Botox those Salivary Glands for a Better Look: A Superior Treatment for Clozapine-Induced Sialorrhea
- 1. Department of Psychiatry and Neuroscience, University of California, USA
- 2. Department of Psychiatry & Human Behavior, University of California, USA
- 3. Psychopharmacology Resource Network, Clinical Operations Division, California Department of State Hospitals, USA
- 4. Department of Psychiatry, Loma Linda University School of Medicine, USA
- 5. Department of State Hospitals - Patton, USA
Abstract
Clozapine is a unique antipsychotic with demonstrated efficacy for treatment-resistant schizophrenia and mania, and aggression, suicide, or psychogenic polydipsia related to schizophrenia. Sialorrhea, a common adverse effect, is often a barrier to clozapine treatment. Anticholinergic agents have limited efficacy and carry elevated risks of constipation, bowel impaction, or ileus. Periodic injection of major salivary glands with botulinum neurotoxin has proven to be a superior treatment option for clozapine-induced sialorrhea (CIS). This mini-review describes the viability and logistics involved in establishing a successful botulinum treatment clinic in a forensic psychiatric hospital, including adequate administrative support and nursing staff, clinician training, educating treatment providers about the availability and effectiveness of botulinum treatment for sialorrhea, and development of a clinic protocol (e.g., procedural elements and relevant rating scales). Finally, botulinum treatment was evaluated to be cost-effective.
Keywords
• Clozapine
• Sialorrhea
• Hypersalivation
• Drooling
• Botulinum Neurotoxin
• Botulinum Toxin
• Treatment
• Clinic
• Salivary Glands
Citation
Arias AW, Cummings MA, Proctor GJ (2025) Botox those Salivary Glands for a Better Look: A Superior Treatment for Clozapine-Induced Sialorrhea. Ann Otolaryngol Rhinol 12(5): 1371.
INTRODUCTION
Clozapine is the sole antipsychotic agent with demonstrated efficacy for treatment resistant schizophrenia and mania, and for nonpsychotic aggression, suicidality and primary polydipsia associated with schizophrenia spectrum disorders. However, clozapine possesses a gamut of adverse effects, especially sialorrhea which affects circa 90% of clozapine-treated patients. Sialorrhea is a significant barrier to clozapine treatment including patients’ desire to discontinue clozapine, social stigmatization, and medical comorbidities (e.g., nocturnal aspiration pneumonia) [1,2]. Clozapine and its active metabolite norclozapine have muscarinic receptor agonist and antagonist properties. The predominately anticholinergic clozapine is thought to be primarily responsible for constipation, while norclozapine’s M1 receptor agonism in salivary glands is posited to cause sialorrhea. As clozapine has valuable benefits, developing sialorrhea management protocols are critical in minimizing treatment refusal and patient morbidity [3].
Because clozapine can cause obstipation, preferred initial treatments for sialorrhea included topical anticholinergic agents (e.g., atropine drops or ipratropium bromide spray) with minimal systemic absorption. Atropine drops or ipratropium bromide are applied to the buccal mucosa, held in the mouth for 15 and 30 seconds, respectively, and rinsed out with 5 ml of water. When topical agents fail, a systemic alternative like glycopyrrolate can be used as it does not cross the blood brain barrier like centrally acting anticholinergics (e.g. benztropine or trihexyphenidyl); however, it can double the risk for potentially fatal ileus [4].
Medical literature from two decades ago described another evidence-based alternative, which involves botulinum toxin injected directly into salivary glands. Botulinum toxins (BT) disable cholinergic neurotransmission by inhibiting proteins involved in vesicle fusion of presynaptic cholinergic neurons. Type A cleaves synaptosome-associated protein (SNAP-25), while type B targets vesicle-associated membrane protein (VAMP). Both have traditionally been used to paralyze muscles in neurological conditions such as dystonia, for treatment of migraine headaches, and for cosmetic purposes. By 2001, papers began noting the use of BT to decrease salivary secretion in patients with neurological conditions associated with sialorrhea such as Parkinson’s disease (PD) or cerebral palsy (CP) [5]. Researchers subsequently examined BT use for CIS as viable alternatives to topical and systemic anticholinergics [6]. Treatment-resistant schizophrenia in both community and forensic settings requiring clozapine therapy compels us to seek an evidence-based treatment for sialorrhea which circumvents serious concerns for constipation, ileus, and bowel obstruction with systemic anticholinergics treatment of CIS [7,8].
