Repetitive Transcranial Magnetic Stimulation in Aphasia and Communication Impairment in Post-Stroke: Systematic Review of Literature
- 1. Group Neurovascular Sciences, Cardiovascular Foundation of Colombia, Colombia
Abstract
Background: Post-stroke aphasia is one of the major disabilities and a risk factor for other complications, is also associated with increased mortality, depression and impairments in communication. Repetitive Transcranial Magnetic Stimulation (rTMS) is an alternative. We propose the review of literature published until now of rTMS.
Methods: We included articles published on PubMed and EMBASE, we included studies if were: randomized controlled blinded clinical trials, meta-analyses or crossover designs of rTMS alone or with speech therapy or any other therapy tested with rTMS. One author included these if: report baseline assessment and one or more posterior assessments; overall results and subtest report; statistically significant results showed by p value. We also assessed the risk of bias of each article.
Main results: We included 15 articles, the average age was62.42 ± 4.04; most used low frequency stimulation (1 Hz), most used a coil of 70mm, 77% applied 10 sessions, most did a Speech language therapy. From overall test result:66% studies improved Aphasia; room subtest results: only 1 study improved listening, speaking and reading; 2 studies improved writing, 80% studies improved naming skills, Two studies improved writing and description, 3 studies improved compression and expression, 46% improved repetition.47% of studies used Boston battery, 33% used Aachen Aphasia Test.
Conclusions: The articles included showed improvement of scale results for aphasia in post-stroke patients, mainly due to improvement in naming and repetition skills.
Keywords
• Aphasia
• Post-stroke
• Repetitive transcranial magnetic stimulation
Citation
Mendoza JA, Silva FA, Pachón MY, Rueda LC, Lopez Romero LA, Pérez M (2016) Repetitive Transcranial Magnetic Stimulation in Aphasia and Communication Impairment in Post-Stroke: Systematic Review of Literature. J Neurol Transl Neurosci 4(3): 1070.
ABBREVIATIONS
rTMS: Repetitive Transcranial Magnetic Stimulation; SLT: Speech Language Therapy; BDAE: Boston Diagnostic Aphasia Examination; AAT: Aachen Aphasia Test; BNT: Boston Naming Test; CCAT: Criteria Cognitive Aptitude Test; KWAB: Western Aphasia Battery; CPNT: Computerized Picture Naming Test; ASRS: Aphasia Severity Rating Scale; ANELT: Amsterdam-Nijmegen Everyday Language Test; HSS: High Sensation Seeking Test; HRQL: Health-Related Quality of Life; SAQOL: Aphasia Quality of Life Scale-39; PRO: Patient Reported Outcomes
INTRODUCTION
Aphasia is one of the major disabilities and a risk factor for other complications in post-stroke patients. The worldwide incidence of stroke is about 217 per 100.000 person years and its prevalence, 715 per 100.000.In high income countries, the aphasia incidence in first-ever ischemic stroke amounted to 43 per 100.000 inhabitants [1,2]. Unlike any other condition, disabilities due to stroke are multiple, affecting: walking, speech, balance; co-ordination, vision, spatial awareness, swallowing; bladder and bowel control [3]. Aphasia as sequel of stroke is associated with increased mortality (36% vs. 16%), depression (70% of aphasic at 3 months) and impairments in communication [4-7]. Social participation for people with aphasia is affected; they communicate with fewer friends, have poor social networks and negative impacts on-quality of life [8-10].
Aphasia is an acquired disorder that affects the ability to use and/or understand language; it involves various components: meaning (semantics) sounds (phonology) and structure (syntax/morphology) [11]. Aphasia has many classifications and these can be used to guide treatment selection. The classic classifications are based on: fluency, language understanding and preserved repeated speech and syndromes (global, Broca, isolated, transcortical motor; Wernicke, transcortical sensory, conduction and anomic), but remain based on elementary clinical characteristics of dichotomies (motor-sensory, expressive-receptive, fluent or nonfluent) [12]. In recent years, researches showed more participation of sub cortical gray matter structures in brain that could explain language impairment in brain areas different of the lobes in post-stroke patients. Pedersen et al, found one year after stroke: global 7%, Broca´s 13%, transcortical motor 1%, wernicke´s 5%, transcortical, conduction 6% and anomic 29% [13]. Language deficits assessment is an important research area, clinical practice and rehabilitation [14]; standardised batteries for aphasia gathered information from different language subtest that assesses: spontaneous speech, comprehension, repetition and naming [14]. This is important to focus aphasia therapy in specific language impairment.
