A Study of Clinical Characteristics and Risk Factors of Mortality Associated with Tigecycline
- 1. Hospital of Obstetrics and Gynecology, Fudan University, China
- 2. Department of Pharmacy, First Affiliated Hospital of Naval Military Medical University, China
- 3. Ciyuan Rehabilitation Hospital, China
- 4. Clinical Pharmacy, Shanghai East Hospital, Tongji University School of Medicine, China
Abstract
In 2010, the FDA warned that tigecycline was associated with an increased risk of death. The risk factors of tigecycline-associated mortality are still controversial. The objective of this study was to obtain the clinical characteristics and to identify risk factors associated with mortality after tigecycline therapy. In an observational retrospective study, we compared the clinical characteristics of patients (non-survivor group) who died within 28 days after tigecycline therapy with those (survivor group) who survived within 28 days after tigecycline therapy. We used a logistic regression model to find risk factors of tigecycline associated mortality. 240 patients were included in the analysis, include 79 in the non-survivor group and161 in the survivor group. The proportion of the kidney failure events and change in creatinine value were greater in the non-survivor group after treated with tigecycline than in the survivor group (p < 0.001). In the multivariate analysis, age (p = 0.019), absolute value of the change of serum creatinine (ΔCre) (p < 0.001), SCr (p = 0.038), APACHE II score (p = 0.031) were independently associated with mortality. An age of ≥ 79 years, ΔCre (μmol/L) ≥ 36, SCr ≥ 112 μmol/L and APACHE II score ≥ 12.7 were selected as the cutoff points for predicting tigecycline associated mortality. The distribution of non-survivors at different ranges of creatinine change showed higher percentages of mortality with high ΔCre after tigecycline treatment. This study identifies the risk factors for tigecycline associated mortality, and draws attention to the fact that there is an increased mortality risk in patients treated with tigecycline.
Keywords
• Tigecycline
• Mortality
• Risk factors
• Hyperphosphatemia
• Creatinine
Citation
Wu S, Wu G, Wang X, Wu H, Lu L (2026) A Study of Clinical Characteristics and Risk Factors of Mortality Associated with Tigecycline. J Family Med Community Health 13(1): 1216.
INTRODUCTION
Tigecycline is a first glycyl tetracycline semisynthetic antibacterial with broad-spectrum activity, and its main indications are complicated intra-abdominal , skin and skin structure infections, and community-acquired pneumonia [1]. With the increasing incidence of Multidrug-Resistant (MDR) bacterial pathogens (e.g. methicillin-resistant Staphylococcus aureus and vancomycin-resistant Enterococcus), it has attracted attention. Because of its low drug resistance rate and minimal drug interactions, tigecycline has become more widely used in the treatment polymicrobial MDR or mixed pathogens infections [2]. But in 2010, the FDA reminded healthcare professionals that the intravenous tigecycline was associated with an increased mortality risk compared to other antibiotics used to treat similar infections [3]. The increased risk was determined using a pooled analysis of tigecycline clinical trials. Although the causes of excessive mortalities in these trials are usually uncertain, most of the mortalities of these seriously infected patients may be related to the progress of infection.
Prior retrospective study showed tigecycline therapy resulted in a significant increase in mortality in carbapenem-susceptible and carbapenem-nonsusceptible Klebsiella pneumoniae bloodstream infections [4]. A meta analyses included 13 randomized controlled trials (10 published and 3 unpublished) showed that tigecycline significantly increased mortality. Overall, tigecycline was associated with a 0.7% absolute or 30% relative increase in mortality. The increased mortality of tigecycline therapy was independent of infection type, trial design, and study size [3]. To date, there is a lack of clinical characteristics and risk factors of tigecycline-associated excess mortality. This retrospective cohort study aims to analyze the clinical features and risk factors of increased mortality after tigecycline treatment in order to provide reference information for the clinical use of tigecycline in antibacterial therapy.
