Characterization of the Left Ventricle after Transcatheter Aortic Valve Implantation in Patients with Cardiovascular Disease
- 1. Department of Cardiology, University Hospital Duesseldorf, Germany
- 2. Department of Cardiology, University of Luebeck, Germany
- 3. Department of Cardiology, Linköping University, Sweden
- 4. Department of Cardiology, UT South Western, Dallas, USA
- 5. Department of Imaging Sciences, King’s College London, UK
Abstract
Background: Aortic stenosis, the most common valve disease in developed countries, can be ameliorated through surgical replacement or transcatheter aortic valve implantation (TAVI). Our aim was to investigate functional recovery of the left ventricle after TAVI, including improvements in regional wall motion and scar formation, in patients with severe aortic stenosis, known cardiovascular disease and heart failure.
Methods: Thirty patients with severe aortic stenosis and cardiovascular disease with heart failure who were referred for TAVI were prospectively enrolled in the study. These patients exhibited discrete midwall fibrosis and/or light fibrosis at the upper and lower insertion points. Before undergoing TAVI, cardiac magnetic resonance imaging (MRI) was performed using a 1.5-Tesla magnetic resonance scanner. A standard balanced steady-state free precession sequence with strain-encoded (SENC) imaging was used to assess left and right ventricular volumes. Late gadolinium enhancement imaging was performed 10 minutes after application of contrast agent. Three-month follow-up imaging was performed using an identical cardiac MRI protocol to characterize the left ventricle.
Results: Three months after TAVI, global (p<0.01) and circumferential strain (p<0.03) were significantly improved. Left ventricular function was significantly increased at follow-up, and volumes were significantly reduced. No new scar formation or fibrotic tissue was detected.
Conclusion: TAVI resulted in functional recovery in patients with cardiovascular disease and known heart failure by decreasing left ventricular volumes and increasing ejection fraction, and no new fibrotic tissue was generated.
CITATION
Elkenhans B, Vieregge I, Henningsson M, Hussain TM, Botnar R, et al. (2023) Characterization of the Left Ventricle after Transcatheter Aortic Valve Implantation in Patients with Cardiovascular Disease. Ann Vasc Med Res 10(4): 1169.
INTRODUCTION
Aortic stenosis is the most common valve disease in developed countries [1], and the onset of symptoms predicts a substantially reduced life expectancy [2]. Because of their advanced age, patients with aortic stenosis often present with comorbidities, such as cardiovascular disease, hypertension, and diabetes. Aortic stenosis can result in continuous pressure overload, and patients with comorbid cardiovascular disease experience left ventricular function impairment and volume increase, which leads to heart failure if not corrected.
Transcatheter aortic valve implantation (TAVI) replaces the stenotic valve with a synthetic replacement; it is recommended for patients who are considered high risk for open-heart surgery, and its use is being investigated in intermediate- and low-risk patients [3]. In comparison with surgical aortic valve replacement (SAVR), TAVI is associated with a similar degree of left ventricular remodeling, but is superior in reducing the valvular pressure gradient and myocardial fibrosis [4]. Furthermore, 6 months after TAVI, left ventricular function, volume, and mass improve significantly [5]. However, although low-flow, low-gradient aortic stenosis does not influence procedural mortality immediately after TAVI, it does increase the 6-month and 1-year mortality, but the survivor’s exhibit haemodynamic and clinical improvements [6]. Furthermore, apical left ventricular function abnormalities may persist in transapical TAVI patients [7].
Currently, cardiac magnetic resonance imaging (MRI) plays a central role in TAVI, quantifying ejection fraction, scar formation, and myocardial mass. In this study, we examined regional and global left ventricular wall motion abnormalities, as assessed by strain-encoded (SENC) MRI, to determine whether regional wall motion abnormalities would be improved 3 months after TAVI in patients with known cardiovascular disease and heart failure without Periprocedural subendocardial scarring.
METHODS
Study population
Institutional Review Board approval was obtained prior to the commencement of recruitment. This study prospectively recruited 30 patients with severe aortic stenosis with known cardiovascular disease and heart failure referred for TAVI at the University Hospital of Duesseldorf, Germany, between December 2012 and March 2015. The diagnosis of severe aortic stenosis was made via transoesophageal echocardiography or transthoracic echocardiography and defined as an aortic valve area <1.0 cm2 or peak velocity >4.0 m/s, respectively. An additional inclusion criterion was a Euro Score >20. The exclusion criteria were any contraindications to cardiac MRI.
