Artificial Intelligence, Primary Care, and the Spectrum of Population Health Outcomes: A Framework for Family Medicine and Community Health
- 1. Department of Medical Education, Anne Burnett Marion School of Medicine at Texas Christian University, USA
Abstract
Background: Artificial Intelligence (AI), digital health tools, and predictive analytics are transforming family medicine and community health, yet frameworks to guide their population-level application remain underdeveloped.
Objective: This special communication proposes a spectrum-of-outcomes framework to help family physicians integrate AI into primary care workflows, with an emphasis on person-centered care, chronic disease management, and community health improvement.
Methods: A self-directed inquiry approach, informed by contemporary methodologies for thematic literature reviews, was used to synthesize open-access literature (2010–2025) from PubMed, Google Scholar, and MEDLINE, prioritizing systematic reviews, scoping reviews, and stakeholder engagement studies relevant to AI in primary care.
Spectrum Framework: The proposed conceptual model links AI tools to population health across four domains: (1) digital health scalability in family medicine; (2) value-based primary care models requiring AI for panel management; (3) EHR infrastructure as an AI substrate; (4) behaviorally-informed metrics (mitigators, factors, outcomes) matched to prevention stages.
Implications: AI amplifies family medicine’s core functions when embedded within stage-matched, equity-focused frameworks. Primary care systems adopting embedded AI design may optimize community health outcomes while addressing persistent implementation barriers, including usability challenges, workflow misalignment, and algorithmic bias.
Keywords
• Artificial Intelligence; Primary Care; Family Medicine; Community Health; Digital Health; Value-Based Care; Population Health; AI Implementation Barriers; Primary Care Workflow Integration; Health Equity; Measurement-Based Care
Citation
Kesling GL (2026) Artificial Intelligence, Primary Care, and the Spectrum of Population Health Outcomes: A Framework for Family Medicine and Community Health. J Family Med Community Health 13(1): 1217.
INTRODUCTION
Family medicine faces unprecedented demands from aging populations, multimorbidity, and social determinants of health, making AI and digital health critical to sustainable primary care delivery [1,2]. From 2010 2025, AI evolved from experimental adjuncts to integral components of family medicine practice—telemedicine for access, predictive algorithms for risk stratification, consumer wearables for remote monitoring, and EHR embedded decision support for guideline adherence [1,2].
Despite this rapid expansion, AI implementation in primary care remains predominantly in developmental stages, with limited real-world deployment beyond ambient scribing and basic clinical decision support [3]. A 2025 scoping review of 73 empirical studies found that while AI tools demonstrated strong technical accuracy, particularly in diagnostic decision support, implementation in routine practice was consistently limited by usability barriers, workflow misalignment, trust concerns, equity gaps, and financial constraints [1]. Critically, stakeholder engagement from family physicians and patients has been notably absent from most AI development processes [4,5].
This special communication synthesizes these transformations through a spectrum-of-outcomes lens tailored to family medicine and community health. The framework addresses sequencing (prevention timing), scale (population distribution), and scope (micro-to-macro flows), with behavioral metrics guiding AI deployment. Unlike broad health system analyses, this work centers family physicians as orchestrators of AI-enabled, whole person care across community contexts.
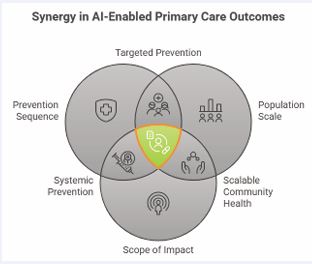
Throughout this manuscript, “equity” refers to health equity across population subgroups—the fair distribution of AI-enabled health benefits regardless of race, ethnicity, socioeconomic status, geography, or digital literacy—unless explicitly stated otherwise in the context of payment or insurance models [6,7] (Figure 1). This figure illustrates the proposed conceptual model organized by prevention sequence (primary, secondary, tertiary), population scale (individual, practice panel, community), and scope of impact (micro to macro). Behavioral metrics (mitigators, factors, outcomes) are mapped to each dimension, with representative measurement instruments indicated.
Figure 1 Framework for Synergy in AI-Enabled Primary Care Outcomes (Kesling, 2026).