MATERIALS AND METHODS
Study Design and Patients
The retrospective cohort study was conducted in in China in the Hongkou Branch of First Affiliated Hospital of Naval Military Medical University Hospital, a tertiary referral hospital. Hospitalized adult patients (≥ 18 years old) with clinical infections who continuously used injection forms of the study tigecycline for ≥ 72 hours treatment duration from 1st January 2020 to 31st December 2020 were included in the study, after receiving approval from the Shanghai Changhai Hospital Ethics Committee (CHEC2022-008). This study did not directly interfere with the patients or show the patients’ name, medical record number, or other personal information. All patients’ records were anonymized and de-identified prior to analyses. Moreover, the study did not adversely affect the rights of patients; therefore, informed consent was not required by the Institutional Review Board in China because of the anonymous and the observational nature of the study. 100 mg tigecycline and a maintenance of 50 mg q12h. Tigacycline can be administered by intravenous infusion or intravenous injection. All patients were treated in the ward especially the Intensive Care Unit (ICU).
The exclusion criteria were as follows:
- Patients who did not receive ≥ 3 days of tigecycline treatment;
- Patients with incomplete clinical laboratory data;
- patients who hospitalization; underwent surgery during
- Patients with liver function and renal function damage caused by any drug except tigecycline during hospitalization.
Data Collection
The data were collected included demographic, medical history information and the results of laboratory tests performed before, during and 28 days after the treatment of tigecycline or before the patient died ,concomitant drugs, and clinical outcomes (eg, bleeding, death) from electronic hospital medical records using a standardised data collection form. The severity of the illness was assessed by using the Acute Physiology and Chronic Health Evaluation II (APACHE II) scoring system calculated [5,6]. Primary outcome measured was 28-day all cause in hospital mortality. Secondary outcomes include time to liver dysfunction, renal failure, and coagulation Function, Including Fibrinogen (FIB) levels, Prothrombin Time (PT), Activated Partial Thromboplastin Time (APTT), Thrombin Time (TT), and Platelet Count (PLT).
Definitions
Mortality was defined as the death that occurred during tigecycline therapy and within 28 days after receiving tigecycline treatment during the same hospitalization. Liver dysfunction was defined as an Alanine Aminotransferase (ALT) or Aspartate Aminotransferase (AST) ≥ 3 times the Upper Limit of Normal (ULN) [7]. Kidney failure was defined as an increase in serum creatinine (SCr) ≥ 265.2 × baseline or an absolute SCr ≥ 353.6 μmol/L with an acute rise of at least 442 μmol/L or decrease in Glomerular Filtration Rate (GFR) ≥ 75% or Urine Output (UO) < 0.3 mL/kg/h × 24 h or anuria × 12 h [8]. We defined hypofibrinogenemia as a fibrinogen plasma concentration < 2.0 g/L [9]. The ΔCre is defined as the absolute value (μmol/L) of the increase or decrease of serum creatinine.
Statistical Analysis
Statistical analysis was performed with SPSS statistics 19.0. The continuous variables are presented as means ± standard deviation or medians (25% to 75% range). We evaluated these variables by the t test or Mann– Whitney U-test (for variables that are not normally distributed). The categorical variables are presented as percentages of the group, and Fisher’s exact test or the Chi-square test was used to evaluate these variables. Multivariable logistic regression models were used to identify independent predictors in association analyses. p < 0.05 was regarded as statistically significant.
RESULTS
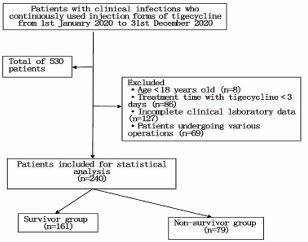
A total of 530 patients were identified during the study period. Following the exclusion of 290 patients, 240 patients were included in this study. Of these patients, 79 (32.9%) patients eventually died during tigecycline therapy and within 28 days after discontinuation of tigecycline therapy and were placed in the non-survivor group, and the other 161 (67.1%) patients were placed in the survivor group (Figure 1).
Figure 1 Flow chart of the process of patient selection.
Table 1: Comparison of baseline characteristics of the two group patients.