Post-stroke aphasia treatment is a challenge that includes: deal with own aphasia and its associated comorbidities as depression, deglutition dysfunction and cognitive deficit. Speech-language therapy is considered the mainstay treatment of aphasia. Medications (piracetam, bromocriptine, dexamfetamine, donezepil,) are also considered, but there is a lack of consensus about their effectiveness and the best time to start with speech language therapy (SLT). In this scenario, repetitive Transcranial Magnetic Stimulation (rTMS) is an alternative for aphasia treatment; however, it remains as an experimental alternative due to lack of epidemiological evidence about its general effectiveness or subtest efficacy, and how it is clinically significant for daily communication. We proposed to carry out a systematic review of literature of studies published for rTMS, with the objective of finding evidence of efficacy and sub-test results that could be useful for individualized patient treatment.
MATERIALS AND METHODS
We performed the search on May 2016, we included articles published until May 2016 on PubMed and EMBASE, using the following terms: (transcranial magnetic stimulation or rTMS) AND (aphasia) AND (Stroke OR post stroke) (clinical trial or meta-analysis). We did not restrict for age or other study characteristics. The studies were selected if they met the inclusion and exclusion criteria: clinical trials, meta-analyses or crossover designs of transcranial magnetic stimulation alone or with speech therapy or any other therapy tested with rTMS. Figure (1),
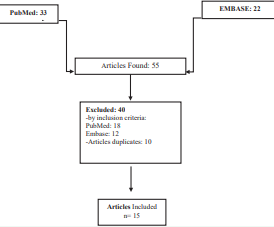
Figure 1 Studies included in the review.
shows the flow chart of the search and the included and excluded studies. The results were filtered to discard duplicate items.
One of the authors selected the articles by title; after that, we reviewed the articles pre-selected to include them in a first step; from this first step, we excluded 10 articles due to duplicate. Other author reviewed the rest of the studies included, and chose those articles considered relevant, and finally took off others that did not meet all of the inclusion criteria. Finally, the articles were put in a spreadsheet designed to describe, classify and check results from each study; all Results of each study were put there to describe general characteristics and the scales used. We included study data, if they were reported as: baseline assessment and one or two posterior assessments; overall results and subtest report; statistically significant results showed by p value. We also did a risk of bias using the PEDro scale of clinical trials: the scale uses different criteria for overall evaluation. Results of risk bias were represented using REVMAN software of Cochrane systematic review Figures (2,3).
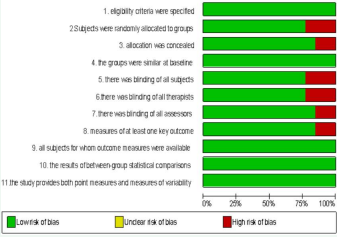
Figure 2 peDro scale risk of bias by criterion: these figures summarize Risks of bias, presented as percentage across all included studies.

Figure 3 Risk of bias by study and criterion: Review authors´ judgements about each risk of bias item for each.
RESULTS
From the studies retrieved and included in the systematic review, (13 clinical trials and 2 meta-analyses) we described the general characteristics of each study. The number of subjects included ranged from 8 to 56. The types of aphasia included were fluent and no-fluent, the average age was 62.42 ± 4.04, the mean time post-stroke included was 20.38 ± 20months, and all studies stimulated contra lateral area. Most of studies did speech-language therapy additional to rTMS. All the studies included sham therapy as comparator and used an aphasia scale to measure outcomes.
