Scrotal Cancer Mimicking a Scrotal Abscess: Case Report and Review of the Literature
- 1. Urology B department, University Hospital Center IBN SINA, University Mohammed V, Morocco.
- 2. Department of Pathology and Cytology, University Hospital Center IBN SINA, University Mohammed V, Morocco.
Keywords
• Scrotal Squamous Cell
• Abscess
• Surgical Excision
ABBREVIATIONS
SCC: Squamous Cell Carcinoma; PUVA: Ultraviolet a Photochemotherapy; HPV: Human Papilloma Virus; PCR: Polymerase Chain Reaction
INTRODUCTION
Scrotal squamous cell carcinoma is an uncommon form of skin cancer, historically recognized as the “chimney sweepers’ disease” first identified by Percivall Pott in the 18th century. Its incidence has significantly decreased in recent decades, largely due to enhanced occupational health standards, better hygiene practices, and stricter control of exposure to carcinogenic substances. Nonetheless, sporadic cases still arise, often in connection with chronic inflammation, HIV infection, prolonged exposure to industrial agents such as mineral oils, soot, or tar, and inadequate personal hygiene.
It typically presents in patients in their 60s, often developing from a pre-existing lesion that gradually increases in size. In approximately 25% of cases, it is associated with inguinal lymphadenopathy. Surgical excision remains the cornerstone of treatment and may be combined with lymph node dissection depending on the disease stage. The aim of our study is to describe the epidemiological, diagnostic, and therapeutic features of this malignancy.
Citation
Amine EM, Anass R, Amine ZM, Yassine AT, Zakia B, et al. (2025) Scrotal Cancer Mimicking a Scrotal Abscess: Case Report and Review of the Literature. J Urol Res 12(2): 1165.
CASE PRESENTATION
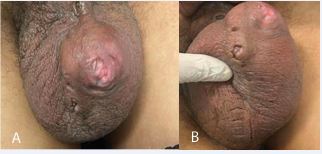
A.Z, a 55-year-old male, employed in the ceramics industry, presented by himself to our clinic, with a solid, hemorrhagic abscessed mass measuring 3 cm in the mid scrotal region, evolving over the past year. The patient also reported associated pruritus (Figure 1 A,B).
Figure 1 A: Clinical aspect of the lesion; B: Inferior view the mass.
The examination didn’t find any lymph node.
Laboratory investigations revealed a mild inflammatory syndrome with White blood cells: 12,000/mm³, C-reactive protein (CRP): 48 mg/L, HIV serology: Negative Scrotal ultrasonography demonstrated a well-defined, oval, heterogeneous echogenic mass with a thick peripheral capsule, richly vascularized, measuring approximately 31 x 29 mm. The mass exhibited cutaneous fistulization and was consistent with an abscessed collection. Associated findings included fat stranding and thickening of the scrotal tunics.
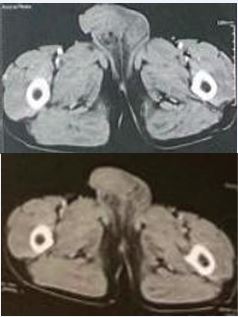
Given the atypical macroscopic appearance and the lesion’s slow progression, a thoracoabdominopelvic CT scan was performed. The scan showed diffuse scrotal edema centered on a 20 mm anterior inter-testicular abscess and revealed a micronodular and bronchial pattern in the lungs, suggestive of granulomatous disease, most likely tuberculosis. No secondary lymph node or visceral involvement was observed (Figure 2).
Figure 2 CT-Scan showing the scrotal mass.
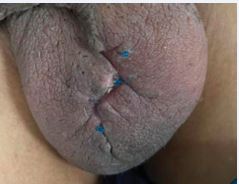
In light of these findings, PCR-based tuberculosis testing was conducted and confirmed the diagnosis of pulmonary tuberculosis. Antitubercular therapy was initiated accordingly. The patient underwent wide local surgical excision of the scrotal lesion without reconstruction, given the localized nature of the mass. The postoperative course was uneventful (Figure 3).
Figure 3 Post-operative aspect of the scrotum (D5).