METHODS: EVIDENCE SYNTHESIS FOR AI IN PRIMARY CARE
Search Strategy and Study Selection
This narrative synthesis employed a self-directed inquiry approach informed by contemporary methodologies for thematic literature reviews in health services research [1,2]. The author conducted systematic, domain-based searches of open-access medical literature published between January 2010 and March 2025, drawing on established search filter development methods and narrative review best practices to ensure methodological rigor while maintaining the flexibility required for rapidly evolving topics such as AI in primary care [1-3].
Databases searched included PubMed, Google Scholar, and MEDLINE, selected for their comprehensive coverage of primary care and digital health literature and their accessibility for self-directed scholarly inquiry [1,2]. The search strategy was informed by validated family medicine search filters developed by Pols et al., which demonstrated 96.8% sensitivity and 74.9% specificity for sensitive filters and 97.4% specificity and 90.3% sensitivity for specific filters in identifying primary care relevant literature.
Representative search terms included primary terms (‘artificial intelligence,’ ‘machine learning,’ ‘primary care,’ ‘family medicine,’ ‘community health,’ ‘digital health’) combined with secondary domain-specific terms (‘telemedicine,’ ‘electronic health records,’ ‘EHR,’ ‘risk stratification,’ ‘chronic disease management,’ ‘health equity,’ ‘health disparities,’ ‘value-based care,’ ‘implementation barriers,’ ‘stakeholder engagement,’ ‘behavioral metrics,’ ‘predictive analytics,’ ‘workflow integration,’ ‘clinical decision support’). Boolean operators (AND, OR) were used to combine terms across domains, following established search syntax conventions for medical literature databases [1-3].
Inclusion Criteria
Were operationalized following contemporary guidance for scoping and narrative reviews: Studies were included if they were (a) peer-reviewed and published in English; (b) empirical in nature, including scoping reviews, systematic reviews, meta-analyses, qualitative stakeholder studies, or implementation science research; (c) directly relevant to AI applications in primary care or family medicine workflows, including diagnostics, chronic disease management, workflow optimization, telemedicine, and population health analytics; and (d) published in family medicine, primary care, digital health, or health informatics journals [1-3].
Exclusion Criteria
Included algorithm-only validation studies without clinical context, pediatric-only populations, secondary or tertiary care contexts not explicitly addressing primary care workflows, nonempirical publications (editorials, protocols, commentaries without substantive synthesis), and non-English studies [1-3].
Quality Appraisal
Recency was prioritized given the rapid evolution of AI technologies, with preference given to publications from 2020 to 2025, while seminal earlier works establishing foundational concepts were also included [1-3]. Evidence was thematically organized through the author’s iterative reading and coding, following approaches for thematic synthesis in health services research. The author employed an inductive-deductive hybrid coding approach, with initial themes derived from the research questions (effectiveness, implementation barriers, health equity considerations, primary care workflow integration) and refined through iterative engagement with the literature [1,2].
Evidence was thematically organized through the author’s iterative reading and coding, following approaches for thematic synthesis in health services research. The author employed an inductive-deductive hybrid coding approach, with initial themes derived from the research questions (effectiveness, implementation barriers, health equity considerations, primary care workflow integration) and refined through iterative engagement with the literature [1,2].
As a single-author, self-directed synthesis, dual independent coding was not performed; however, to mitigate potential subjectivity and selection bias, thematic categories were systematically cross-referenced against the domain structures and taxonomies reported in recent high-quality scoping reviews of AI in primary care, ensuring alignment with consensus frameworks emerging in the field [1-3]. This self-directed inquiry approach, while not exhaustive, provides a methodologically informed synthesis that maps AI’s practical utility for family physicians and identifies critical gaps in the equitable deployment of AI across community health contexts.