We also described the therapeutic application of repetitive transcranial magnetic stimulation (Table 1,2),
|
Table 1: Characteristics of included studies. |
|||||||||
|
Study |
n |
Aphasia Type |
Mean age |
Mean time post-stroke (months) |
Study design |
Site of stimulation |
Experimental |
Control |
Outcome measurement |
|
Hara et al [15]. |
50 |
FA / NFA |
60.3 |
55.9±38.0 |
CT |
CL-IL* |
rTMS |
— |
SLTA |
|
Rubi-fessen et al [16]. |
30 |
FA/ NFA |
NI |
NI |
CT |
CL |
rTMS |
Sham |
AAT, FIM, Anelt A-scale |
|
Li Y et al [19]. |
132 |
NFA/ ALL TYPE |
NA |
— |
MA |
CL |
rTMS |
Sham |
BNT, BADE, CCAT, AAT |
|
Otal et al [42]. |
183 |
ALL TYPE |
NI |
NI |
MA |
CL |
rTMS |
Sham |
AAT, BDAE, CPNT PNT |
|
Yoon et al [21]. |
20 |
NFA |
Active60.46 |
6,80 ± 2,39 5,20 ± 2,67 |
CT |
CL |
rTMS |
Sham |
K-WAB |
|
Wang et al [20]. |
45 |
FA/NFA |
TMSsyn 61.3 |
TMSsyn16.8±6.4 TMSsub15.7±8.5 |
|
CL |
Syn(rTMS + naming training) together/ Sub(rTMS + naming training)separate |
Sham + sync |
CCAT |
|
Tsai et al [17]. |
56 |
NFA |
Experimental 62.3 Sham 62.8 |
Experimental 17.8 ± 7.2 |
CT |
CL |
rTMS |
Sham |
CCAT |
|
Khedr et al [18]. |
30 |
NFA |
57,3 |
Experimental: 2 |
CT |
CL |
rTMS |
Sham |
HSSASRS |
|
Barwood et al [22]. |
12 |
ALL TYPE |
Active 60.8 |
Active 18.46 |
CT |
CL |
rTMS |
Sham |
BNT – BDAE
|
|
Seniow et al [23]. |
40 |
ALL TYPE |
60.7 |
Active:1.10 |
CT |
CL |
rTMS |
Sham |
BDAE |
|
Thiel et al [44]. |
30 |
FA/ NFA |
Active: 71.2 |
Active: 1.23 |
CT |
CL |
rTMS |
Sham |
AAT |
|
Waldowsi et al [45]. |
26 |
NFA |
Active: 62.31 |
NI |
CT |
CL |
rTMS |
Sham |
CPNT ASRS BDAE |
|
Szaflarski et al [46]. |
8 |
NFA |
54.4 |
63.3 |
CT |
IL |
rTMS |
Sham |
SFT, COWAT BNT, BDAE |
|
Weiduschat et al [47]. |
10 |
NFA |
66.6 |
Active: 45.2 |
CT |
CL |
rTMS |
Sham |
AAT |
|
Barwood et al [43]. |
12 |
NFA |
Active 60.8 |
18.36 |
CT |
CL |
rTMS |
Sham |
BNT – BDAE |
|
Abbreviations: FA: Fluent Aphasia; NFA: Nonfluent Aphasia; CT: Clinical Trial; CL: Contralesional, IL: Ipsilesional; SLTA: the Standard Language Test of Aphasia; AAT: Aachen Aphasia Test; FIM: The Functional Independence Measure; Anelt A-scale: Amsterdam-Nijmegen Everyday Language Test; BNT: Boston Naming Test; BDAE: Boston Diagnostic Aphasia Examination; CCAT: Criteria Cognitive Aptitude Test; AAT: Aachen Aphasia Test; PNT: Philadelphia Naming Test; K-WAB: Korean-version of the Western Aphasia Battery; ASRS: Aphasia Severity Rating Scale; HSS: High Sensation Seeking Test; CPNT: Computerized Picture Naming Test; SFT: Semantic Fluency Test; COWAT: Controlled Oral Word Association Test; Syn: Synchronous; Sub: Subsequent, rTMS: Repetitive Transcranial Magnetic Stimulation; NA: No Apply; NI: No information. *Ipsilesional or contralesional depend on results by functional magnetic resonance imaging. |
|||||||||
|
Table 2: Protocols for repetitive transcranial magnetic stimulation. |
||||||||||
|
Study |
1 Hz |
Coil |
Stimulator |
Frequency |
# Sessions |
Length |
Intensity* |
Localization |
sham localization |
SLT After rMTS |
|