|
Variable |
Total (n = 240) |
Non-survivor group (n = 79) |
Survivor group (n = 161) |
p |
|
Age (years) (mean ± SD) |
71.3 ± 10.4 |
78.3 ± 13.9 |
68.0 ± 11.2 |
0.022 |
|
Sex (male, n (%)) |
168 (70.0) |
59 (74.9) |
109 (67.7) |
0.184 |
|
Admission type |
||||
|
Sepsis or septicemia |
77 (32.1) |
28 (35.4) |
49 (30.4) |
0.312 |
|
COPD (%) |
30 (12.5) |
12 (15.2) |
18 (11.2) |
0.729 |
|
Post-surgery |
26 (10.8) |
7 (8.9) |
19 (11.8) |
0.178 |
|
Trauma |
24 (10.0) |
9 (11.4) |
15 (9.3) |
0.216 |
|
Solid organ tumour |
25 (10.4) |
8 (10.1) |
17 (10.6) |
0.182 |
|
Cerebrovascular accident |
20 (8.3) |
6 (7.6) |
14 (8.7) |
0.165 |
|
Other, n (%) |
38 (15.8) |
9 (11.4) |
29 (18.0) |
0.108 |
|
Comorbidity |
||||
|
Hypertension |
178 (74.2) |
53 (67.1) |
125 (77.6) |
0.221 |
|
Diabetes |
155 (64.6) |
38 (48.1) |
117 (72.7) |
0.106 |
|
Coronary heart disease |
152 (63.3) |
54 (68.4) |
98 (60.9) |
0.732 |
|
Carcinoma |
61 (25.4) |
15 (19.0) |
46 (28.6) |
0.235 |
|
Chronic liver disease |
25 (10.4) |
6 (7.6) |
19 (11.8) |
0.118 |
|
Chronic renal failure |
36 (15.0) |
14 (17.7) |
22 (13.7) |
0.216 |
|
Other |
136 (56.7) |
47 (59.5) |
89 (55.28) |
0.542 |
|
Laboratory findings when starting to use tigecycline |
|
|||
|
WBC (×109) |
13.8 ± 9.3 |
11.6 ± 8.4 |
14.5 ± 9.8 |
0.216 |
|
Hemoglobin (g/L) |
127.4 ± 75.6 |
114.6 ± 54.6 |
133.1 ± 68.5 |
0.218 |
|
Albumin (g/L) |
42.6 ± 9.7 |
39.5 ± 8.6 |
43.7 ± 8.6 |
0.328 |
|
ALT |
29.85 ± 35.652 |
31.01 ± 47.835 |
28.18 ± 29.969 |
0.195 |
|
AST |
44.72 ± 73.52 |
47.7 ± 89.14 |
43.15 ± 39.906 |
0.127 |
|
PT/s |
13.80 ± 1.6 |
13.2 ± 1.5 |
14.1 ± 1.8 |
0.527 |
|
APTT/s |
38.69 ± 8.8 |
39.5 ± 8.7 |
38.3 ± 9.5 |
0.486 |
|
TT/s |
14.26 ± 5.7 |
15.4 ± 3.1 |
13.7 ± 4.5 |
0.426 |
|
FBG/g·L-1 |
3.8 ± 2.1 |
3.8 ± 1.9 |
3.8 ± 2.2 |
0.562 |
|
PLT/×109·L-1 |
245.72 ± 164.86 |
227.2 ± 165.6 |
254.8 ± 162.6 |
0.376 |
|
SCr (μmol/L) |
75.4 ± 37.5 |
78.3 ± 45.5 |
73.1 ± 31.7 |
0.164 |
|
Therapeutic regimen |
||||
|
Loading dose: 100 mg (%) |
198 (82.5) |
70 (88.6) |
128 (79.5) |
|
|
200 mg (%) |
42 (17.5%) |
9 (11.4) |
33 (20.5) |
|
|
Maintenance dose: 50 mg Q12H (%) |
224 (93.3) |
72 (91.1) |
152 (94.4) |
|
|
100 mg q12h (%) |
16 (6.67) |
7 (8.9) |
9 (5.6) |
|
|
Treatment duration (d) (25% to 75% range) |
8.32 (6.48?17.23) |
11.25 (7.18?20.18) |
7.34 (5.87?17.18) |
0.017 |
|
APACHE II score |
11.2 ± 3.6 |
14.8 ± 4.0 |
9.5 ± 2.7 |
0.025 |
|
Daily fluid balance (all intake minus all output) [median in mL (IQR)] |
312 (− 298 to 1054) |
328 (− 364 to 1106) |
259 (− 196 to 988) |
0.235 |
The baseline and clinical characteristics in non survivor group and survivor group are presented in (Table 1). In the univariate analysis, the ages of the non-survivor group and survivor group were 78.3 ± 13.9 and 68.0 ± 11.2 years old (p = 0.022), the medians of treatment duration were 11.25(7.18 ? 20.18) and 7.34(5.87 ? 17.18) day (p = 0.017), and the APACHE II score were 14.8 ± 4.0 and 9.5 ± 2.7 (p = 0.025), respectively. There were no significant differences between the two groups in sex, admission type, comorbidity, laboratory findings and daily fluid balance when starting to use tigecycline.
