Left Atrial Pressure and Volume in Patients with Ischemic Stroke
- 1. Department of Medical Genetics, Haukeland University Hospital, Norway
- 2. Department of Neurology, University of Bergen, Norway
Abstract
Aim: To explore the relation between left atrial pressure (LAP) and volume (LAV) in ischemic stroke patients with or without atrial fibrillation (AF). We hypothesized that there is an association between high LAP and atrial fibrillation.
Methods: All patients admitted to the Stroke unit, Haukeland University hospital are registered in the Norstroke database. A subgroup of the patients was referred to an out clinic cardiologist for transthoracic echocardiography based on the judgments by the clinician in charge of the patients during the hospital stay. The transthoracic echocardiography included measurement of LAV, LAP and ejection fraction (EF). Risk factors and MRI findings were registered. Univar ate, multivariate and lowess smoother curve analyses were performed.
Results: In total, 138 patients were referred to the cardiologist and 23 (17%) had documented AF. Linear regression analysis showed that LAP was independently associated female sex, hypertension and increasing LAV (all P<0.05). Linear regression analysis showed that LAV was independently associated with male sex, AF and increasing LAP (all P<.05). Lowess smoother curves showed U-shaped relations between LAV and both AF and old cerebral infarction on MRI.
Conclusion: We did not find an association between LAP and AF even though LAP correlated with LAV. However, we found U-shaped relations between LAV and both AF and old cerebral infarction on MRI.
Keywords
• Left atrial pressure
• Ejection fraction
• Norstroke
• Atrial pressure
Citation
Gradek G, Naess H (2020) Left Atrial Pressure and Volume in Patients with Ischemic Stroke. J Neurol Transl Neurosci 6(1): 1086.
INTRODUCTION
Ischemic stroke is often associated with atrial fibrillation (AF) and these strokes tend to be large [1]. Anticoagulation is more effective than antiplatelet treatment as to prevention of ischemic stroke in patients with AF. Paroxysmal AF is often difficult to detect even on long-term Holter monitoring [2], and the clinician needs to consider the possibility of silent AF.
Many risk factors for paroxysmal AF have been identified. These include high age, hypertensive heart disease, diabetes mellitus, obesity, hypertension, smoking, congestive heart failure, elevated troponin, and mitral valve disease [3-6]. In ischemic stroke patients, paroxysmal AF is associated with brain natriuretic peptide, troponin, left atrial dilatation, embolic strokes in multiple arterial territories, and myocardial infarction [7,8]. Factors associated with paroxysmal AF may help the clinician select ischemic stroke patients with possible paroxysmal AF for anticoagulation. Several scores have been constructed to determine the probability of underlying paroxysmal AF in ischemic stroke patients [9-11].
Little is known about left atrial pressure (LAP) in patients with ischemic stroke. Elevation in LAP may reduce the left atrial appendage peak early and late emptying velocities [12], possibly contributing to thrombus formation in the left atrial appendage. Increased LAP seems to be associated with AF [13], and recurrence of AF after ablation [14]. We hypothesized that increasing LAP is associated with AF. Left atrial dilatation is associated with AF. We aimed to explore the relation between left atrial volume (LAV) and AF in patients with ischemic stroke.
METHODS
All consecutive patients with acute ischemic stroke (the index stroke) admitted to the Center for neurovascular diseases, Department of Neurology, Haukeland University Hospital between January 2011 and December 2017 were prospectively registered in a database (The Bergen NORSTROKE Registry). Ischemic stroke was defined in accordance with the Baltimore-Washington Cooperative Young Stroke Study Criteria comprising neurological deficits lasting more than 24 hours because of ischemic lesions or transient ischemic attacks where CT or MRI showed infarctions related to the clinical findings [15].
A subgroup of the patients were referred to an out clinic cardiologist for transthoracic echocardiography. No predetermined criteria for referral were specified. Referral was based on the judgment by the clinician in charge of the patients during the hospital stay (mostly because of long waiting time for in-hospital echocardiography).
The transthoracic echocardiography (Philips i33 or GE 95) included measurement of LAV, LAP and ejection fraction (EF). Pathological values were defined as LAV≥53 mL/m² for females, LAV≥59 mL/m² for males and LAP≥13 mmHg for both sexes.
Variables in the Norstroke Registry included The National Institute of Health Stroke Scale (NIHSS) score on admission. Risk factors were defined according to a predefined protocol: angina pectoris, myocardial infarction, peripheral atherosclerosis, hypertension, diabetes mellitus, heart disease (including cardiomyopathy, heart failure), aortic valve replacement (both mechanical and biological valves) and smoking. Current smoking was defined as smoking at least one cigarette per day. Diabetes mellitus was considered present if the patient was on glucose-lowering diet or medication. Hypertension, angina pectoris, myocardial infarction and peripheral artery disease were considered present if diagnosed by a physician any time before stroke onset. AF needed ECG confirmation.