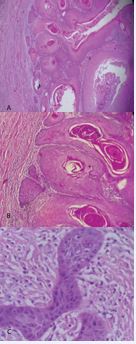
Histopathological analysis confirmed a diagnosis of verrucous squamous cell carcinoma, measuring 2.5 cm in greatest dimension and infiltrating the reticular dermis, with a maximal tumor thickness estimated at 0.6 cm. The surgical margin was clean. The tumor was classified as pT2 according to the AJCC 8th edition staging system (Figure 4 A,B,C).
The patient didn’t receive any adjuvant treatment. At a 1-year postoperative follow-up, the patient was clinically well. A follow-up CT scan showed no evidence of recurrence or metastasis.
Figure 4 A: HE staining: low magnification; B: HE staining: medium magnification; C: HE staining: high magnification.
The patient didn’t receive any adjuvant treatment. At a 1-year postoperative follow-up, the patient was clinically well. A follow-up CT scan showed no evidence of recurrence or metastasis.
DISCUSSION
Scrotal cancer is an exceptionally rare form of malignancy, with a reported incidence of approximately 1.5 new cases per million people each year in Western nations [1]. In a retrospective study of 471 cases spanning from 1973 to 2002, Wright et al. identified the most frequent histological subtypes as squamous cell carcinoma (32%), extramammary Paget’s disease (21%), basal cell carcinoma (18%), and sarcomas (18%). Notably, squamous cell carcinoma appeared significantly more common in Black patients compared to White patients (69% vs. 31%) [2]. The first cancer to be directly associated with occupational exposure was scrotal SCC. Over the past 20 years, occupational risk factors have not been linked to an elevated incidence of scrotal SCC due to significant improvements in working environments and applicable laws.
Among the known risk factors for SCC are: [3].
- Jobs like tar and paraffin workers and chimney sweepers. Additionally, jobs involving exposure to cutting and mineral oils, printing, and metalworking, including those in the automotive and aircraft industries, auto repair shops, commercial printers, aluminum and shale oil production, pitching, engineering, steel manufacturing, and cavalry.
- Exposure to metals that cause cancer, such as nickel, chromium, and arsenic.
- Persistent mechanical discomfort.
- Persistent inflammatory conditions (such as surgical scars and chronic lymphoedema). lifestyle choices (such as smoking and inadequate personal hygiene).viruses, such as HPV.
- Ionizing radiation exposure.
- Exposure to iatrogenic substances, such as Fowler’s solution, tar, coal, PUVA, radiation, and nitrogen mustard.
- Immunosuppression (such as post-transplant immunosuppression and acquired and inherited immunodeficiency).
To ascertain the histology of the scrotal cancer, an excisional biopsy is necessary for the lesions. A thorough physical examination and cross-sectional imaging techniques, like CT scanning or MRI, can be used to evaluate non-localized disease and metastases; however, MRI is superior for assessing soft tissue lesions. If there is probable involvement of the pelvic lymph nodes or elsewhere, PET scanning should be taken into consideration [4,5].
The penis or testes may be invaded by advanced illness. Histologic assessment confirms this diagnosis, and sampling should be done in multiple sites to ascertain the depth of invasion and the boundaries of extension. The lymphatic drainage pattern of the scrotum is identical to that of the penis. From the inguinal lymph nodes to the pelvic lymph nodes, tumors typically spread in a progressive manner [4].
It’s interesting to note that the scrotal lymphatics don’t seem to leak into the ipsilateral superficial inguinal lymph nodes after crossing the median raphe. As a result, tumors that do not affect the median raphe hardly ever spread to the other inguinal location.23 The clinical workup for scrotal SCC is fairly similar to that of penile carcinoma due to similarities in site and histology. Chest radiographs and pelvic and/or abdominal CT scans are examples of routine imaging tests. When necessary, additional tests such bone scans and chest CT or PET- CT may be performed [4,5].
In cases of invasive scrotal SCC, lymph node metastases cannot be accurately determined by physical examination or cross-sectional imaging methods [6]. The simplest method of confirming metastasis is fine needle aspiration cytology. However, the test is only useful if the results are positive. An inflammatory response may cause the inguinal lymph nodes to expand. An excision biopsy is recommended if a lymph node is still palpable following antibiotic treatment and the fine-needle aspiration cytology is negative [7]. Which wasn’t the case of our patient who didn’t had any lymph node.