USES FOR BOTULINUM TOXIN
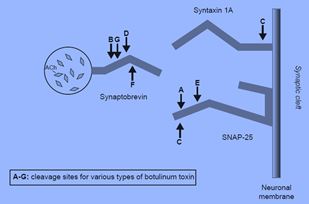
The 7 immunologically distinct BT serotypes work via distinct mechanisms to inhibit the SNARE complex causing flaccid paralysis of target muscles in dystonia and for cosmetic applications (Figure 1). In the U.S., four type A toxins (onabotulinumtoxinA, abobotulinumtoxinA, incobotulinumtoxinA, prabotulinumtoxinA) and one type B toxin (rimabotulinumtoxinB) are approved for cosmetic purposes, migraine headaches, and muscular dystonia/ spasms [9-14]. BT provides transient relief of symptoms lasting between 3 to 6 months.
Figure 1 Target sites for BT types on SNARE proteins.Adapted from Barr, et al. (2005). Botulinum neurotoxin detection and differentiation by mass spectrometry, Emerging Infectious Diseases, 11, 1578-1583 [20].
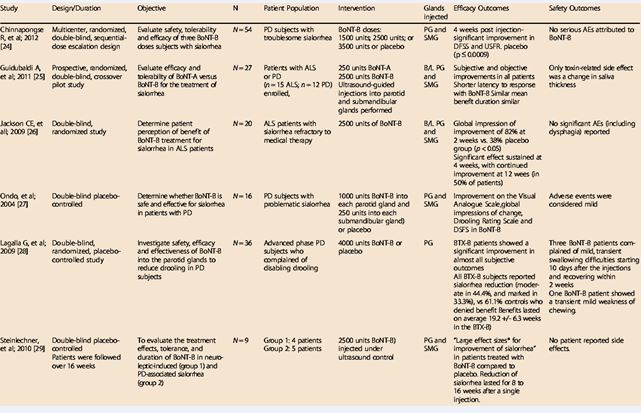
In 2001, the first studies documented BT injection into salivary glands for Parkinsonian sialorrhea to inhibit release of acetylcholine from synaptic vesicles of post-ganglionic parasympathetic fibers containing cholinergic receptors in the salivary glands [5-15]. Further research noted that botulinum injections to the parotid and submandibular glands were highly effective in sialorrhea related to neurological conditions for 3 to 6 months. Benefits for medication-induced sialorrhea were subsequently documented, including easy administration and no systemic effects [6-17] (Table 1).
Table 1 RimabotulinumtoxinB in sialorrhea: systematic review of clinical trials [17].
BOTULINUM NEUROTOXIN FOR CIS
Both BT types have substantial data for sialorrhea, including notable case reports for CIS. CIS may be greater in severity due to constant stimulation from norclozapine’s M1 agonist properties, but the literature demonstrated that BT could reduce hypersalivation from all causes. Adverse effects from BT include pain or bruising at the site of inoculation, rare flu-like symptoms, headache, nausea, redness, dry mouth (<5 % of cases), and unlikely possibility of temporary muscle paresis if toxin spreads beyond the injection area. Locally injected BT has virtually no risk of systemic adverse effects (e.g., breathing problems, trouble swallowing, muscle weakness, or slurred speech observed in botulism).