According to the results in (Table 2), comparison of clinical laboratory findings of patients between two groups, there were no significant difference in the ALT, PT between before and after treated with tigecycline, there were significant difference in the TT, APTT, FBG, PLT between before and after treated with tigecycline. The SCr was significant difference before and after treated with tigecycline only in the non-survivor group and no significant difference in the survivor group.
Table 2: Comparison of clinical laboratory findings of two groups before and after treated with tigecycline.
|
Variable |
Non-survivor groups (n = 79 ) |
p |
Survivor groups (n = 161 ) |
p |
||
|
before |
after |
before |
after |
|
||
|
ALT |
31.01 ± 47.835 |
52.11 ± 138.694 |
0.106 |
28.18 ± 29.969 |
35.46 ± 42.462 |
0.315 |
|
AST |
47.7 ± 89.14 |
107.48 ± 205.132 |
0.032 |
43.15 ± 39.906 |
62.69 ± 109.034 |
0.045 |
|
PT/s |
13.2 ± 1.5 |
14.8 ± 2.4 |
0.528 |
14.1 ± 1.8 |
14.5 ± 2.3 |
0.483 |
|
APTT/s |
39.5 ± 8.7 |
51.3 ± 10.4 |
0.0086 |
38.3 ± 9.5 |
49.5 ± 11.4 |
0.0057 |
|
TT/s |
15.4 ± 3.1 |
21.5 ± 8.1 |
0.0052 |
13.7 ± 4.5 |
19.5 ± 7.4 |
0.0017 |
|
FBG/g·L-1 |
3.8 ± 1.9 |
2.1 ± 1.2 |
0.0038 |
3.8 ± 2.2 |
2.5 ± 1.3 |
0.0023 |
|
PLT/×109·L-1 |
227.2 ± 165.6 |
174.8 ± 135.6 |
0.0017 |
254.8 ± 162.6 |
182.7 ± 165.6 |
0.0019 |
|
SCr (μmol/L) |
78.3 ± 45.5 |
119.7 ± 75.8 |
0.0016 |
73.1 ± 37.5 |
76.7 ± 27.8 |
0.528 |
ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; PT: Prothrombin Time; APTT: Activated Partial Thromboplastin Time; TT: Thrombin Time; FIB: Fibrinogen; PLT: Platelet Count; SCr: Serum Creatinine
According to the results in (Table 3), comparison of liver dysfunction events and hypofibrinogenaemia events between two groups, there were no significant difference after treated with tigecycline. The proportion of the kidney failure events and change in creatinine value were greater in the non-survivor group (p < 0.001) after treated with tigecycline than in the survivor group.
Table 3: Summary of adverse events of two groups after treated with tigecycline.
|
Variable |
Non-survivor groups (n = 79) |
Survivor groups (n = 161) |
p |
|
Liver dysfunction events (n/n %) |
22 (27.8) |
32 (19.9) |
> 0.05 |
|
Hypofibrinogenaemia events (n/n %) |
36 (45.6) |
69 (42.9) |
> 0.05 |
|
Kidney failure events (n/n %) |
11 (13.9) |
0 (0) |
< 0.01 |
|
ΔCre value (mean ± SD) |
70.33 ± 43.36 |
12.95 ± 11.53 |
< 0.01 |
According to the results in (Table 4), multivariate analysis (stepwise approach) identified that age (p = 0.019), ΔCre (p < 0.001), SCr (p = 0.038), APACHE II score (p = 0.031) were independent predictors of tigecycline associated mortality. Other conventional markers were poor predictors of tigecycline associated mortality in that they did not reach the threshold of statistical significance.