Domain 1: AI and Digital Health Tools in Family Medicine
Recent scoping reviews identify four primary AI application domains in primary care: (1) early intervention and decision support (29% of studies); (2) chronic disease management (22%); (3) operations and patient management (16%); and (4) acceptance and implementation experiences (33%) [1]. Diagnostic decision support emerged as the most technically accurate application, with AI tools demonstrating strong performance in identifying conditions such as hypertension, diabetes, and cardiovascular risk [1,2]. However, stakeholder priorities diverge from current development focus. A Canadian deliberative dialogue with primary care patients, providers, and health system leaders found that stakeholders prioritized
AI for documentation, practice operations, and triage tasks to improve efficiency while maintaining person-centered relationships and access [4]. Complex AI-driven clinical decision support was viewed as impactful but recognized as carrying potential risks requiring careful implementation [4] Similarly, UK general practitioners emphasized concerns about diagnostic accuracy, AI errors, industry influence, increased workload from unnecessary testing, lack of evidence base, and accountability gaps [8].
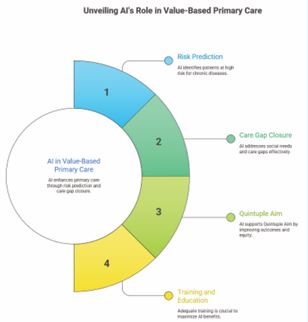
Telemedicine and remote monitoring represent established AI-enabled applications. AI-enabled telemedicine demonstrated equivalence or superiority to in-person care for diabetes glycemic control, heart failure management, and mental health in primary care settings [1,2]. Consumer wearables and AI-driven remote monitoring shifted chronic care from episodic to continuous, enabling family physicians to intervene earlier in disease trajectories [1]. Predictive algorithms embedded in EHRs now identify high-risk patients for proactive outreach, critical for value-based panel management [1,2]. Implementation reality: Community health centers report workflow disruptions and digital divides, underscoring the equity imperative for family medicine [1-6]. AI succeeds when matched to practice readiness and patient digital literacy [1-5] (Figure 2).
Figure 2 Use of Artificial Intelligence (AI) in Value-Based Primary Care (Kesling, 2026).
Domain 2: Value-Based Primary Care and AI Risk Stratification Value-based care models in primary care demand AI for population-level risk prediction and care gap closure—family physicians managing 2,000+ patient panels require automated identification of uncontrolled chronic disease and social needs [1,2]. AI panel management tools stratify patients by risk, prioritize interventions, and track quality metrics, aligning payment with outcomes [2]. AI has the potential to facilitate the achievement of the Quintuple Aim (better patient outcomes, population health, and health equity at lower costs while preserving clinician well-being), but inattention to primary care training in the use of AI-based tools risks the opposite, imposing harm and exacerbating inequalities [1,2]. The impact of AI-based tools on these aims will depend heavily on the decisions and skills of primary care clinicians; therefore, appropriate medical education and training will be crucial to maximize potential benefits and minimize harms [1,2].
Ten transformational AI applications in primary care include: (1) automated documentation and scribing; (2) clinical decision support for diagnosis; (3) risk stratification and population health management; (4) patient triage and scheduling optimization; (5) medication management and adherence monitoring; (6) chronic disease remote monitoring; (7) patient communication and education; (8) care coordination across settings; (9) quality measurement and reporting; and (10) operational efficiency and workflow optimization [1,2].
Family medicine opportunity: AI bridges the volume-to-value transition by automating documentation, risk adjustment, and patient outreach, freeing physicians to focus on relationship-centered care [1,2]. However, poorly implemented AI risks pushing humanity to the margins; done wisely, AI can free up physicians’ cognitive and emotional space for patients and shift the focus away from transactional tasks toward personalized care [1-9].
Domain 3: EHR Infrastructure as AI Substrate in Primary Care EHR adoption has reached critical mass, with 96% of hospitals and 78% of office-based physicians adopting EHRs by 2023, and 70% of hospitals achieving interoperability, enabling primary care data sharing [2]. Patient portals reached 75% offer rate, with 57% active use—critical data streams for AI model training in community practices [2]. Family medicine benefits from EHR reminders that improve immunization rates, depression screening, and statin adherence; AI-augmented clinical decision support further refines these outcomes at scale [2]. A systematic review found that EHRs had a positive impact on healthcare quality, with improvements in adherence to guideline-based care, enhanced surveillance and monitoring, and decreased medication errors [2].