For patients with severe mental illness who may be wary about injections into the face, we considered patient comfort during injection and resistance to BT itself when selecting which BT to use for CIS. As some literature indicates greater pain with rimabotulinumtoxinB injections compared to BT A, we chose incobotulinumtoxinA which imparts minimal pain when administered with a 30-gauge needle [16]. Primary resistance to the toxin itself occurs at a rate of 6.25% in non-cosmetic applications, sometimes appearing after the first treatment [18,19]. Secondary resistance can arise due to development of antibodies to the toxin if BT is used for conditions with only partial response (e.g., congenital ptosis, myasthenia gravis, etc.) and other treatment variables (e.g., high dosing frequency dosing, improper storage or transport) in about 5 – 10% of non-cosmetic cases, resulting in partial or no response to subsequent BT treatments.
For the reasons above and its lower risk for immunogenicity (i.e., it is the only BT with no induction of secondary resistance), we selected incobotulinumtoxinA based which also has FDA approval for sialorrhea [18,19].
LOGISTICS FOR CLINIC SET-UP
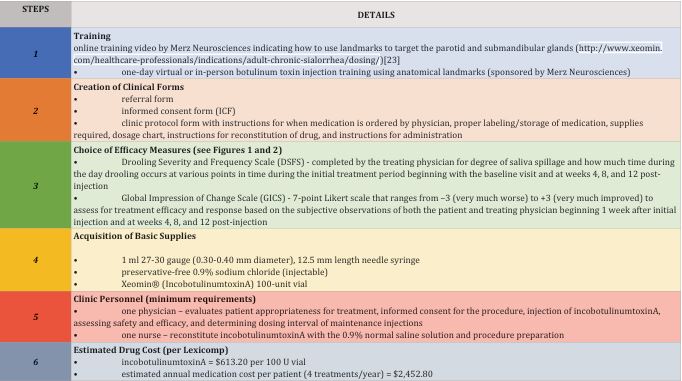
Establishing a BT sialorrhea clinic involves the following, as noted in Table 2.
Table 2: Steps for Establishing a BT Sialorrhea Clinic
Training, involving a brief video illustrating the anatomical landmarks (Table 2), and live training with an experienced neurologist, can be completed in a day. Ultrasound guidance is not necessary for most cases but could be considered in those with gland atrophy [6-17]. Paperwork could be limited to three forms: referral,
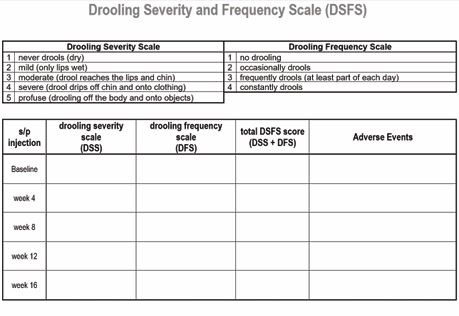
informed consent (ICF) and clinic protocol. The ICF is standardized to include what conditions BT is FDA approved to treat, description of the procedure, risks/ benefits, and signatures of the patient and administering physician. The clinic protocol includes the requisite basic supplies and instructions for handling and administering BT correctly. Objective and subjective efficacy scales can be used to determine efficacy. Objective measurements of unstimulated salivary flow rate using a digital scale calibrated to the thousandth of a gram and SalivaBio® Oral Swabs is important for clinical research but is not practical for routine treatment of CIS in a clinic (time consuming, requiring ~1.25 hours). Subjective scales (e.g., Drooling Severity and Frequency Scale and Global Impression of Change Scale) are sufficient to measure efficacy of BT for CIS [20,21] (Figures 2 and 3).
Figure 2 Drooling Severity and Frequency Scale (Department of State Hostpials – Patton).
Figure 3 Global Impression of Change Scale (Department of State Hostpials – Patton).