Cardiac MRI protocol
Patients with severe aortic stenosis and known cardiovascular disease with heart failure were scanned on a 1.5-Tesla scanner (Achieva, Philips, Best, the Netherlands) with a 32-channel coil. To image the entire left and right ventricles, multislice, multiphase cine imaging was performed using a standard steady- state free precession pulse sequence in short-axis view (voxel size 1.48 × 1.48 × 8.0 mm3, repetition time (TR) 3.2, echo time (TE) 1.59, flip angle 60°). Late gadolinium enhancement (LGE) imaging (voxel size 1.37 × 8.0 × 8.0 mm3, TR 3.8, TE 1.86, flip angle 15°) was performed following a Look-Locker sequence (inversion time scout) 10 minutes after the administration of gadoterate meglumine contrast agent (Dotarem, 0.2 mmol/l, Guerbet, Villepinte, France). The images were analysed at the scanner workstation (EWS, release 3.2.2, Philips) to quantify left and right ventricular volumes, myocardial mass, and myocardial scarring. SENC was conducted [8] in four- and two-chamber views to investigate longitudinal strain. Three months after TAVI, patients were followed up using an identical cardiac MRI protocol.
Cardiac MRI image analysis
Endocardial and epicardial borders were manually contoured at end-diastole and end-systole to allow the calculation of ventricular volumes and mass according to the formula (epicardial volume – endocardial volume) × myocardial density (1.05 g/cm3), and values were indexed to body surface area. To measure longitudinal strain, the left ventricle was divided into 12 segments and analysed with dedicated SENC software (Diagnosoft MAIN version 1.06, Diagnosoft Inc. Palo Alto, Calif.) [8]. LGE areas were measured and indexed for normal myocardium and then interpreted by calculating the segmental extent of LGE on short- axis planes at baseline to assess the transmural extent of defects.
Statistical analysis
Data were analysed using Graph Pad Prism (version 5, LaJolla, CA 92037, USA). The chi-square test and Fisher’s exact test were used for normally distributed variables. Student’s t-test and the Mann-Whitney U test were used for nonparametric variables. P-values less than 0.05 were regarded as statistically significant.
RESULTS
Our study population comprised 20 patients with known coronary artery disease. Six patients had previously undergone coronary artery bypass grafting (CABG), and 13 had previously undergone percutaneous coronary intervention (PCI). Two patients had peripheral artery disease (PAD), and one patient had cerebral sclerosis. One patient had previously had a stroke, and 7 patients had atrial fibrillation. Thirteen patients presented with hypertension, and 7 patients had diabetes. There were 5 patients with chronic obstructive lung disease Table 1.
Table 1: Number and percentage of the study population with different comorbidities
|
Comorbidity |
N=30 |
% |
|
CVD |
20 |
66.67 % |
|
MI |
0 |
0.00 % |
|
Previous CABG |
6 |
20.00 % |
|
Previous PCI |
13 |
43.33 % |
|
PAD |
2 |
6.67 % |
|
cAVK |
1 |
3.33 % |
|
Stroke |
1 |
3.33 % |
|
Atrial fibrillation |
7 |
23.33 % |
|
Hypertension |
13 |
43.33 % |
|
Diabetes mellitus |
7 |
23.33 % |
|
COPD |
5 |
16.67 % |
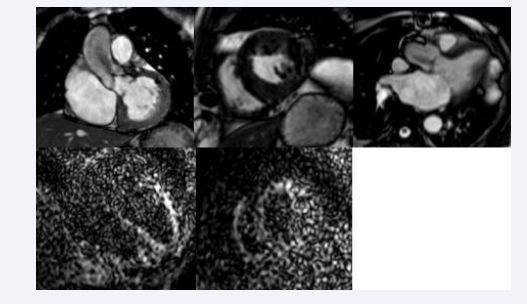
Prior to TAVI, patients with severe aortic stenosis were investigated with a standardized imaging protocol, which included cine imaging in four-, three-, and two-chamber view, and in short-axis view.
