Pulmonary Involvement in Systemic Lupus Erythematosus: A Case Report
- 1. Department of Pulmonology, Hôpital 20 août 1953, Morocco
CITATION
El Khattabi W, Nahidi M, Arfaoui H, Msika S, Bougteb N, et al. (2023) Pulmonary Involvement in Systemic Lupus Erythematosus: A Case Report. Clin Res Pulmonol 9(3): 1064.
INTRODUCTION
Systemic lupus erythematosus (SLE) is a connective tissue disease characterized by multi-visceral involvement and a variety of clinical, biological and radiological signs. Systemic lupus erythematosus can affect several organs, including the lung. All lung anatomical compartments can be affected, dividing lesions into pleural, infiltrative pulmonary, bronchial and vascular involvement.
We report a case of lung parenchymal involvement revealing systemic lupus erythematosus.
OBSERVATION
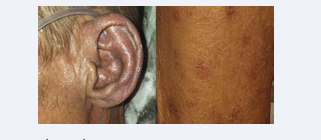
The patient is 71 years old, married with 6 children, a former quarry worker, retired and living in a rural area. He is a chronic smoker, weaned off 20 packs/year 16 years ago. He is a type 2 diabetic on metformin and has been hypertensive for 3 months on amlodipine. He underwent an operation for acute appendicitis, with a simple postoperative course. He was consulted 3 months ago for a symptomatology consisting of progressive dyspnoea that became worse with the slightest effort, a cough associated with whitish sputum, feverish sensations and a decline in general condition. On clinical examination, the patient was polypneic with a respiratory rate of 23 cycles per minute, oxygen saturation 89% on room air, and a digital hippocratic features. Respiratory examination revealed bilateral crackling rales at the lung bases, and mucocutaneous examination revealed finely scaly erythematous lesions on the extremities, achromic lesions on the fingers, a poikylodermal appearance on the left auricle, and ictyosiform scales on both lower limbs. The rest of the physical examination was unremarkable. (Figure 1: skin lesions).
Figure 1: Skin involvement
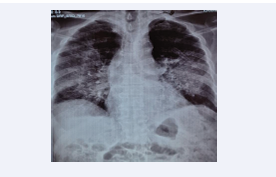
A frontal chest X-ray revealed bilateral butterfly wing opacities (Figure 2).
Figure 2: Frontal chest x-ray
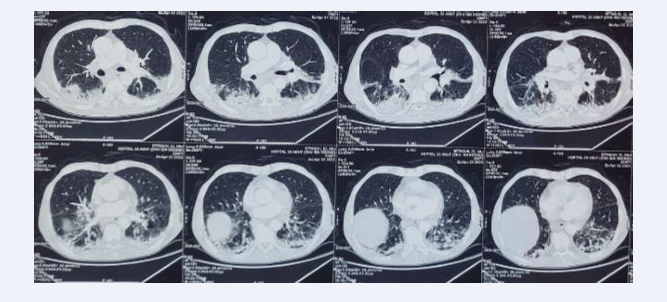
The initial chest CT scan revealed foci of alveolar condensation with bilateral posterobasal air bronchograms, as well as ground glass nodules and micronodules, the largest on the right middle lobe measuring 7.7mm in long axis (Figure 3).
Figure 3 Chest CT : bilateral postero-basal condensation foci
The ECG was normal, the pro-BNP was normal and the echocardiogram showed good bi-ventricular function with a 60% LVEF and non elevated LVEP, ruling out the possibility of PAO, especially as the patient showed no clinical improvement on furosemide and oxygen therapy. Arterial blood gas confirmed hypoxia, with PaO2 at 52.4 mmhg and no abnormalities in pH or capnia.
The patient also received amoxicillin-clavulanic acid without clinical or laboratory improvement.
