Platelet Counts and Imbalanced Proportions of Phosphatidylinositol and Phosphatidylcholine in Plasma Membranes of Hematopoietic Cells in Patients with Paroxysmal Nocturnal Hemoglobinuria
- 1. Institute of Hematology and Transfusion Medicine, Poland
- 2. Department of Biochemistry and Biopharmaceuticals, National Medicines Institute, Poland
Abstract
Paroxysmal nocturnal hemoglobinuria (PNH) is an acquired clonal hematopoietic stem cell disorder characterized by the absence of glycosylphosphatidylinositol (GPI)-anchored proteins due to mutations in the PIG-A gene. This deficiency leads to intravascular hemolysis, bone marrow failure, and an increased risk of thrombotic events. This study investigates the relationship between phosphatidylinositol (PI) and phosphatidylcholine (PC) in hematopoietic cells of PNH patients and their association with clinical outcomes, specifically platelet counts and thrombotic risk. We analyzed blood samples from 22 PNH patients and 14 healthy controls, quantifying lipid content using high-performance liquid chromatography. Our results reveal a significant increase in the PI/PC molar ratio in PNH patients, reflecting decreased levels of PC in cellular membranes, which correlates with thrombocytopenia. These findings suggest that phospholipid imbalances are linked to both the functional deficiencies of hematopoietic cells and the heightened thrombotic tendency seen in PNH. Ultimately, this study underscores the potential for targeted therapies aimed at addressing lipid metabolism in managing PNH and mitigating thrombotic risks. Future investigations could explore the clinical implications of dietary or pharmacological interventions designed to restore lipid balance in these patients.
Keywords
• Paroxysmal nocturnal hemoglobinuria
• Thrombosis
• Platelets
• Phosphatidylinositol
• Phosphatidylcholine
• Phospholipid imbalance
Citation
Nowak J, Szlendak U, Jaworska M, Ros?on M, Mendek-Czajkowska E, et al. (2026) Platelet Counts and Imbalanced Proportions of Phosphati dylinositol and Phosphatidylcholine in Plasma Membranes of Hematopoietic Cells in Patients with Paroxysmal Nocturnal Hemoglobinuria. Ann Vasc Med Res 13(1): 1195.
INTRODUCTION
Paroxysmal is nocturnal hemoglobinuria (PNH) recognized as an acquired clonal hematopoietic stem cell disorder characterized by the absence of glycosylphosphatidylinositol (GPI)-anchored proteins, predominantly due to somatic mutations in the PIG-A gene located on the X chromosome. This genetic alteration results in the risk of intravascular hemolysis, bone marrow failure, and increased thrombotic events. While the mutation itself initiates the clonal expansion of defective hematopoietic cells, several additional factors play crucial roles in this process, including immune evasion, the presence of supportive microenvironmental factors, and alterations in cell signaling mechanisms. Chatzidavid et al., highlighted that while PNH arises from a singular mutation, factors such as a hypercoagulable state may perpetuate the expansion of these abnormal cells [1]. The absence of GPI-anchored proteins inhibits the normal functions of immune cells that would typically target and eliminate abnormal cells [2,3]. In healthy cells, phosphatidylinositol is converted to glycosylated phosphatidylinositol; however, this conversion is halted in PNH cells due to PIG-A mutation [4]. This disruption not only affects cellular signaling pathways but also alters the lipid composition of plasma membranes. PI content in cells is fine regulated by highly specific enzyme, the phosphatidylinositol transfer protein (PITP) that is able to deliver PI to plasma membranes at the expense of equimolar quantity of phosphatidylcholine (PC) [5]. The imbalance in PI and PC in hematopoietic cells may play a role in this context, as alterations in phospholipid composition can influence membrane stability and cellular signaling, thereby affecting platelet activation. Thrombosis is a significant complication associated with PNH, with patients experiencing a higher risk of thrombotic events such as venous thromboembolism. One of the primary mechanisms by which thrombocytopenia occurs in PNH is through complement-mediated destruction of platelets. Notably, Gralnick et al., highlighted that activated platelets release procoagulant factors, which may amplify the risk of thrombosis in the presence of intravascular hemolysis [6]. The dynamic between platelet activation and consumption in a hypercoagulable state creates a vicious cycle wherein thrombosis leads to further depletion of platelets and worsening thrombocytopenia [7,8]. Given this context, this study was designed to investigate the proportional relationship between PI and PC in hematopoietic cells of PNH patients, focusing on how these cellular changes relate to clinical outcomes, particularly platelet counts and thrombotic risk. Understanding these relationships is crucial for developing targeted therapies aimed at reducing thrombosis incidence in PNH patients.
MATERIALS AND METHODS
Study Design
To assess the phospholipid content of PI and PC in nucleated hematopoietic cells, a total of 22 PNH patients were included in the study alongside 14 healthy controls. All patients signed informed consent for the project. The patients were tested at the Institute of Hematology and Transfusion Medicine, Warsaw from March 2018 to March 2020. All patients were treated with eculizumab. The patients were diagnosed clinically based on laboratory findings of hemolysis and PNH-specific assays, including FLAER tests for erythrocytes and granulocytes. The research was conducted in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki). The research project was approved by the Bioethics Committee of the Institute of Hematology and Transfusion Medicine.