Table 4: Multivariate analysis of factors for tigecycline associated mortality.
|
Variable |
Cutoff |
β |
SE (β) |
Odds Ratio |
95%CI |
p-value |
|
Age |
79 |
0.68 |
0.2593 |
1.7723 |
1.13 - 3.17 |
0.019 |
|
ΔCre (μmol/L) |
36 |
1.1025 |
0.2964 |
3.028 |
1.68 - 5.25 |
< 0.001 |
|
SCr |
112 |
0.1847 |
0.068 |
1.046 |
1.16 - 1.37 |
0.038 |
|
APACHE II score |
12.7 |
0.5063 |
0.1869 |
2.475 |
2.04 - 21.68 |
0.031 |
We selected the cutoff point with the largest Youden index. An age of ≥ 79 years, ΔCre (μmol/L) ≥ 36, SCr ≥ 112 and APACHE II score ≥12.7 were selected as the cutoff points for predicting tigecycline associated mortality (Table 5).
Table 5: The sensitivity and specificity of the best cutoff point of Age, Δ cre, SCr and APACHE II as a discriminator of the probability of tigecycline associated mortality.
|
Variable |
Sensitivity |
Specificity |
|
Age≥79 |
0.562 |
0.418 |
|
ΔCre (μmol/L)≥36 |
0.784 |
0.583 |
|
SCr≥112 |
0.475 |
0.382 |
|
APACHE II score≥12.7 |
0.832 |
0.627 |
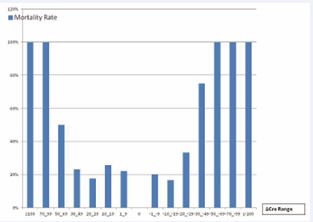
The mortality distribution according to ΔCre had been displayed in (Figure 2). Moreover, the distribution of survivors and non-survivors at different ranges of creatinine change in the studies showed higher percentages of mortality in patients with high ΔCre (include increased higher serum creatinine and decreased lower serum creatinine), after tigecycline treatment. Among all the enrolled patients, there were 142 patients with increased serum creatinine and 45 patients with decreased serum creatinine, 53 patients serum creatinine no change after treatment with tigecycline.
Figure 2 Mortality distribution of all the enrolled patients according to ΔCre.
DISCUSSION
To the best of our knowledge, this is the first study to investigate mortality after tigecycline treatment. In this retrospective observational study, we report results of patients after tigecycline treatment which focused on common clinical indicators associated with 28 day mortality. The age, ΔCre, Serum Cr and APACHE II score were independently associated with 28 day mortality. ΔCre was the most sensitive factors for 28 day mortality based on the logistic regression analysis of the present study.
Tigecycline associated hypofibrinogenaemia and coagulopathy have been reported [5-7]. In this study, the overall incidence of hypofibrinogenaemia was 43.8%, non-survivor group and survivor group were 45.6% and 42.9% respectively. There was no difference in hypofibrinogenaemia events, TT, APTT, FBG and PLT after treatment with tigecycline between two groups. These findings are in agreement with prior reports [7,8]. However, hypofibrinogenemia and coagulation parameter were not a risk factor for tigecycline associated 28 day mortality.
According to the multivariate analysis, we identified age, ΔCre, SCr, APACHE II score as independent risk factors for tigecycline associated mortality. Age is a biopsychosocial marker of aging because of its relation to the sum of age-related deterioration in organ function, which may convert into higher mortality risk over time. Prior study found that patients 75 years and older were at increased risk of death [9]. Our study can better reflect the effect of tigecycline on mortality in elderly patients. These results were consistent with those previous studies [4]. APACHE II score was most strongly associated with worsening infection, poor prognosis and hospital mortality [10]. In the present study, the patients treated with tigecycline were mainly the elderly over 70 years old, most of them had high APACHE II score. According to our study, worse patient’s condition could be more prone to developing mortality after the tigecycline treatment. Our findings are consistent with increased risk of death when tigecycline is used may be explained by a worsening infection or potential complications [2].
According to our study, ΔCre and SCr were found to be related to tigecycline associated 28-day mortality. A significant increase in the kidney failure events (p < 0.001) were observed in non- survivor group. To the best of our knowledge, this is the first study to investigate the association between mortality and serum creatinine and variation in serum creatinine. In our study, ΔCre showed the greatest discriminatory power compared to other factors. This suggests that the ΔCre plays a major role in determining the death outcomes after tigecycline usage. The relationship between ΔCre and tigecycline associated 28 day mortality showed a U-shaped distribution. Either the increase of serum creatinine or the decrease of serum creatinine after tigecycline usage increased the mortality of patients. Whether the increase of serum creatinine is more than 50 μmol/L or the decrease of serum creatinine is more than 70 μmol/L, the mortality rate of patients could reach 100%.