Hara et al [15]. |
YES |
70-mm |
MagPro R30 |
Every day |
10 |
40 min |
90% |
IFG-STG (left or right) |
— |
YES |
|
Rubi-fessen et al [16]. |
YES |
70-mm |
Magstim Rapid |
NI |
15 |
20 min |
90% |
PT-IFG (right) |
Vertex |
YES |
|
Yoon et al [21]. |
YES |
NI |
MagPro® |
Every day |
20 |
20 min |
90% |
IFG (right) |
— |
YES |
|
Wang et al [20]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
20 min |
90% |
PT (right) |
same-sham-coil |
YES |
|
Tsai et al [17]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
10min |
90% |
PT IFG(right) |
Same less 5% |
YES |
|
Khedr et al [18]. |
YES |
90-mm |
Magstim Rapid |
Every day |
10 |
NI |
80% |
PT– Pop (right) |
Same no-contact |
YES |
|
Barwood et al [22]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
20 min |
90 % |
PT– Pop (right) |
same no contact |
No |
|
Seniow et al [23]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
20 min |
90% |
PT (right) |
same-sham coil |
YES |
|
Thiel et al [44]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
20 min |
90% |
PT- PIG (right) |
Vertex |
YES |
|
Waldowski et al [45]. |
YES |
70-mm |
Magstim Rapid |
Everyday |
15 |
30 min |
90% |
PT - POp (right) |
same- Sham coil |
YES |
|
Szaflarski et al [46]. |
NO |
70-mm |
Magstim Rapid2 |
Every day |
10 |
NI |
80% |
left Broca’s |
— |
No |
|
Weiduschat et al [47]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
20 min |
90% |
IFG (right) |
Vertex |
YES |
|
Barwood et al [43]. |
YES |
70-mm |
Magstim Rapid |
Every day |
10 |
20 min |
90% |
PT(right) |
Same-Sham coil |
No |
|
Abbreviations:*: % of each individual patient's motor threshold intensity; IFG: Inferior Frontal Gyrus; STG: Superior Temporal Gyrus; PT: Pars Triangularis; POp: Pars Operculus; PIG: Posterior Inferior Gyrus; NI: No Information. |
||||||||||
most of studies included, used low frequency stimulation (1 Hz) only Szaflarski study used 50 Hz every 200 milliseconds in two second trains, repeated every 10 seconds over 200 seconds for a total of 600 pulses. Related to the coil, most of studies used a 70mm coil; Khedr et al used a 90mm coil and one study did not report it. 11 of 13 (without meta-analyses) used the Magstim® stimulator; 2 studies reported use of magPro® stimulator. Related to the number of sessions, 77% (10/13) reported10 stimulations every day (except weekends), two studies 15 days, and one study 20 sessions. Authors also reported length of each stimulation, 73% of studies stimulate 20 minutes in average; Hara et al reported 40 minutes of stimulation each session, Waldowski et al., stimulate 30 minutes, two studies did not report time of each stimulation session. Of stimulation sites, all of them stimulate the contra lateral area Broca’s area. Sham strategy for stimulation were some: stimulation in vertex, same place but a sham coil; same localization but less than 5% of stimulation intensity and same site but without skull contact. All studies performed a SLT following the stimulation, the type, length and intensity depended of each investigator.