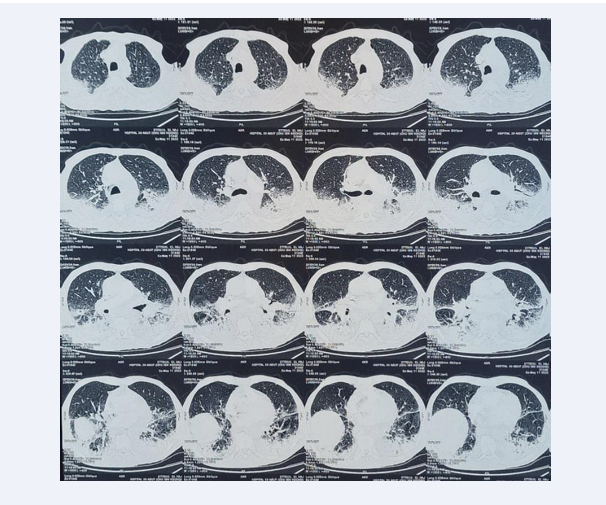
CRP remained elevated at 33.9 mg/L, but the infectious disease work-up, including sputum cytobacteriological examination, was negative. Blood count revealed regenerative normocytic normochromic anemia with hemoglobin at 10.6 g/dL. The rest of the blood count was normal. The D-dimer level was elevated to 890µg/L with no pulmonary embolism on angioscan, which showed, however, worsening foci of alveolar condensation (Figure 4).
Figure 4: Thoracic angioscan: increased scannographic lesions
At this stage, the possibility of infection became less likely, and the presence of anemia prompted us to perform a positive Coombs test, without any biological signs of hemolysis. The diagnosis of systemic lupus erythematosus thus became increasingly likely, despite her advanced age. In addition, 24-hour proteinuria was elevated to 0.62g/24hr, and the urine ionogram showed mild hypocalciuria and slight hyperphostaturia, possibly consistent with associated renal damage.
The Addis count, however, showed no hematuria. Blood ionogram was normal. Protein electrophoresis revealed polyclonal hypergammaglobulinemia. CRP kinetics showed an ascending trend, with a CRP of 55.6 mg/l at follow-up, despite antibiotic therapy, the spectrum of which was broadened by the addition of a quinolone. Flexible bronchoscopy was deferred due to hypoxia. Finally, the anti-nuclear antibody (ANA) assay was positive at 1280, with a mottled appearance. Similarly, anti native DNA was positive at 80. Anti-ENA is positive for anti-SSA and anti-SCL 70. At this stage, the diagnosis of SLE was based on autoimmune hemolysis, proteinuria > 0.5g/24h and positive anti-DNA antibodies.
Skin biopsy reinforced the diagnosis, demonstrating lichenoid dermatitis in favor of systemic lupus erythematosus.
After a multidisciplinary discussion and a pre-therapeutic work-up, the patient was put on corticosteroids at a dose of 1mg/ kg/day and transferred to the internal medicine department for further management, with a good clinical and biological evolution.
DISCUSSION
Thoracic involvement in systemic lupus erythematosus is many and varied. There are an estimated 30 pleuropulmonary manifestations in systemic lupus erythematosus [1].
Pleural involvement is the most common, but lung parenchymal involvement in the form of diffuse interstitial lung disease is rare. Parenchymal involvement rarely reveals systemic lupus erythematosus, and its diagnosis should only be considered after ruling out infectious or embolic causes, which are frequent in systemic lupus erythematosus and have a poor prognosis.
The respiratory lesions of lupus are less well known than those of the skin, kidneys and joints, given the diversity of clinical and radiological presentations. The prognosis of these disorders has been modified by the use of new immunosuppressive therapies.