We found that a decrease in serum creatinine after tigecycline therapy was frequent. In this case, creatinine was not diluted because there was no difference in fluid balance. Our study is in agreement with prior reports: Thongprayoon’s research showed that a baseline serum creatinine < 0.4 mg/dl was seen to increase the mortality risk, even compared with those patients with serum creatinine > 1.5 mg/dl [11]. Kang et al., in a study of 486 ICU patients on the effect of changes in serum creatinine after admission ICU, reported that not only an increase in serum creatinine, but also a decrease in serum creatinine was associated with mortality in critically [12]. Decrease in serum creatinine contents can be explained decrease in creatinine generation rate which represents muscle mass [13]. These findings suggest these patients in non survivor group had a more serious course of disease and more complicated treatment course. John et al., found that critical illness is associated with significant falls in serum creatinine from admission to discharge [14]. From those findings and muscle relationship, we reasonably speculated that a lower serum creatinine might be indicative of patient’s lower muscle mass or malnutrition, thus defining lower serum creatinine as a marker of lesser capacity to respond to the critical situation. The patients in non survivor group had more comorbidities than the survivor group and needed more infusions or diuretics which could dilute the serum creatinine concentration and provide a falsely lowered serum creatinine. Until now, these disease factors were known to be harmful to patients. A decrease reflects the patient’s worse physical condition [15]. The increase of serum creatinine reflected the deterioration of the patient’s renal function in non-survivor group, which led to the increase of patients’ mortality. The decrease of serum creatinine reflected the worse clinical diseases status of the patients in non-survivor group, which also led to the increase of the patient’s mortality rate after tigecycline therapy.
The cause of tigecycline associated mortality is unclear. In terms of tigecycline, it has anticancer properties, including cell cycle arrest, apoptosis and oxidative stress and also inhibit mitochondrial oxidative phosphorylation, cell proliferation, migration, invasion and angiogenesis. Another mechanism of its antitumour effects is inducing autophagy [16]. Autophagy in eukaryotic cells is a constitutive process in which damaged macromolecules and organelles are degraded and recycled and to maintain intracellular homeostasis and cell integrity. Paradoxically, autophagy can contribute to cell damage but may also serve to protect cells. Dysregulation of autophagyis involved in the pathogenesis of many of metabolic and age-related diseases. Elderly individuals are particularly sensitive to the dysregulation [17]. These studies suggested that the balance of autophagy activation is important for maintaining people’s health and recovering from diseases. In the patients we studied, the continuous usage of tigecycline could activate autophagy in the patients. It is obvious that our patient’s autophagy may have been out of balance due to the combined influence of various factors.
Recent evidence suggests that autophagy is implicated in cell injury during renal diseases [18]. Autophagy is now widely recognized as a renoprotective cellular response in acute kidney injury of various causes. However, persistent activation of autophagy would promote renal injury [19]. A previous study reported that the activation of autophagy markedly improved renal function in young rats, but was ineffective in aged rat kidneys. Aged kidneys are refractory to autophagy activation in a rat model of renal ischemia reperfusion injury [20]. Our results show that the mortality was significantly related to the kidney failure events and decline in renal function in non-survivor group compared with survivor group. Because the patients in the non survivor group were older, we speculate that tigecycline activated autophagy had no renal protective effect in this group, but appeared renal damage. Autophagy is thought to help maintain muscle mass, but excess autophagy will aggravate muscle atrophy. Under nutrient deprivation skeletal muscle becomes one of the organs with the highest rate of autophagy flux. In recent years, it has been confirmed that various conditions and disease states ranging from fasting, systemic diseases to drug effects can activate autophagy of skeletal muscle [21]. Mitophagy could be triggered by some drugs such as dexamethasone and induced skeletal muscle atrophy [22]. Previous studies demonstrated that autophagy process is activated in the muscles of mice with chronic kidney disease undergoing muscle catabolism [17,21,23]. One of the best validated indicator in chronic kidney disease is muscle atrophy which is related to increased mortality. Muscle atrophy occurs within days to weeks after autophagy activation [23]. The decrease of muscle quality and quantity ultimately aggravates diseases and increases morbidity and mortality. It is reported that increased muscle atrophy is associated with a three-fold increase in 4–6 year mortality [24]. We speculate that tigecycline-activated abnormal autophagy was accompanied by muscle loss which led to a decrease in creatinine production in the non-survivor group after treatment with tigecycline. Therefore, some patients in the non-survivor group showed serum creatinine decrease after tigecycline usage, which was not the improvement of renal function, but the worsening of the disease and eventually died.
