakori P. Struvite and Triple Phosphate Renal Calculi. Med Surg Urol. 2021; 10: 257.
- Eisner BH, Deshmukh SM, Lange D. Struvite Stones. Urinary Stones. 2014; 48-56.
- Claes DJ, Jackson E. Cystinuria: Mechanisms and management. Pediatric nephrology (Berlin, Germany). 2012; 27: 2031-2038.
- D’Ambrosio V, Capolongo G, Goldfarb D, Gambaro G, Ferraro PM. Cystinuria: An update on pathophysiology, genetics, and clinical management. Pediatric nephrology (Berlin, Germany). 2022; 37: 1705-1711.
- Chillarón J, Font-Llitjós M, Fort J, Zorzano A, Goldfarb DS, Nunes V, et al. Pathophysiology and treatment of cystinuria. Nature reviews. Nephrol. 2010; 6: 424-434.
- Sadiq S, Cil O. Cystinuria: An overview of diagnosis and medical management. Turkish Arch Pediatr. 2022; 57: 377-384.
- Bawari S, Sah AN, Tewari D. Urolithiasis: An Update on Diagnostic Modalities and Treatment Protocols. Ind J Pharmaceutical Sci. 2017; 79: 164-174.
- Barbas C, García A, Saavedra L, Muros M. Urinary analysis of nephrolithiasis markers. Journal of chromatography. B, Analytical technologies in the biomedical and life sciences. 2002; 781: 433-455.
- Abou-Elela A. Epidemiology, pathophysiology, and management of uric acid urolithiasis: A narrative review. J Adv Res. 2017; 8: 513- 527.
- Maalouf NM. Uric Acid Stones. Urinary Stones. 2014; 26-35.
- Tamborino F, Cicchetti R, Mascitti M, Litterio G, Orsini A, Ferretti S, et al. Pathophysiology and Main Molecular mechanisms of urinary stone formation and recurrence. Int J Mol Sci. 2024; 25: 3075.
- Roedel MM, Nakada SY, Penniston KL. Sulfamethoxazole-induced sulfamethoxazole urolithiasis: A case report. BMC Urol. 2021; 21: 133.
- Varghese V, Rodriguez R, Self S, Velez JCQ. Atazanavir crystal-induced chronic granulomatous interstitial nephritis. Kidney Int Reports. 2020; 5: 1106-1110.
- Spritzer SD, Bravo TP, Drazkowski JF. Topiramate for treatment in patients with migraine and epilepsy. Headache. 2016; 56: 1081-1085.
- Welch BJ, Graybeal D, Moe OW, Maalouf NM, Sakhaee K. Biochemical and stone-risk profiles with topiramate treatment. Am J Kidney Diseases: The official J National Kidney Foundation. 2006; 48: 555- 563.
- Barnett SM, Jackson AH, Rosen BA, Garb JL, Braden GL. Nephrolithiasis and Nephrocalcinosis from topiramate therapy in children with epilepsy. Kidney Int Rep. 2018; 3: 684-690.
- Chase AM, Hines L, Ellis E, Jain R, Quarrier SO. Sulfamethoxazole stone in a patient with extensive history of urolithiasis and recurrent urinary tract infections. Urol Case Rep. 2021; 39: 101812.
- Morgan K, Donelan W, Andre M, Janelle J, Canales B, Bird VG. N4- acetyl-sulfamethoxazole stone in a patient on chronic trimethoprim/ sulfamethoxazole therapy: A case report and literature review. Am J Clin Exp Urol. 2024; 12: 296-300.
- Nasr SH, Milliner DS, Wooldridge TD, Sethi S. Triamterene crystalline nephropathy. American J kidney diseases: The official J National Kidney Foundation. 2014; 63: 148-152.
- Sorensen MD. Calcium intake and urinary stone disease. Translational Androl Urol. 2014; 3: 235-240.
- Assimos DG. Re: Vitamin D and Calcium Supplementation accelerates randall’s plaque formation in a murine model. J Urol. 2020; 203: 458- 459.
- Assimos DG. Re: Ascorbic acid supplements and kidney stone incidence among men: A prospective study. J Urol. 2013; 190: 1268- 1269.
- Grases F, Costa-Bauza A, Roig J, Rodriguez A. Xanthine urolithiasis: Inhibitors of xanthine crystallization. PloS one. 2018; 13: e0198881.
- Chou YH, Huang CN, Li WM, Huang SP, Wu WJ, Tsai CC, et al. Clinical study of ammonium acid urate urolithiasis. Kaohsiung J Med Sci. 2012; 28: 259-264.
- Lahyani M, Rhannam Y, Slaoui A, Touzani A, Karmouni T, Elkhader K, et al. Bilateral kidney matrix stones: A rare case. The Pan African MedJ. 2016; 25: 102.
- Bai Y, Wang Y, Kang M, Gabe CM, Srirangapatanam S, Edwards A, et al. Organic matrix derived from host-microbe interplay contributes to pathological renal biomineralization. ACS Nanoscience Au. 2023; 3: 335-346.
- Caliskan Y, Buck J, Lombardo L, Bastani B. Silica Urinary Stones: A case report and a brief review of literature. Iran J Kidney Dis. 2022; 16: 266-268.
- Flythe JE, Rueda JF, Riscoe MK, Watnick S. Silicate nephrolithiasis after ingestion of supplements containing silica dioxide. American Jkidney diseases: The official J National Kidney Foundation. 2009; 54:127-130.
- Millet-Herrera JL, Méndez-Molina R, Milke-Garcia AM, Cruz-May TN, Mendez-Dominguez N, Flores-Tapia J P. Calcium carbonate urolithiasis in a pediatric patient: A Case Report. Cureus. 2023; 15; e47873.
- Torricelli FC, De SK, Gebreselassie S, Li I, Sarkissian C, Monga M. Dyslipidemia and kidney stone risk. J Urology. 2014; 191: 667-672.
- Tan Z, Hong J, Sun A, Ding M, Shen J. Causal effects of circulating lipids and lipid-lowering drugs on the risk of urinary stones: A Mendelian randomization study. Front Endocrinol. 2023; 14: 1301163.
