Thrombolytic Use in Acute Ischemic Stroke Patients with Concurrent Infective Endocarditis: Emphasis on History, Physical Exam, Outcomes, and Review of Literature
- 1. CentraCare Neurosciences, CentraCare St. Cloud Hospital, USA
- 2. Department of Neurology, Texas Tech Health Science Center, USA
Abstract
Background: Intravenous (IV) alteplase is not recommended in acute ischemic stroke patients with concurrent infective endocarditis (AIS-IE) due to high rates of hemorrhagic conversion, death, and disability.
Methods: We report a case in which a patient received IV alteplase and was later diagnosed with possible infective endocarditis. Previously published literature regarding similar cases and articles on PubMed was also reviewed.
Case summary: A 36-year-old male patient with no history of hypertension, diabetes, a trial fibrillation, hyper lipidemia, or other classic risk factors for stroke presented to the emergency room (ER) with a sudden onset of left hemiparesis, numbness, and left facial droop. NIHSS at admission was 10. His blood pressure was 137/100 mmHg, temperature 98.1 F, respiratory rate 23, and pulse 105. His WBC count was 13.8 K/UL. Physical exam was negative for cardiac murmurs, fever, and vascular stigmata associated with IE. Patient had a history of migraine headaches, but denied headache at the time of presentation. Per the patient’s history, he had suffered a low-grade fever for the past few days which he had attributed to West Nile fever. Patient’s history was negative for seizures and stroke. Initial computed tomography (CT) of the head was negative for any bleeding. Patient was assessed and administered IV alteplase (0.9mg/kg) within the American Heart Association’s (AHA) time frame recommendations (67 minutes from onset of symptoms). The next day the patient’s hemiparesis resolved with NIHSS of 0. Considering his reported history of low-grade fevers in conjunction with multiple small infarcts revealed on his MRI, infective endocarditis (IE) was investigated as a possible cause of the embolic stroke. Blood cultures were positive for gram positive cocci in clusters. Follow-up imaging revealed asymptomatic hemorrhagic conversion. IV Penicillin G was initiated and prescribed for six weeks.
Conclusion: While evaluating young AIS patients for IV alteplase, who present without classic stroke risk factors, a thorough history and clinical exam with a specific focus on the signs and symptoms of IE, systemic inflammatory response syndrome (SIRS), and other embolic diseases can help reduce hemorrhagic complications. Absence of fever, vascular stigmata, and cardiac murmur at presentation does not necessarily exclude the possibility of IE. Afebrile patients who report a recent history of fever warrant caution before treatment with thrombolytics.
Keywords
• Acute ischemic stroke
• Alteplase
• Infective endocarditis
• Intracerebral hemorrhage
Citation
Miran MS, Roering L, Maud A, Peterson M, Shea K, et al. (2017) Thrombolytic Use in Acute Ischemic Stroke Patients with Concurrent Infective Endocarditis: Emphasis on History, Physical Exam, Outcomes, and Review of Literature. J Neurol Transl Neurosci 5(1): 1077
ABBREVIATIONS
ICH: Intracerebral Hemorrhage; SAH: Subarachnoid Hemorrhage; AIS-IE: Acute Ischemic Stroke Patients with Concurrent Infective Endocarditis; WBC: White Blood Cell; MRI: Magnetic Resonance Imaging; NIHSS: National Institute of Health Stroke Scale
INTRODUCTION
IV alteplase is not recommended in AIS-IE due to high rates of hemorrhagic conversion, death, and disability.
CASE PRESENTATION
A 36-year-old male patient with no history of hypertension, diabetes, a trial fibrillation, hyperlipidemia, or other classic risk factors for stroke presented to the ER with a sudden onset of left hemiparesis, numbness, and left facial droop. Symptoms began while the patient was at work. Upon presentation, his NIHSS was 10, blood pressure 137/100mmHg, temperature 98.1 F, respiratory rate 23, and pulse 105. His white blood cell count was 13.8 K/UL. Physical exam was negative for cardiac murmurs, fever, and vascular stigmata associated with IE. Patient had a history of migraine headaches, but denied headache at the time of presentation. Per the patient’s history, he had suffered a low-grade fever for the past few days which he attributed to West Nile fever. Patient’s history was negative for seizures and stroke. Initial computed tomography (CT) scan of the head was negative for any bleeding. The patient was assessed and administered IV alteplase (0.9mg/kg) within the AHA’s recommended time frame (67 minutes from onset of symptoms).
The next day the patient’s hemiparesis resolved with NIHSS 0. Considering his reported history of low-grade fevers in conjunction with multiple small infarcts revealed on his magnetic resonance imaging (MRI), infective endocarditis (IE) was investigated as a possible cause of the embolic stroke. A transesophageal echocardiogram (TEE) revealed bicuspid aortic valve with moderate aortic insufficiency. TEE did not reveal any vegetation. Blood cultures were positive for gram positive cocci in clusters and positive for streptococcus mitis. IV Penicillin G was prescribed for six weeks. Follow-up imaging revealed numerous asymptomatic small hemorrhagic foci scattered throughout both cerebral hemispheres, the right thalamus, and the cerebellar hemispheres which, given the patient’s history, most likely represented acute hemorrhagic transformation of small non-hemorrhagic embolic infarcts (Figure 1).