With a nearly 90% efficacy rating, we anticipated a significant lowering of DSFS scores between the pre- and post-injection ratings [16]. Most clinics already have or can easily acquire basic supplies (syringes with smaller gauge needles, straight forceps, rubbing alcohol, cotton/gauze pads, and normal saline solution). The most significant cost is for incobotulinumtoxinA. Based on U.S. wholesale pricing, the annual cost for anticholinergic agents used for sialorrhea approximates $704.10 annually, while incobotulinumtoxinA costs approximately $2,500 when dosed quarterly (Wholesale Acquisition Price = $613.20 per 100 U vial). However, the risk and cost for hospitalization to treat aspiration pneumonia or ileus is elevated with ineffective anticholinergic treatment ($27,853 with a daily rate of $3,979 in California; figures based on information from the 2021 American Hospital Association Annual Survey in the Becker’s Hospital CFO Report) [22,23]. Hence, providing BT for CIS is cost-effective.
BT CLINIC PROTOCOL
To evaluate baseline sialorrhea severity, we recommend discontinuing all systemic anticholinergics for sialorrhea one week prior to initial appointment; all anticoagulants and aspirin 5 days prior to appointment date to reduce injection site bleeding; and all topical medications for sialorrhea 48 hours before initial appointment. Male patients should shave the jawline area prior to the appointment. After injections, patients should resume anticoagulants and aspirin. Because there may be no improvement for up to two weeks, topical/systemic sialorrhea treatments should be resumed for 14 days after the first injection to mitigate risk for aspiration pneumonia during the immediate lag period and then discontinued.
The treating psychiatrist or physician injector should rate the DSFS and GICS prior to injections. After ratings are obtained, the injector administers 30 U into each parotid gland and 20 U into each submandibular gland (100 U incobotulinumtoxinA per treatment). Initial follow up visits for ratings are scheduled in 4-week intervals with the first maintenance injection visit scheduled at 12 weeks after initial injections. Based on individual patient response, the interval between injections could be adjusted from every 4 to 20 weeks.
DISCUSSION
This is the first systematic protocol for the establishment of a clinic treating CIS with BT, which circumvents complications of anticholinergic agents. As noted above, staff physicians could be trained to deliver the injections in a single day.
Cost Analysis
Sialorrhea treatment with incobotulinumtoxinA is cost-effective. As noted above, no specialized supplies are required for a BT clinic: syringes with smaller gauge needles, straight forceps, rubbing alcohol, cotton/gauze pads, and normal saline solution. The most significant supply cost for this type of sialorrhea treatment clinic is the medication itself, incobotulinumtoxinA. Per the California Department of General Services Wholesale Acquisition Cost, the annual cost for incobotulinumtoxinA is approximately $2500 when dosed quarterly (Wholesale Acquisition Price = $613.20 per 100 U vial). Although the annual cost of incobotulinumtoxinA for sialorrhea is 3 to 4 times the cost of anticholinergic agents (10% efficacy), it is 9 times more effective. A BT Clinic can operate with one physician and one nurse as described above.
All patients were educated about the procedure, underwent the procedure, and tolerated it well except for one who was unaware of the reason for the appointment and refused the injections. None who underwent this procedure reported any discomfort, and all injections performed to date have been well tolerated without any complications.
Take-Home Points
- BT offers evidence-based method to avoid adverse effects of anticholinergics which may not be effective.
- Physicians can be easily trained to administer BT injections. No severe complications, such as injury to facial nerve, have been observed.
- Bilateral injections into the parotid and submandibular glands are indicated and safe for CIS.
- All but one of 33 patients who received BT injections tolerated well with no adverse effects; the one patient had transient perioral swelling.
REFERENCES
- Maher S, Cunningham A, O’Callaghan N, Byrne F, Mc Donald C, McInerney S, et al. Clozapine-induced hypersalivation: an estimate of prevalence, severity and impact on quality of life. Ther Adv Psychopharmacol. 2016; 6: 178-184.
- Meyer JM. The Clozapine Handbook. United States of America by Sheridan Books, Inc., Cambridge University Press. 2019
- Cummings MA, Proctor GJ, Arias AW. Dopamine antagonist antipsychotics in diverted forensic populations. CNS Spectr. 2020; 25: 128-135.