- Levento?lu Emre, Kuzucu Selin, Fidan Kibriya, Soylemezoglu Oguz. A rare side effect of a frequent medicine in children: Quartz Stone. Clinical Case Reports: Open Access. 2021; 4.
- Taguchi S, Nose Y, Sato T, Kobayashi T, Takaya K, Ishikawa A, HommaY. Silicate urolithiasis during long-term treatment with zonisamide. Case Rep Med. 2013; 629381.
- Dessombz A, Kirakoya B, Coulibaly G, Ouedraogo RW, Picaut L, Weil R, et al. High prevalence of opaline silica in urinary stones from burkina faso. Urology. 2015; 86: 1090-1095.
- Ghahramani N. Silica nephropathy. Int J Occup Environ Med. 2010; 1: 108-115.
- Lusco MA, Fogo AB, Najafian B, Alpers CE. AJKD Atlas of Renal Pathology: 2,8-Dihydroxyadeninuria. American J kidney diseases: The official J National Kidney Foundation. 2017; 69: e15-e16.
- Zaidan M, Palsson R, Merieau E, Cornec-Le Gall E Garstka A, Maggiore U, et al. Recurrent 2,8-dihydroxyadenine nephropathy: A rare but preventable cause of renal allograft failure. American journal of transplantation: Official journal of the American society of transplantation and the American society of transplant surgeons. 2014; 14: 2623-2632.
- Lee AJ, Yoo EH, Bae YC, Jung SB, Jeon CH. Differential identification of urine crystals with morphologic characteristics and solubility test. J Clin laboratory Analysis. 2022; 36: e24707.
- Sun XY, Xu M, Ouyang JM. Effect of crystal shape and aggregation of calcium oxalate monohydrate on cellular toxicity in renal epithelial cells. 2017.
- Simhadri PK, Rout P, Leslie SW. Urinary crystals identification andanalysis. In StatPearls. StatPearls Publishing. 2024.
- Shastri S, Patel J, Sambandam KK, Lederer ED. Kidney Stone Pathophysiology, Evaluation and Management: Core Curriculum 2023. American J Kidney diseases: The official J National kidney foundation. 2023; 82: 617-634.
- Bawari S, Sah AN, Tewari D. Urolithiasis: An Update on Diagnostic Modalities and Treatment Protocols. Ind J Pharmaceutical Sci. 2017; 79: 164-174.
- Paliouras C, Tsampikaki E, Alivanis P, Aperis G. Pathophysiology of Nephrolithiasis. Nephrology Res Rev. 2012; 4: 58-65.
- Kok DJ, Boellaard W, Ridwan Y, Levchenko VA. Timelines of the “free- particle” and “fixed-particle” models of stone-formation: Theoretical and experimental investigations. Urolithiasis. 2017; 45: 33-41.
- Khan SR. Histological aspects of the “fixed-particle” model of stoneformation: Animal studies. Urolithiasis. 2017; 45: 75-87.
- Gupta M, Bhayana D, Sikka D. Pathophysiology of calcium containing renal stones. Int J Phys Chem Mathematical Sci. 2012; 1: 1-7.
- Tsai Li-Hsien, Chang Chao-Hsiang, Chen Szju-Ju, Chen Wen-Chi. Randall’s Plaque, the Origin of Nephrolithiasis: Where Do We Stand Now? Urological Sci. 2019; 30: 200-205.
- Hsi RS, Ramaswamy K, Ho SP, Stoller ML. The origins of urinary stone disease: Upstream mineral formations initiate downstream Randall’s plaque. BJU Int. 2017; 119: 177-184.
- Wang Z, Zhang Y, Zhang J, Deng Q, Liang H. Recent advances on the mechanisms of kidney stone formation (Review). Int J Molecular Med. 2021; 48: 149.
- CARR RJ. A new theory on the formation of renal calculi. Br J Urol. 1954; 26: 105-117.
- Kapoor D, Vyas RB, Dadarwal D. Nephrolithiasis – an updated review in relation to diagnosis, prevention and treatment. Open Access J Trans Med Res. 2017; 1: 37-42.
- Sun Y, Sun H, Zhang Z, Tan F, Qu Y, Lei X, et al. New insight into oxidative stress and inflammatory responses to kidney stones: Potential therapeutic strategies with natural active ingredients. Biomed Pharmacother. 2024; 179: 117333.
- Xu Z, Yao X, Duan C, Liu H, Xu H. Metabolic changes in kidney stone disease. Front Immunol. 2023; 14: 1142207.
- Thongboonkerd V, Yasui T, Khan SR. Editorial: Immunity and Inflammatory response in kidney stone disease. Front Immunol. 2021; 12: 795559.
- Yuan D, Yang J, Wu W, Amier Y, Li X, Wan W, et al. The immune factors have complex causal regulation effects on kidney stone disease: A mendelian randomization study. BMC Immunol. 2024; 25: 34.
- Singhto N, Kanlaya R, Nilnumkhum A, Thongboonkerd V. Roles of macrophage exosomes in immune response to calcium oxalate monohydrate crystals. Front Immunol. 2018; 9: 316.
- Mehta M, Goldfarb DS, Nazzal L. The role of the microbiome in kidney stone formation. Int J Surg (London, England). 2016; 36: 607-612.
- Miller AW, Penniston KL, Fitzpatrick K, Agudelo J, Tasian G, LangeD. Mechanisms of the intestinal and urinary microbiome in kidney stone disease. Nature Rev. Urol. 2022; 19: 695-707.
- Liu M, Zhang Y, Wu J, Gao M, Zh, Z, Chen H. Causal relationship between kidney stones and gut microbiota contributes to the gut- kidney axis: A two-sample Mendelian randomization study. Front Microbiol. 2023; 14: 1204311.
- Negri AL, Spivacow FR. Kidney stone matrix proteins: Role in stone formation. World J Nephrol. 2023; 12: 21-28.
- Shah HN, Kharodawala S, Sodha HS, Khandkar AA, Hegde SS, Bansal MB. The management of renal matrix calculi: A single-centre experience over 5 years. BJU Int. 2009; 103: 810-814.