Persistent barriers: Alert fatigue, poor usability, and data silos undermine AI potential [1,2]. Interoperable health information exchanges remain primary care’s best hope for comprehensive risk profiles [2]. The greatest opportunities for AI integration in primary care are centered on enhancing comprehensive service delivery, particularly by improving diagnostic accuracy, optimizing screening programs, and advancing early disease prediction [2]. However, the most significant challenges arise in stewardship and resource generation functions, with key concerns about data security and privacy, technical performance issues, and limited data accessibility [2].
Domain 4: Spectrum-of-Outcomes Framework for AI in Family Medicine
The proposed Spectrum-of-Outcomes Framework is a conceptual model developed through this self-directed inquiry to guide the integration of AI tools into primary care workflows. It synthesizes evidence from recent reviews and stakeholder studies to operationalize AI deployment across three dimensions tailored to family medicine:
Sequence: Primary (wellness), secondary (early disease), and tertiary (complexity) prevention trajectories, with AI-timed nudges (e.g., annual wellness visits triggering risk algorithms) [1,2].
Scale: Distributional equity across practice panels and communities, using AI-generated Population Health Index visualizations [6,7].
Scope: Micro (patient self-efficacy) to macro (community health) flows, supported by interoperable data and predictive tools [1,2].
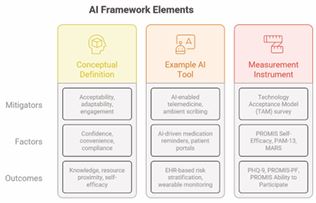
Behavioral metrics: Structure AI deployment through three operationally defined categories:
Mitigators are variables or interventions that facilitate AI adoption and sustained engagement, including acceptability, adaptability, and workflow fit. AI tools must align with family medicine workflows and clinical reasoning to achieve sustained adoption [1-10].
Factors are attributes influencing the effectiveness of AI tools at the patient-clinician interface, including confidence, convenience, and compliance. Patient-facing AI builds trust in recommendations when designed with stakeholder input [4,5].
Outcomes are quantifiable changes in patient health status, behavior, or system-level performance attributable to AI integration, including knowledge acquisition, resource proximity, and self-efficacy. AI connects patients to community resources and enables proactive population health management [6,7] (Table 1).
Table 1 Maps each framework element to its operational definition, example AI element, and recommended measurement instrument.
This framework is intended to serve as a foundation for future empirical validation and refinement, not as a prescriptive or empirically tested intervention. It guides stage-matched AI interventions—from consumer wearables that promote self-efficacy to practice-level algorithms that optimize prevention cascades [1,2].
DISCUSSION: EMBEDDED AI DESIGN FOR FAMILY MEDICINE
AI transforms family medicine from reactive firefighting to predictive relationship management, but only within structured frameworks like the spectrum-of-outcomes model. “Embedded design” integrates AI into core workflows—risk prediction within EHRs, telehealth within scheduling, wearables within panel management—making improvement a system property rather than an add-on project [1,2].
Participatory co-design emerged as the most frequently voiced strategy for overcoming implementation barriers [10]. The effective deployment of AI in primary health care requires a match between the AI tools being developed and the needs of primary health care practitioners and patients [5]. Currently, the majority of AI development targeted toward potential application in primary care is being conducted without the involvement of these stakeholders [4,5].
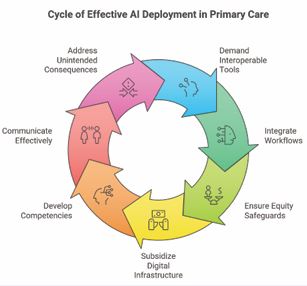
Practice implications: Family physicians should demand interoperable AI tools with validated primary care algorithms, workflow integration, and equity safeguards [1,2]. Community health centers require subsidized digital infrastructure to close access gaps [6]. Six domains of competency for effective deployment of AI-based tools in primary care include: (1) foundational knowledge (what is this tool?); (2) critical appraisal (should I use this tool?); (3) medical decision making (when should I use this tool?); (4) technical use (how do I use this tool?); (5) patient communication (how should I communicate with patients regarding the use of this tool?); and (6) awareness of unintended consequences (what are the “side effects” of this tool?) [1,2] (Figure 3).
