Use of Steroid Injection to Alleviate Posterior Glottic Stenosis
- 1. Department of Otolaryngology, The University of Tennessee Health Science Center, USA
- 2. The University of Tennessee Health Science Center, School of Medicine, USA
ABSTRACT
Posterior glottis stenosis (PGS) represents an interesting dilemma to the airway surgeon. Diagnosis of this rare condition can be difficult, often mimicking bilateral vocal fold paralysis. Management can also be challenging, with several potential therapeutic options. After receiving IRB approval, pediatric Otolaryngology patient records at a single institution in Memphis, TN were reviewed from 1/1/2013 to 7/1/2014 and 4 patients were identified with PGS. Serendipitously, each these 4 patients represented a distinct grade, I-IV, of PGS. Our case series presents these pediatric patients, all with confirmed PGS on operative direct laryngoscopy that were treated with Triamcinolone 40 mg/ 1mL injectable solution a variable number of times, based on therapeutic response. All 4 patients showed improvement or resolution of synechiae causing the PGS on subsequent laryngoscopy. Triamcinolone injection has previously been shown to be a viable first-line therapeutic option for the treatment of low-grade (type I-II) PGS. Based on our limited series, a trial of steroid injection may represent a standalone therapeutic option worth trial for higher grade (type III-IV) PGS prior to more aggressive surgical treatment such as cartilage grafting, which is the standard practice at this time.
KEYWORDS
• Posterior glottis stenosis
• Scar • Synechia
• Steroid
• Kenalog
• Triamcinolone
CITATION
Tompkins JJ, Rosen P, McLevy JD, Thompson JW (2017) Use of Steroid Injection to Alleviate Posterior Glottic Stenosis. Ann Otolaryngol Rhinol 4(2): 1163.
INTRODUCTION
Posterior glottis stenosis (PGS) represents an interesting dilemma to the airway surgeon. Diagnosis of this rare condition can be difficult, often mimicking bilateral vocal fold paralysis. Management can be challenging, with several potential therapeutic options, including steroid injection, which has traditionally been used for low-grade stenosis. PGS is defined as the development of scar tissue between posterior glottic structures. The involved structures include the posterior third of the vocal folds, arytenoids, posterior commissure with the interarytenoid muscle, cricoarytenoid joints, as well as the cricoid lamina and the overlying mucosa. The adhesions commonly lead to the partial or total fixation of the vocal folds, potentially resulting in airway compromise [1,2]. Additionally, the immobility of the vocal folds can make phonation challenging for patients [3]. Prolonged intubation with associated trauma is the most common cause of PGS. Reports have been as high as 14% in patients intubated for longer than 10 days. Many factors contribute to the development of PGS, including traumatic intubation, large endotracheal tube size, movement of endotracheal tube, prolonged duration of intubation, repetitive extubations and intubations, and local infection [2]. The treatment of recurrent respiratory papillomatosis, abnormal sixth brachial arch development, and radiotherapy has also been associated with PGS [4-6]. PGS requires differentiation from other forms of bilateral vocal cord immobility, with bilateral vocal cord paralysis being the most frequently encountered cause of immobility. Both can have a similar presentation of stridor for a prolonged period that quickly worsens to dyspnea, or the gradual progression of dyspnea over the course of a few months. One can usually make the distinction with the medical history, laryngoscopic examination, and laryngeal electromyography [6]. While both have the same treatment goal of airway patency, the actual therapeutic action is quite different [7] Use of the classification system developed by Bogdasarian et al. [8] Table (1)
Table 1: Classification of Posterior Glottic Stenosis (Bogdasarian et al. [8]).
| Type I | Glottis- interarytenoid scar, normal posterior commissure |
| Type II | Interarytenoid scar and posterior commissure scar |
| Type III | Posterior commissure scar involving a cricoarytenoid joint |
| Type IV | Posterior commissure scar involving both cricoarytenoid joints |
has become a helpful guide when determining the treatment method to employ. More advanced levels of PGS have traditionally required more complex therapeutic interventions [1]. Modalities are categorized into intralesional injections, endolaryngeal procedures, open surgical procedures. Corticosteroids have been injected as a sole treatment, or to augment the effects of laser excision of synechiae [2,9].Type I PGS has traditionally involved laser or endolaryngeal excision with postoperative stenting. Moderately advanced levels of stenosis, including types II-III involve scarring too extensive to for this method. The higher grade PGS can be surgically repaired with the endoscopic dilatation, micro trapdoor flap technique, subtotal arytenoidectomy, laryngofissure with posterior cricoidotomy, and Rethi’s technique (laryngofissure and posterior cricoidotomy with resection of scar tissue, graft placement and molding) [6,10-14].