- Sohnel O, Grases F. Urinary supersaturation as a diagnostic measure in urolithiasis. World J Clin Urol. 2017; 6: 40-43.
- Kachkoul R, Benjelloun Touimi G, El Mouhri G, El Habbani R, LahrichiPathophysiological aspects of renal stone formation and stone types. Notulae Scientia Biologicae. 2023; 15: 11462.
- Phillips R, Hanchanale VS, Myatt A, Somani B, Nabi G, Biyani CS. Citrate salts for preventing and treating calcium containing kidney stones in adults. The Cochrane database of systematic reviews. 2015; CD010057.
- Wang Z, Zhang Y, Zhang J, Deng Q, Liang H. Recent advances on the mechanisms of kidney stone formation (Review). Int J Mol Med. 2021; 48: 149.
- Stoller ML, Meng MV, Abrahams HM, Kane JP. The primary stone event: A new hypothesis involving a vascular etiology. J Urology. 2004; 171: 1920-1924.
- Basavaraj DR, Biyani CS, Browning AJ, Cartledge JJ. The role of urinary kidney stone inhibitors and promoters in the pathogenesisof calcium containing renal stones. Eau-ebu Update Series. 2007; 5: 126-136.
- Balaji KC, Menon M. Mechanism of stone formation. The Urologic clinics of North America. 1997; 24: 1-11.
- Anthony RM, Davidson S, MacLeay JM, Brejda J, Werness P, Jewell DE. Comparison of two software programs used to determine the relative supersaturation of urine ions. Front Vet Sci. 2023; 10: 1146945.
- Lande MB, Varade W, Erkan E, Niederbracht Y, Schwartz GJ. Role of urinary supersaturation in the evaluation of children with urolithiasis. Pediatric Nephrol. 2005; 20: 491-494.
- Parks JH, Coward M, Coe FL. Correspondence between stone composition and urine supersaturation in nephrolithiasis. Kidney Int. 1997; 51: 894-900.
- Kachkoul R, Benjelloun Touimi G, El Mouhri G, El Habbani R, LahrichiPathophysiological aspects of renal stone formation and stone types. Notulae Scientia Biologicae. 2023; 15: 11462.
- Chaiyarit S, Thongboonkerd V. Mitochondrial Dysfunction and Kidney Stone Disease. Front Physiol. 2020; 11: 566506.
- Khan SR, Pearle MS, Robertson WG, Gambaro G, Canales BK, Doizi S, et al. Kidney stones. Nature reviews. Disease primers. 2016; 2: 16008.
- Govindaraj A, Selvam R. An oxalate-binding protein with crystal growth promoter activity from human kidney stone matrix. BJU Int. 2002; 90: 336-344.
- Schepers MSJ, van der Boom BG, Romijn JC, Schröoderand FH, Verkoelen CF. Urinary Crystallization Inhibitors Do Not Prevent Crystal Binding. J Urology. 2002; 167: 1844-1847.
- Wu L, Xue X, He C, Lai Y, Tong L. Cell death related molecules and targets in the progression of urolithiasis (Review). Int J Mol Med. 2024; 53: 52.
- Tamborino F, Cicchetti R, Mascitti M, Litterio G, Orsini A, Ferretti S, et al. Pathophysiology and Main Molecular mechanisms of urinary stone formation and recurrence. Int J Mol Sci. 2024; 25: 3075.
- Pfau A, Knauf F. Update on Nephrolithiasis: Core Curriculum 2016. Am J Kidney Diseases: The official J National Kidney Foundation. 2016; 68: 973-985.
- Ratkalkar VN, Kleinman JG. Mechanisms of stone formation. Clinical reviews in bone and mineral metabolism. 2011; 9: 187-197.
- Prakash, R., Arunachalam, Narayanasamy. Prevalence and socio- demographic status on kidney stone patients in Thanjavur district, Tamil Nadu, India. Int J Commun Med Public Health. 2019.
- Ferraro PM, Bargagli M, Trinchieri A, Gambaro G. Risk of kidney stones: Influence of Dietary Factors, Dietary Patterns, and Vegetarian-Vegan Diets. Nutrients. 2020; 12: 779.
- Nirumand MC, Hajialyani M, Rahimi R, Farzaei MH, Zingue S, Nabavi SM, Bishayee A. Dietary Plants for the Prevention and Management of Kidney Stones: Preclinical and Clinical Evidence and Molecular Mechanisms. Int J Mol Sci. 2018; 19: 765.
- Lin BB, Lin ME, Huang RH, Hong YK, Lin BL, He XJ. Dietary and lifestyle factors for primary prevention of nephrolithiasis: A systematic review and meta-analysis. BMC Nephrol. 2020; 21: 267.
- Legay C, Haeusermann T, Pasquier J, Chatelan A, Fuster DG, Dhayat N, et al. Differences in the food consumption between kidney stone formers and nonformers in the swiss kidney stone cohort. J of renal nutrition: the official J Council on Renal Nutrition of the National Kidney Foundation. 2023; 33: 555-565.
- Siener R. Nutrition and Kidney Stone Disease. Nutrients. 2021; 13: 1917.
- Xu C, Zhang C, Wang, XL, Liu TZ, Zeng XT, Li S, et al. Self-Fluid Management in Prevention of Kidney Stones: A PRISMA- Compliant Systematic Review and Dose-Response Meta-Analysis of Observational Studies. Med. 2015; 94: e1042.
- Courbebaisse M, Travers S, Bouderlique E, Michon-Colin A, Daudon M, De Mul, A, et al. Hydration for adult patients with nephrolithiasis: Specificities and Current Recommendations. Nutrients. 2023; 15: 4885.
- Jadoon S, Wang J, Mahmood Q, Li XD, Zeb BS, Naseem I, Ditta A. Association of nephrolithiasis with drinking water quality and diet in Pakistan. Environmental Eng Manag J (EEMJ). 2020; 19
- Ferraro PM, Taylor EN, Gambaro G, Curhan GC. Soda and other beverages and the risk of kidney stones. Clinical journal of the American Society of Nephrology: CJASN. 2013; 8: 1389-1395.