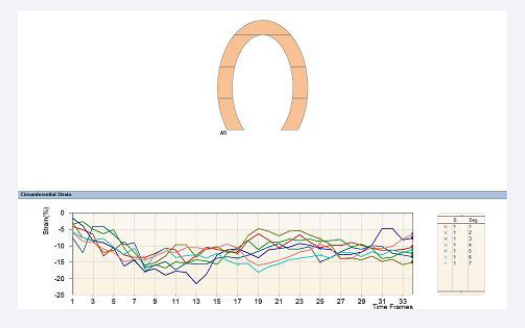
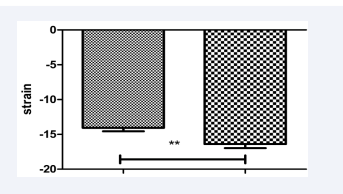
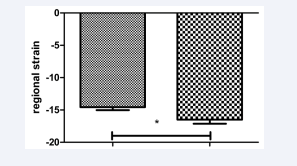
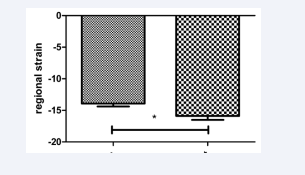
SENC images in four-chamber view, two-chamber view and short-axis view were also acquired. Three months post-TAVI, global strain was significantly increased in both the four-chamber (p<0.01) and two-chamber views (p<0.01), and regional strain was significantly elevated in the four-chamber (p<0.03) and two- chamber views (p<0.02) Figure 1a, 1c, 1d.
Figure 1a: Cine loop images from cardiac MRI in a patient with aortic stenosis (slide one), short-axis view (slide two) and three-chamber view (slide three). SENC imaging in four-chamber view (slide four) and short-axis view (slide five) one day pre-TAVI.
Figure 1c: Shows SENC analysis in four-chamber view one day pre-TAVI for the left ventricle.
Figure 1d: SENC analysis in two-chamber view for the left ventricle one day pre-TAVI.
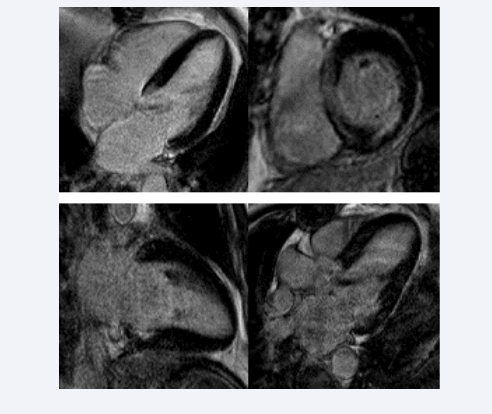
Late gadolinium enhancement images were acquired in short-axis view, four-chamber view, two-chamber view and three-chamber view pre-TAVI Figure 1b.
Figure 1b: Late gadolinium enhancement images obtained one day pre-TAVI. Upper left: short-axis view; upper right: four-chamber view; lower left: two-chamber view; lower right: three-chamber view
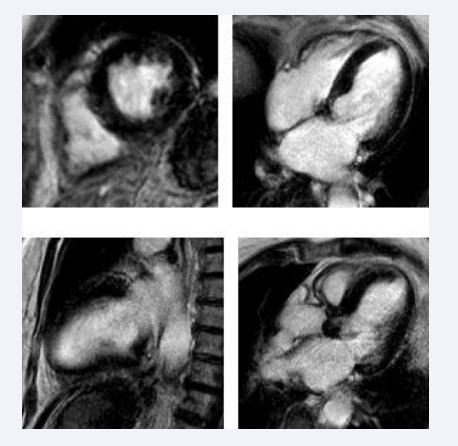
Three months post-TAVI, patients were imaged with an identical protocol. (Figure 2a -c) shows
Figure 2a: Late gadolinium enhancement images obtained one month post-TAVI. In the upper left image, no subendocardial scarring or midwall fibrosis is seen. The upper right image shows inferolateral subendocardial scarring and discrete midwall septal fibrosis in a short-axis view. The lower left image presents a two-chamber view showing no fibrosis or endomyocardial scarring. The lower right image presents a three-chamber view, again showing no subendocardial scarring or fibrosis.
Figure 2b: SENC analysis one month post-TAVI performed in four-chamber view
Figure 2c: SENC analysis one month post-TAVI performed in two-chamber view.
discrete midwall fibrosis in the short-axis view in the septal wall and subendocardial scarring on the inferolateral wall with 25 % transmurality.