Some limitations of this study have to be acknowledged. First, our study is a single-center retrospective analysis and does not represent the average status of all Chinese tertiary university hospitals. Second, the clinical data were the data of several points rather than the changes of clinical data of each patient during the whole disease progression, dynamic monitoring of patient conditions was insufficient and a confounding bias might have been unavoidable. When collecting clinical data, some data was missing and some patients had to be excluded from the analysis. Selection bias may be present and missing data may have affected the results. Third, the risk evaluation criteria have not been verified externally or included in intervention studies. Therefore, it is necessary to verify the clinical value of the criteria in future clinical trials and further randomized controlled trials with larger cohorts are warranted.
Despite these limitations, we identified several clinical features and risk factors of tigecycline associated mortality. We believe that our data truly reflected valuable and reliable information related to mortality associated with tigecycline in this study. Our study will also contribute to raising clinicians’ awareness toward the various risk factors related to tigecycline associated mortality, thus more patients at risk for tigecycline associated mortality can be identified by clinicians and more patients may get more appropriate treatment earlier and avoid death.
CONCLUSIONS AND IMPLICATIONS
The tigecycline associated mortality is emerging as a serious health care issue associated with difficult-to-treat infections and elderly patients. This study demonstrated that age, ΔCre, SCr, APACHE II score represent significant risk factors for the development of tigecycline associated mortality. The results of our studies show that the relation-ship between ΔCre and tigecycline associated mortality showed a U-shaped distribution. We conclude that reduced renal function is increased by tigecycline exposure and contributes to tigecycline associated mortality. Collectively, these data allow us to make a bold speculation that tigecycline associated mortality may be related to abnormal autophagy activated by tigecycline. This information can help clinicians stratify risk of tigecycline-associated mortality for selective monitoring of clinical indicators and optimal treatment regimen management.
Ethics Approval
The study was approved by the First Affiliated Hospital of Naval Military Medical University (CHEC2022-008) and informed consent was not required because of the anonymous and the observational nature of the study.
Competing Interests
The manuscript is approved by all authors for publication. I would like to declare that the work described was original research that has not been published previously, and not under consideration for publication elsewhere, in whole or in part. All the authors listed have approved the manuscript that is enclosed. The authors declare no conflict of interest.
Author Contributions
Gao Wu, Shuxie Wu and Hanbin Wu were involved in the conception of the study; Gao Wu, Xuebin Wang ,Hong Jiang collected and analyzed the data; Hanbin Wu assessed and interpreted the results and prepared figures; Shuxie Wu wrote the first draft; Gao Wu, Shuxie Wu and Hanbin Wu revised the draft.
Availability of Data and Material
We declared that materials described in the manuscript, including all relevant raw data, will be freely available to any scientist wishing to use them for non-commercial purposes, without breaching participant confidentiality.
REFERENCES
- Starzengruber P, Thriemer K, Haque R, Khan WA, Fuehrer HP, Siedl A, et al. Antimalarial activity of tigecycline, a novel glycylcycline antibiotic. Antimicrob Agents Chemother. 2009; 53: 4040-4042.
- Yaghoubi S, Zekiy AO, Krutova M, Gholami M, Kouhsari E, Sholeh M, et al. Tigecycline antibacterial activity, clinical effectiveness, and mechanisms and epidemiology of resistance: narrative review. Eur J Clin Microbiol Infect Dis. 2021; 41: 1003-1022.
- Prasad P, Sun J, Danner RL, Natanson C. Excess Deaths Associated with Tigecycline After Approval Based on Noninferiority Trials. Clin Infect Dis. 2012; 54: 1699-1709.
- Xiao T, Yu W, Niu T, Huang C, Xiao Y. A retrospective, comparative analysis of risk factors and outcomes in carbapenem-susceptible and carbapenem-nonsusceptible Klebsiella pneumoniae bloodstream infections: tigecycline significantly increases the mortality. Infect Drug Resist. 2018; 11: 595-606.