- Rodak K, Kokot I, Kratz EM. Caffeine as a factor influencing the functioning of the human body-friend or foe? Nutrients. 2021; 13: 3088.
- Gamage KN, Jamnadass E, Sulaiman SK, Pietropaolo A, Aboumarzouk O, Somani BK. The role of fluid intake in the prevention of kidney stone disease: A systematic review over the last two decades. Turk J Urol. 2020; 46: S92-S103.
- Hall PM. Nephrolithiasis: Treatment, causes, and prevention. Cleveland Clinic J Med. 2009; 76: 583-591.
- Carvalho M. Urinary pH in calcium oxalate stone formers: does it matter? J brasileiro de nefrologia. 2018; 40: 6-7.
- Shane E, Irani D. Chapter 26. Hypercalcemia: Pathogenesis, Clinical Manifestations, Differential Diagnosis, and Management. Primer on the metabolic bone diseases and disorders of mineral metabolism. 2006.
- Lemann J Jr, Worcester EM, Gray RW. Hypercalciuria and stones. Am J Kidney Dis. 1991; 17: 386-91.
- Ticinesi A, Nouvenne A, Maalouf NM, Borghi L, Meschi T. Salt and nephrolithiasis. Nephrology, dialysis, transplantation: Official publication of the European Dialysis and Transplant Association - European Renal Association. 2016; 31: 39-45.
- Stechman MJ, Loh NY, Thakker RV. Genetic causes of hypercalciuric nephrolithiasis. Pediatric Nephrol (Berlin, Germany). 2009; 24: 2321-2332.
- Gameel A, Kassem A, Kamel A, Abdelghaffar N. Hypercalciuria as a risk Factor For pediatric Urolithiasis in Fayoum. Fayoum University Medical J. 2023; 12: 39-49.
- Damasio PC, Amaro CR, Cunha NB, Pichutte AC, Goldberg J, Padovani CR, Amaro JL. The role of salt abuse on risk for hypercalciuria. Nutrition J. 2011; 10: 3.
- Bedford JL, Barr SI. Higher urinary sodium, a proxy for intake, is associated with increased calcium excretion and lower hip bone density in healthy young women with lower calcium intakes. Nutrients. 2011; 3: 951-961.
- Letavernier E, Daudon M. Vitamin D, Hypercalciuria and Kidney Stones. Nutrients. 2018; 10: 366.
- Chen K, Xie Y, Zhao L, M, Z. Hyperthyroidism-associated hypercalcemic crisis: A case report and review of the literature. Med. 2017; 96: e6017.
- Kohri K, Kodama M, Umekawa T, Ishikawa Y, Katayama Y, Takada M, et al. Calcium oxalate crystal formation in patients with
hyperparathyroidism and hyperthyroidism and related metabolic disturbances. Bone Mineral. 1990; 8: 59-67.
- Summers R, Macnab R. Thyroid, parathyroid hormones and calcium homeostasis. Anaesthesia Intensive Care Med. 2017; 18: 522-526.
- Khan M, Jose A, Sharma S. Physiology, Parathyroid Hormone. In StatPearls. StatPearls Publishing. 2022.
- Chen K, Xie Y, Zhao L, Mo Z. Hyperthyroidism-associated hypercalcemic crisis: A case report and review of the literature. Med. 2017; 96: e6017.
- de Arruda PF, Gatti M, de Arruda JG, Fácio FN, Jr Spessoto LC, de Arruda LF., et al. Familial mixed nephrocalcinosis as a cause of chronic kidney failure: two case reports. J Med Case Rep. 2014; 8: 355.
- Mustaqeem R, Arif A. Renal Tubular Acidosis. In StatPearls. StatPearls Publishing. 2023.
- Palmer BF, Kelepouris E, Clegg DJ. Renal tubular acidosis and management strategies: A Narrative Review. Adv Therapy. 2021; 38: 949-968.
- Buckalew VM, Jr. Nephrolithiasis in renal tubular acidosis. J Urol. 1989; 141: 731-737.
- Magni G, Unwin RJ, Moochhala SH. Renal Tubular Acidosis (RTA) and kidney stones: Diagnosis and management. Arch Esp Urol. 2021; 74: 123-128.
- Giglio S, Montini G, Trepiccione F, Gambaro G, Emma F. Distal renal tubular acidosis: A systematic approach from diagnosis to treatment. J Nephrol. 2021; 34: 2073-2083.
- Uribarri J, Oh MS, Pak CY. Renal stone risk factors in patients with type IV renal tubular acidosis. Am J kidney diseases: The official J National Kidney Foundation. 1994; 23: 784-787.
- Hawkins-van der Cingel G, Walsh SB, Eckardt KU, Knauf F. Oxalate Metabolism: From kidney stones to cardiovascular disease. Mayo Clinic Proceedings. 2024; 99: 1149-1161.
- Owino C, Mutugi A, Tang J. Hyperoxaluria - A Major Metabolic Risk for Kidney Stone Disease. R I Med J (2013). 2023; 106: 14-19.
- Demoulin N, Aydin S, Gillion V, Morelle J, Jadoul M. Pathophysiology and Management of Hyperoxaluria and Oxalate Nephropathy: A Review. Am J Kidney Diseases: The official J National Kidney Foundation. 2022; 79: 717-727.
- Bhasin B, Ürekli HM, Atta MG. Primary and secondary hyperoxaluria: Understanding the enigma. World J Nephrol. 2015; 4: 235-244.
- Sadiq S, Cil O. Cystinuria: An Overview of Diagnosis and Medical Management. Turkish Arch Pediatr. 2022; 57: 377-384.
- Leslie SW, Sajjad H, Nazzal L. Cystinuria. In StatPearls. StatPearls Publishing. 2023.
- Clark CS, Gnanappiragasam S, Thomas K, Bultitude M Cystinuria: An Overview of Challenges and Surgical Management. Front Surg. 2022; 9: 812226.