Sample Collection and Processing
Peripheral blood samples were collected from PNH patients and healthy controls. Plasma membrane phospholipids were extracted using a modified Folch- based lipid extraction protocol [9]. This method allows for the effective separation of lipid components from cellular debris while preserving the integrity of the phospholipids for subsequent analysis.
Phospholipid Analysis
Extracted phospholipid fractions were analyzed utilizing high-performance liquid chromatography (HPLC) with charged aerosol detection (CAD) [10]. The concentrations of phosphatidylinositol and phosphatidylcholine were quantified and expressed as nmoles per 106 cells, facilitating the calculation of the molar ratios of PI to PC in cellular membranes.
RESULTS
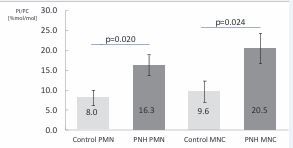
Our findings indicated a significant increase in the PI/PC molar ratio within the hematopoietic cells of PNH patients compared to healthy controls (Figure 1). Specifically, the mean ratio in polymorphonuclear (PMN) cells was 16.3 ± 2.6 %mol/mol versus 8.0 ± 2.0 %mol/mol in controls (p=0.020). In mononuclear cell (MNC) fractions, this ratio was 20.5 ± 3.8 %mol/mol in PNH patients versus 9.6 ± 2.7 %mol/mol in controls (p=0.024).
The notable imbalance in PI/PC was predominantly attributed to a reduction in the absolute content of PC in the cellular membranes of PNH patients (Table 1). Furthermore, the proportion of FLAER (-) cells in PNH was directly correlated with the PC content in PMN cells (R=0.53, p=0.020). Notably, platelet (PLT) counts exhibited a strong positive correlation with PC content in the MNC subset (R=0.55, p<0.01).
Figure 1 Molar proportions of PI/PC in PMN and MNC cells in PNH patients (mean ± SE). Legend: MNC, mononuclear cells; PMN, polymorphonuclears; PC, phosphatidylcholine; PI, phosphatidylinositol; PNH, paroxysmal nocturnal hemoglobinuria; SE, standard error.
Table 1: PI and PC content in PNH patients and controls (mean values, ng/106 cells and p).
|
Cells |
PI PNH |
PI Control |
p |
PC PNH |
PC Control |
p |
PI/PC PNH |
PI/PC Control |
p |
|
PMN |
0,31 |
0,32 |
0,90 |
1,70 |
3,15 |
0,00077 |
16,28 |
8,04 |
0,020 |
|
MNC |
0,36 |
0,33 |
0,90 |
2,14 |
3,98 |
0,0019 |
20,47 |
9,63 |
0,024 |
Additionally, both white blood cell (WBC) and PLT counts tended to decrease as PI/ PC molar ratios increased (R>0.39, p<0.09).
DISCUSSION
The results from this investigation elucidate a significant imbalance in phosphatidylinositol (PI) and phosphatidylcholine (PC) proportions within the plasma membranes of hematopoietic cells in patients with PNH. Reduced ratios of PI to PC in the membranes of hematopoietic cells from patients with PNH can significantly affect the survival of pathological cells. This alteration is linked to the impaired synthesis of glycosylphosphatidylinositol (GPI) anchors stemming from mutations in the PIGA gene, which plays a crucial role in the production of GPI-anchored proteins essential for cellular signaling and membrane integrity [11-13].
In normal cells, PI serves as a precursor for various signaling molecules, particularly phosphatidylinositol 3,4,5-trisphosphate (PIP3), which is pivotal for cell survival and proliferation. PIP3 is generated by the action of phosphoinositide 3-kinase (PI3K) on PI, and its presence in the cell membrane activates key pathways involved in cell growth and survival, including the AKT pathway, which inhibits apoptotic mechanisms [14]. Moreover, Szlendak et al. (4), suggested that compensatory mechanisms in PNH can lead to increased concentration and activity of PIP3 in GPI-negative clone cells, possibly as a response to signaling substrate imbalance. The dysregulation of this signaling pathway is concerning as it could promote PNH cell survival, facilitating the persistence of mutant hematopoietic cells. This suggests a complex interplay where even pathological cells, lacking regular regulatory GPI-anchored proteins such as CD55 and CD59, find ways to thrive under certain stress conditions through enhanced PI signaling. The enlarged relative proportions of PI may pose a mechanistic basis for enhanced anti-apoptotic signaling within these cells, potentially prolonging their lifespan and contributing to clonal domination over normal hematopoietic cells. Such alterations in cellular lipid composition may also have implications on cellular interactions and signaling processes in the hematopoietic microenvironment.