Surgery is the main therapeutic option for scrotal cancer. In order to treat all types of histology, the cancer must be surgically removed. Chemotherapy and radiation therapy are examples of adjuvant treatments [8].
The patient’s age, the size, grade, and stage of the tumor, as well as the extent of surgery, all affect the prognosis for scrotal SCC. According to reports, one of the most significant predictors of local and/or regional tumor control is the state of the surgical margin.
Additionally, it has been noted that even with a 2 cm margin for wide excision, the surgical margin remains favorable [9].
Every patient should have a history and physical examination, including a skin examination, every three months for two years, then every six months for three years, and finally once a year for the rest of their lives [10]. During consultations, it is necessary to discuss patient education regarding sun protection and skin self examination for regional disease recurrence.
CONCLUSION
The epidemiology of scrotal SCC has evolved recently, despite the fact that it was once thought to be an occupational disease. These days, the chance of getting this malignancy is greatly increased by iatrogenic disorders such PUVA, immunosuppression, and HPV infection. The cornerstone of the scrotal SCC treatment plan is surgery. The original lesion should be removed, and regional lymph nodes should be staged and treated using a risk- stratified strategy.
DECLARATION
Ethical Approval
Ethics approval has been obtained to proceed with the current study Ethical approval for this study (Ethical Committee N009-24) was provided by the Ethical Committee Ibn University Hospitals, Rabat Morocco on 22 January 2024.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of the journal.
Scare Guidelines
The work has been reported in line with the SCARE criteria [11].
REFERENCES
- Verhoeven RH, Louwman WJ, Koldewijn EL, Demeyere TB, Coebergh JW. Scrotal cancer: incidence, survival and second primary tumours in the Netherlands since 1989. Br J Cancer. 2010; 103: 1462-1466.
- Wright JL, Morgan TM, Lin DW. Primary scrotal cancer: disease characteristics and increasing incidence. Urology. 2008; 72: 1139-1143.
- Debashis Sarkar. A Systematic Review of Scrotal Squamous Cell Carcinoma. EMJ Urol. 2019; 7: 68-74.
- Chung BI et al., “Surgical, radiology, and endoscopic anatomy of the male pelvis,” Wein AJ et al. (eds.), Campbell-Walsh Urology 10th edition, Philadelphia: Elsevier. 2015; 3: 1630.
- Graafland NM, Leijte JA, Valdés Olmos RA, Hoefnagel CA, Teertstra HJ, Horenblas S. Scanning with 18F-FDG-PET/CT for detection of pelvic nodal involvement in inguinal node-positive penile carcinoma. Eur Urol. 2009; 56: 339-345.
- Hughes B, Leijte J, Shabbir M, Watkin N, Horenblas S. Non-invasive and minimally invasive staging of regional lymph nodes in penile cancer. World J Urol. 2009; 27: 197-203.
- Zhu Y, Zhang HL, Yao XD, Zhang SL, Dai B, Shen YJ, et al. Development and evaluation of a nomogram to predict inguinal lymph node metastasis in patients with penile cancer and clinically negative lymph nodes. J Urol. 2010; 184: 539-545.
- Dai B, Kong YY, Ye DW, Xu XW, Yao XD, Zhang SL. Basal cell carcinoma of the scrotum: clinicopathologic analysis of 10 cases. Dermatol Surg. 2012; 38: 783-790.
- Ng LG, Yip SK, Tan PH. Extramammary Paget’s disease of scrotum. Urology. 2001; 58: 105.
- Huang JH, Broggi M, Osunkoya AO, Master VA. Surgical Management of Primary Scrotal Cancer. Urol Clin North Am. 2016; 43: 531-544.
- Sohrabi C, Mathew G, Maria N, Kerwan A, Franchi T, Agha RA; Collaborators. The SCARE 2023 guideline: updating consensus Surgical CAse REport (SCARE) guidelines. Int J Surg. 2023; 109: 1136- 1140.