- Abrahams HM, Meng MV, Stoller ML. Urinary Stone Inhibitors. In: Stoller ML, Meng MV. (eds) Urinary Stone Disease. Current Clinical Urology. Humana Press. 2007
- Sakhaee K, Nicar M, Hill K, Pak CY. Contrasting effects of potassium citrate and sodium citrate therapies on urinary chemistries and crystallization of stone-forming salts. Kidney Int. 1983; 24: 348-352.
- Zuckerman JM, Assimos DG. Hypocitraturia: pathophysiology and medical management. Rev Urol. 2009; 11: 134-144.
- Domrongkitchaiporn S, Stitchantrakul W, Kochakarn W. Causes
of hypocitraturia in recurrent calcium stone formers: focusing on urinary potassium excretion. Am J Kidney Dis. 2006; 48: 546-554.
- Shin S, Srivastava A, Alli NA, Bandyopadhyay BC. Confounding risk factors and preventative measures driving nephrolithiasis global makeup. World J Nephrol. 2018; 7: 129-142.
- Howles SA, Thakker RV. Genetics of kidney stone disease. Nat Rev Urol. 2020; 17: 407-421.
- Singh P, Harris PC, Sas DJ, Lieske JC. The genetics of kidney stone disease and nephrocalcinosis. Nature reviews. Nephrol. 2022; 18: 224-240.
- Sorokin I, Mamoulakis C, Miyazawa K, Rodgers A, Talati J, Lotan Y. Epidemiology of stone disease across the world. World J Urol. 2017; 35: 1301-1320.
- Rule AD, Roger VL, Melton LJ 3rd, Bergstralh EJ, Li X, Peyser PA, et al. Kidney stones associate with increased risk for myocardial infarction. J Am Society Nephrol. 2010; 21: 1641-1644.
- Alblowi S, Safdar O, Aboulola N, Alharazy D, Najem N. Renal stone prevalence and risk factors in Jeddah and Riyadh. J Family Med Primary Care. 2022; 11: 2839-2845.
- Yongzhi L, Shi Y, Jia L, Yili L, Xingwang Z, Xue G. Risk factors for urinary tract infection in patients with urolithiasis-primary report of a single center cohort. BMC Urology. 2018; 18: 45.
- Rule AD, Bergstralh EJ, Melton LJ 3rd, Li X, Weaver AL, Lieske JC. Kidney stones and the risk for chronic kidney disease. Clin J Am Society Nephrol. 2009; 4: 804-811.
- Gkentzis A, Kimuli M, Cartledge J, Traxer O, Biyani CS. Urolithiasis in inflammatory bowel disease and bariatric surgery. World J Nephrol., 2016; 5: 538-546.
- Rizzato G, Colombo P. Nephrolithiasis as a presenting feature of chronic sarcoidosis: A prospective study. Sarcoidosis Vasc Diffuse Lung Dis. 1996; 13: 167-172.
- McPhail EF, Gettman MT, Patterson DE, Rangel LJ, Krambeck AE. Nephrolithiasis in medullary sponge kidney: evaluation of clinical and metabolic features. Urol. 2012; 79: 277-281.
- Ciacci C, Spagnuolo G, Tortora R, Bucci C, Franzese D, Zingone F, et al. Urinary stone disease in adults with celiac disease: prevalence, incidence and urinary determinants. J Urol. 2008; 180: 974-979.
- Rasool M, Mousa T, Alhamadani H, Ismael A. Therapeutic potential of medicinal plants for the management of renal stones: A review, Baghdad J Biochem Appl Biol Sci. 2022; 3: 69-98.
- Mishra NK. Renal calculi (kidney stones) and it’s therapy. Indo Global J Pharm Sci. 2019; 9: 1-4.
- Eyre KS, Lewis F, Cui H, Grout E, Mihai R, Turney BW, Howles SA. Utility of blood tests in screening for metabolic disorders in kidney stone disease. BJU Int. 2021; 127: 538-543.
- Leslie SW, Sajjad H, Bashir K. 24-Hour Urine Testing for Nephrolithiasis: Interpretation and Treatment Guidelines. In StatPearls. StatPearls Publishing. 2024.
- Dunmire B, Lee FC, Hsi RS, Cunitz BW, Paun M, Bailey MR, et al. Tools to improve the accuracy of kidney stone sizing with ultrasound. J Edourology. 2015; 29: 147-152.
- McCarthy CJ, Baliyan V, Kordbacheh H, Sajjad Z, Sahani D, Kambadakone A. Radiology of renal stone disease. Int J Surg. 2016; 36: 638-646.
- Tzou DT, Usawachintachit M, Taguchi K, Chi T. Ultrasound use in urinary stones: Adapting Old Technology for a Modern-Day Disease. J Endourology. 2017; 31: S89-S94.
- Al-Shawi MM, Aljama NA, Aljedani R, Alsaleh MH, Atyia N, Alsedrah A, Albardi M. The role of radiological imaging in the diagnosis and treatment of urolithiasis: A Narrative Review. Cureus. 2022; 14: e33041.
- Johnston R, Lin A, Du J, Mark S. Comparison of kidney-ureter-bladder abdominal radiography and computed tomography scout films for identifying renal calculi. BJU Int. 2009; 104: 670-673.
- Bhojani N, Paonessa JE, El Tayeb MM, Williams JC, Jr. Hameed TA, Lingeman JE. Sensitivity of Noncontrast Computed Tomography for Small Renal Calculi with Endoscopy as the Gold Standard. Urol. 2018; 117: 36-40.
- Euler A, Wullschleger S, Sartoretti T, Müller D, Keller EX, Lavrek, D, et al. Dual-energy CT kidney stone characterization-can diagnostic accuracy be achieved at low radiation dose? Euro Radiol. 2023; 33: 6238-6244.
- ?wi?to? D, Grzywi?ska M, Czarniak P, Go??biewski A, Durawa A, Teodorczyk J, et al. The Emerging role of MR Urography in imaging megaureters in children. Front Pediatrics. 2022; 10: 839128.
- Mehta SR, Annamaraju, P. Intravenous Pyelogram. In StatPearls. StatPearls Publishing. 2023.