Our case series presents 4 pediatric patients with interarytenoid synechiae causing PGS. We pose that a trial of Triamcinolone injection represents a viable first-line therapeutic option for the lysis of all stages (Bogdasarian and Olson Stage I-IV) of interarytenoid synechiae.
METHODS
Study Design
Case series: After obtaining IRB approval, we performed a retrospective review of pediatric patients with PGS due to interarytenoidsynechiae treated with Triamcinolone injection from 1/1/2013 to 7/1/2014. Inclusion criteria: age less than 18 years, confirmed PGS on operative direct laryngoscopy with images, treated with Triamcinolone 40 mg/ 1mL injectable solution. All patients that received multiple injections of Triamcinolone received therapeutic treatment within 1-2 months intervals.
RESULTS
Four patients were identified that met inclusion criteria. All 4 Bogdasarian and Olson stages of PGS were represented. Three patients had tracheostomy tubes at time of evaluation and diagnosis. Causes of stenosis included prolonged intubation in all 3 tracheotomy patients, and intubation trauma in the remaining patient (Table 2).
Table 2: Causes and Severity of PGS.
| Patient | Age at Diagnosis | Cause | Severity | Location | Tracheostomy |
| 1 | 20 months | Prolonged intubation at 4 months due to respiratory failure |
Type I | 1-2mm posterior interarytenoid scar band, normal posterior commissure |
Yes, respiratory failure at 4 months |
| 2 | 23 months | Ex-26 week premature infant, prolonged intubation |
Type II | Subglottic stenosis extending to and involving posterior commissure |
Yes, respiratory failure at 4 months |
| 3 | 35 months | Traumatic intubation due to respiratory failure (RSV and vascular ring) |
Type III | Posterior commissure scar involving leftcricoarytenoid joint, left true vocal cord immobile |
No |
| 4 | 13 years | Ex-26 week premature infant, previous tracheostomy tube |
Type IV | Posterior commissure scar involving both cricoarytenoid joints |
Yes, prolonged intubation Decannulated 2004, replaced 6/12/13. |
Each patient was treated with varying amounts of Triamcinolone 40 mg/ 1 mL injectable solution, based on synechiae extent and the area treated (Figure 1).
Figure 1: Kenalog Injection. Posterior larynx pictured with Kenalog injection needle placed at posterior glottis interarytenoid scar band.
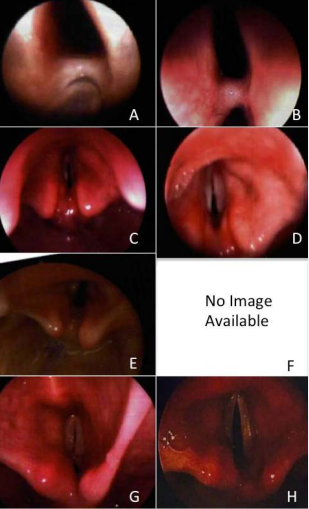
All 4 patients showed improvement of synechiae on subsequent laryngoscopy (Figure 2).
Figure 2: Kenalog Pre-injection and Post-injection Comparison Images (A) Patient 1: initial, stage I, (B) Patient 1: final showing improved/ stable, (C) Patient 2: initial, stage II, (D) Patient 2: final showing improved/resolved, (E) Patient 3: initial, stage III, (F) Patient 3: no image exists due to flexible fiberoptic laryngoscopy use for final exam which showed resolution of scar band, (G) Patient 4: initial, stage IV, (H) Patient 4: final showing improved/resolved.
Table 3.