- Hu J, Xiao YH, Zheng Y, Lai YX, Fang XL, Fang Q. Clinical characteristics and risk factors of tigecycline-associated hypofibrinogenaemia in critically ill patients. Eur J Clin Pharmacol. 2020; 76: 913-922.
- Treml B, Rajsic S, Hell T, Fries D, Bachler M. Progression of Fibrinogen Decrease during High Dose Tigecycline Therapy in Critically Ill Patients: A Retrospective Analysis. J Clin Med. 2021; 13: 10: 4702
- Brandtner A, Bachler M, Fries D, Hermann M, Ruehlicke J, Fux V, et al. Tigecycline Interferes with Fibrinogen Polymerization Independent of Peripheral Interactions with the Coagulation System. Antibiotics (Basel). 2020; 14: 9: 84
- Wang J, Pan Y, Shen J, Xu Y. The efficacy and safety of tigecycline for the treatment of bloodstream infections: a systematic review and meta-analysis. Ann Clin Microbiol Antimicrob. 2017; 16: 24
- Mayer-Oakes SA, Oye RK, Leake B. Predictors of mortality in olderpatients following medical intensive care: the importance of functional status. J Am Geriatr Soc. 1991; 39: 862-868.
- Ishikawa D, Takehara Y, Takata A, Takamura K, Sato H. Combination of dirty mass volume and APACHE II score predicts mortality in patients with colorectal perforation. World J Emerg Surg. 2021; 30: 16-17
- Thongprayoon C, Cheungpasitporn W, Kittanamongkolchai W, Harrison M, Kashani K. Prognostic importance of low admission serum creatinine concentration for mortality in hospitalized patients. Am J Med. 2017; 130: 545-554.
- Kang HR, Lee SN, Cho YJ, Jeon JS, Noh H, Han DC, Park S, et al. A decrease in serum creatinine after ICU admission is associated with increased mortality. PLoS One. 2017; 12: e0183156.
- Wyss M, Kaddurah-Daouk R. Creatine and creatinine metabolism. Physiol Rev. 2000; 80: 1107-213
- Prowle JR, Kolic I, Purdell-Lewis J, Taylor R, Pearse RM, Kirwan CJ. Serum creatinine changes associated with critical illness and detection of persistent renal dysfunction after AKI. Clin J Am Soc Nephrol. 2014; 9: 1015-23.
- Diago CAA, Señaris JAA. Should we pay more attention to low creatinine levels? Endocrinol Diabetes Nutr (Engl Ed). 2020; 67: 486- 492.
- Dong Z, Abbas MN, Kausar S, Yang J, Li L, Tan L, Cui H. Biological Functions and Molecular Mechanisms of Antibiotic Tigecycline in the Treatment of Cancers. Int J Mol Sci. 2019; 20: 3577
- Triolo M, Hood DA. Manifestations of Age on Autophagy, Mitophagy and Lysosomes in Skeletal Muscle. Cells. 2021; 10: 1054.
- Lenoir O, Tharaux PL, Huber TB. Autophagy in kidney disease and aging: lessons from rodent models. Kidney Int. 2016; 90: 950-964.
- Lin TA, Wu VC, Wang CY. Autophagy in Chronic Kidney Diseases. Cells. 2019; 8: 61.
- Diao C, Wang L, Liu H, Du Y, Liu X. Aged kidneys are refractory to autophagy activation in a rat model of renal ischemia-reperfusion injury. Clin Interv Aging. 2019; 14: 525-534.
- Franco-Romero A, Sandri M. Role of autophagy in muscle disease. Mol Aspects Med. 2021; 82: 101041.
- Zhang YY, Gu LJ, Huang J, Cai MC, Yu HL, Zhang W, et al. CKD autophagy activation and skeletal muscle atrophy-a preliminary study of mitophagy and inflammation. Eur J Clin Nutr. 2019; 73: 950-960.
- Neel BA, Lin Y, Pessin JE. Skeletal muscle autophagy: a new metabolic regulator. Trends Endocrinol Metab. 2013; 24: 635-643.
- Zhang YY, Yang M, Bao JF, Gu LJ, Yu HL, Yuan WJ. Phosphate stimulates myotube atrophy through autophagy activation: evidence of hyperphosphatemia contributing to skeletal muscle wasting in chronic kidney disease. BMC Nephrol. 2018; 19: 45