- Yoldas M, Kuvvet Yoldas T. Fluoroscopy Is Essential in Retrograde Intrarenal Surgery. Int J Clinical practice. 2023; 8896681.
- Tabibi A, Akhavizadegan H, Nouri-Mahdavi K, Najafi-Semnani M, Karbakhsh M, Niroomand AR. Percutaneous nephrolithotomy with and without retrograde pyelography: A randomized clinical trial. Int Braz J Urol. 2007; 33: 19-24.
- Taylor AT. Radionuclides in nephrourology, part 1: Radiopharmaceuticals, quality control, and quantitative indices. J Nuclear Med. 2014; 55: 608-615.
- Wang L, Li KP, Yin S, Yang L, Zhu PY. Contrast-enhanced ultrasound versus conventional ultrasound-guided percutaneous nephrolithotomy in patients with a non-dilated collecting system: Results of a pooled analysis of randomized controlled trials. BMC Urology. 2023; 23: 93.
- Vicentini FC, El Hayek KKR, Szwarc M, Perrella R. Kuriki P, Cohen D, et al. Ultrasound guided endoscopic combined Intrarenal surgery - 10 steps for the success. Int Braz J Urol. 2022; 48: 874-875.
- Khan FN, Haider MF, Singh MK, Sharma P, Kumar T, Neda EN. A comprehensive review on kidney stones, its diagnosis and treatment with allopathic and ayurvedic medicines. Urology Nephrology Open Access J. 2019.
- Amer T, Osman B, Johnstone A, Mariappan M, Gupta A, Brattis N, et al. Medical expulsive therapy for ureteric stones: Analysing the evidence from systematic reviews and meta-analysis of powered double-blinded randomised controlled trials. Arab J Urology. 2017; 15: 83-93.
- Ingimarsson JP, Krambeck AE, Pais VM., Jr. Diagnosis and Management of Nephrolithiasis. The Surgical clinics of North America. 2016; 96: 517-532.
- McAteer JA, Evan AP. The acute and long-term adverse effects of shock wave lithotripsy. Semin Nephrol. 2008; 28: 200-213.
- Reynolds LF, Kroczak T, Pace KT. Indications and contraindications for shock wave lithotripsy and how to improve outcomes. Asian J Urol. 2018; 5: 256-263.
- Lautz J, Sankin G, Zhong P. Turbulent water coupling in shock wave lithotripsy. Physics Med Biol. 2013; 58: 735-748.
- Jain A, Shah TK. Effect of air bubbles in the coupling medium on efficacy of extracorporeal shock wave lithotripsy. Eur Urol. 2007; 51: 1680-1687.
- Elmansy HE, Lingeman JE. Recent advances in lithotripsy technology and treatment strategies: A systematic review update. Int J Surgery. 2016; 36: 676-680.
- D’Addessi A, Vittori M, Racioppi M, Pinto F, Sacco, E, Bassi P. Complications of extracorporeal shock wave lithotripsy for urinary stones: to know and to manage them-a review. Scientific World J. 2012; 619820.
- Butt AU, Khurram M, Ahmed A, Hasan Z, Rehman A, Farooqi MA. Extracorporeal shock wave lithotripsy. J Coll Physicians Surg Pak. 2005; 15: 638-641.
- McAteer JA, Evan AP. The acute and long-term adverse effects of shock wave lithotripsy. Sem Nephrol. 2008; 28: 200-213.
- Bahilo-Mateu P, Budia-Alba A. Extracorporeal shockwave lithotripsy in the management of urinary stones: New concepts and techniques to improve outcomes. Asian J Urol. 2024; 11: 143-148.
- Ponraj, Perumal. Endoscopy. Nature Proceedings. 2012.
- Paul E, Sasikumar P, Gomathi S, Abhishek A, Selvam GS. Recombinant lactic acid bacteria secreting oxdc as a novel therapeutic tool for the prevention of kidney stone disease. Multifunctional systems for combined delivery. Biosensing and Diagnostics. 2017; 327-345.
- De Coninck V, Keller EX, Somani B, Giusti G, Proietti S, Rodriguez- Socarras, M, et al. Complications of ureteroscopy: A complete overview. World J Urol. 2020; 38: 2147-2166.
- Ganpule AP, Vijayakumar M, Malpani A, Desai MR. Percutaneous Nephrolithotomy (PCNL) a critical review. Int J Surg. 2016; 36: 660- 664.
- Malik I, Wadhwa R. Percutaneous Nephrolithotomy: Current Clinical Opinions and Anesthesiologists Perspective. Anesthesiol Res Practice. 2016; 2016: 9036872.
- Michel MS, Trojan L, Rassweiler JJ. Complications in percutaneous nephrolithotomy. Eur Urol. 2007; 51: 899-906.
- Rashid AO, Fakhulddin SS. Risk factors for fever and sepsis after percutaneous nephrolithotomy. Asian J Urol. 2016; 3: 82-87.
- Boutros CS, Loftus AW, Bassiri A, Davis LE, Vince R, Sinopoli J, et al. A rare cause of colon perforation after percutaneous nephrolithotomy-a case report and review of the literature. Case Rep Surg. 2024; 2024: 4475216.
- Türk C, Pet?ík A, Sarica K, Seitz C, Skolarikos A, Straub M, Knoll T. EAU Guidelines on Interventional Treatment for Urolithiasis. Eur Urol. 2016; 69: 475-482.
- Kijvikai K. The role of laparoscopic surgery for renal calculi management. Therap Adv Urol. 2011; 3: 13-18.
- Van Cleynenbreugel B, K?l?ç Ö, Akand M. Retrograde intrarenalsurgery for renal stones - Part 1. Turk J Urol. 2017; 43: 112-121.
- K?l?ç Ö, Akand M, Van Cleynenbreugel B. Retrograde intrarenalsurgery for renal stones - Part 2. Turk J Urol. 2017; 43: 252-260.
- Aghamir SMK. Successful Retrograde Intrarenal Surgery (RIRS) for a 2-centimeter stone in a Chronic Renal Failure (CRF) patient. Int J Surg Case Rep. 2021; 87: 106375.