Results of all studies included are shown in Table (3);
|
Table 3: Effect of repetitive transcranial magneticstimulation in the treatment of aphasia post- stroke according to the literature review. |
|||||||||||
|
Study |
Outcome measurement |
? Measure overall P value |
Listening |
Speaking |
Reading |
Writing |
Naming |
Compression |
Expression |
Repetition |
Description |
|
Hara et al. [15] |
SLTA |
CL df:5.9p<0.01 IL df:6.6p<0.01 |
CL df:0.9 p>0.05 IL df:1.6 p<0.05 |
CL df:1.7 p<0.01 IL df:2.8 p<0.01 |
CL df:0.7 p<0.01 IL df:0.5 p>0.05 |
CL df:1.4 p<0.05 IL df:1.6 p<0.01 |
No |
No |
No |
No |
No |
|
Rubi-fessen et al. [16] |
AAT FIM Anelt A |
Real df:6.20 p<.001 Sham df:3.26 p<.001 |
No
|
No |
No |
Real df:4.7 p<.001 Sham df:1.87 p=0.007 |
Real df:6.47 p<.001 Sham df:2.07 p=0.057 |
Real df:4.40 p<.001 Sham df:1.60 p=0.045 |
Rea dfl:0.53p=0.013 Sham df:0.27 p=0.052 |
Real df:2.93 p=0.005 Sham df:2.34p<.001 |
No |
|
Li Y et al. [19] |
BNT, BADE, CCAT, AAT |
No |
No |
No |
No |
No |
SMD:0.51(0.16–0.86) |
SMD:0.31 (–0.14–0.75) |
No |
SMD: 0.31 (–0.04–0.65) |
No |
|
Otal et al. [42] |
AAT, BDAE, CPNT PNT |
No |
No |
No |
No |
No |
SMD: 0.51 p=0.0003 |
No |
No |
No |
No |
|
Yoon et al. [21] |
K-WAB |
No |
No
|
No |
No |
No |
Real, df=17 p=0.039 |
No |
No |
Real df=12.2 p=0.042 |
No |
|
Wang et al. [20] |
CCAT |
SYN= Post1 df: 1.6 p<0.05 |
No |
No |
No |
No |
SYN= Post1 df:20% p<0.05 Post2 df:26.1% p<0.05 |
No |
SYN= Post1df:2 p<0.05
|
No |
SYN= Post1 df:1.3 p<0.05
|
|
Tsai et al. [17] |
CCAT |
Post1 df:1.2 p<0.001 Post2 df:1.7 p<0.05 |
No
|
No |
No |
No |
Post1 df:1.2 p<0.05 |
No |
Post1 df:1.4 p<0.01 Post2 df:1.5 p<0.05 |
Post1df: 0.9 p<0.05 Post2 df: 1 p<0.05 |
Post1df:0.8 p<0.05 Post2 df:1.4 p<0.05 |
|
Khedr et al. [18] |
HSS ASRS |
df=1.6 p= 0.004 |
No
|
No |
No |
No |
df=2.1 p=0.01 |
df=1.7 p=0.04 |
No |
df=1.5 p=0.002 |
No |
|
Barwood et al. [22] |
BNT BDAE |
df=4.9 p< 0.05 |
No |
No |
No |
No |
df=3.9 p< 0.05 |
No |
No |
df = 4.01 p< 0.05 |
No |
|
Seniow et al. [23] |
BDAE |
No |
No |
No |
No |
No |
Anterior-follow up Real df=64.9 Sham df=40.4 p=0.03 |
No |
No |
Posterior-post Real df=5.2 Sham df=2.1 p=0.02 Severe-follow up Real df=7.7 Sham df=3.5 p=0.016 |
No |
|
Thiel et al. [44] |
AAT |
df=16.2 p=0.003 |
No |
No |
No |
No |
df=5.2 p=0.002 |
No |
No |
No |
No |
|
Waldowski et al. [45] |
CPNT ASRS BDAE |
No |
No |
No |
No |
No |
df=13 p=0.016 |
No |
No |
No |
No |
|
Szaflarski et al. [46] |
SFT COWAT BNT BDAE |
No |
No |
No |
No |
No |
No |
No |
No |
No |
No |
|
Weiduschat et al. [47] |
AAT |
df=19.8 p=0.002 |
No |
No |
No |
No |
No |
No |
No |
No |
No |
|
Barwood et al. [43] |
BNT BDAE |
df=10 p< 0.01 |
No |
No |
No |
No |
df=10 p< 0.01 |
No |
No |
No |
No |
|