Investigations included cerebral MRI and or CT, Holter monitoring, ECG, and carotid Duplex sonography. Blood analyses including troponin were performed on admission. Isolated acute ischemic lesions on MRI were defined as lacunar infarctions if <1.5 cm and located subcortical or in the brainstem. All other acute ischemic lesions were defined as non-lacunar infarctions. Acute ischemic lesions in more than one arterial territory (left or right anterior circulation or posterior circulation) were registered. Old infarctions were registered.
The study was approved by the local ethics committee (REK Vest).
STATISTICS
Chi-square and students t-test analyses were performed when appropriate. Pearson’s correlation analyses were performed between LAV or LAP and different factors. Stepwise backward linear regression analyses with ULA or ULP as dependent variable and significant findings in (Table 2) were performed. Sex and age were kept in all models. Separate lower smoother curves analyses were performed between LAV and both old cerebral infarction on MRI and atrial fibrillation. STATA 15.0 (Statacorp 4905 Lake way Drive, College Station, Texas 77845 USA) was used for analyses.
RESULTS
In total, 2573 patients with ischemic stroke were admitted during the study period. Among these 138 patients were referred to the out clinic cardiologist. (Table 1)
|
Table 1: Characteristics of ischemic stroke patients referred or not to out clinic cardiologist. |
||||
|
S.no |
|
Referred patients |
Not referred patients |
P |
|
N (%) |
N (%) |
|||
|
1 |
Male |
83 (60) |
1461 (57) |
0.44 |
|
2 |
Female |
55 (40) |
11 (43) |
|
|
3 |
Age mean (standard deviation) |
69 (9.8) |
71 (15.5) |
0.16 |
|
4 |
Hypertension |
72 (52) |
1381 (54) |
0.72 |
|
5 |
Diabetes mellitus |
19 (14) |
414 (16) |
0.47 |
|
6 |
Atrial fibrillation |
23 (17) |
757 (29) |
0.001 |
|
7 |
Prior atrial fibrillation |
9 (7) |
486 (19) |
<.001 |
|
8 |
New atrial fibrillation |
14 (10) |
271 (11) |
0.52 |
|
9 |
Smoking |
46 (34) |
611 (25) |
0.03 |
|
10 |
Prior cerebral infarction |
8 (6) |
263 (10) |
0.11 |
|
11 |
Prior myocardial infarction |
14 (10) |
352 (14) |
0.23 |
|
12 |
Angina pectoris |
9 (7) |
283 (11) |
0.1 |
|
13 |
Peripheral atherosclerosis |
4 (3) |
160 (6) |
0.11 |
|
14 |
TOAST |
|
|
0.06 |
|
15 |
Atherosclerosis |
9 (7) |
229 (9) |
|
|
16 |
Cardiac embolism |
35 (26) |
831 (33) |
|
|
17 |
Small vessel disease |
23 (17) |
297 (12) |
|
|
18 |
Other determined cause |
2 (2) |
101 (4) |
|
|
19 |
Unknown cause |
67 (49) |
1096 (67) |
|
|
20 |
MRS 0-2 |
127 (92) |
1358 (53) |
<.001 |
|
21 |
MRS 3-5 |
11 (8) |
1215 (47) |
|
|
22 |
Pathological troponin |
27 (18) |
1024 (46) |
<.001 |
|
23 |
Non-lacunare infarction on MRI |
95 (70) |
1618 (75) |
0.23 |
|
24 |
Lacunar infarction on MRI |
40 (30) |
539 (25) |
0.23 |
|
25 |
Old infarction |
42 (30) |
897 (40) |
0.03 |
|
26 |
MACI |
7 (5) |
274 (13) |
0.01 |
|
TOAST: Trial of Org 10172 in Acute Stroke Treatment classification; MRS: Modified Rankin scale score; MACI: Acute Infarctions in Multiple Arterial Territories |
||||
shows demographics of the patients referred compared to the patients not referred to the out clinic cardiologist. The most important differences were that AF was less frequent and the stroke less severe in the referred patients. Otherwise, there were only minor differences.
Patients referred to out clinic cardiologist
Mean LAV was 26.4 mL/m² (standard derivation (SD) 13.2) and mean LAP was 15.0 mmHg (SD 4.3). Three (13%) patients with documented AF were treated with anticoagulants prior to the stroke.