- Lee JY, Jeon SH. Robotic flexible ureteroscopy: A new challenge inendourology. Investigative and Clin Urol. 2022; 63: 483-485.
- Ganpule AP, Prashant J, Desai MR. Laparoscopic and robot-assisted surgery in the management of urinary lithiasis. Arab J Urol. 2012; 10: 32-39.
- Reddy K, Gharde P, Tayade H, Patil M, Reddy LS, Surya D. Advancements in Robotic Surgery: A comprehensive overview
of current utilizations and upcoming frontiers. Cureus. 2023; 15: e50415.
- Siddiqui KM, Albala DM. Robotic-assisted surgery and treatment of urolithiasis. Int J Surg. 2016; 36: 673-675.
- Courbebaisse M, Travers S, Bouderlique E, Michon-Colin A, Daudon M, De Mul A, et al. Hydration for Adult Patients with Nephrolithiasis: Specificities and Current Recommendations. Nutrients. 2023; 15: 4885.
- Penniston KL, Steele TH, Nakada SY. Lemonade therapy increases urinary citrate and urine volumes in patients with recurrent calcium oxalate stone formation. Urol. 2007; 70: 856-860.
- Siener R. Nutrition and Kidney Stone Disease. Nutrients. 2021; 13: 1917.
- Sorensen MD. Calcium intake and urinary stone disease. Translational Androl Urol. 2014; 3: 235-240.
- Mitchell T, Kumar P, Reddy T, Wood KD, Knight J, Assimos DG, et al. Dietary oxalate and kidney stone formation. American journal of physiology. Renal Physiol. 2019; 316: F409-F413.
- Gul Z, Monga M. Medical and dietary therapy for kidney stone prevention. Korean J Urology. 2014; 55: 775-779.
- Grygiel B, Puszczewicz M. Diet in hyperuricemia and gout: Myths and facts. Reumatologia. 2014; 52: 269-279.
- Lin BB, Lin ME, Huang RH, Hong YK, Lin BL, He XJ. Dietary and lifestyle factors for primary prevention of nephrolithiasis: A systematic review and meta-analysis. BMC Nephrol. 2020; 21: 267.
- Mao W, Zhang L, Sun S, Wu J, Zou X, Zhang G, Chen, M. Physical activity reduces the effect of high body mass index on kidney stones in diabetes participants from the 2007-2018 NHANES Cycles: A cross-sectional study. Front Public Health. 2022; 10: 936552.
- Hirotsu C, Tufik S, Andersen, ML. Interactions between sleep, stress, and metabolism: From physiological to pathological conditions. Sleep science (Sao Paulo, Brazil). 2015; 8: 143-152.
- Cheng L, Zhang K, Zhang Z. Effectiveness of thiazides on serum and urinary calcium levels and bone mineral density in patients with osteoporosis: a systematic review and meta-analysis. Drug design, development and therapy. 2018; 12: 3929-3935.
- Trinchieri A, Esposito N, Castelnuovo C. Dissolution of radiolucent renal stones by oral alkalinization with potassium citrate/potassium bicarbonate. Arch Ital Urol Androl. 2009; 81: 188-191.
- Du L, Zong Y, Li H, Wang Q, Xie L, Yang B, et al. Hyperuricemia and its related diseases: mechanisms and advances in therapy. Signal transduction and targeted therapy. 2024; 9: 212.
- Bhatt NP, Deshpande AV, Starkey MR. Pharmacological interventions for the management of cystinuria: A systematic review. J Nephrol. 2024; 37: 293-308.
- Jota Baptista CV, Faustino-Rocha AI, Oliveira PA. Animal Models in Pharmacology: A brief history awarding the nobel prizes for physiology or medicine. Pharmacol. 2021; 106: 356-368.
- Mukherjee P, Roy S, Ghosh D, Nandi SK. Role of animal models in biomedical research: A review. Laboratory animal Res. 2022; 38: 18.
- Hickman DL, Johnson J, Vemulapalli TH, Crisler JR, Shepherd R. Commonly used animal models. Principles of animal research for graduate and undergraduate students. 2017; 117-175.
- Chen SJ, Chiu KY, Chen HY, Lin WY, Chen YH, Chen WC. Animal models for studying stone disease. Diagnostics (Basel, Switzerland). 2020; 10: 490.
- T zou DT, Taguchi K, Chi T, Stoller ML. Animal models of urinary stone disease. Int J Surg. 2016; 36: 596-606.
- Hosszu A, Kaucsar T, Seeliger E, Fekete A. Animal models of renal pathophysiology and disease. In: Pohlmann, A., Niendorf, T. (eds) Preclinical MRI of the Kidney. Methods in Molecular Biology, vol 2216. Humana, New York, NY. 2021.
- Grases F, Prieto RM, Costa-Bauzá A. In vitro models for studying renal stone formation: A clear alternative. Altern Lab Anim. 1998; 26: 481-503.
- Achilles W. In vitro crystallisation systems for the study of urinary stone formation. World J Urol. 1997; 15: 244-251.
- Achilles W, Freitag R, Kiss B, Riedmiller H. Quantification of crystal growth of calcium oxalate in gel and its modification by urinary constituents in a new flow model of crystallization. J Urol. 1995; 154: 1552-1556.
- Ibis F, Yu TW, Penha FM, Ganguly D, Nuhu MA, van der Heijden AEDM, et al. Nucleation kinetics of calcium oxalate monohydrate as a function of pH, magnesium, and osteopontin concentration quantified with droplet microfluidics. Biomicrofluidics. 2021; 15: 064103.
- Bigelow MW, Wiessner JH, Kleinman JG, Mandel NS. Calcium oxalate crystal attachment to cultured kidney epithelial cell lines. J Urol. 1998; 160: 1528-1532.
- Khan A, Wang W, Khan SR. Calcium oxalate nephrolithiasis and expression of matrix GLA protein in the kidneys. World J Urol. 2014; 32: 123-130.
- Huang MY, Chaturvedi LS, Koul S, Koul HK. Oxalate stimulates IL-6 production in HK-2 cells, a line of human renal proximal tubular epithelial cells. Kidney Int. 2005; 68: 497-503.