- Nielsen J, Meyer JM. Risk factors for ileus in patients with schizophrenia. Schizophr Bull. 2012; 38: 592-598.
- Porta M, Gamba M, Bertacchi G, Vaj P. Treatment of sialorrhoea with ultrasound guided botulinum toxin type A injection in patients with neurological disorders. J Neurol Neurosurg Psychiatr. 2001; 70: 538-540.
- Steinlechner S, Klein C, Moser A, Lencer R, Hagenah J. Botulinum toxin B as an effective and safe treatment for neuroleptic-induced sialorrhea. Psychopharmacology (Berl). 2010; 207: 593-597.
- Elkis H. Treatment-resistant schizophrenia. Psychiatr Clin North Am. 2007; 30: 511-533.
- Martin A, O’Driscoll C, Samuels A. Clozapine use in a forensic population in a New South Wales prison hospital. Aust N Z J Psychiatry. 2008; 42: 141-146.
- Shaterian N, Shaterian N, Ghanaatpisheh A, Abbasi F, Daniali S, Jahromi MJ, et al. Botox (OnabotulinumtoxinA) for Treatment of Migraine Symptoms: A Systematic Review. Pain Res Manag. 2022; 2022: 3284446.
- Evolus I. Jeuveau Package Insert. prabotulinum toxin type a. Newport Beach, California, U.S.A. 2023.
- Allergan I. Botox Cosmetic Package Insert. onabotulinumtoxina. Chicago, Illinois, U.S.A. 2024
- Ipsen Biopharmaceuticals I. Dysport Package Insert. botulinum toxin type a. Cambridge, Massachusetts, U.S.A. 2024
- Merz North AMerica, I. (2024). Xeomin Package Insert. incobotulinumtoxina. Raleigh, North Carolina, U.S.A.
- Solstice Neurosciences L. Myobloc Package Insert. rimabotulinumtoxinb. Rockville, Maryland, U.S.A. 2024
- Friedman A, Potulska A. Botulinum toxin for treatment of parkinsonian sialorrhea. Neurol Neurochir Pol. 2001; 35: 23-27.
- Petracca M, Guidubaldi A, Ricciardi L, Ialongo T, Del Grande A, Mulas D, et al. Botulinum Toxin A and B in sialorrhea: Long-term data and literature overview. Toxicon. 2015; 107: 129-140.
- Dashtipour K, Bhidayasiri R, Chen JJ, Jabbari B, Lew M, Torres- Russotto D. RimabotulinumtoxinB in sialorrhea: systematic review of clinical trials. J Clin Mov Disord. 2017; 4: 9.
- Kessler KR, Benecke R. The EBD test--a clinical test for the detection of antibodies to botulinum toxin type A. Mov Disord. 1997; 12: 95-99.
- Dobryansky M, Korsh J, Shen AE, Aliano K. Botulinum toxin type A and B primary resistance. Aesthet Surg J. 2015; 35: NP28-30.
- Jost WH, Friedman A, Michel O, Oehlwein C, Slawek J, Bogucki A, et al. SIAXI: Placebo-controlled, randomized, double-blind study of incobotulinumtoxinA for sialorrhea. Neurology. 2019; 92: e1982-e1991.
- Jost WH, Friedman A, Michel O, Oehlwein C, Slawek J, Bogucki A, et al. Long-term incobotulinumtoxinA treatment for chronic sialorrhea: Efficacy and safety over 64 weeks. Parkinsonism Relat Disord. 2020; 70: 23-30.
- Leung JG, Hasassri ME, Barreto JN, Nelson S, Morgan RJ 3rd. Characterization of Admission Types in Medically Hospitalized Patients Prescribed Clozapine. Psychosomatics. 2017; 58: 164-172.
- Merz Neurosciences. “Adults with Chronic Sialorrhea: Where to Inject and Injection Guidelines”. 2020.