Figure 3 Cycle of Effective AI Deployment in Primary Care (Kesling, 2026).
Real-World AI Failures and Implementation Risks
Real-world examples of AI failures in primary care are increasingly documented and must inform responsible deployment. General practitioners in London reported that AI-driven decision support tools prompted unnecessary patient tests and failed to account for population-specific clinical nuances, increasing workload and undermining trust [8]. AI triage tools have been shown to perpetuate disparities across age, ethnicity, language, and socioeconomic status, with algorithmic bias and undertriage resulting in certain patient groups receiving suboptimal care [6,7]. Automated documentation systems, including ambient scribing tools, have sometimes resulted in increased in-basket volume and asynchronous work, contributing to clinician burnout rather than alleviating it. Qualitative syntheses reveal that both clinicians and patients are concerned about the loss of empathy, connection, and trust in the physician-patient relationship when AI tools are introduced without sufficient stakeholder engagement or co-design [8-10]. Data privacy and security concerns are also prominent, with clinicians and patients expressing apprehension about the quality, accessibility, and stewardship of data used to train and operate AI systems [2].
Embedded vs. Bolt-On AI Design
Embedded AI refers to tools seamlessly integrated into core clinical systems, such as EHRs, scheduling platforms, or telehealth modules that operate within the natural flow of clinical work and provide real-time support without requiring clinicians to exit their primary workspace. For example, a hypertension risk prediction algorithm embedded within the EHR can trigger a preventive care reminder during rooming, enabling the clinician to act on actionable insights at the point of care. In contrast, bolt-on AI designs involve external applications added onto existing systems, often requiring separate logins, manual data entry, or parallel workflows. A hypertension risk flag that appears only in a separate analytics dashboard after the patient visit may be overlooked, reducing its utility and impact. The literature consistently emphasizes that embedded AI designs are preferable for primary care, as they support relational care models, align with clinical reasoning, and minimize disruption, while bolt-on tools often disrupt practice, increase cognitive load, and exacerbate unintended consequences [1-10] (Figure 4,5).
Figure 4 Summarizes Enhancing Healthcare with Integrated AI.
Figure 5 Current Challenges and Proposed Solutions to Address Cardiovascular Health Disparities in Artificial Intelligence–Based Tools undefined.
Equity Considerations
Without intentional equity-focused design, AI tools risk reflecting and amplifying existing biases in data, algorithms, and clinical practice patterns [6-11]. A systematic scoping review specifically examining AI and health disparities in primary care identified six domains of concern: (1) access barriers; (2) trust erosion; (3) dehumanization of care; (4) differential agency for self-care; (5) algorithmic bias perpetuating disparities; and (6) external system effects [6]. Strategies for equity-centered AI deployment include: community engagement throughout the AI lifecycle; bias mitigation through diverse training datasets and algorithmic fairness testing; investment in digital infrastructure for vulnerable populations; and equity-first design principles applied at every stage of AI development and deployment [6-11].
Research Gaps and Limitations
This synthesis has several limitations inherent to its self-directed inquiry approach. As a narrative, thematic review, it does not claim the exhaustiveness of a protocol-driven systematic review or meta-analysis. The literature was sampled using public domain search engines rather than through a protocol-registered, comprehensive search, and thematic organization was performed by a single author, introducing potential selection bias and subjectivity. To mitigate these risks, thematic categories were systematically cross-referenced against domain structures reported in recent high-quality scoping reviews [1-3].
The Spectrum-of-Outcomes Framework is a proposed conceptual model that has not yet been empirically validated; it is intended to serve as an inflection point for ongoing discussion and future research, not as a prescriptive or empirically tested intervention. The heterogeneity of study designs in the included literature, the predominance of small-scale feasibility studies, and the rapid pace of AI development further limit the generalizability of the findings. Future mixed-methods pilots and implementation trials are needed to test the framework’s feasibility, acceptability, and impact on patient outcomes in diverse primary care settings [1-3]. Family medicine-led trials must validate AI impact on patient self-efficacy, care coordination, and cost reduction [1,2]. Longitudinal studies should track spectrum metrics across diverse primary care populations [2-6]. Future research should prioritize pragmatic trials, co-design with primary care professionals, and anticipatory planning using future methods to ensure responsible and equitable implementation [1-10].