Abbreviations: CL: Contralateral; IL: Ipsilateral; df: Difference From Baseline To Posterior Assessment; No: no Statistical Significance; SMD: Standard Mean Difference; BNT: Boston Naming Test; SLTA: The Standard Language Test of Aphasia; AAT: Aachen Aphasia Test; FIM: The Functional Independence Measure; Anelt A-scale Amsterdam-Nijmegen Everyday Language Test; BNT: Boston Naming Test; BDAE: Boston Diagnostic Aphasia Examination; CCAT: Criteria Cognitive Aptitude Test; AAT: Aachen Aphasia Test; PNT: Philadelphia Naming Test; K-WAB: Korean-version of the Western Aphasia Battery; ASRS: Aphasia Severity Rating Scale; HSS: High Sensation Seeking Test; CPNT: Computerized Picture Naming Test; SFT: Semantic Fluency Test; COWAT: Controlled Oral Word Association Test |
|||||||||||
by test results, 60% (9/15) studies showed overall improvement of aphasia and 6 did not. Results were also described for sub-scale tests in aphasia if they were described and had statistical significance. We included the following: listening, speaking, reading, writing, naming, compression, expression; repetition and description assessments. For listening, speaking and reading; only 1 study showed improvement [15] and 2 studies reported improvement in writing skills [15,16]. 80% (12/15) studies showed improvement in naming skills. Two studies reported improvement for writing and description [15,16] and [17,18], respectively, 3 studies showed improvement for compression and expression [16,18-19] and [16-17,20] respectively), 47% (7/15) reported improvement for repetition [16-19, 21-23].
Related to scales used on this studies, 47% (7/15) of studies used or reported results with Boston Diagnostic Aphasia Examination (BDAE), 33% of studies used Aachen Aphasia Test (AAT) or Boston Naming Test (BNT), 20% (3/15) usedCriteria Cognitive Aptitude Test(CCAT) and 13% (2/15) used Korean-version of the Western Aphasia Battery(KWAB), Computerized Picture Naming Test (CPNT) and Aphasia Severity Rating Scale (ASRS).From overall test results the scales used were BDAE 30% (3/10), CCAT and AAT 20% (2/10), and 10% (1/10) with Amsterdam-Nijmegen Everyday Language Test (ANELT), SLTA and High Sensation Seeking Test(HSS). Listening, speaking and reading skills only improve in Hara´s et al., study; using SLAT scale, writing results improved in Hara et al and Rubi-fessen et al., using SLTA and Anelt A scale.
DISCUSSION
Most of the studies reported improvement of aphasia post-stroke with repetitive transcranial magnetic stimulation. There were improvements in results of scales from baseline score to post-stimulation assessment score, which could be due to rTMS alone or synchronic naming training and complementary to speech therapy in aphasia post-stroke. Aphasia is one of the most challenging consequences of stroke. In this scenario, repetitive transcranial magnetic stimulation emerged as a promising alternative to improve communication skills in patients that until now only speech therapy has showed to be effective to maximize the natural recovery after stroke [24].