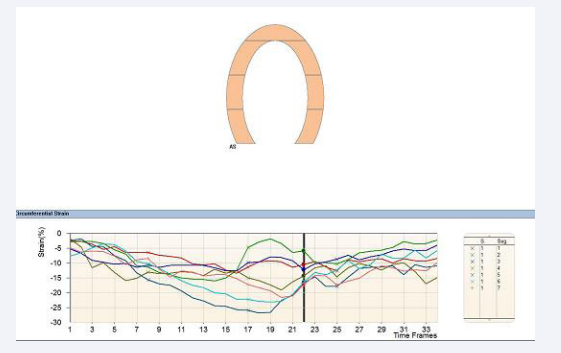
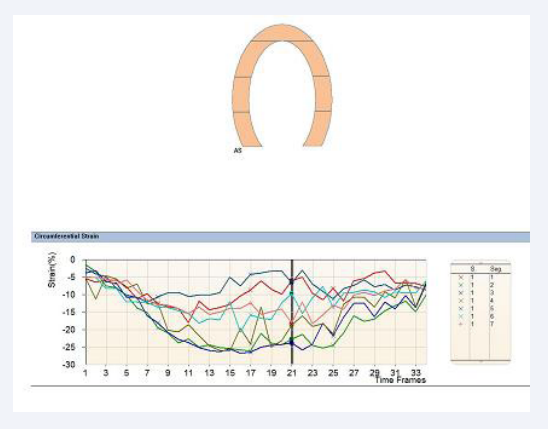
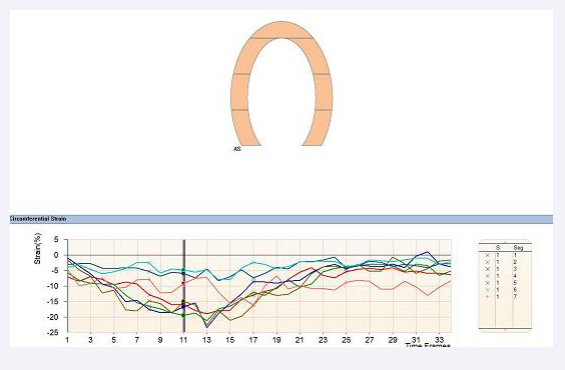
Global and regional strain was improved (Figure 3a-d).
Figure 3a: Four chamber view global strain of the left ventricle. Left column: one day prae TAVI, right column: one month post TAVI. p<0.01.
Figure 3b: Two chamber view global strain of the left ventricle. Left column: one day prae TAVI, right column: one month post TAVI. p<0.01.
Figure 3c: Four chamber view circumferential strain of the left ventricle. Left column: one day prae TAVI, right column: one month post TAVI. p<0.03.
Figure 3d: Two chamber view circumferential strain of the left ventricle. Left column: one day prae TAVI, right column: one month post TAVI. p<0.02.
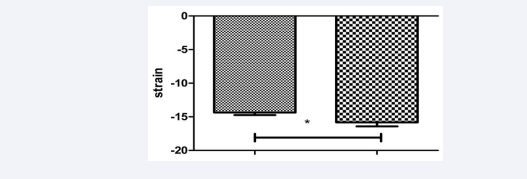
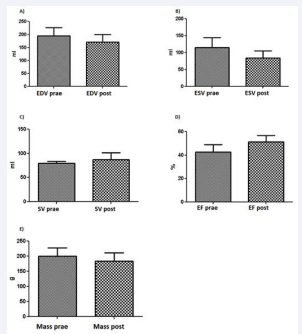
At the 3-month follow-up, left ventricular volumes were significantly reduced post-TAVI [Figure 4], and left ventricular function was improved Figure 4.
Figure 4: EDV: enddiastolic volume, ESV: endsystolic volume, SV: stroke volume, EF: ejection fraction
a) End-diastolic volumes one day pre-TAVI and one month post-TAVI.
b) End-systolic volumes one day pre-TAVI and one month post-TAVI.
c) Stroke volume one day pre-TAVI and one month post-TAVI.
d) Ejection fraction one day pre-TAVI and one month post-TAVI.
e) Myocardial mass one day pre-TAVI and one month post-TAVI.
DISCUSSION
The use of cardiac MRI with SENC imaging and conventional LGE to assess left ventricular volume and ejection fraction revealed significant improvements in patients with severe aortic stenosis and cardiovascular disease with heart failure undergoing TAVI despite the high prevalence of comorbidities, suggesting that aortic valve replacement stabilizes coronary artery disease as well. Regional wall motion abnormalities improved significantly, a finding in line with previous research comparing left ventricular mechanics of patients undergoing TAVI to those undergoing SAVR [9]. Furthermore, although others have reported apical left ventricular function abnormalities at the 3-month follow-up after TAVI [7], in our study, ejection fraction increased and left ventricular volume and myocardial mass had decreased at that time point. This finding mirrors that of Merten [5], who described a significant improvement in left ventricular function, volume, and mass 6 months after TAVI [4]. Observed that TAVI was comparable to SAVR regarding left ventricular remodelling and superior at reducing the valvular pressure gradient and myocardial fibrosis.