The diagnosis of systemic lupus erythematosus is based on the EULAR/ACR2019 classification criteria:
- Fever (2 points)
- Leukopenia (3 points)
- Thrombocytopenia (4 points)
- Autoimmune hemolysis (4 points)
- Delirium (2 points)
- Psychosis (3 points)
- Convulsion (5 points)
- Non-scarring alopecia (2 points)
- Oral erosion (2 points)
- Skin lesions of discoid or subacute lupus (4 points) and acute lupus (6 points)
- Pleural or pericardial effusion (5 points)
- Acute pericarditis (6 points)
- Joint involvement (6 points)
- Proteinuria greater than 0.5g per 24h (4 points)
- Lupus glomerulonephritis class 2 or 5 (8 points) and class 3 and 4 (10 points)
- Anticardiolipin or circulating anticoagulant (2 points)
- Low complement fraction c3 and c4 (4 points) and individually (3 points)
- Presence of anti-DNA and anti-Sm antibodies (6 points)
A total score greater than or equal to 10 confirms the existence of systemic lupus, with 96% sensitivity and 93% specificity.
Our patient had an EULAR 2019 score of 18, allowing the diagnosis of systemic lupus erythematosus to be retained.
Acute lupus pneumonia is an uncommon manifestation: 2-10% [2]. It manifests itself in an acute clinical picture of dyspnoea with hypoxia, cough, fever, chest pain and inconstant haemoptysis. X-rays and chest CT scans show non-specific lesions that should rule out infectious pneumonia or alveolar hemorrhage: uni- or bilateral infiltrates, predominantly at the bases, and sometimes pleural effusions. Treatment is based on high-dose corticosteroid therapy (1-2 mg/kg/d prednisone) [3].
Intra-alveolar haemorrhage is rare in systemic lupus erythematosus (1-5.4%), but alters the prognosis of SLE (mortality: 40%) [4]. It is associated with hemoptysis of great or moderate abundance, respiratory distress and a picture of hemosiderosis in its chronic form, with the possibility of underlying visceral damage [5].
Our patient’s clinical picture was, however, progressive and without hemoptysis, which made the possibility of alveolar hemorrhage less likely, especially as alveolar hemorrhage in lupus, unlike that in vasculitis, develops very rapidly [6]. Diffuse interstitial lung disease is the third most common parenchymal manifestation of systemic lupus erythematosus, accounting for 3 to 10% of cases. It is characterized by chronicity and low-growth. It is rarely severe, but may be accompanied by multi-visceral damage. The radiological picture shows interstitial damage, predominantly at the bases. Functional tests show a restrictive syndrome with reduced DLCO and hypoxemia. Histology is non-specific. Corticosteroid therapy slows progression to pulmonary fibrosis [3].
Pulmonary hypertension is a serious complication of systemic lupus erythematosus, occurring in 2.8 to 14% of cases [7], with an average survival from diagnosis of around two years [8].
CONCLUSION
Lung involvement in systemic lupus erythematosus is not exceptional, but is less frequent than pleural involvement. Certain parenchymal disorders, such as alveolar hemorrhage and References.
REFERENCES
2. Meyer O. Systemic lupus erythematosus. Encycl Méd Chir Rhumatologie Orthopédie. 2005; 2: 1-32.
3. Fellrath JM, Sauty A. Lupus erythematosus and respiratory involvement. Rev Med Suisse. 2000; 58: 477-480.
4. Artiguesa N, Martin-Silvaa N, Moreraa J. Dyspnea and anemia in a lupus patient: consider intra-alveolar hemorrhage. Rev Méd Int. 2006; 27: 337-338.
5. Picard C, Parrot A, Mayaud, Cadranel J. Intra-alveolar hemorrhages of immune origin in adults. Rev Mal Respir. 2006; 23: 3S61-3S73.
6. Santos-Ocampo AS, Mandell BF, Fessler BJ. Alveolar hemorrhage in systemic lupus erythematosus: presentation and management. Chest. 2000; 118: 1083-1090.
7. Cottin V. Pulmonary arterial hypertension associated with connectivites. Rev Mal Respir. 2006; 23: 13S61-13S72.