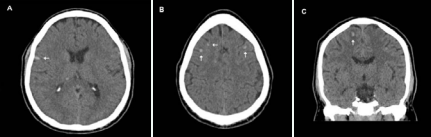
Figure 1 Small multi focal bilateral hemorrhages (arrows) on computed tomography (CT) scans (A, B, and C)
The largest hemorrhage measured ≈ 7 mm. Magnetic resonance angiography and CT angiography of intracranial and extra cranial vasculature was negative for any stenosis, aneurysm or dissection.
Patient was discharged with a peripherally inserted central catheter (PICC) line with orders for weekly labs draws. Patient’s modified Rankin scale at discharge was 2. At follow-up appointment, patient had recovered completely and resumed normal physical activity. Ultimately, the patient did not suffer from a symptomatic hemorrhagic conversion or any other complications resulting from alteplase use in IE after following the treatment plan (Figure 2).
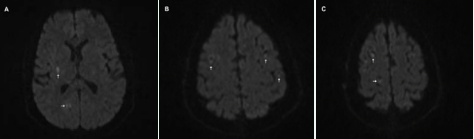
Figure 2 Small multi-focal bilateral ischemia (arrows) on Magnetic Resonance Imaging (MRI) scans (A, B, and C).
DISCUSSION
IV alteplase is the current standard treatment for AIS if given within first 3 hours (and up to 4.5 hours in selected eligible patients) from the onset of symptoms per recommendations of AHA [1]. However, the use of IV alteplase in patients with IE is not recommended by AHA (* Class III; level of evidence C) and is associated with high rate of complications. Previous studies have associated mycotic aneurysms and micro abscesses with much higher rates of hemorrhages in AIS-IE patients [2-4]. Diagnosing IE at the time of AIS and within the window of IV alteplase is challenging as classical signs and symptoms of IE (fever, peripheral vascular stigmata, and cardiac murmur) may not be evident [5]. The diagnostic workup for IE includes history, physical exam, imaging/TEE, blood cultures, and investigation of risk factors using the modified Duke criteria [5]. History and physical exam are the most critical aspects that can help a physician evaluate an AIS patient for IE prior to IV alteplase as the other aspects of the full diagnostic workup may not be able to be completed within the designated time period (3.0-4.5 hours from onset of symptoms). Evaluating components of systemic inflammatory response syndrome (SIRS) may be used to make a clinical judgement within the alteplase window [6,7]. Body temperature of < 36°C or > 38° C, heart rate > 90, respiratory rate > 20, and white blood cell count < 4000/mm or > 12,000/mm or > 10% bands has been previously studied and associated with poor outcome [6,7]. However, the elevated white cell count may independently predict the stroke severity in the absence of IE and may not necessarily be a component of systemic/embolic process [8]. Additionally, looking for conjunctival hemorrhages, Janeway lesions, Osler nodes, and Roth spots is of clinically significant along with auscultation for murmurs and rubs [5]. Subsequently, in younger AIS patients (< 40 years of age) who have an absence of other classic risk factors of stroke (carotid stenosis, hypertension, hyper lipidemia, etc), a detailed history and physical exam can help reduce the rate of complications resulting from use of IV alteplase in AIS-IE patients. Additionally, a febrile patients who report a recent history of fever also warrant a detailed history and exam as this report may be clinically significant in diagnosing possible IE (Table 1).
Current literature supports evidence of adverse outcomes in AIS-IE patients receiving IV alteplase. Asaithambi et al., performed a similar study in 2013 using the Nationwide Inpatient Sample [2]. This study reported higher rates of post-thrombolytic intracranial hemorrhage (20% vs 6.5 %, p=0.006), increased seizures (7.2% vs 1.7%, p=0.3), longer hospital length of stay (14 ± 10vs 7 ± 8, p=0.006), and lower rates of favorable outcomes (10% vs 37%, p=0.01) in the IE group as compared to patients without IE who received IV alteplase. Similarly, Walker et al., in 2012 reported 11 AIS patients with IE where 4 out of 11 received IV alteplase and 7 did not [9]. The mortality rate among the thrombolytic receiving group as compared to the non-thrombolytic group of patients was substantially higher (75% vs 29 %) [9]. Additionally, all 4 IE patients receiving IV alteplase revealed hemorrhagic transformation upon follow-up imaging [9]. Walker et al., also noticed an association between anemia and leukocytosis in these patients at the time of presentation [9]. Only 33% patients in this study were confirmed febrile while an additional 28% patients had reported a history of fever before the event much like our patient [9].