We identified a lack of standardization of the rTMS´s protocols, which can lead to heterogeneity in the effect of the therapy or the dilution of it; in addition, the limited size of sample of studies contributes to the heterogeneity in results. We also identified lack of homogenous uses of scales in the studies included, however; it is because the origin of each one, and the use of specific designed scale by language. Boston battery is a generic scale for aphasia, but CCAT and Anelt A scale were designed for specific languages (Chinese and Netherlands). Boston scale was used more frequently than others in this study; some authors have reported its limitations (poor psychometric properties, lack of standardization and inadequate norms), but it is still the most used test by neuropsychologists [25,26]. From subtest results the body of evidence shows improvement on naming results (12 studies), following by repetition (7 studies) and less by compression, expression and description. There is a lack of evidence by listening, speaking and reading (1 study); the split out of studies from sub-test results is useful to describe strength and weakness of rTMS, and possible language skills to improve with SLT. The overall test results are in the same line of other authors; Wong et al, in their systematic review of studies on the effectiveness of rTMS, found improvement in post-stroke aphasia with or without conventional rehabilitation [27], Ren et al., in their meta-analysis also reported a positive effect on language recovery in patients with post-stroke aphasia [28], but Gadenz et al, in a systematic review of randomized controlled trials, reported uncertain benefits in neurologic disorders related to communication; the authors analysed 3 studies about dysphagia 1 about dysarthria in Parkinson´s disease and 1 about linguistic deficits in Alzheimer´s disease, which could underestimate rTMS effects [29].
Most of studies included, stimulated the contralateral Broca´s area (only Szaflarski et al., stimulated ipsilateral using excitatory intermittent theta-burst stimulation) this on base of theories about post-stroke aphasia recover, which proposed that brain may use two strategies to recover speech-relevant regions (the structural repair of primarily speech-relevant regions or the activation of compensatory areas) [30]. Karbe et al., found that brain recruit right-hemispheric regions for speech processing when the left-hemispheric centers were permanently impaired as compensatory mechanisms. On this way, Thiel et al., analysed the potential to compensate damage of left-hemisphere by other brain zones using functional neuroimaging and transcranial magnetic stimulation in patients with brain tumors as a model; they found that time is factor which determines successful integration of right-brain [31], But post-stroke recovery is also based on plastic changes in the central nervous system and activation of perilesional zones and reactivation of impaired zones and this could be done by rTMS [32]. The use of rTMS in contralesional brain zones is explained by the inhibition of over-activation in the Broca´s homologous to the lesion that lead to inhibition on perilesional brain zones [33]. One of the first works on rTMS in aphasia post-stroke was done by Naeser et al., they used a 1 Hz 1200 pulses at 90% of motor threshold in anterior part of Broca´s homologue, this area was identified on each patient by MRI scan; they reported improve in picture naming at 2 months post-rTMS [34], and this due to improve in inter hemispheric modulation of semantic procession for picture naming [32]; author remarks the possibly modulating the distributed bi-hemispheric language network for naming; however, is possible that patient´s improved naming scores are related to their having learned the test items, this is one of the issues in neuropsychological test re-test assessment.
From our study results, some questions emerged; first, is the test scores the best way to measure aphasia? Second, post-stroke aphasia population have to be measured also as Health-related quality of life? Changing in naming outcomes is enough to improve quality of life? Third, what are the most useful marginal improvements to communicate?
First, there is a lack of consensus about the most suitable aphasia scale, but the use of Boston test is widely accepted for clinical trials [14]. Second, the recommendation for future clinical trials is the inclusion of Health-related quality of life (HRQL) and also the inclusion of people with aphasia and report separately to determine the main predictors of HRQL [35]. The improvements on some sub-test scores are insufficient to improve basic communication skills in real life. Third, it could be possible that marginal changes in speaking, listening, writing and reading made the real difference for patients. We considered that more investigations could be useful in this field.
As a weakness of this study we identify the heterogeneity of the scales used by the studies to measure outcomes in aphasic patients; compare studies results by scale changes from baseline results to post stimulation results, is a task for this kind of studies. Changes in mean were reported for the majority of studies included in the systematic review; we avoid measuring the magnitude of its median changes for results, our focus was reporting differences on scales results. As a weakness we also identified the differences in inclusion and exclusion criteria, following of patients included, time post-stroke, use of SLT and other coadjutants in stroke. The only one study included in the systematic review that showed improve on speaking, reading and writing skills, was the Hara et al., study, on it the SLTA scale was used for measure study outcomes-no other study used it. The SLTA was developed for classification of severity of aphasia of Japanese speakers in 1975; this test includes specific tasks and assessments considered complicated, but focused on daily communications skills [36]. There is a lack of studies published that included assessment only for this scale-different to the Japanese language-is impossible to assume that speaking, reading and writing skills improve in Japanese population with rTMS by this study along. Some authors propose an ecological approach aimed at restoring the patient ability to communicate in daily context, for this approach propose of new alternatives for help them is a validate alternative.
