Pronuclear Microinjection of Rabbit Fertilized Eggs
- 1. Department of Biotechnology and Food Science, Research Institute for Animal Production-NAFC, Slovak University of Agriculture, Slovak Republic
Abstract
Although, the first transgenic animal was produced more than 30 years ago, there are still many factors limiting efficiency of transgenesis. One of them is the low rate of transgene incorporation into the genome of microinjected eggs. This mini-review summarizes recent research methods based on pronuclear microinjection of rabbit fertilized eggs in order to reach higher efficiency of transgene integration and expression.
Citation
Chrenek P (2017) Pronuclear Microinjection of Rabbit Fertilized Eggs. JSM Cell Dev Biol 5(1): 1020.
Keywords
Pronuclear microinjection; Transgene; Animal; Integration.
INTRODUCTION
Genetic engineering, recombinant DNA technology, genetic modification/manipulation (GM) and gene splicing are terms that are applied to the manipulation of genes, generally implying that the process is outside the organism’s natural reproductive process. It involves the isolation, manipulation and reintroduction of DNA into cells or model organisms, usually to express a protein. Gene transfer involves two distinctly separate processes. The first step in the process must provide a mechanism by which genetic information can be transported from extracellular space, across biological membranes, and into the nucleus so that the incoming genetic information comingled with the genome of the target organism. The second step in the process affords a means for the new genetic information to become part of the target genome.
The mutual contributions of developmental biology and genetic engineering permitted rapid development of the techniques for the creation of transgenic animals. DNA microinjection was the first successful technique tested on laboratory mouse [1] and then on various other species such as rat, rabbit, sheep, pigs, birds and fish. Since 1981, when the term “transgenic” was introduced, there has been rapid development in the use of genetically engineered animals, as investigators have found an increasing number of applications for the technology.
Gene transfer can be realized through:
a) the fertilized eggs and embryo
b) the germ cells (sperm and oocyte)
c) the gonads (ovary and testis)
d) the somatic cells
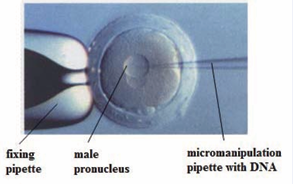
This method involves the direct microinjection of a chosen gene construct (plasmid based or artificial chromosome type transgene) from another member of the same species or from a different species, into the pronucleus of a fertilized ovum. It is one of the first methods that proved to be effective in mammals. Microinjection into mouse and rabbit one cell embryos is relatively simple due to the visible pronucleus (Figure 1).
Figure 1: Pronuclear microinjection of rabbit fertilized egg.
DNA single microinjection
This method involves the direct microinjection of a chosen gene construct (a single gene or a combination of genes) from another member of the same species or from a different species, into the pronucleus of a fertilized ovum. It is one of the first methods that proved to be effective in mammals. Microinjection in mice and rabbit eggs is relatively simple, due to better visualization of pronucleus. However, this does not hold good for embryos of rat and other higher species of animals, where it is much more complex. Total amount of microinjected DNA is about 1-2 pl, which at a concentration of 1-2 ng/μl represents around 200-400 copies of DNA, depending on the type of gene construct. Eggs are fixed on one side with the help of holder pipette and are microinjected from the other end using a microinjection syringe of several micrometres in size automatically or with manual microinjector. Microinjection process causes a visible increase in the volume of the injected pronucleus. After microinjection followed by short-time or long-time embryo culture under in vitro conditions, embryos are transferred to the oviduct or the uterus of the hormonally treated female or using vasectomized males. Most of the gene constructs were prepared using cDNA for the studied gene. Under ideal circumstances and thanks to promoter, targeted expression of the gene in the desired tissue at sufficient level in proper time was achieved, and thereby the ability of transgenic individual to carry the integrated gene to offspring was confirmed. Number of model animals for the purpose of transgenesis, are probably used in order to introduce and integrate gene constructs of different sizes ranging from 10-20 kb using plasmids to about 250-300 kb using YAC, BAC and MAC vectors. The insertion of DNA is, however, a random process, and there is a high probability that the introduced gene will not insert itself into a site on the host DNA that will permit its expression. A major advantage of this method is its applicability to a wide variety of species.
DNA double microinjection
On the same principle as microinjection of foreign DNA into pronucleus lies the method which facilitates the transfer of foreign gene into both the pronuclei of the fertilized egg, which was successfully used for production of transgenic mice [2]. This approach was extrapolated by Chrenek et al. [3], (2005) on a larger species thereby increasing the transgene integration efficiency upon microinjection of DNA into both pronuclei of rabbit embryos. In case of double microinjection, degradation of embryos (lysis, fragmentation, and irregular division) takes place, based on the information as above. Obtained results also showed that double microinjection does not cause significant embryo degeneration. Success of integration of foreign gene using this method was almost 30% higher than that obtained by the previous method.
Cytoplasmic injection
However, using the established method of DNA-ligandpolylysine conjugate to transfect mammalian cells (DNAtransferrin-polylysine) transfecting avian cells, it was suggested that this might provide a means for successful cytoplasmic injection. Cytoplasmic injection of DNA/polylysine mixtures in producing transgenic animals, as well as effect of DNA concentrations and lysine-to-phosphate ratios influences transgene efficiency.
Thus, cytoplasmic injection of DNA is possible by this method, although there is a loss of efficiency over the standard pronuclear injection procedure. This method allows injection of gene into zygote even when the pronuclei are not visible (sheep, pigs, cattle). There is also the unexplored avenue that injection of DNA/polylysine mixtures anywhere in the zygote (including the pronucleus) may yield transgenic animals, thereby allowing trainee injectors a chance to be productive while acquiring the skills necessary to complete pronuclear injection proper.
Nuclear transfer
Nuclear transfer, which involves the transfer of each nucleus (genetically modified) from a multicellular embryo into an enucleated metaphase II oocyte, has been developed in several species. Nuclear transfer has become an active field of research over the last decade, culminating in reports over the past two years of live offspring from differentiated cells of sheep, cattle and mice. Dozens of laboratories are producing calves and lambs whose genetic parents are transgenic somatic cells, often derived from adults, transfected via electroporation. In spite of the fact that research orients towards modification of somatic cell genome through homologous recombination with the aim of producing so called “knock-out” (targeted gene is blocked) or “knock-in” (which refers to the integration of the transgene to specific site in the cell). After selecting the positive cells (“knockout” or “knock-in”), these may serve as a donor of nucleus mainly for the purpose of cloning. As the production of cloned (or transgenic) animals is demanding, combination of the techniques of transgenesis, cloning and also chimerism allows achieving up to 30% better results. The principle depends on the utilization of transgenic somatic cells (or nucleus) for the purpose of cloning. Blastomeres isolated from cloned embryo together with early transgenic embryos are subsequently used for the purpose of chimera. In this manner, chimeric cloned transgenic animals can be obtained.