Interestingly, there are rare case reports found within the current literature that suggest favorable outcomes in AIS-IE patients treated with thrombolytics [10]. One of them was a 12-year-old female patient whose case was reported by Tan et al. in 2008 [10,11]. The author argued against the absolute contraindication of thrombolytics use in all cases, especially for the pediatric age group. The author suggested a possible therapeutic use for thrombolytics in select childhood IE-related AIS. In a separate case report in 2007, Junna et al., reported a 56-year-old male patient with a favorable outcome after receiving thrombolytic who was subsequently diagnosed with IE [11]. However, the literature evidence of unfavorable outcomes is far more prevalent and supports the rationale for contraindicating the use of thrombolytic use in such patients (Table 1).
|
Table 1: Characteristics and outcomes of patients with AIS-IE treated with thrombolytics. |
|||||
|
Study |
Age/Gender |
Confirmed fever upon presentation |
Cardiac murmur present upon exam |
Outcome |
Complications |
|
Tversky et al., 2016 |
57M |
NA *Patient reports recent fever prior to admission. |
NA |
Unfavorable |
Severe multifocal ICH |
|
Tan et al., 2008 |
12 F |
Febrile |
absent |
Favorable |
None |
|
Asaithambi et al., 2013 n=222 |
59 ± 18 (mean±SD) 46 % F |
NA |
NA |
Lower rate of favorable outcome in AIS-IE group (10% vs 37%, p=0.01) |
Higher rate of ICH in AIS-IE group (20% vs 6.5%, p= 0.006) |
|
Walker et al., 2012 |
56.6±16.5 (mean±SD) 50% F
|
Afebrile
|
Absent in 66% patients |
Higher mortality (75% vs 29%) and 100% hemorrhagic conversion rate) |
ICH, SAH, and death |
|
Bhuva et al., 2009 |
46 M |
A febrile |
Absent |
Unfavorable |
ICH, death |
|
Buva et al., 2009 |
65 F |
A febrile |
Present |
Unfavorable |
ICH, SAH |
|
Buva et al., 2009 |
61 M |
A febrile |
Absent |
Unfavorable |
ICH, SAH |
|
Junna et al., 2007 |
56 M |
A febrile *Patient reports recent fever prior to admission. |
NA |
favorable |
None |
|
Sontenini et al., 2009 |
70 M |
Febrile, |
Present murmur present on exam |
favorable |
None |
|
Our case |
36 M |
Afebrile *Patient reports recent fever prior to admission. |
Absent |
Favorable |
Asymptomatic hemorrhagic conversion |
|
Ong et al., 2013 |
68 M |
A febrile |
Absent |
Unfavorable |
ICH, SAH |
|
Abbreviations: ICH: Intracerebral Hemorrhage; SAH: Subarachnoid Hemorrhage; M: Male; F: Female; AIS-IE: Acute Ischemic Stroke Patients with Concurrent Infective Endocarditis |
|||||
As in our case, the absence of fever upon admission, vascular stigmata, or a heart murmur on initial exam made the diagnosis of IE very challenging (Table 1) [5]. Hence, there is a need to meticulously examine patient and exclude potential and definite IE in all suspicious patients presenting with AIS symptom prior to thrombolytic therapy by looking for conjunctival hemorrhages, Janeway lesions, Osler nodes, Roth spots, SIRS components, and other risk factors (i.e. poor dentition, IV drug abuse, and mechanical heart valves) [5-7]. In cases where a diagnosis of possible IE is suspected and the AHA timeframe for IV alteplase can still be met, it may be beneficial to complete other investigations prior to administering IV alteplase. However, larger sample studies are needed to determine guidelines for this practice. Endovascular therapy and/or mechanical thrombectomy may prove to be a comparatively safer option for AIS-IE patients as the systemic effects of fibrinolysis can be avoided by these procedures [3,12,13]. Dababneh et al., in 2012, and Kang et al., in 2013 have reported successful endovascular interventions for AIS-IE cases [12,13]. However, the scientific evidence for this treatment modality is not enough since it has not been probed in randomized clinical trials.
* Class III: “Conditions for which there is evidence and/or general agreement that the procedure or treatment is not useful/ effective and in some cases may be harmful”; Level of evidence C: “Consensus opinion of experts, case studies, or standard of care” [14-17].
CONCLUSION
A detailed history, physical exam, and investigating SIRS components can reduce the rate of complications resulting from thrombolytics use in AIS-IE patients. Endovascular treatment may be a comparatively safer option; however, this course of treatment still needs to be thoroughly elucidated. Absence of fever, vascular stigmata, and cardiac murmur does not necessarily exclude the possibility of IE in identified high-risk (i.e. poor dentition, IV drug abuse, mechanical valves etc) patients.








































































































