CONCLUSIONS
AI positions family medicine to deliver 21st-century population health through continuous, comprehensive, coordinated care at scale. The proposed Spectrum-of-Outcomes Framework provides family physicians with a practical lens to deploy digital tools equitably, matching interventions to prevention stages and behavioral readiness.
Primary care practices embracing embedded AI design—where prediction, context, digital capacity, and disciplined workflow decisions interweave—will lead community health transformation through 2050 [1,2]. However, successful AI integration in primary care requires a coordinated, multidimensional approach, with stewardship, resource generation, and financing playing key roles in enabling service delivery [2]. Addressing existing knowledge gaps, examining interactions among these dimensions, and fostering a collaborative approach in developing AI solutions among stakeholders are essential steps toward achieving an equitable and efficient AI-driven primary care system [2-10].
Family Medicine and Community Health must champion this integration to fulfill its person-centered mission amid technological acceleration, ensuring that AI augments, not subverts, the patient-physician relationship [1-9].
ACKNOWLEDGMENTS
The author gratefully acknowledges the thoughtful and constructive feedback provided by the reviewers, whose expertise strengthened this manuscript. Their emphasis on methodological transparency, framework validation, and equity-centered implementation has been invaluable in refining this work.
REFERENCES
- Katonai G, Arvai N, Mesko B. AI and Primary Care: Scoping Review. J Med Internet Res. 2025; 27: e65950.
- Yousefi F, Dehnavieh R, Laberge M, Gagnon MP, Ghaemi MM, Nadali M, et al. Opportunities, challenges, and requirements for Artificial Intelligence (AI) implementation in Primary Health Care (PHC): a systematic review. BMC Prim Care. 2025; 26: 196.
- Iannone S, Kaur A, Johnson KB. Artificial Intelligence in Outpatient Primary Care: A Scoping Review on Applications, Challenges, and Future Directions. Journal of General Internal Medicine. 2026; 41: 364-373.
- Upshaw TL, Craig-Neil A, Macklin J, Gray CS, Chan TCY, Gibson J, et al. Priorities for Artificial Intelligence Applications in Primary Care: A Canadian Deliberative Dialogue with Patients, Providers, and Health System Leaders. J Am Board Fam Med. 2023; 36: 210-220.
- Terry A, Lizotte D, Brown J, Ryan B, Kueper J, Meredith L, et al. Is primary health care ready for artificial intelligence? Stakeholder perspectives: Worth the risk as long as you do it well. Ann Fam Med. 2022; 20: 2905.
- d’Elia A, Gabbay M, Rodgers S, Kierans C, Jones E, Durrani I, et al. Artificial intelligence and health inequities in primary care: a systematic scoping review and framework. Fam Med Community Health. 2022; 10: e001670.
- Bright TJ, Norris KC. Artificial Intelligence and Health Equity. Annu Rev Med. 2026; 77: 525-536.
- Razai MS, Al-Bedaery R, Bowen L, Yahia R, Chandrasekaran L, Oakeshott P. Implementation challenges of artificial intelligence (AI) in primary care: Perspectives of general practitioners in London UK. PLoS One. 2024; 19: e0314196.
- Sahni NR, Carrus B. Artificial Intelligence in U.S. Health Care Delivery.N Engl J Med. 2023; 389: 348-358.
- Darcel K, Upshaw T, Craig-Neil A, Macklin J, Steele Gray C, Chan TCY, et al. Implementing artificial intelligence in Canadian primary care: Barriers and strategies identified through a national deliberative dialogue. PLoS One. 2023; 18: e0281733.
- Ahluwalia M, Sehgal S, Lee G, Agu E, Kpodonu J. Disparities in Artificial Intelligence-Based Tools Among Diverse Minority Populations: Biases, Barriers, and Solutions. JACC Adv. 2025; 4: 101742.