- Wang Q, Hu H, Dirie NI, Lu Y, Zhang J, Cui, L., et al. High concentration of calcium promotes mineralization in NRK-52E Cells Via Inhibiting the Expression of Matrix Gla Protein. Urology. 2018; 119: 161.
- Petrovic A, Kizivat T, Curci? I, Smoli? R, Smolic M. In vitro cell culture models of hyperoxaluric states: Calcium oxalate and renal epithelial cell interactions. Crystals. 2021; 11: 735.
- Sarigul N, Korkmaz F, Kurultak ?. A new artificial urine protocol to better imitate human urine. Scientific Rep. 2019; 9: 20159.
- Burns JR, Finlayson B, Gauthier J. Calcium oxalate retention in subjects with crystalluria. Urologia Int. 1984; 39: 36-39.
- Crestani T, Crajoinas RO, Jensen L, Dima LL, Burdeyron P, Hauet T, et al. A sodium oxalate-rich diet induces chronic kidney disease and cardiac dysfunction in rats. Int J Mol Sci. 2021; 22: 9244.
- Khan SR, Finlayson B, Hackett RL. Experimental induction of crystalluria in rats using mini-osmotic pumps. Urological Res. 1983; 11: 199-205.
- Iqbal A, Glagola JJ, Nappe TM. Ethylene Glycol Toxicity. In StatPearls. StatPearls Publishing. 2022.
- Price CJ, Kimmel CA, Tyl RW, Marr MC. The developmental toxicity of ethylene glycol in rats and mice. Toxicol Appl Pharmacol. 1985; 81: 113-127.
- Booth ED, Dofferhoff O, Boogaard PJ, Watson WP. Comparison of the metabolism of ethylene glycol and glycolic acid in vitro by precision-cut tissue slices from female rat, rabbit and human liver. Xenobiotica; The fate of foreign compounds in biological systems. 2004; 34: 31-48.
- Ahmad Y, Kissling S, Torrent C, Chiche JD, Liaudet L, Ltaief Z. The three biological gaps and hyperoxaluria in ethylene glycol poisoning: Case presentation and review. Eur Rev Med Pharmacol Sci. 2021; 25:6295-6299.
- Knight J, Jiang J, Assimos DG, Holmes RP. Hydroxyproline ingestion and urinary oxalate and glycolate excretion. Kidney Int. 2006; 70: 1929-1934.
- Khan SR, Glenton PA, Byer KJ. Modeling of hyperoxaluric calcium oxalate nephrolithiasis: Experimental induction of hyperoxaluria by hydroxy-L-proline. Kidney Int. 2006; 70: 914-923.
- Yang B, Veneziano D, Somani BK. Artificial intelligence in the diagnosis, treatment and prevention of urinary stones. Current Opin Urol. 2020; 30: 782-787.
- Yuan Q, Zhang H, Deng T, Tang S, Yuan X, Tang W, et al. Role of artificial intelligence in kidney disease. Int J Med Sci. 2020; 17: 970- 984.
- Maleki Varnosfaderani, S., & Forouzanfar, M. The Role of AI in Hospitals and Clinics: Transforming Healthcare in the 21st Century. Bioengineering (Basel, Switzerland). 2024; 11: 337.
- Shabaniyan T, Parsaei H, Aminsharifi A, Movahedi MM, Jahromi AT, Pouyesh S, et al. An artificial intelligence-based clinical decision support system for large kidney stone treatment. Australasian Phy Engineering Sci Med. 2019; 42: 771–779.
- Ahmed F, Abbas S, Athar A, Shahzad T, Khan WA, Alharbi M, et al. Identification of kidney stones in KUB X-ray images using VGG16 empowered with explainable artificial intelligence. Scientific Rep. 2024; 14: 6173.
- Mahmoodi F, Andishgar A, Mahmoudi E, Monsef A, Bazmi S, TabriziR. Predicting symptomatic kidney stones using machine learning algorithms: Insights from the Fasa Adults Cohort Study (FACS). BMC Res. 2024; 17: 318.
- Sánchez C, Larenas F, Arroyave JS, Connors C, Giménez B, Palese MA, et al. Artificial Intelligence in Urology: Application of a machine learning model to predict the risk of urolithiasis in a general population. J Endourol. 2024; 38: 712-718.
- Anastasiadis A, Koudonas A, Langas G, Tsiakaras S, Memmos D, Mykoniatis I. et al. Transforming urinary stone disease management by artificial intelligence-based methods: A comprehensive review. Asian J Urol. 2023; 10: 258-274.
- Streeper NM, Lehman K, Conroy DE. Acceptability of Mobile Health Technology for Promoting Fluid Consumption in Patients with Nephrolithiasis. Urology. 2018; 122: 64-69.
- Scales CD, Jr Desai AC, Harper JD, Lai HH, Maalouf NM, Reese PP, et al. Prevention of Urinary Stones With Hydration (PUSH): Design and Rationale of a Clinical Trial. Am J Kidney Dis: The official J National Kidney Foundation. 2021; 77: 898-906.
- Tang L, Li W, Zeng X, Wang R, Yang X, Luo G, et al. Value of artificial intelligence model based on unenhanced computed tomography of urinary tract for preoperative prediction of calcium oxalate monohydrate stones in vivo. Ann Transl Med. 2021; 9: 1129.
- Panthier F, Melchionna A, Crawford-Smith H, Phillipou Y, Choong S, Arumuham V, et al. Can artificial intelligence accurately detect urinary stones? A Systematic Review. J Endourol. 2024; 38: 725-740.
- Dhudum R, Ganeshpurkar A, Pawar A. Revolutionizing Drug Discovery: A comprehensive review of AI Applications. Drugs and Drug Candidates. 2024; 3: 148-171.
- Ghosh A, Choudhary G, Medhi B. The pivotal role of artificial intelligence in enhancing experimental animal model research: A machine learning perspective. Indian J Pharmacol. 2024; 56: 1-3.
- Tsvetanov F. Integrating AI Technologies into remote monitoring patient systems. Engineering Proceedings. 2024; 70: 54.