Wang et al., include synchronous verbal training during rTMS; they investigate the efficacy with a simultaneous picture-naming activity and demonstrated a superior language for verbal expression also [20]. This study showed change from baseline assessment to post 1 and post 2, for conversation and expression there were changes in all of groups, but exist doubt regard to changes from baseline assessment and evaluations until 90 days’ post-stroke with or without any intervention. Lazar et al., study, reported that patients improved by approximately 70% of the maximum potential recovery with language therapy; it was considered as standard care from this results, and alternatives of standard care that could be useful [37]. From this study, authors consider that coadjutants to speech therapy could be used and it could be added value to standard care (SLT) [24]. Thus many gap data come from these results, severely of aphasia, the time and the quality of speech therapy; this was other issues found in studies included in our study.
Hills in her study outline about targets of therapy and important aspects of recovery; she recalls for evaluations of speech articulation, reading writing and grammatical skills. The purpose of these results is improved communications assessments, and guide for toward communication enhancement programs and others (book clubs, social organizations) [24]. For an inclusion of a holistic outcome measure in post-stroke aphasic patients, the inclusion of different assessment close related to aphasia may be included as emotional distress, activity limitations, social factors, quality of life, cognitive function, and so on. From this perspective authors designed alternative measure tools, one of them is aphasia quality of life scale-39 (SAQOL), this stroke-specific scale incorporates patients´ views about their own health perception. Hilari et al., tested the feasibility and psychometric evaluation of this scale: 87% were able to complete the SAQOL, and derivate a version that identified 4 sub domains (physical, psychosocial, communication and energy) to Resume it [35]. Many other authors have evaluated quality of life in stroke survivors. The overall results are a decrease in QL that depends on demographic factors, comorbidity, disability and psychosocial factors [38,39]. Frances et al., measured outcomes related to quality of life in patients with post-stroke and aphasia. They used modified outcome measure included the stroke impact scale, the 36-item from medical outcomes, reintegration to normal living scale and activity card sort. They concluded with recommendation for future investigations and practitioners the inclusion of outcomes that assess self-reported quality of life, as an understanding of consequences of stroke [40]. There are movements towards self-reported outcomes, patient preferences and approaches of a holistic measure of results in clinical practice to focus new treatments or fitting current alternatives; from this view patient reported outcomes (PRO) could help to capture patient perspective -determine which health outcome are relevant from them [41-47].
CONCLUSION
Our study shows benefits measured by aphasia scales with rTMS; in overall and sub-test scores there were improve from baseline to post-evaluations, sub-test scores showed improve mainly in naming, and less for other sub-test; neither study reported improve in basic communication skills. Our results support the use of rTMS in patients with aphasia post-stroke; chronic and early. We considered that minimal improvements in patients with aphasia are relevant in a condition that is difficult and considered a task-due to the complexity of it. We also considered that more research has to be done focused on quality of life changes with rTMS as a holistic assessment from patient perspective and incorporate it in the health system if it shows benefits for daily life of patients with aphasia post-stroke.
ACKNOWLEDGMENT
This work was supported by a grant from the Science and Technology Institute. Initials: COLCIENCIAS. Colombia. Code Number: 5020-53-731809. Funding by COLCIENCIAS.








































































































