Table 3: Treatment with Injectable Triamcinolone. Patients 1 and 4 received multiple injections. Subsequent injections were given at the time of follow-up laryngoscopy.
| Patient | Severity | Triamcinolone 0mg/1mL | Follow-up Laryngoscopy Results |
| 1 | Type I | 1: 0.3mL 2: 0.4mL 3: 0.3mL 4: 0.6mL |
Direct laryngoscopy at 2 months: Stable Direct laryngoscopy at 2 months: Improved Direct laryngoscopy at 2 months: Stable |
| 2 | Type II | 0.5mL | Direct laryngoscopy at 1 month: Improved |
| 3 | Type III | 0.1mL | Flexible laryngoscopy at post-op day #7: Resolution of synechiae, bilateral true vocal cords mobile |
| 4 | Type IV | 1: 0.25mL 2: 0.7mL 3: 0.6mL 4: 1mL |
Direct laryngoscopy at 2 months: Stable Direct laryngoscopy at 1 month: Improved Direct laryngoscopy at 1 month: Improved |
Patient 1 showed improvement of the type I adhesion. Patients 2-4 experienced resolution of types II IV synechiae. There were no post-operative complications encountered in this series.
DISCUSSION
PGS remains an elusive diagnosis and a challenging condition to treat. One must first differentiate this rare condition from the more common diagnoses that present in a similar fashion. After confirmation of PGS, one enters into a difficult therapeutic paradigm. These children often require tracheostomy, due to the narrowed airway. After confirming or producing a safe airway, one is then tasked with treating a difficult condition, for which no gold standard therapeutic option exists.
We present a series of 4 patients that were evaluated and treated at a freestanding, tertiary care, pediatric hospital from 1/1/2013 to 7/1/2014. Serendipitously, these patients were found to represent each of the 4 Bogdasarian and Olson stages of PGS. Three of the 4 patients had tracheostomy tubes placed prior to initiation of therapy. All 3 of those patients had developed PGS as a result of prolonged intubation. The first patient experienced interarytenoid scar due to prolonged intubation at 4 months of age due to respiratory failure. Patient 2 was an ex-26 week premature infants with prolonged intubation that developed subglottic stenosis involving the posterior commissure. Patient 3 did not have a tracheostomy tube. She was a 2-year-old female with a remote history of double aortic arch repair at 9 days old. She presented at 2 years of age in respiratory distress, ultimately found to be due to a combination of respiratory syncytial virus and vascular compression. The patient was intubated and subsequently found to have subglottic necrosis extending to and involving the posterior true vocal cords after developing post-extubation respiratory distress. A revision aortopexy was performed prior to Triamcinolone injection therapy. The necrosis abated and the left true vocal cord was treated for immobility due to synechiae. Patient 4 was a full term infant with multiple congenital anomalies including hydrocephalus, Tetrology of Fallot, pulmonary atresia, and duodenal atresia that was intubated due to respiratory failure at 4 months of age. He was decannulated during infancy, but continued to experience stridor and dyspnea with exertion. The decision was made to replace the tracheostomy tube prior to therapeutic intervention. His type 4 posterior commissure scar band resolved with a series of 5 Triamcinolone injections spaced 1 to 2 months apart. His true vocal cords are now passively mobile with instrumentation, but remain neurologically nonfunctional.
All 4 patients were treated conservatively with a trial of Triamcinolone injection before embarking on more extensive PGS lysing procedure. All 4 showed improvement or resolution on subsequent examination. All 3 patients with tracheostomy tube remain tracheostomy tube dependent. Two of these patients require supplemental oxygen due to their pulmonary status, and the final patient’s treatment for posterior glottis stenosis revealed neurologically inert vocal cords, thus preventing decannulation. Voice quality was not routinely assessed due to a number of factors, including age, neurologic status, and respiratory limitations. The patient that did not have a tracheostomy tube, patient 3, improved after one treatment and was found to have normal mobility of the previously immobile left true vocal cord on bedside examination using a flexible laryngoscope. The robust response of the grade 3 patient may be due to the quick intervention between the initial injury and the steroid infection procedure. Alternatively, the long-standing scar in the patient with grade 4 stenosis showed incremental improvement over a series of 5 injections.
CONCLUSION
Triamcinolone injection represents a viable first-line therapeutic option for the treatment of all stages (I-IV) of interarytenoid synechiae causing posterior glottis stenosis. It is a useful therapeutic option for monotherapy of low-grade PGS, and a useful adjunct, or possible monotherapy, in the treatment of higher stage PGS.