Cutaneous Diagnosis of Systemic Disseminated Fusarium Infection in a Pediatric Patient with Leukemia during Hematopoietic Stem Cell Transplantation
- 1. Department of Child Health, Sultan Qaboos University Hospital, Oman
- 2. Father Muller Medical College, India
Abstract
Background: Skin involvement in fusariosis can present as rapidly progressive disseminated lesions.
Case presentation: We report a 12-year old boy with leukemia with fever on day+31 post-hematopoietic stem-cell transplant. Despite antibiotics, fever continued with emergence of tender scattered papules over back, breakage in skin integrity in interdigital space (right 3rd-4th toes) within next week. Amphotericin-B (3mg/kg) was started in view of suspected fungal infection.
On day+39, patient was started on reconditioning-protocol for second transplant as he failed the primary transplant. Over next 3-4 days, generalized skin lesions appeared in different stages of evolution. Despite high inflammatory markers, blood cultures remained negative.
Skin biopsy performed a week after onset of lesions revealed septate hyphae. Skin culture revealed heavy growth of fusarium with species identification (Fusarium keratoplasticum-sensitive to Amphotercin-B).
CT scan revealed focal abnormalities namely thyroid, pulmonary, splenic and renal hypodense lesions favoring diagnosis of disseminated fusarium infection. Fever and skin lesions showed slow resolution over next few weeks. Clinical course was complicated by graft-versus-host disease of gut/skin, cytomegalovirus viremia & adenoviremia; with recurrent episodes of soft tissue swellings at different sites suggesting frequent showering of septic emboli by underlying invasive fungal infection; necessitating continuation of Amphotericin-B (5 mg/kg). Patient succumbed to the disease 13 months post-transplant.
Conclusion: Disseminated fusariosis should be considered as a possible differential in the diagnosis of immunocompromised patients with fever; and the appearance of cutaneous lesions should warrant a skin biopsy/culture, as skin might be the only diagnostic source. Prognosis is poor with death without antifungal therapy.
Keywords
• Disseminated fusariosis
• Skin lesions
• Immunocompromised
CITATION
Tony S, Mevada R (2024) Cutaneous Diagnosis of Systemic Disseminated Fusarium Infection in a Pediatric Patient with Leukemia during Hematopoietic Stem Cell Transplantation. Ann Pediatr Child Health 12(2): 1333.
INTRODUCTION
Invasive fungal infections can cause high mortality and morbidity in patients with hematologic malignancies, prolonged neutropenia post-chemotherapy and/or severe T-cell immunodeficiency [1,2]. Fusarium species represent the second most frequent mold-causing invasive fungal infections and cause a broad spectrum of infections, from superficial (keratitis, onychomycosis) to invasive and disseminated (pneumonia, fungemia, severe skin lesions) [2-5].
Skin infection may occur either as primary or metastatic site In immunocompromised patients, skin involvement can present as rapidly progressive disseminated lesions at various stages of evolution. Skin might be the only source of diagnosis for majority of them [3]. Since fusariosis is still a rare infection, problems related to it lie in difficult diagnosis and treatment1 .
We report a pediatric patient with acute myeloid leukemia (AML) post-chemotherapy undergoing hematopoietic stem-cell transplantation (HSCT) who presented with cutaneous lesions as a manifestation of underlying disseminated fusariosis. Skin biopsy/culture identified Fusarium keratoplasticum and proved to be diagnostic in this immunocompromised patient.
CASE PRESENTATION
We report a 12-year old boy with AML on voriconazole prophylaxis with fever on day +31 post-HSCT. Tazocin was initiated after septic work-up. Within next 24 hours, patient developed septic shock requiring fluid boluses and antibiotics were upgraded (Meropenem, Amikacin and Vancomycin).
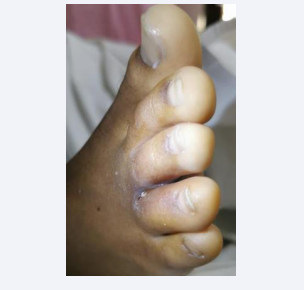
Fever spikes continued with emergence of new tender scattered papules on skin over back, extremities and whitish area of tenderness with breakage in skin integrity in interdigital space between right 3rd and 4th toes within the next week (Figure 1). In view of strong clinical suspicion of fungal foot infection, Amphotericin-B was added (3 mg/kg) to the antibiotic regimen in addition to voriconazole.
Figure 1: Whitish cutaneous lesion with skin disintegration interdigital space.
On day + 39, patient was restarted on conditioning-protocol for a second transplant as he failed the primary transplant. Over next 3-4 days along with persistent fever, new painful papular lesions appeared on scalp and behind left ear with central necrosis followed by similar lesions in different stages of evolution on left thigh, axilla and chest; hence Posaconazole was added, Voriconazole discontinued; simultaneously Amphotericin-B dosage was increased (5mg/kg/day). Inflammatory markers were high (absolute neutrophil count 3000 cells/cmm and C-Reactive Protein 300 mg/L), despite negative blood cultures and unremarkable echocardiography.
Skin biopsy performed a week after onset of initial skin lesions revealed septate hyphae. Skin culture eventually revealed heavy growth of fusarium. Isolate was sent to the mycology reference laboratory in Bristol and identified as Fusarium keratoplasticum which is part of the old Fusarium solani complex. Minimum inhibitor concentrations (MICs) to various antifungals was conducted (sensitive to Amphotericin-B, intermediate sensitivity to azoles); and Posaconazole was discontinued.