Table 2
|
Table 2: Correlation between left atrial volume, left atrial pressure and different factors. |
|||||
|
S.no |
|
Left atrial volume |
Left atrial pressure |
||
|
|
P |
|
P |
||
|
1 |
Male versus female |
0.14 |
0.07 |
0.09 |
0.25 |
|
2 |
Age |
0.14 |
0.07 |
0.2 |
0.01 |
|
3 |
Hypertension |
0.23 |
0.003 |
0.35 |
<.001 |
|
4 |
Diabetes mellitus |
0.18 |
0.02 |
0.14 |
0.08 |
|
5 |
Smoking |
-0.1 |
0.21 |
-0.05 |
0.5 |
|
6 |
Prior cerebral infarction |
0.08 |
0.33 |
0.14 |
0.1 |
|
7 |
Prior myocardial infarction |
0.12 |
0.12 |
0.14 |
0.09 |
|
8 |
Angina pectoris |
0.01 |
0.9 |
0.1 |
0.23 |
|
9 |
Peripheral atherosclerosis |
-0.04 |
0.6 |
0.01 |
0.94 |
|
10 |
Atrial fibrillation |
0.44 |
<.001 |
0.16 |
0.06 |
|
11 |
Chronic atrial fibrillation |
0.36 |
<.001 |
-0.09 |
0.28 |
|
12 |
Paroxysmal atrial fibrillation |
0.18 |
0.02 |
0.13 |
0.11 |
|
13 |
New atrial fibrillation |
0.33 |
<.001 |
0.2 |
0.03 |
|
14 |
Left atrial pressure |
0.44 |
<.001 |
- |
- |
|
15 |
Left atrial volume |
- |
- |
0.44 |
<.001 |
|
16 |
Ejection fraction |
-0.04 |
0.62 |
0.13 |
0.12 |
|
17 |
Old infarction on MRI |
0.04 |
0.6 |
-0.04 |
0.66 |
shows correlation analyses between LAP and different factors. Increasing LAP correlated with increasing age, hypertension, new atrial fibrillation and LAV. Linear regression with LAP as dependent variable showed that increasing LAP was independently associated with female sex (beta=0.22, P=0.003), hypertension (beta=0.31, P<0.001) and increasing LAV (beta=0.36, P<0.001), R-squared=0.33. LAP was not independently associate with AF when replacing LAV with AF in the above linear regression model (beta=0.1, P=0.23).
Table 2 shows correlation analyses between LAV and different factors. Increasing LAV correlated with hypertension, diabetes mellitus, AF and LAP (all P<0.05). Linear regression with LAV as dependent variable showed that increasing LAV was independently associated with male sex (beta=0.21, P=0.005), AF (beta=0.38, P<0.001) and increasing LAP (beta=0.40, P<.001), R-squared=0.37.
Figure 1
Figure 1 Lowess smoother curve showing relation between atrial fibrillation or not and left atrial volume.
shows a U-shaped relation between LAV and the presence or not of AF. The correlation (r) between AF versus sinus rhythm and LAV<19 was r=0.33, P=0.03. The correlation between AF versus sinus rhythm and LAV ≥19 was r=0.45, P<0.001.
Figure 2
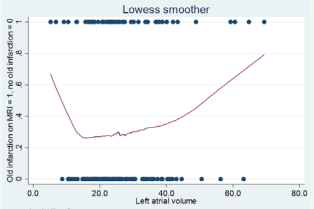
Figure 2 Lowess smoother curve showing relation between old infarction or not on MRI and left atrial volume.
shows a U-shaped relation between LAV and the presence or not of old infarctions on MRI. The correlation (r) between old infarctions and LAV17 was r=0.18, P=0.05.
DISCUSSION
We found no correlation between left atrial pressure and atrial fibrillation in total. Our hypothesis was thus falsified. This is contrary to a recent study that found an association between increased left atrial pressure and recurrent atrial fibrillation after cardio version [13]. Different patient populations may explain the discrepancy. However, there was a weak association between increasing left atrial pressure and new atrial fibrillation in our patients.
We found that increasing left atrial pressure was independently associated with increasing age, prior hypertension and increasing left atrial volume.
Overall, we found that increasing left atrial volume correlated with documented atrial fibrillation. Several other studies have found the same association between left atrial volume and atrial fibrillation both in patients with and without cerebral infarction [16,17]. On multivariate analyses we found that left atrial volume was independently associated with both atrial fibrillation and left atrial pressure.
Based on the correlation between left atrial volume and left atrial pressure it is surprising that left atrial pressure did not correlate with atrial fibrillation. This suggests that there is a subgroup of patients with both high left atrial pressure and large left atrial volume not prone to atrial fibrillation. A larger study is needed to explore this possibility further.
However, lower smoother curves nuanced the relation between left atrial volume and atrial fibrillation reported above. It showed a U-shaped relation between the presence of atrial fibrillation or not and left atrial volume. Both arms of the U-curve showed significant albeit opposite relation between atrial fibrillation and left atrial volume. This suggests that both in the low and the high range of left atrial volume, there is an association with atrial fibrillation in ischemic stroke patients.
Lower smoother curves also showed at U-shaped relation between old infarction or not on MRI and left atrial volume. Both arms of the U-curve showed significant albeit opposite relation between old infarction and left atrial volume. A possible explanation is that untreated atrial fibrillation is associated with old infarctions. A postdoc analysis showed this to be the case among patients with low left atrial volume, but not with high left atrial volume.
The most important limitation of this study is the relatively low number of patients, and this study should be considered as a pilot study.
In conclusion, we did not find an association between left atrial pressure and atrial fibrillation even though left atrial pressure correlated with left atrial volume. However, we found U-shaped relations between left atrial volume and both atrial fibrillation and old cerebral infarction on MRI. (Figures: 1,2) (Tables: 1,2).








































































































