Phosphatidylcholine deficiency in the membranes of hematopoietic cells, including platelets, can significantly impact the survival of these cells and the risk of thrombosis. PC is a key component of cell membranes, and its deficiency leads to impaired cell membrane properties, negatively impacting physiological functions. The first aspect of the impact of PC deficiency is its impact on cell membrane integrity. PC is essential for maintaining membrane elasticity, and its deficiency can lead to increased cell susceptibility to damage. Studies
have shown that deficiencies of phospholipids, including PC, can lead to impaired platelet protective mechanisms, increasing their susceptibility to activation by coagulant factors such as thrombin [15]. Changes in membrane composition can also lead to increased release of procoagulant microparticles, which can promote further hemostatic activation and accelerate the thrombotic process [16]. In summary, this study advances the understanding of membrane lipid dynamics in PNH and establishes a significant association between lipid imbalances and clinical markers of disease severity, such as thrombosis and platelet counts. Further research is warranted to explore the effects of dietary or pharmacological modulation of these lipid components in PNH management.
REFERENCES
- Chatzidavid S, Giannakopoulou N, Diamantopoulos PT, Gavriilaki E, Katsiampoura P, Lakiotaki E, et al. JAK2V617F positive polycythemia vera with paroxysmal nocturnal hemoglobinuria and visceral thromboses: a case report and review of the literature. Thromb J. 2021; 19: 16.
- Raza A, Ravandi F, Rastogi A, Bubis J, Lim SH, Weitz I, et al. A prospective multicenter study of paroxysmal nocturnal hemoglobinuria cells in patients with bone marrow failure. Cytometry B Clin Cytom. 2014; 86: 175-182.
- Ruggiero G, Terrazzano G, Becchimanzi C, Sica M, Andretta C, Masci AM, et al. GPI-defective monocytes from paroxysmal nocturnal hemoglobinuria patients show impaired in vitro dendritic cell differentiation. J Leukoc Biol. 2004; 76: 634-640.
- Szlendak U, Krzymieniewska B, Mendek-Czajkowska E, Rogatko- Koro? M, Witkowska A, W?odarska J, et al. Persistent imbalance, anti-apoptotic, and anti-inflammatory signature of circulating C-C chemokines and cytokines in patients with paroxysmal nocturnal hemoglobinuria. Cytokine. 2022; 150: 155780.
- Wirtz KW. Phospholipid transfer proteins. Annu Rev Biochem. 1991; 60: 73-99.
- Gralnick HR, Vail M, McKeown LP, Merryman P, Wilson O, Chu I, et al. Activated platelets in paroxysmal nocturnal haemoglobinuria. Br J Haematol. 1995; 91: 697-702.
- Hillmen P, Muus P, Dührsen U, Risitano AM, Schubert J, Luzzatto L, et al. Effect of the complement inhibitor eculizumab on thromboembolism in patients with paroxysmal nocturnal hemoglobinuria. Blood. 2007; 110: 4123-4128.
- Moyo VM, Mukhina GL, Garrett ES, Brodsky RA. Natural history of paroxysmal nocturnal haemoglobinuria using modern diagnostic assays. Br J Haematol. 2004; 126: 133-138.
- FOLCH J, LEES M, SLOANE STANLEY GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957; 226: 497-509.
- Ros?on M, Jaworska M, Anuszewska EL. Determination of Glycerophospholipids in Biological Material Using High-Performance Liquid Chromatography with Charged Aerosol Detector HPLC-CAD-A New Approach for Isolation and Quantification. Molecules. 2022; 27: 3356.
- Rahman K, Gupta R, Harankhedkar S, Gupta T, Sarkar MK, NityanandS. Utility of CD157 as a Common Leukocytes Marker for Paroxysmal Nocturnal Hemoglobinuria Screening in a Single Tube Five Color Combination. Indian J Hematol Blood Transfus. 2018; 34: 304-309.
- Bessler M, Mason PJ, Hillmen P, Miyata T, Yamada N, Takeda J, et al. Paroxysmal nocturnal haemoglobinuria (PNH) is caused by somatic mutations in the PIG-A gene. EMBO J. 1994; 13: 110-117.
- Röth A, Dührsen U. Treatment of paroxysmal nocturnal hemoglobinuria in the era of eculizumab. Eur J Haematol. 2011; 87: 473-479.
- Wang H, Liu H, Li L, Chen Y, Liu Z, Li L, et al. Long noncoding RNA FAM157C contributes to clonal proliferation in paroxysmal nocturnal hemoglobinuria. Ann Hematol. 2023; 102: 299-309.
- Beckett CS, Kell PJ, Creer MH, McHowat J. Phospholipase A2-catalyzed hydrolysis of plasmalogen phospholipids in thrombin-stimulated human platelets. Thromb Res. 2007; 120: 259-268.
- Nordöy A, Hamlin JT, Chandler AB, Newland H. The influence of dietary fats on plasma and platelet lipids and ADP induced platelet thrombosis in the rat. Scand J Haematol. 1968; 5: 458-473.