To evaluate systemic spread, Computerized Tomography (CT) scan was done which revealed focal abnormalities namely thyroid, pulmonary (multiple nodules with halo signs predominantly located at the periphery of the lungs), splenic and renal hypodense lesions which were most-likely related to the known underlying fungal infection suggestive of disseminated fusarium infection, in addition Fine Needle Aspiration from thyroid lesion confirmed fusarium.
Magnetic Resonance Imaging done in view of persistent foot infection revealed osteomyelitis of navicular bone. Patient underwent surgical debridement and pus revealed growth of fusarium. Fever and skin lesions showed gradual resolution, antibiotics were downgraded and discontinued after 6 weeks; with continuation of Amphotericin-B.
Although patient had complete chimerism, his clinical course was complicated by graft-versus-host disease (GVHD) of gut/ skin, cytomegalovirus viremia, and adenoviremia; together with recurrent episodes of soft tissue swellings and inflammation at different sites suggesting intermittent showering of septic emboli by the underlying invasive fungal infection.
He underwent frequent imaging for follow-up of flare-up of disseminated lesions with isolation of fusarium from affected sites. Six months post-transplant, CT scan revealed bilateral subpleural nodules in the lungs, interval reduction in the size and number of splenic lesions and stable thyroid lesion.
In view of prolonged administration of immunosuppression (flare up of GVHD), low lymphocyte count (300 cell/cmm), recurrent swellings with inflammation, the consensus was to continue antifungal therapy for 1-1.5 years post-transplant. Thirteen months post-transplant patient developed respiratory infection with respiratory failure, severe systemic inflammation and cytokine storm and succumbed to the disease.
DISCUSSION
Skin involvement in fusariosis can represent either a primary site of infection, usually as cellulitis of toes, or a manifestation of metastatic infection in patients with disseminated fusariosis. Patients at risk for disseminated fusariosis include those with acute leukemia, prolonged neutropenia and patients undergoing HSCT. Disseminated disease is the most frequent and challenging clinical form of fusariosis, accounting for approximately 70% of all cases of fusariosis in immunocompromised patients [5].
Fusarium solani (50%) is the most common species causing infection, followed by Fusarium oxysporum (20%) and Fusarium verticillioidis and Fusarium moniliforme (10% each) [2].
The most frequent pattern of disseminated disease in severely immunocompromised patients includes combination of cutaneous lesions and positive blood cultures, with or without involvement of other sites (sinuses, lungs, and others). Fusarial skin lesions can involve practically any site, with predominance in extremities, and evolve rapidly, usually over a few days [5].
Lesions at different stages of evolution (papules, nodules, and necrotic lesions) may be present. Patients with disseminated disease typically have multiple erythematous papular or nodular and painful lesions, frequently with central necrosis giving the lesions an echthyma gangrenosum-like appearance. Skin lesions might be the single source of diagnosis in majority of patients with such lesions [5].
Since fusariosis is still a rare infection, problems related to it lie in difficult diagnosis and treatment [1]. The definite diagnosis of Fusarium infection requires Fusarium species isolation either from blood or tissue culture (such as the skin, lungs and sinuses) [6]. In presence of disseminated skin lesions, the presence of the fungus in blood cultures may increase to 56%. In majority of cases, fungemia is likely to develop in a median of five days (range, one to 10 days) following the appearance of skin lesions7 . However, in the present case, blood culture did not yield Fusarium species despite disseminated skin lesions.
Skin biopsies are extremely easy to perform and confirm clinical suspicion [5]. Biddeci et al., reported diagnostic skin lesion culture tests in 55% of the cases [4]. Similarly, in our patient skin culture was diagnostic, and the pathogen identified was Fusarium kaerotoplasticum.
Identifying the specific pathogen is critical, because Fusarium species have variable susceptibilities to the antifungal agents that are currently used [2]. Nucci et al., suggested high-dose amphotericin-B or liposomal amphotericin-B since specific Fusarium species may be resistant to azoles. In our patient, the isolate confirmed adequate MIC of 1.0 mg/L to Amphotericin-B, hence single-agent therapy was continued [8].
The prognosis is very poor with death without antifungal therapy, as dissemination is known to occur especially in immunocompromised hosts with hematologic malignancies and chemotherapy treatment [1]. Mortality from fusarial infections in immunocompromised patients ranges from 50% to 80% [9]. As seen in our patient, the prognosis is considerably worse in patients with pulmonary infiltrates [10].
The greatest challenge to clinicians is the early diagnosis of the Fusarium infection and preemptive treatment; requiring a strong clinical suspicion on the basis of constellation of clinical and laboratory findings.
CONCLUSION
Disseminated fusariosis is a challenging clinical form of fusariosis and is associated with a high mortality in immunocompromised patients. It should be considered as a possible differential in the diagnosis of immunocompromised patients with fever; and the appearance of cutaneous lesions should warrant a skin biopsy/culture, as skin might be the only diagnostic source. Apart from diagnosis, specific species identification is important in guiding antifungal therapy to prevent mortality in these patients.
DECLARATIONS
Authors’ contributor Statements: S. Tony designed the study and wrote the manuscript; R. Mevada assisted in drafting the manuscript and critically reviewed the manuscript. Both authors agreed upon the final version of the manuscript and have reviewed and agreed upon the manuscript content.
Informed consent from legal guardian was obtained for data collection and publication.