Myocardial infarction is an infrequent complication of TAVI, particularly in comparison with SAVR [10], fortunately, no myocardial infarctions were reported during TAVI or during the three-month follow-up period in our patients. We also detected no new scar formation and only very light midwall fibrosis and discrete fibrosis at the upper and lower insertion points at 3 months. Reverse remodelling occurs immediately after TAVI to a degree dependent on the amount of fibrotic or subendocardial scarring before the procedure [11]. Because patients with subendocardial scarring were excluded from our study, we conclude that remodelling was ameliorated 3 months post-TAVI in our study population.
Cardiac MRI plays a central role in patient assessment in TAVI. A compelling advantage of cardiac MRI over computed tomography is the ability to investigate left and right ventricular volume, aortic and valve diameters, and myocardial scarring or fibrosis with a single method. Using cardiac MRI, we found that aortic valve replacement with TAVI leads to improved left ventricular function, with decreased volumes, increased global and regional strain, increased ejection fraction, and improved wall motion abnormalities despite the presence of comorbid cardiovascular disease.
LIMITATIONS
This study included only selected patients due to contraindications of cardiac MRI, e.g., implantation of devices such as permanent pacemakers and implantable cardio vertex- defibrillators.
CONCLUSION
TAVI is beneficial for elderly patients with cardiovascular comorbidities because it promotes functional recovery of the heart. Further studies should address whether cardiovascular disease needs to be treated prior to the procedure.
REFERENCE
- Nkomo VT GJ, Skelton TN, Gardin JM, Gottdiener JS, Scott CG, Sarano ME. Burden of valvular heart dieseases: A population based study. Lancet. 2006; 368: 1005-10011.
- Otto CM. Valvular aortic stenosis: Disease severity and timing of intervention. J A Coll Cardiol. 2006; 47: 2141-2151.
- Voigtländer L, Seiffert M. Expanding TAVI to Low and Intermediate Risk Patients. Front Cardiovasc Med. 2018; 5: 92.
- Fairbairn TA, Steadman CD, Mather AN, Motwani M, Blackman DJ, Plein S, et al. Assessment of valve haemodynamics, reverse ventricular remodelling and myocardial fibrosis following transcatheter aortic valve implantation compared to surgical aortic valve replacement: A cardiovascular magnetic resonance study. Heart. 2013; 99: 1185-1191.
- Merten C, Beurich HW, Zachow D, Mostafa AE, Geist V, Toelg R, et al. Aortic regurgitation and left ventricular remodeling after transcatheter aortic valve implantation: A serial cardiac magnetic resonance imaging study. Circ Cardiovasc interv. 2013; 6: 476-483.
- Elhmidi Y, Piazza N, Krane M, Deutsch MA, Mazzitelli D, Lange R, et al. Clinical presentation and outcomes after transcatheter aortic valve implantation in patients with low flow/low gradient severe aortic stenosis. Catheter Cardiovascular Interv. 2014; 84: 283-290.
- Meyer CG, Frick M, Lotfi S, Altiok E, Koos R, Kirschfink A, et al. Regional left ventricular function after transapical vs. Transfemoral transcatheter aortic valve implantation analysed by cardiac magnetic resonance feature tracking. Eur Heart J Cardiovasc Imaging. 2014; 15: 1168-1176.
- Neizel MK, Lossnitzer D, Kuehl H, Hoffmann R, Ocklenburg C, Giannitsis, et al. Impact of systolic and diastolic deformation indexes assessed by strain-encoded imaging to predict persistent severe myocardial dysfunction in patients after acute myocardial infarction at follow-up. J Am Coll Cardiol. 2010; 56: 1056-1062.
- Nucifora G, Tantiongco JP, Crouch G, Bennetts J, Sinhal A, Tully PJ, et al. Changes of left venricular mechanics after trans-catheter aortic valve implantation and surgical aortic valve replacement for severe aortic stenosis: A tissue tracking cardiac magnetic resonance study; Int J Cardiol. 2017; 228: 184-190.
- Dobson LE, Musa TA, Uddin A, Fairbairn TA, Swoboda PP, Ripley DP, et al. Post-prodecural myocardial infarction following surgical aortic valve replacement and transcatheter aortic valve implantation; EuroIntervention. 2017; 2: e153-e160.
- Dobson LE, Musa TA, Uddin A, Fairbairn TA, Swoboda PP, Erhaiem B, et al. Acute Reverse Remodelling After Transcatheter Aortic Valve Replacement: A Link Between Cardiac Fibrosis And Left Ventricular Mass Regression. Can J Cardiol. 2016; 12: 1411-1418.